Abstract
Background:
Therapeutic hypothermia can improve neurological status in cardiac arrest survivors.
Objectives:
We investigated the association between the timing of inducing therapeutic hypothermia and neurological outcomes in patients who experienced out-of-hospital cardiac arrest.
Methods:
We evaluated data from 116 patients who were comatose after return of spontaneous circulation and those who received therapeutic hypothermia between January 2013 and April 2017. The primary endpoint was good neurological outcomes during index hospitalization, defined as a cerebral performance category score of 1 or 2. Therapeutic hypothermia timing was defined as the duration from the return of spontaneous circulation to hypothermia initiation. We analyzed the effect of early hypothermia induction on neurological results.
Results:
In total, 112 patients were enrolled. The median duration to hypothermia initiation was 284 min (25th–75th percentile, 171–418 min). Eighty-two (69.5%) patients underwent hypothermia within 6 h, and 30 (25.4%) had good neurological outcomes. The rates of good neurological outcomes by hypothermia initiation time quartile (shortest to longest) were 28.3%, 34.5%, 14.8%, and 28.6% (p = 0.401). The good neurologic outcomes did not differ between hypothermia patients within 6 h or after (26.5% vs 26.7%, p = 0.986). Short low-flow time and bystander resuscitation were associated with good neurological outcomes (p = 0.044, confidence interval: 0.027–0.955), but the timing of hypothermia initiation was not (p = 0.602, confidence interval: 0.622–1.317).
Conclusion:
A shorter low-flow time was associated with good neurological outcomes in out-of-hospital cardiac arrest patients who experienced hypothermia. However, inducing hypothermia sooner, even within 6 h, did not improve the neurological outcomes. Thus, as current guidelines recommend, initiating hypothermia within 6 h of recovery of spontaneous circulation is reasonable.
Introduction
Neurological impairment is an important and critical complication to consider for patients who are successfully resuscitated after cardiac arrest. 1 Therapeutic hypothermia (TH) has been proposed in previous studies as a method for improving neurological status in cardiac arrest survivors,2,3 and several studies have shown that TH can reduce neurological complications in unconscious patients who experienced out-of-hospital cardiac arrest (OHCA).4,5 The Hypothermia after Cardiac Arrest Study Group 6 analyzed patients who recovered after cardiac arrest due to ventricular fibrillation and concluded that TH can improve their neurological outcomes and reduce mortality.
On the basis of prior studies, TH has been included as a treatment option for cardiac arrest in the American Heart Association’s guidelines. 7 Regardless of initial rhythm (i.e. shockable or not) and the location where cardiac arrest occurred (i.e. in hospital or out of hospital), well-controlled TH (32°C–34°C for at least 24 h) is strongly recommended in comatose patients following successful resuscitation. 8
Recently, a randomized clinical trial 9 reported promising efficacy for TH in cardiac arrest. However, several factors in daily clinical practice, including the time interval between the occurrence of cardiac arrest and the initiation of TH, can affect the efficacy of TH. Unlike in controlled trials, the time interval in practice could vary according to the hospital’s emergency triage protocol, resuscitative techniques, and the specific device used to initiate cooling.
Several studies have revealed that a longer “down time” (i.e. the no-flow time plus the low-flow time) is related to worse neurological outcomes and lower survival rates for OHCA patients, which is presumably due to the longer duration of cerebral ischemia. 10 A study using registry data showed that a down time lasting longer than 30 min was associated with very low survival at 1 month. 11 Another study showed that a shorter period between cardiac arrest and achievement of mild hypothermia and the coldest temperature was associated with good neurological outcomes. 12 However, it is unclear whether the time interval between the occurrence of cardiac arrest and the initiation of TH is associated with neurologic outcomes.
Thus, this study aimed to investigate the association between the time to TH initiation and neurological outcomes in patients who experienced OHCA and return of spontaneous circulation (ROSC). We hypothesized that early initiation of TH, which would decrease the time interval between cardiac arrest and the coldest temperature, would be associated with better neurological outcomes.
Methods
Study design and participants
We retrospectively evaluated data from a group of consecutive OHCA patients treated at a single tertiary educational center between January 2013 and April 2017. The hospital has an extensive regional emergency medical and trauma center with 1300 inpatient beds. The study protocol was approved by an institutional review board and ethics committee waived informed consent due to the study’s retrospective nature. The study was performed in accordance with the Helsinki Declaration. Patients or the public were not involved in the design or planning of our research. The study population consisted of all patients older than 19 years admitted for OHCA who were comatose after successful ROSC and those who underwent TH. Comatose patients with neurological problems, such as cerebral hemorrhage and extensive stroke, were excluded, as hypothermia is contraindicated in these patients.
In total, 480 patients with OHCA were treated at the emergency department during the study period. Of those, 357 were successfully resuscitated and experienced ROSC, and 116 patients who were comatose after successful ROSC underwent TH. After excluding four of these patients because of an unknown hypothermia initiation time, 112 patients were ultimately included in the study. Patients were divided into quartile groups according to the time to TH initiation (Figure 1).
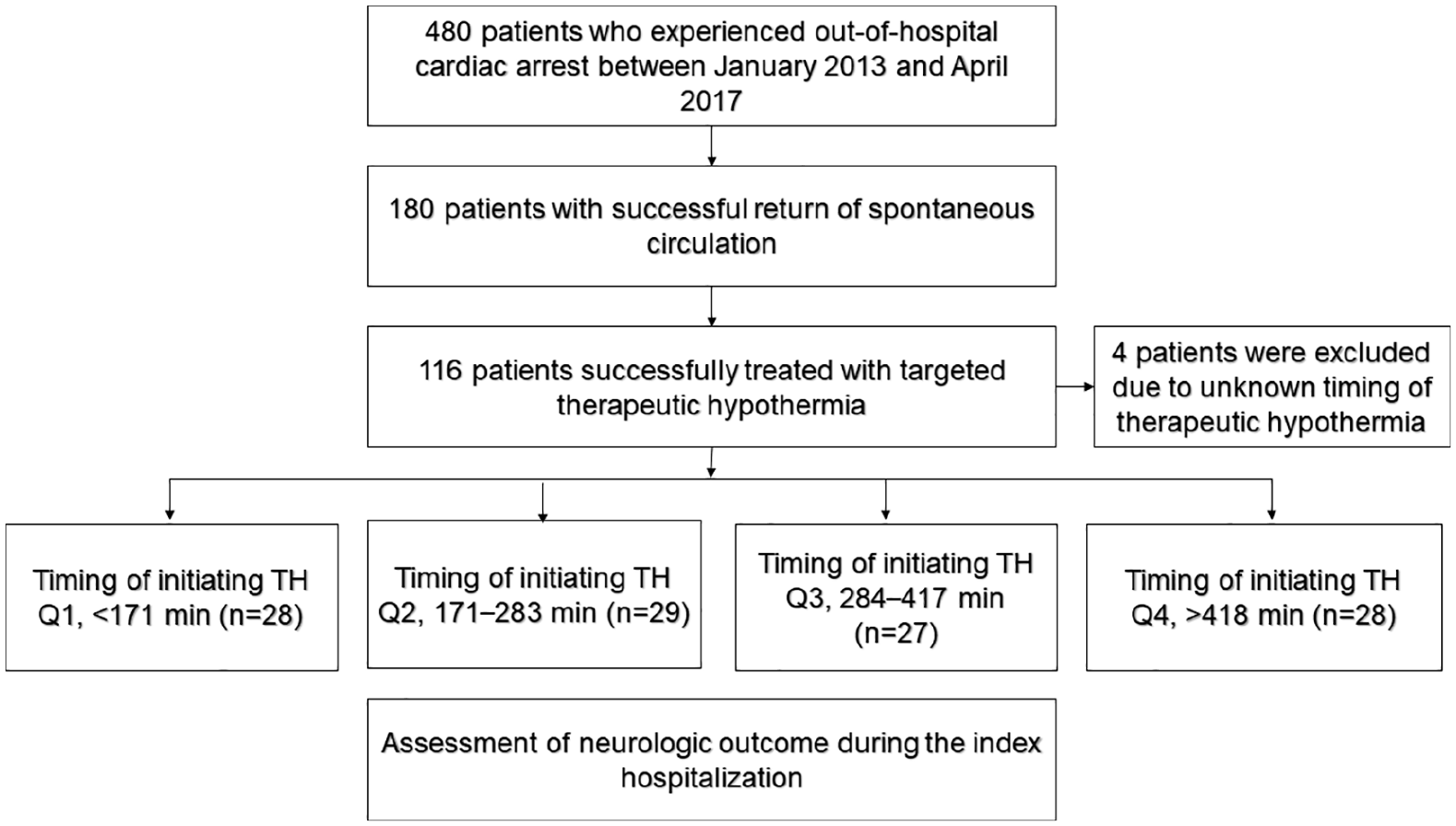
Study flow diagram.
Definitions and endpoints
The timing of TH was calculated as the time interval between ROSC and TH initiation. No-flow time was defined as the interval from the detection of cardiac arrest to the beginning of resuscitation. Low-flow time was defined as the time interval from resuscitation (by bystander or emergency medical services) to ROSC. Sustained ROSC was defined as the restoration of a palpable pulse for at least 20 min. Down time was defined as the time span between lack of pulse and ROSC, thus comprising no-flow time and low-flow time. The primary endpoint was good neurological outcomes during the index hospitalization. The neurological condition of patients who survived until discharge was evaluated using the cerebral performance category (CPC) and Glasgow Coma Scale (GCS) scores, as measured by a neurologist during consultation and recorded in the discharge summary. The CPC scores were as follows: 1 (no significant impairment), 2 (moderate impairment but able to complete activities of daily living), 3 (severe impairment but conscious), 4 (vegetative state or coma), and 5 (death). A good neurological outcome was defined as a CPC score of 1 or 2. Mild impairments were generally defined as those associated with GCS scores of 13–15; moderate impairments with GCS scores of 9–12; and severe impairments with GCS scores of 8 or less.
TH
The ARCTIC SUN® Temperature Management System (Medivance Inc., Louisville, CO, USA), designed to monitor and control a patient’s temperature within a range of 32°C–38.5°C, was used in this study. The apparatus consists of an electronic module and disposable ARCTICGEL™ pads used to achieve the target TH set according to our protocol. In our TH protocol, body temperature (BT) was lowered to 34°C over 4 h and maintained for 20 h. Rewarming was initiated after 24 h, during which BT was increased slowly for 12 h and then continued at normothermia (37°C) for up to 12 h, for a total TH duration of 48 h.
Statistical analysis
Data are expressed as mean values ± standard deviations or medians and interquartile ranges (25th–75th percentile) for continuous variables and as numbers and percentages for categorical variables. Between-group comparisons were performed using a one-way analysis of variance or Kruskal–Wallis, chi-square, or Fisher’s exact tests, as appropriate. Logistic regression analysis was used to identify predictors of good neurological outcomes. The variables were as follows: age, sex, prior coronary artery disease, comorbidity, transfer from another hospital, location of cardiac arrest, witnessed or not, bystander cardiopulmonary resuscitation, pulse rate, first type of rhythm, and low-flow time and timing of therapeutic HT. A multiple logistic regression model was constructed using a stepwise method for all analyses; complete data sets were used, and the level of statistical significance was set at p < 0.05. All analyses were performed using SPSS 22.0 for Windows (SPSS Inc., Chicago, IL, USA).
Results
The median duration to initiating TH was 284 min (range: 171–418 min). In total, TH was initiated in 82 patients (73.2%) within 6 h of ROSC. There were 30 patients (26.8%) who showed good neurological outcomes (CPC scores of 1 or 2).
Baseline characteristics according to the timing of TH initiation
The patients were divided into quartile groups according to the TH initiation time, as shown in Table 1. There were no significant differences in age, sex, hypertension status, the presence or absence of diabetes, or the presence of other comorbidities between groups. The rates of witnessed cardiac arrest (i.e. OHCA but in the presence of another person), cardiopulmonary resuscitation (CPR) performed by a bystander, initial shockable or non-shockable rhythm, low-flow time, and duration of hypothermia did not differ between groups. The target temperature for hypothermia and the time to reach target temperature were also not different between groups. Patients transferred from other hospitals had longer intervals to TH initiation. Initial vital signs were slightly different between groups, but the difference between the highest and lowest mean arterial blood pressure was only 2 mm Hg. The initial BT was higher in patients with delayed initiation of TH (36.6°C) and lower in patients with early initiation of TH (35.8°C). Laboratory test results revealed higher lactic acid concentrations and a lower pH in patients with early initiation of TH, while all other values were not significantly different.
Baseline characteristics according to timing of therapeutic hypothermia initiation.
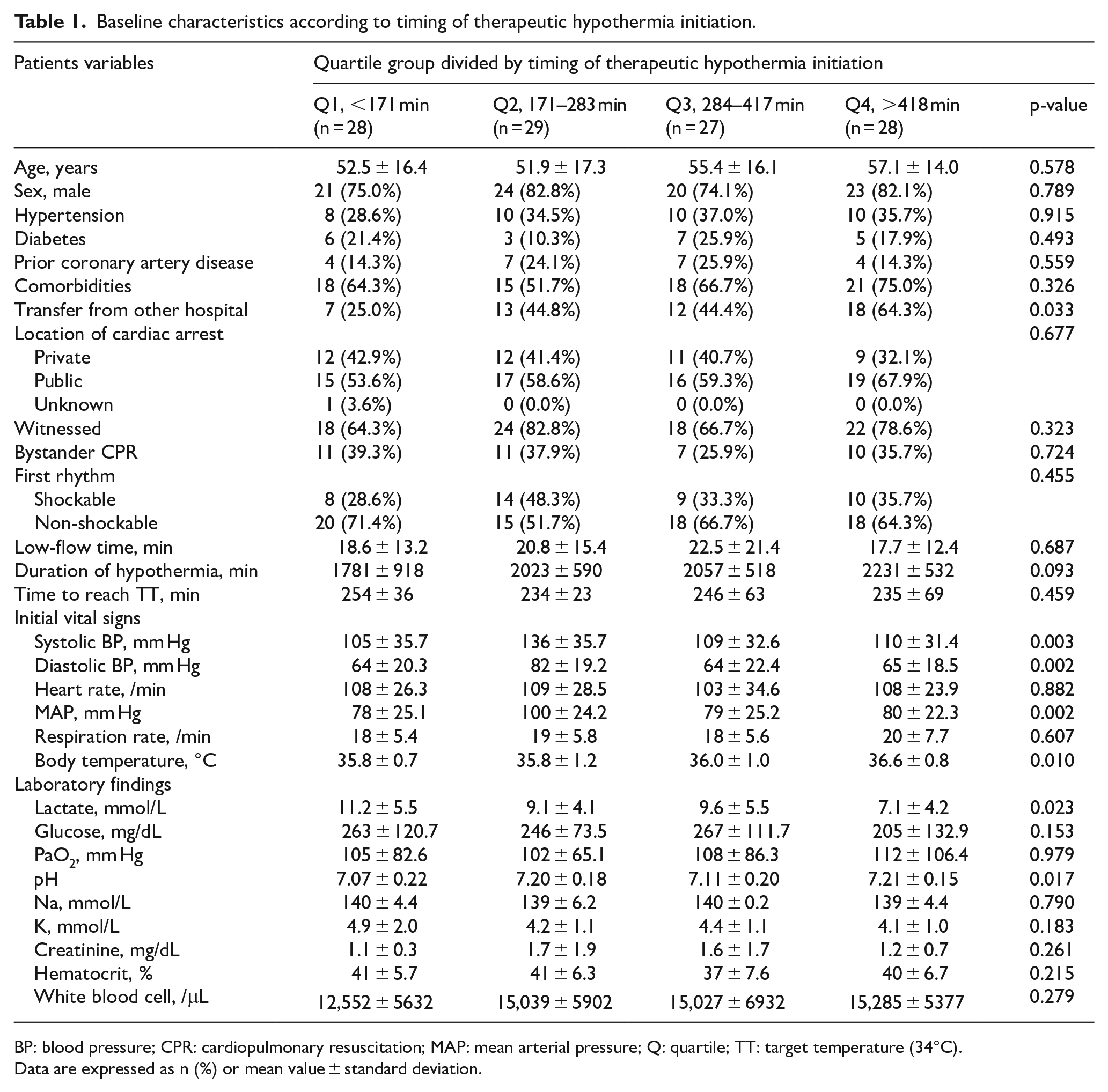
BP: blood pressure; CPR: cardiopulmonary resuscitation; MAP: mean arterial pressure; Q: quartile; TT: target temperature (34°C).
Data are expressed as n (%) or mean value ± standard deviation.
Neurological outcomes according to the time to TH initiation
We evaluated the rate of good neurological outcomes based on the time to TH initiation (Table 2). The overall rates of good neurological outcomes for quartiles 1 through 4 were 28.3%, 34.5%, 14.8%, and 28.6%, respectively, (Figure 2). Early TH initiation was not associated with a tendency toward either increased or decreased neurological outcomes. The rate of good neurological outcomes was higher in patients with an initial shockable rhythm than in those with an initial non-shockable rhythm (39% vs 19%). The rate of good neurological outcomes based on the time to TH initiation did not differ between patients with initial shockable and non-shockable rhythms. The neurologic outcomes related to TH within 6 h are presented in Supplementary Table s1, which shows that the neurologic outcomes did not differ between patients that received TH within or after 6 h (CPC 1–2: 26.8% vs 26.7%, p = 0.986 in overall patients with any type of rhythm; 40% vs 36.4%, p = 0.833 in patients with shockable rhythm; 20% vs 23.5%, p = 0.757 in patients with non-shockable rhythm).
Neurologic outcomes according to timing of therapeutic hypothermia initiation and initial rhythm.
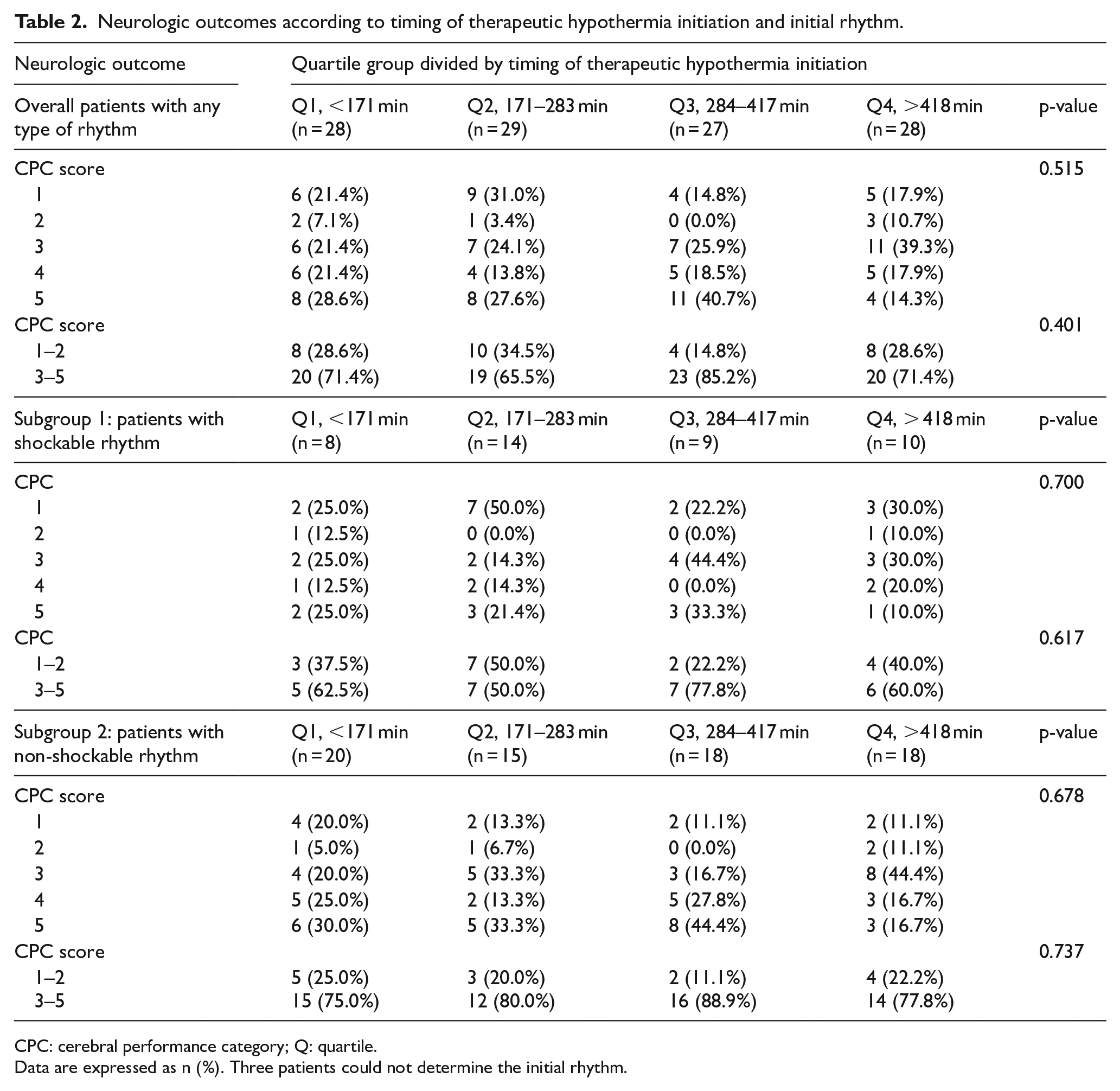
CPC: cerebral performance category; Q: quartile.
Data are expressed as n (%). Three patients could not determine the initial rhythm.
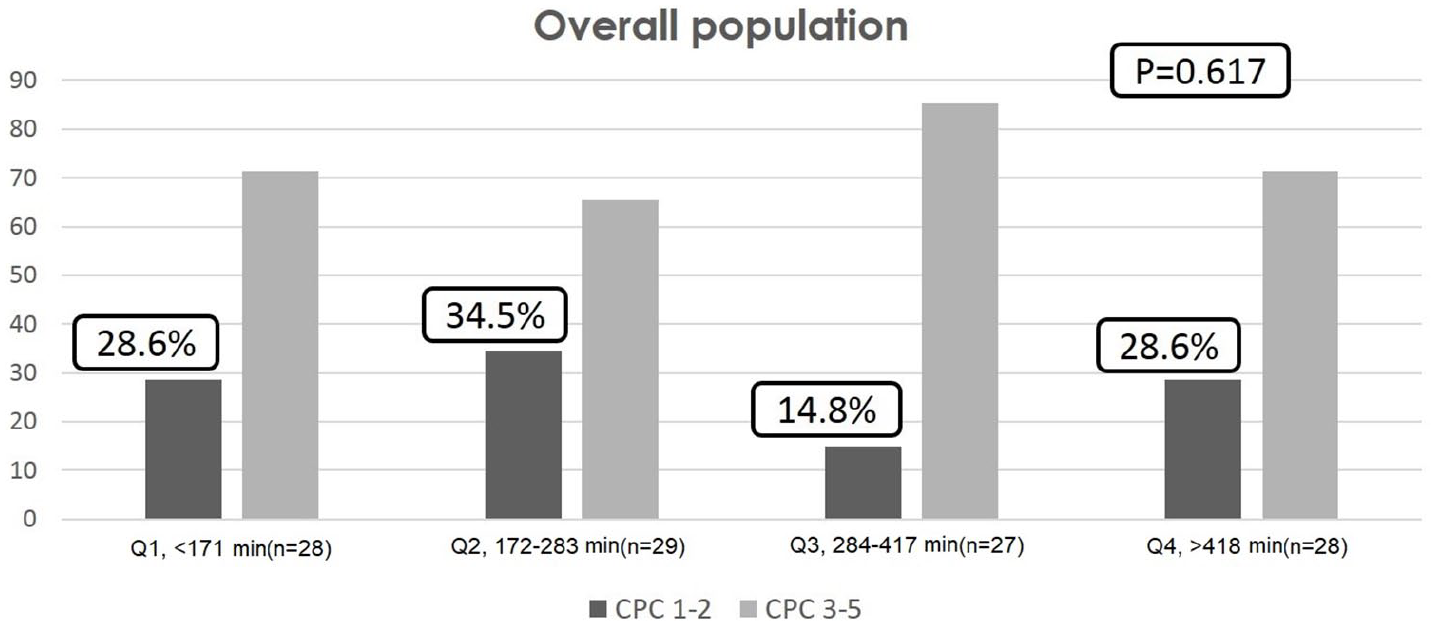
Neurological outcomes according to the timing of therapeutic hypothermia initiation.
Independent predictors of good neurological outcomes
The logistic regression analysis showed that the performance of bystander CPR (odds ratio (OR) = 3.451, 95% confidence interval (CI) = 1.116–10.67, p = 0.032) and a shorter low-flow time (OR = 0.162, 95% CI = 0.027–0.955, p = 0.044) were statistically significant predictors of good neurological outcomes. However, early TH within 6 h (OR = 0.905, 95% CI = 0.622–1.317, p = 0.602) or continuous variable (OR = 1.000, 95% CI = 0.998–1.001, p = 0.635) was not a statistically significant predictor (Figure 3). In addition, TH as a quartile group was not an independent predictor, as shown in the Supplementary file (Table s2).
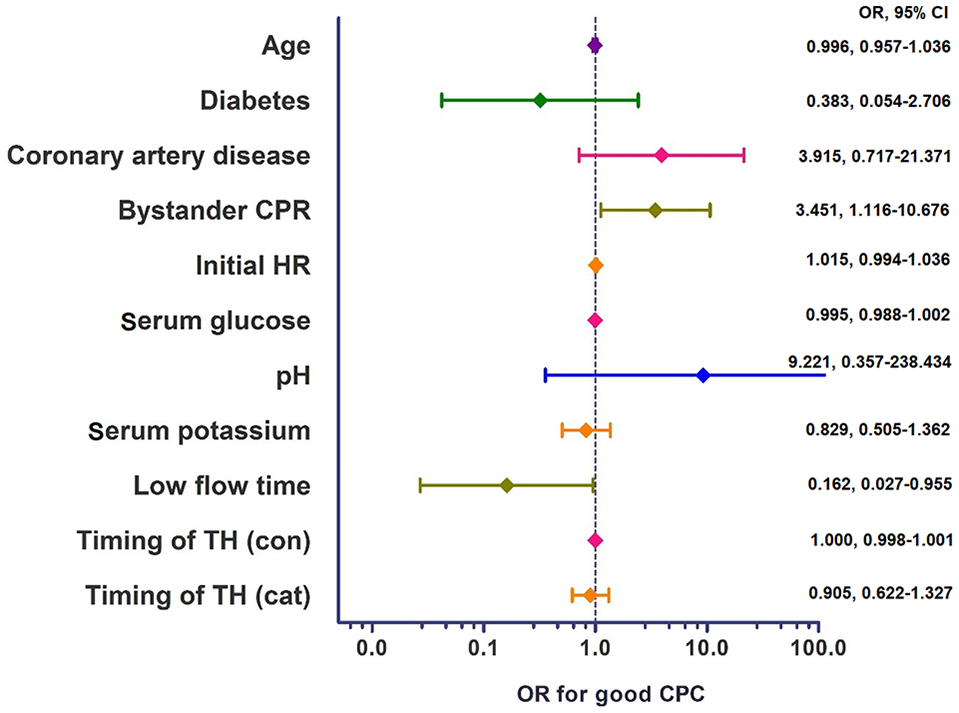
Predictors of good neurological outcomes.
Discussion
In our study, we divided the length of time from ROSC to the initiation of TH into four quartiles to investigate the association between early TH induction and neurological outcomes. We did not observe any statistically significant associations between the time to TH initiation and good neurological outcomes. There were also no significant associations between good neurological outcomes and initial cardiac rhythms. Logistic regression analysis revealed that CPR performed by a bystander and shorter low-flow times were independent predictors of good neurological outcomes, but early TH was not.
In an animal study, early hypothermia initiation had a positive effect on neurological status. 13 Colbourne et al. 14 demonstrated a neuroprotective effect of hypothermia initiated 6 h after brain ischemia in rats. Two prospective randomized studies also showed the effectiveness of early initiation of hypothermia in humans.4,6 In a study by Bernard, hypothermia was achieved within 2 h, and in another study by the Hypothermia after Cardiac Arrest Study Group, hypothermia was achieved at a median interval of 8 h. Hence, the American Heart Association’s guidelines recommend that TH should be performed within 6 h of ROSC. 7
Wolff et al. 12 studied the association between earlier hypothermia initiation and neurological outcomes. The primary endpoints were good or bad CPC scores and serum neuron-specific enolase concentration as a marker of brain damage after cardiac arrest. Multivariate regression analysis of data from 49 consecutive patients revealed that the time to coldest temperature, CPR performed by a bystander, and younger age were significantly associated with good neurological outcomes and lower concentrations of neuron-specific enolase. There were some differences between that study and ours. The time interval from cardiac arrest to the coldest temperature during hypothermia was used in that study, while the time interval from ROSC to TH initiation was used in ours. The time interval from cardiac arrest to the coldest temperature includes down time, the time interval from ROSC to hypothermia initiation, and the actual cooling time to the lowest temperature. A shorter down time is a well-known predictor of good neurological outcomes. Thus, whether the good neurological outcomes in the study by Wolff et al. were because of a shorter down time alone or a shorter down time plus early initiation of TH is unclear. The cooling systems used were also different; Wolff et al. used an endovascular system (Icy, Alsius Corp., Irvine, CA, USA), while we used an external cooling system (ARCTIC SUN).
Nielsen et al. 15 investigated outcomes of TH using the Swedish Cardiac Arrest Registry (SCAR) and found that advanced age, a longer down time, poor neurological status at admission, no witness to the cardiac arrest, and asystole were factors related to poor outcomes, while time to initiation of TH and time to target temperature were not. Time to target temperature and time to lowest temperature were similar in our quartile groups because we controlled the cooling speed and target temperature similarly in our patients. We hypothesized that time to hypothermia initiation would be an independent predictor of good neurological outcomes; however, it was not.
Kim et al. 10 found that a longer down time was inversely correlated with good neurological outcomes in OHCA survivors. They also found that an initial shockable rhythm was more associated with a less rapid drop in neurological than was a non-shockable rhythm, as down time increased. Their results are consistent with ours, as we found that a shorter low-flow time and bystander CPR were associated with good neurological outcomes. Both shorter low-flow time and bystander CPR are related to shorter down time; for instance, a person who receives early CPR from a bystander will have a shorter no-flow time, and therefore a shorter down time.
Two previous studies using data from SCAR and cardiac arrest cases witnessed by Emergency Medical Services in Sweden showed that ventricular fibrillation was an independent predictor of a good survival rate.11,15 Early cerebral blood perfusion can be achieved via defibrillation in patients with a shockable rhythm; therefore, neurological impairment can be reduced. We did not find this result in our study, possibly because down time was not significantly different between patients with shockable and non-shockable rhythms.
A recent study by Nordberg et al. 16 that used a trans-nasal evaporative cooling method found no significant difference in improvement in survival with good neurologic outcome at 90 days, between the early hypothermia intervention group (105 min) and the conventional treatment group (182 min). Trans-nasal evaporation after resuscitation reduces brain temperature faster than does conventional hypothermia. Based on their results, we hypothesized that a faster induction of hypothermia of the brain is perhaps insufficient to ensure neurologic protection following an ischemic, hypo-perfusion insult. We also hypothesized that neurologic outcomes could be more dependent on core BT than on brain temperature and related to the metabolic process, and neither our results nor Nordberg et al. obtained statistically significant results should not discourage future hypothermia studies.
This study has some limitations. First, the sample size was small. However, based on the rarity of comatose patients who are successfully resuscitated following OHCA and the cost of TH, it is difficult to generate large sample sizes in a single center. Second, our sample of patients was from a single tertiary university hospital and not from a multicenter trial. Third, down time is known to be an important predictor of good neurological outcomes, but the no-flow time was missing from our data as it was not included in the medical records. Down time has more clinical influence than does low-flow time on neurological outcomes and survival rates in patients with OHCA. Fourth, the time intervals from ROSC to TH initiation were not widely dispersed. Fifth, nearly one-third of the patients did not undergo TH within 6 h. Finally, the retrospective nature of this study is an essential limitation.
In conclusion, although a shorter low-flow time and a higher bystander CPR rate were associated with good neurological outcomes in OHCA patients who underwent TH following ROSC in this observational retrospective study, we could not find a causal association between early hypothermia initiation and good CPC scores. A prospective, well-structured, multicenter, larger-sized study is needed to verify our results, as it is unclear if early hypothermia inductions are correlated with good CPC scores.
Supplemental Material
Supplementary_material – Supplemental material for Timing of inducing therapeutic hypothermia in patients successfully resuscitated after out-of-hospital cardiac arrest
Supplemental material, Supplementary_material for Timing of inducing therapeutic hypothermia in patients successfully resuscitated after out-of-hospital cardiac arrest by Doo Youp Kim, Jin Sup Park, Sun Hak Lee, Jeong Cheon Choe, Jin Hee Ahn, Hye Won Lee, Jun Hyok Oh, Jung Hyun Choi, Han Cheol Lee, Kwang Soo Cha and Taek Jong Hong in Hong Kong Journal of Emergency Medicine
Footnotes
Acknowledgements
The authors thank Editage for English language editing.
Author contributions
J.S.P. researched and conceived the study. D.Y.K. and J.S.P. wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by clinical research grant from the Pusan National University Hospital (2018) and also supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2017R1D1A1B03033520).
Availability of data and materials
Data sharing is not applicable to this article as no data sets were generated or analyzed during this study.
Informed consent
Informed consent was not sought for this study because of the study’s retrospective nature.
Ethical approval
Ethical approval for this study was obtained from the Institutional Review Board of Pusan National University.
Human rights
This study was performed according to the Helsinki Declaration.
Trial registration
It is not applicable, as this study was not a clinical trial.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
