Abstract
Background:
0.9% saline commonly used for resuscitation of septic patients might induce biochemical changes leading to detrimental effects. Ringer’s lactate being a balanced crystalloid might be beneficial in such a scenario.
Objectives:
We undertook this study to explore in detail the effect of these fluids in the resuscitation of septic patients, and risks and benefits these two fluids would have on the overall prognosis of patients.
Methods:
This was an open-label randomized controlled trial undertaken in emergency medical services attached to the department of medicine at a tertiary care teaching hospital. One hundred sixty adult (⩾18 years old) medical patients admitted with the diagnosis of sepsis fulfilling eligibility criteria were included. They were randomly assigned to receive 0.9% saline or ringer’s lactate. These fluids were given for the initial 24 h only, and after then, the type of fluid given was based on treating physician discretion. Various biochemical parameters were measured at baseline and various time points during the hospital stay. The primary outcome was to find out the incidence of hyperchloremia at 24 h from the time of randomization and during the hospital stay. The secondary outcomes were incidence of acute kidney injury, need for renal replacement therapy; differences in pH, bicarbonate, serum lactate, coagulation parameters, sequential organ failure assessment scores at various time points; and hospital/30-day mortality.
Results:
The baseline characteristics in both groups were comparable. At admission, each group had a serum chloride value which was comparable (p value: 0.595); however, at 24 and 48 h, a statistically significant difference was noticed, with 0.9% saline group having a higher mean serum chloride value (113.66 ± 10.04 v/s 108.98 ± 8.04 mEq/L, p value: 0.001 at 24 h) and (114.75 ± 9.51 v/s 111.12 ± 7.84 mEq/L, p value: 0.022 at 48 h). At 24 and 48 h post-randomization, the incidence of hyperchloremia was significantly higher in the 0.9% saline group (at 24 h, 0.9% saline: 75.0% v/s Ringer’s lactate: 48.8%, p value: 0.001 and at 48 h, 0.9% saline: 77.2% v/s Ringer’s lactate: 60.3%, p value: 0.022), although there was no difference in the incidence of hyperchloremia recorded during the hospital stay. Acute kidney injury incidence at 24 and 48 h post-randomization was significantly higher in the 0.9% saline group (at 24 h, 0.9% saline: 23.8% v/s Ringer’s lactate: 10.0%, p value: 0.020 and at 48 h, 0.9% saline: 29.1% v/s Ringer’s lactate: 15.4%, p value: 0.039). No significant differences in other secondary outcomes were observed.
Conclusion:
Higher incidence of hyperchloremia and a higher rate of acute kidney injury at 24 and 48 h after randomization were noted in the 0.9% saline group.
Keywords
Introduction
Sepsis is primarily a dysregulated, potentially life-threatening host response to an infection. Initial fluid resuscitation in these patients in a state of hypoperfusion or frank shock carries significant prognostic importance. Unfortunately, the question of ideal fluid for resuscitation remains a long-standing debate with every fluid having its own merits and demerits. Various types of acid–base imbalances are known to occur in these patients, but the most commonly seen is metabolic acidosis. It occurs due to renal dysfunction and hypoperfusion leading to elevated lactate levels. Elevated chloride level induced by large volume resuscitation also contributes to iatrogenic metabolic acidosis. 1 Till date 0.9% saline is the most preferred crystalloid used for resuscitation; however, the subsequent hyperchloremic metabolic acidosis induced is a poorly recognized disorder, which on several occasions leads to severe detrimental effects. 2 Ringer’s lactate (RL) being a balanced crystalloid solution contains sodium and chloride in more physiological levels as compared to 0.9% saline and hence does not induce an excess of these ions. Apart from the effects induced by metabolic acidosis, several studies have shown complications due to excess chloride like renal vasoconstriction (reducing glomerular filtration rate), deranged coagulation parameters, and induction of a proinflammatory state due to release of various cytokines.3–5
Due to inadequate data available on the harmful effects of 0.9% saline, especially from the Indian subcontinent, and due to the absence of any standardized guidelines, 0.9% saline continues to be used as a resuscitation fluid of choice. Hence, trials are required to elucidate in detail the benefits of balanced crystalloids over other intravenous fluids, especially widely used 0.9% saline. We tried to evaluate the benefit of a balanced solution, RL over 0.9% saline in the initial fluid resuscitation of patients with sepsis in emergency services. Though many studies have been done in the past regarding an ideal intravenous fluid, there is a paucity of literature in the management of sepsis from the Indian subcontinent, and a randomized controlled study would be the best way to assess any benefit of RL over 0.9% saline if any.
Material and methods
Trial design
This was an open-label randomized controlled trial (RCT) undertaken in emergency medical services attached to the department of medicine at a tertiary care teaching hospital from July 2017 to December 2018. It compared the effect on biochemical, acid–base, and renal function parameters of 0.9% saline-based fluid resuscitation with RL-based resuscitation. This trial conforms to standards of the code of ethics mentioned in the Declaration of Helsinki. The study was approved by the institutional ethics committee. It was retrospectively registered at Clinical Trials Registry of India.
Participants
One hundred sixty adult (⩾ 18 years old) medical patients brought to emergency services of a large tertiary care center with an admission diagnosis of sepsis participated. Sepsis was defined as potentially life-threatening organ dysfunction caused by a dysregulated host response to infection. Organ dysfunction was defined as the change in sequential organ failure assessment (SOFA) score of ⩾2 points. Patients less than 18 years, pregnant, having baseline elevated serum chloride and potassium levels, already received fluid resuscitation before reporting to emergency medical services for a period exceeding 24 h, receiving or expected to receive renal replacement therapy (RRT) within 6 h, on maintenance hemodialysis for chronic kidney disease, presenting in acute heart failure, and not willing to give written informed consent were excluded from the study.
Interventions
Eligible study subjects were randomly assigned to 0.9% saline (NS group) or RL (RL group). Informed consent was obtained from the patient or next of kin. The set bottles were instituted by the staff nurse on duty. The volume of fluids to be administered was not set at a particular value/rate, but was guided as per treating physician’s discretion as well as surviving sepsis campaign guidelines taking into consideration physiological variables like mean arterial pressure (MAP), central venous pressure (CVP), inferior vena cava (IVC) diameter, urine output, and so on. Allocated fluids were given to patients according to the study protocol for the first 24 h after randomization, post this interval subjects could receive any type of intravenous fluid on the discretion of treating physician.
Outcome measures
The primary outcome measure was the incidence of hyperchloremia at 24 h from the time of randomization and during the hospital stay. Hyperchloremia was defined as a serum chloride value of ⩾110 mEq/L. Secondary outcome measures included the incidence of acute kidney injury (AKI) at 24 and 48 h from the time of randomization and during their hospital stay; the difference in pH between groups at 24 and 48 h after randomization and the lowest pH documented during their hospital stay; the difference in coagulation parameters at 24 and 48 h after randomization and worst parameters documented during their hospital stay; the difference in serum lactate levels at 24 and 48 h after randomization and the highest serum lactate documented during their hospital stay; change in SOFA scores from admission to 24 and 48 h after randomization; and need for RRT and hospital/30-day mortality.
Sample size
Based on a previously undertaken study, the expected incidence of hyperchloremia in the 0.9% saline group was 15%, and we expected that hyperchloremia incidence would decrease in the RL group to 3%. To achieve 75% power to detect this difference or effect size of 12% with a significance level of 5%, 75 subjects were required in each group. With a withdrawal/nonevaluable rate of 5%, 80 subjects per group were recruited leading to a total sample size of 160 subjects.
Randomization, allocation concealment, and blinding
The study subjects were randomized by block randomization in blocks of 10 with 1:1 allocation ratio. Allocation concealment was done using sequentially numbered sealed opaque envelopes with the name of the group written in a slip kept inside the envelope. Once the patient was found to be fulfilling all eligibility criteria, the envelope was opened and the patient was resuscitated with the type of intravenous fluid written in slip for the next 24 h. The name and CR No. of the patient were also superscribed over the envelope, so that audit trail is made. It was an open-label trial.
Definitions
The standard reference values of various laboratory parameters which were studied in this trial were: serum chloride 102–109 mEq/L, pH 7.35–7.45, HCO3 22–26 mEq/L, serum lactate 0.5–2.0 mmol/L, serum creatinine for male 0.6–1.2 mg/dL and female 0.5–0.9 mg/dL, prothrombin time 13–16 s, and international normalized ratio (INR) 0.8–1.1. AKI in our study is defined as per the Kidney Disease Improving Global Outcomes (KDIGO) guidelines. 6
Statistical analysis
Statistical analyses were conducted in an intention-to-treat fashion. Continuous variables, distributed normally were expressed as mean ± SD or median and interquartile range (IQR) if the distribution was skewed. Categorical variables were expressed in frequencies and proportions. Differences between means of continuous normally distributed variables were interpreted with the Student t-test and that of non-normally distributed variables with the Mann–Whitney rank-sum test. Categorical variables were interpreted with the chi-square test. The level of significance was expressed as probability values (p value). A two-sided p value < 0.05 was considered statistically significant. Statistical analysis was done using the statistical software ‘SPSS version 26.0’ (SPPS Corp, Chicago, IL, USA).
Results
This open-label randomized controlled study included 160 (⩾18 years old) patients of sepsis admitted to emergency medical services of a tertiary care hospital during study duration of one and half years from July 2017 to December 2018. Four hundred eighteen patients were assessed for eligibility and ultimately 160 patients were recruited, 80 patients in each group. Details of enrolment have been provided in the consort flow diagram (Figure 1). There were 85 (53.1%) males and 75 (46.9%) females in the study cohort. Mean age of study subjects was 43.46 ± 17.99 years in the RL group and 42.44 ± 19.37 years in the NS group. The mean (SD) and median (IQR) amount of fluid received during the first 24 h in the 0.9% saline group were 3.66 ± 0.79 L and 3.50 (3.00–4.00) L, respectively. Seventy (87.5%) patients received more than 2500 mL of intravenous fluids in 24 h. The group of patients, who received RL, mean (SD) and median (IQR) amount of fluid received in 24 h was 3.74 ± 0.92 and 3.50 (3.00–4.00) L, respectively. In this group, the number of patients who received more than 2500 mL of fluid was 68 (85%). Recruited patients in our study were from all over Northern India, including from states of Punjab, Haryana, Jammu and Kashmir, Uttar Pradesh, Bihar, and Himachal Pradesh. Both groups had comparable baseline characteristics. Details of the same are provided in Table 1.
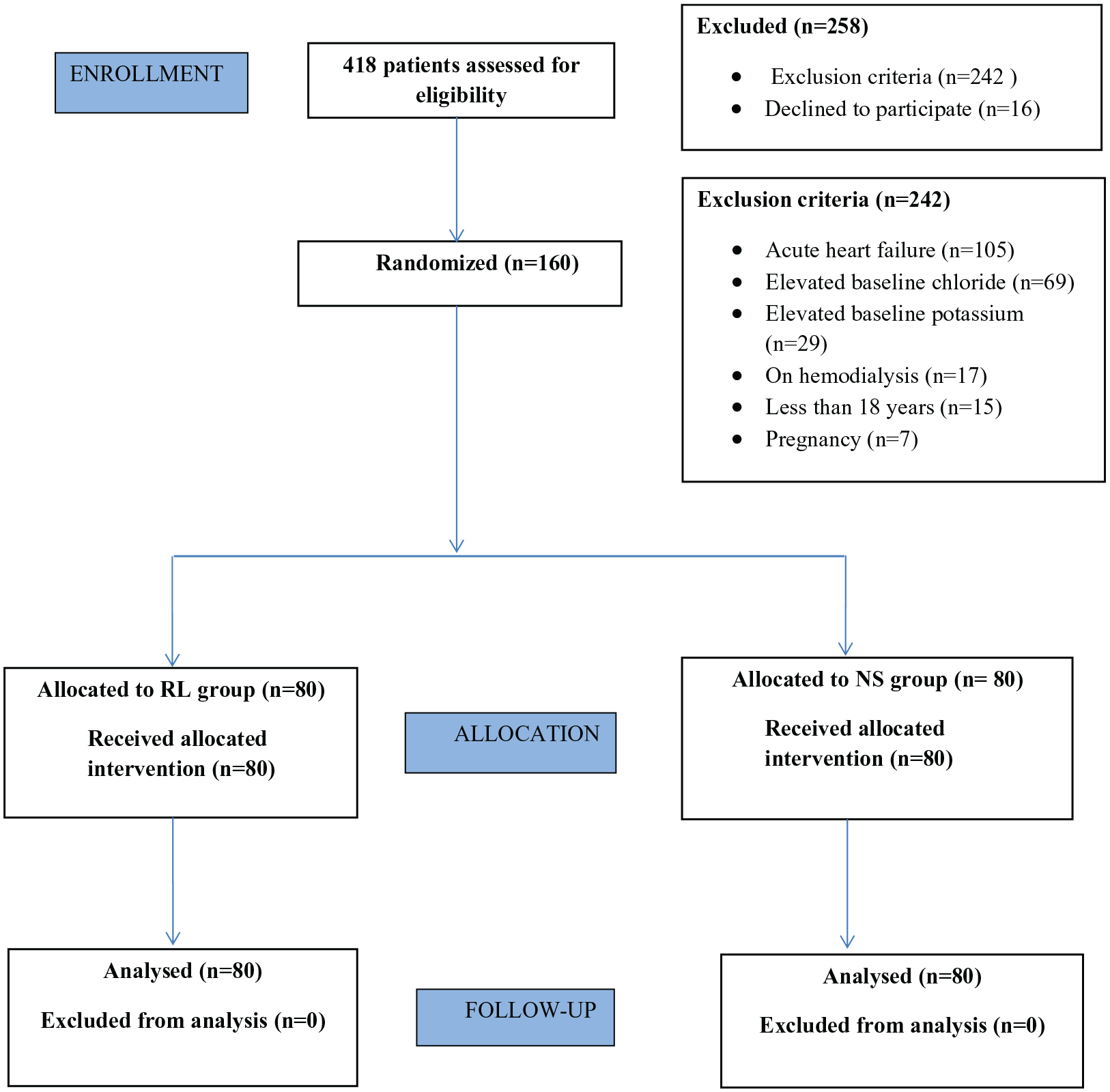
Consort flow diagram of trial.
Baseline characteristics of the patients.
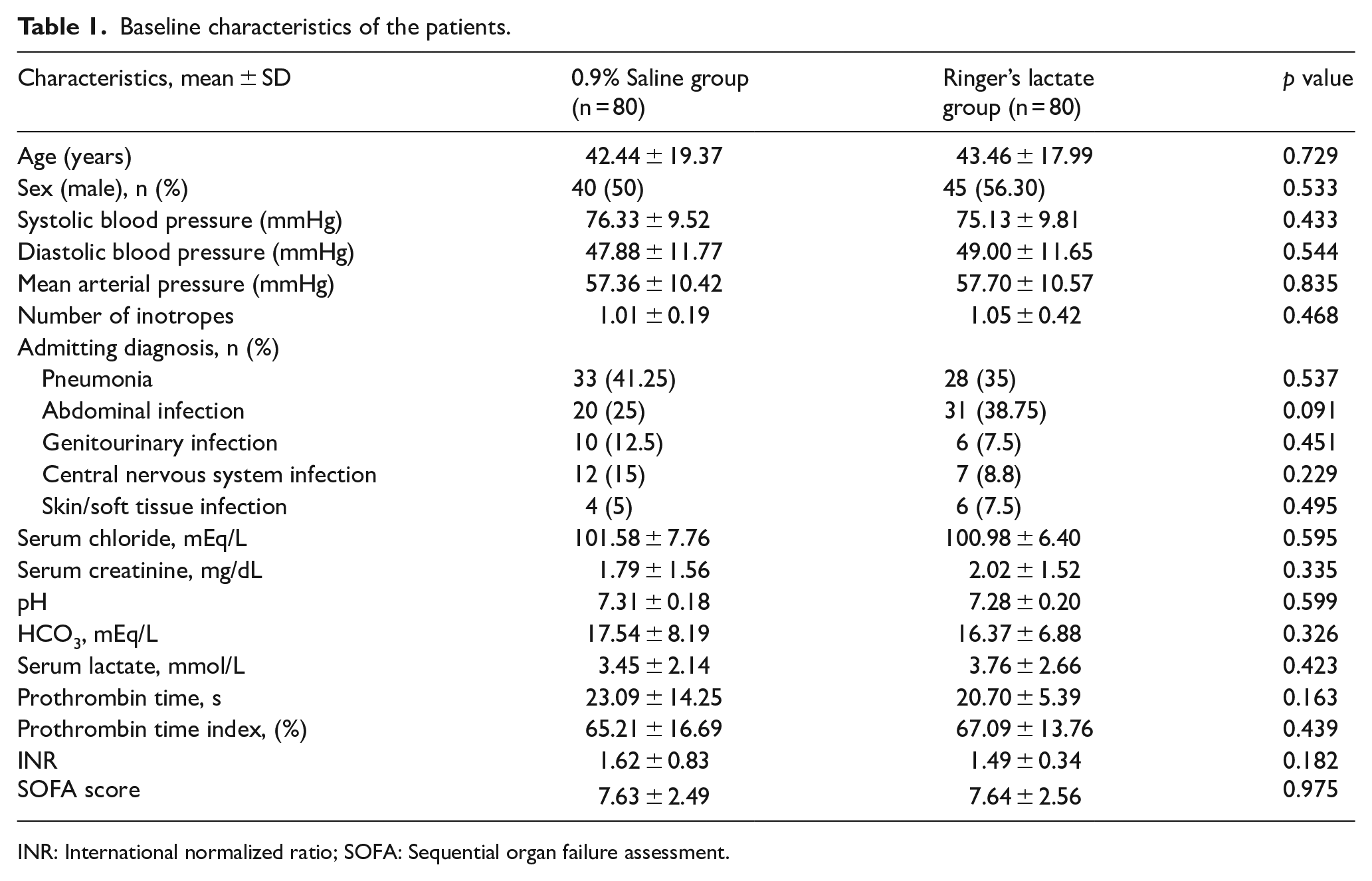
INR: International normalized ratio; SOFA: Sequential organ failure assessment.
Primary outcome
The primary outcome measure was the incidence of hyperchloremia at 24 h from the time of randomization and during the hospital stay. Hyperchloremia in the index study was defined as any value greater than or equal to 110 mEq/L. The mean baseline value for serum chloride was 101.58 ± 7.76 mEq/L in the NS group and 100.98 ± 6.40 mEq/L in the RL group (p value: 0.595). Serial values of serum chloride were measured at 12, 24, 48 h and the highest value during the hospital stay and compared between the two groups. Hyperchloremia at 12 h post-randomization was seen in 75 patients, out of which 27 (33.8%) belonged to the RL group and 48 (60.0%) to the NS group (p value: 0.001). At 24 h, the incidence was significantly higher in the NS group ((NS: 75.0%) v/s (RL: 48.8%); p value: 0.001). There was though no difference in the incidence of hyperchloremia based on the highest chloride level recorded during the hospital stay. These findings are further elaborated in Figures 2 and 3.
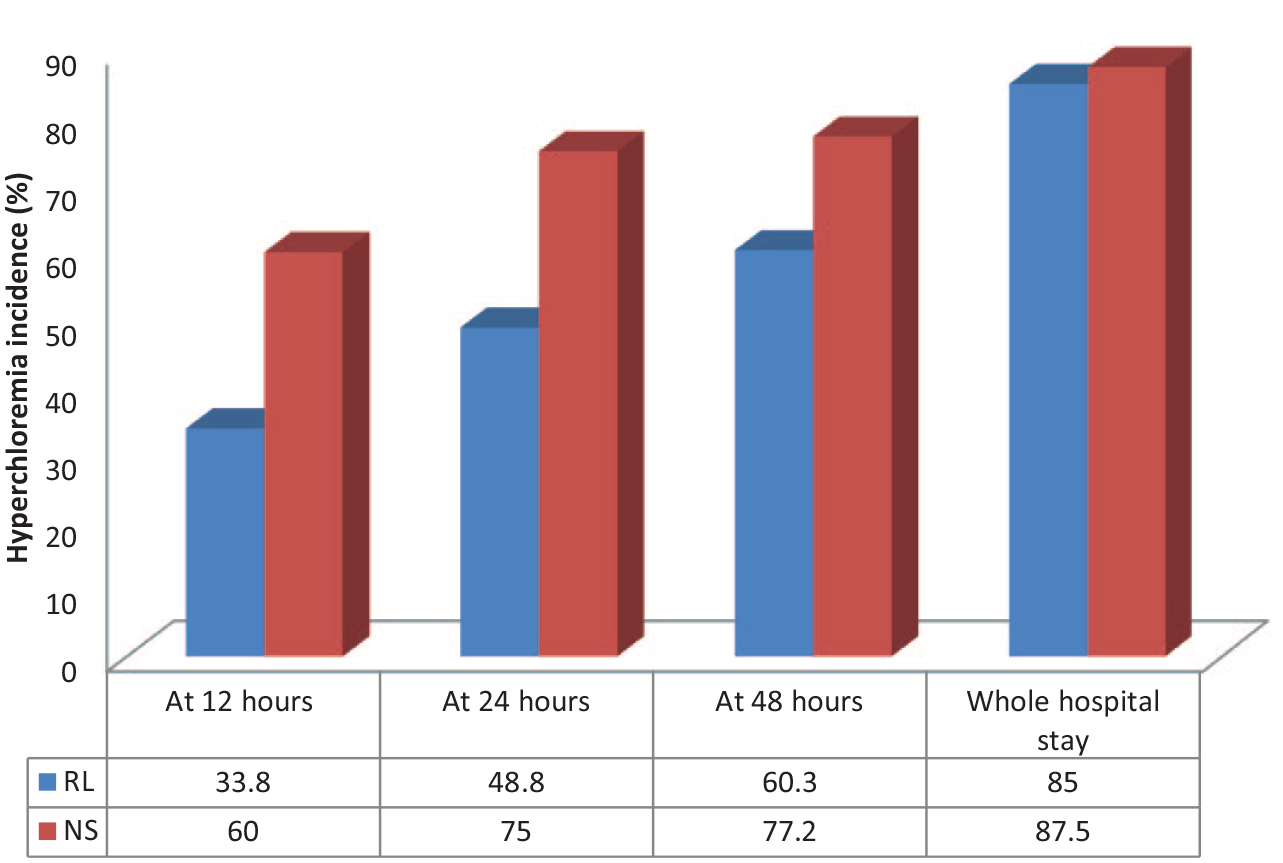
Incidence of hyperchloremia in cohorts of patients resuscitated with Ringer’s lactate (RL) and 0.9% saline (NS) at various time points during their hospital stay.
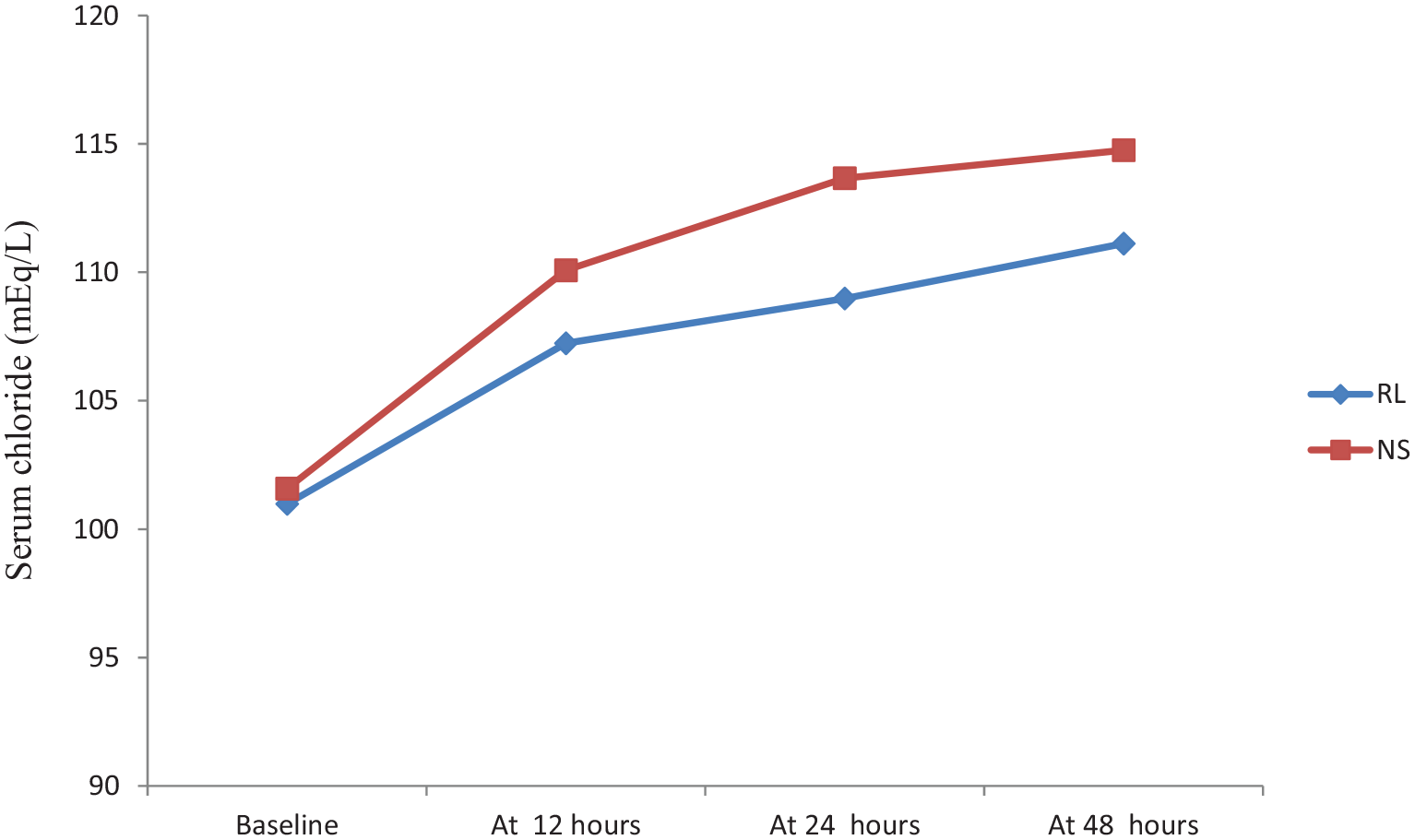
Trends of serum chloride in cohorts of patients resuscitated with Ringer’s lactate (RL) and 0.9% saline (NS) at various time points during their hospital stay.
Secondary outcomes
The incidence of AKI in the whole cohort was 16.9% at 24 h, 22.29% at 48 h, and 33.12% during the hospital stay. AKI at 24 h post-randomization was seen in 27 patients, out of which 8 (10%) belonged to the RL group and 19 (23.8%) belonged to the NS group. At 48 h, AKI was seen in 35 patients, out of which 12 (15.4%) belonged to the RL group and 23 (29.1%) were in the NS group. Overall AKI during hospital stay was seen in 53 patients, in which 21 (26.6%) belonged to the RL group and 32 (40.5%) were in the NS group. The analysis showed significantly higher AKI incidence in the 0.9% saline group at 24 (p value: 0.020) and 48 h (p value: 0.039) intervals (Figure 4). Though the trend of lower pH values at 12, 24, 48 h, and throughout hospital stay was noted in the NS group, this difference was not significant (Figure 5). Serum bicarbonate values did not differ significantly between two groups; however, a steep rise in values was observed in the first 12 h of fluid resuscitation in the RL group. There was no significant difference in serum lactate between the two groups (Figure 6). Similar observations were recorded in prothrombin time between the two groups (Figure 7). The mean SOFA score of the NS group at baseline was 7.63 ± 2.49, while it was 7.64 ± 2.56 in the RL group. SOFA scores at 24 (NS: 8.80 ± 4.48, RL: 8.04 ± 4.18; p value: 0.268) and 48 h (NS: 8.63 ± 4.79, RL: 7.36 ± 4.39; p value: 0.084) were not significantly different among two groups; however, trends of higher SOFA score were noted in the NS group. Out of the 160 patients, 24 patients (15%) required RRT during the hospital stay. Ten (12.5%) patients belonged to the RL group and 14 (17.5%) belonged to the NS group (p value: 0.376). Fifty-three (33.1%) patients died during the hospital stay and 107 (66.9%) recovered and were discharged. Even though there was no significant difference in in-hospital mortality between the two groups (p value: 0.240), there was a trend of slightly higher mortality rate in the 0.9% saline group [30 (37.5%) v/s 23 (28.8%)]. A similar trend of higher 30-day mortality was noted in the 0.9% saline group [35 (43.8%) v/s 29 (36.3%)], but the difference was not significant (p value: 0.418).
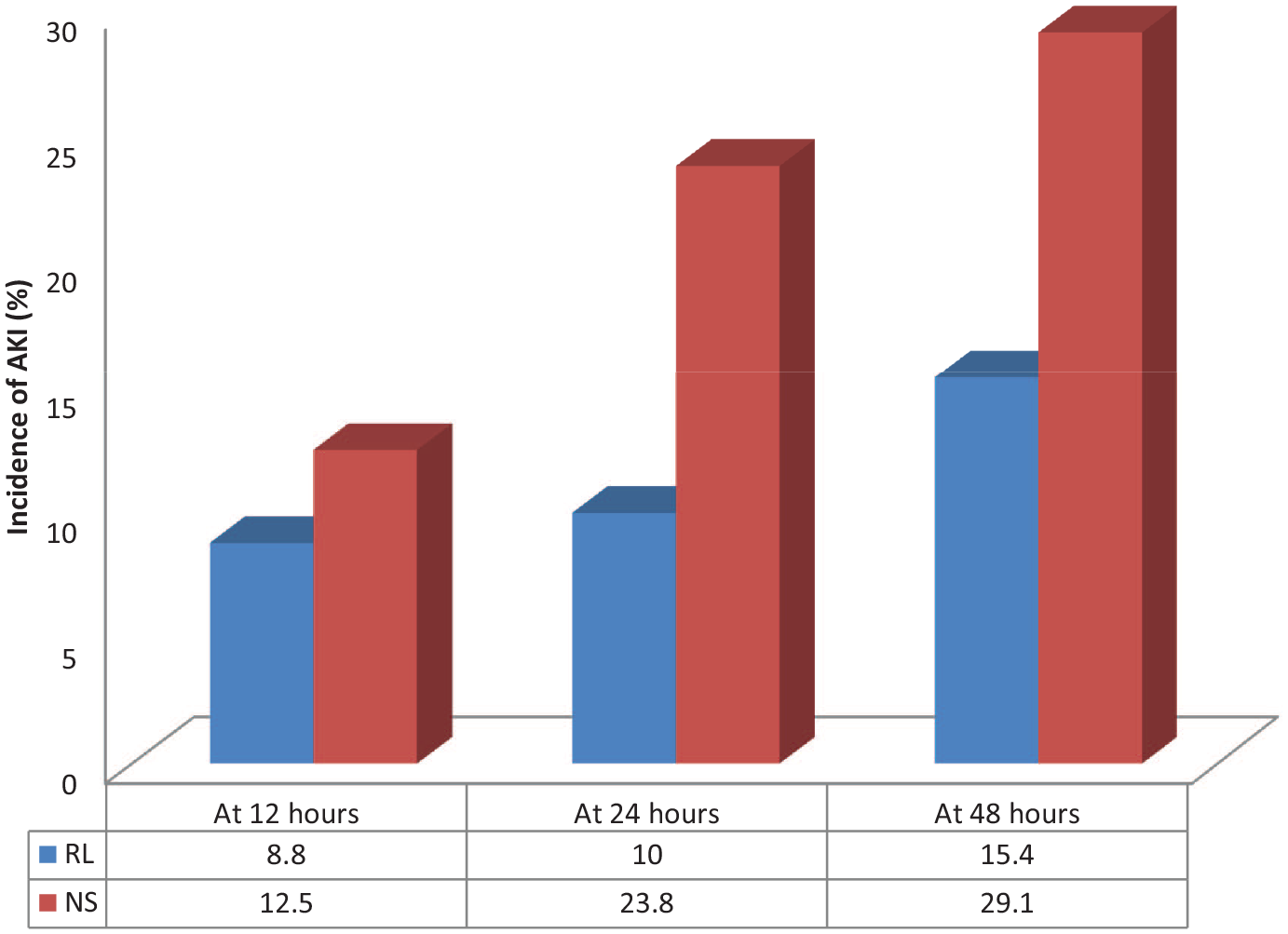
Incidence of acute kidney injury (AKI) in cohorts of patients resuscitated with Ringer’s lactate (RL) and 0.9% saline (NS) at various time points during their hospital stay.
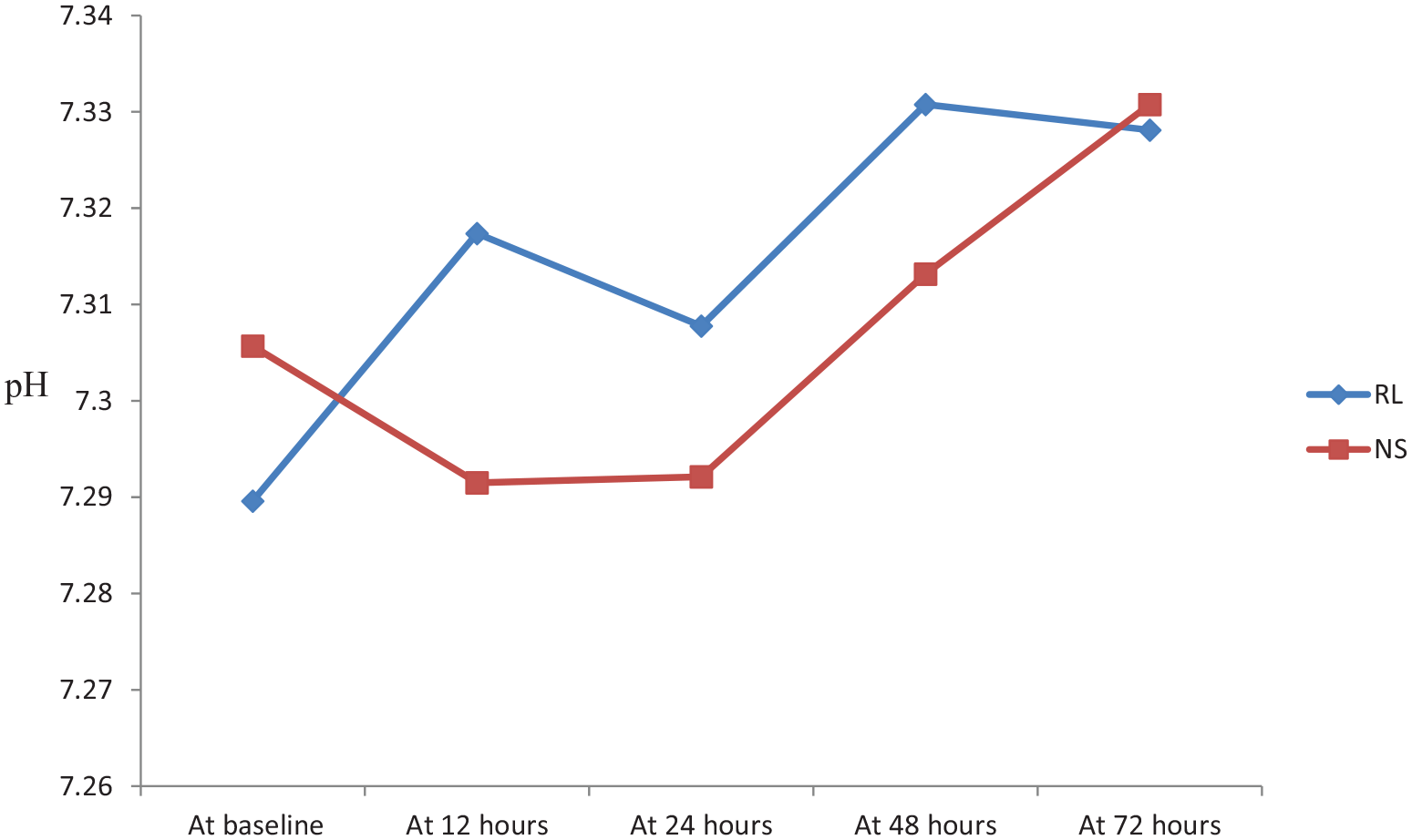
Trends of pH in cohorts of patients resuscitated with Ringer’s lactate (RL) and 0.9% saline (NS) at various time points during their hospital stay.
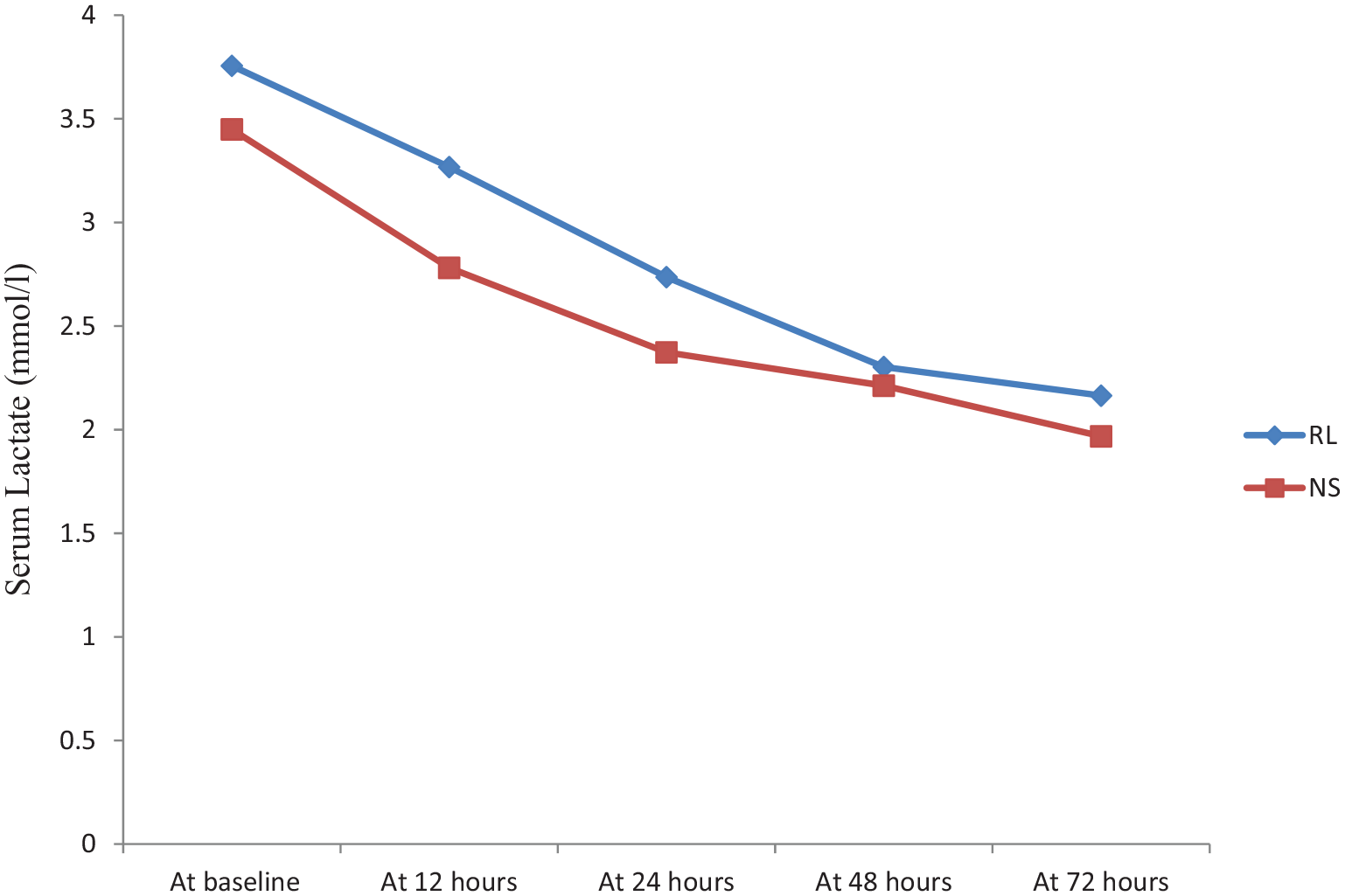
Trends of serum lactate level in cohorts of patients resuscitated with Ringer’s lactate (RL) and 0.9% saline (NS) at various time points during their hospital stay.
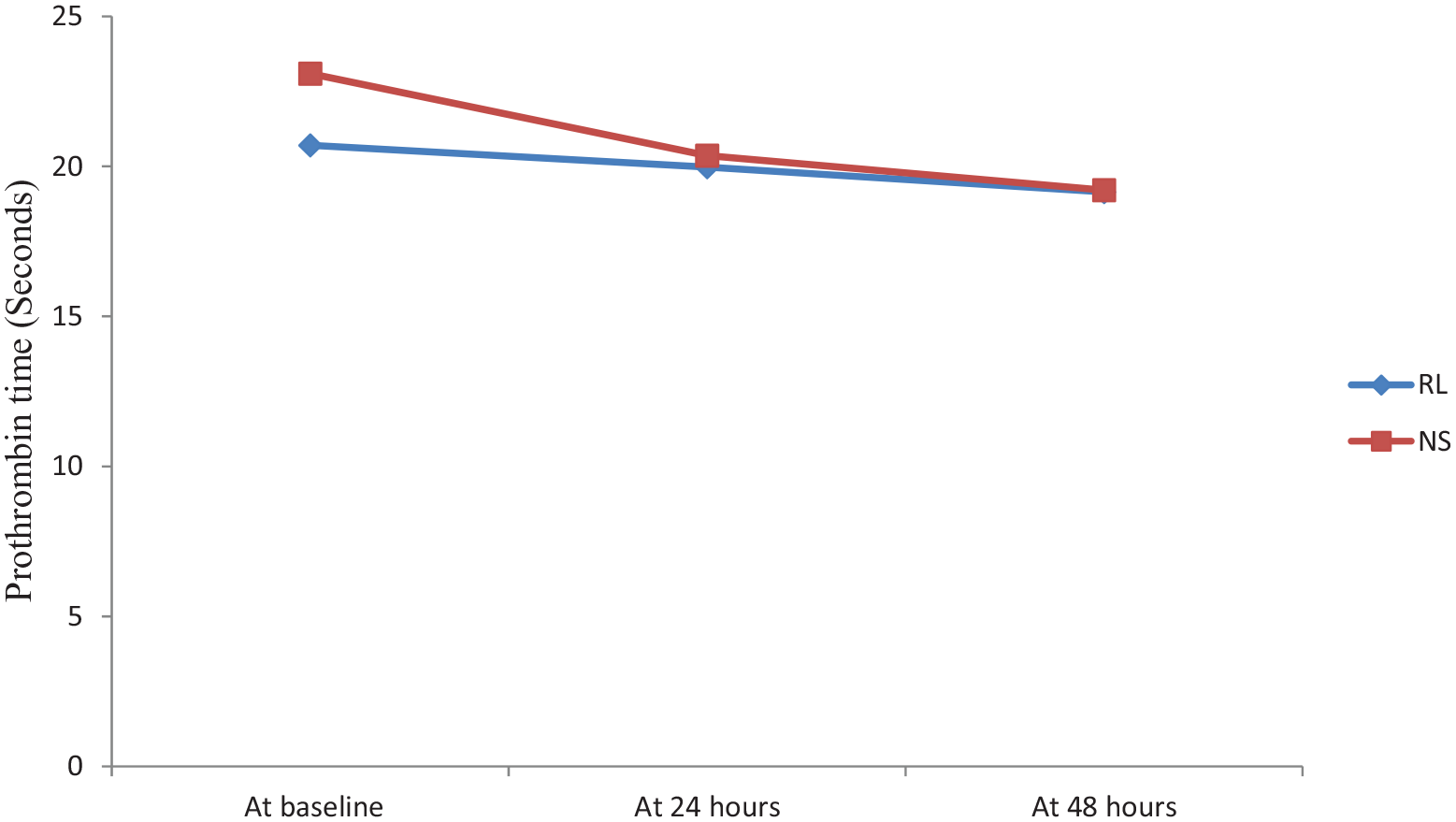
Trends of prothrombin time (PT) in cohorts of patients resuscitated with Ringer’s lactate (RL) and 0.9% saline (NS) at various time points during their hospital stay.
Discussion
This open-label randomized controlled study looked at the incidence rate of hyperchloremia at 24 h post-randomization in cohorts of patients with sepsis admitted in emergency medical services receiving either 0.9% saline or RL for fluid resuscitation. The incidence was significantly higher in the NS group [(NS: 75.0%) v/s (RL: 48.8%); p value: 0.001]. A significantly higher AKI incidence was also noted in the 0.9% saline group at 24 and 48 h intervals as well, though no difference in hospital/30-day mortality was observed.
It is a well-acknowledged fact that fluid resuscitation plays a pivotal role in the management of sepsis, but till date, there is no consensus regarding the choice of intravenous fluid to be used for resuscitation, especially in patients presenting in emergency medical services with sepsis/septic shock from the Indian subcontinent. The literature from the West suggests that resuscitation with 0.9% saline may be associated with biochemical changes, even organ dysfunction in the form of AKI and coagulopathy. A study conducted by Yunos et al. 3 showed a decrease in the mean chloride level from 104.9 ± 4.9 to 102.5 ± 4.6 mmol/L (p < 0.001) while using a chloride restrictive strategy with balanced crystalloids. Scheingraber et al. 7 showed that 2-h perioperative infusion of 30 mL/kg/h 0.9% saline resulted in the rise in serum chloride values from the baseline value of 104 to 115 mmol/L at 2 h as compared to increase of only two points in patients receiving RL (from 104 to 106 mmol/L). A study conducted by Mane 8 in 26 patients showed the mean values of chloride post 6 h of resuscitation with 0.9% saline was 122 mmol/L and with lactated Ringers was 101 mmol/L. Our study also mirrored similar differences in the mean serum chloride values in two study groups. The incidence of hyperchloremia at 24 h was much higher in the 0.9% saline group. This significant difference in the serum chloride levels persisted up to 48 h from the time of randomization. A study done by Raghunathan et al. 9 showed higher serum chloride levels to be associated with increased mortality. It has been postulated that increased delivery of chloride ions to macula densa causes constriction of the renal vessels and subsequent kidney injury. A study done by Chaudhury et al. 10 using 12 healthy volunteers demonstrated sustained hyperchloremia with 0.9% saline compared to plasma-Lyte (p < 0.0001). This study also showed higher reduction in mean renal artery flow velocity (p value: 0.045) and perfusion of cortical tissue (p value: 0.008) from baseline after 0.9% saline infusion, and not after Plasma-Lyte infusion. Yunos et al. 3 conducted an ICU study and showed a higher percentage of AKI and failure as per the RIFLE classification (14.0% vs 8.4%; p value: < 0.001) and the higher need for RRT (10.0% vs 6.3%; p value: 0.005) during the chloride-liberal intravenous fluid strategy period. In our study, a significant difference was noticed between the two groups concerning the incidence of AKI by the KDIGO criteria at 24 h (23.8% v/s 10.0%, p value: 0.020) and 48 h (29.1 v/s 15.4, p value: 0.039). Among the 24 patients requiring RRT in our study, 10 belonged to the RL group (12.5%) and 14 belonged to the NS group (17.5%) showing no significant difference (p value: 0.376). The increased concentration of chloride with 0.9% saline infusion has shown to reduce the strong ion difference and induce a state of nonanion gap metabolic acidosis. Various animal and human studies have shown a difference in pH to occur with lower documented pH being recorded in the 0.9% saline group. Scheingraber et al. 7 in a dose–response study showed that 2-h perioperative infusion of 30 mL/kg/h 0.9% saline resulted in a significant lowering of pH from 7.41 to 7.28 as compared to lactated Ringer’s solution. A slight difference in pH values between two groups was observed in our study as well, with the NS group having lower documented pH values at 12, 24, and 48 h though it was not significantly different (p value > 0.05). It is postulated that 0.9% saline increases the time needed for clot formation which is attributed to the iatrogenic acidosis. A study conducted by Smith et al. to evaluate the outcomes of volume resuscitation on coagulation parameters found no variations in the profile of coagulation after 6 h of fluid resuscitation. However, two parameters not routinely measured, endogenous thrombin potential (ETP) and thromboelastography (TEG) were done. It showed no difference between the two groups in ETP parameters, but TEG results had shown that time from initial clot formation was shorter and fibrin build-up was faster in the plasma-lyte group. 4 Our study assessed the basic coagulation parameters and found no statistical difference; however, more studies with a focus on the sonoclot analyzer may reveal important differences. The landmark SPLIT trial revealed no mortality difference in patients resuscitated and maintained with buffered or unbuffered intravenous fluids (p = 0.40). 11 Another study with 3116 patients with systemic inflammatory response syndrome (SIRS) done by Shaw et al. 12 showed that balanced crystalloids (Plasma-Lyte or Normosol), caused a lower frequency of increase in chloride levels, a lower rate of metabolic acidosis, a shorter duration of hospital stay and lower mortality rates compared to 0.9% saline. In our study, among the 53 patients who died, 23 (43.4%) belonged to the RL group and 30 (56.6%) belonged to the 0.9% saline group, showing no significant difference. Similar observations were seen even in 30-day mortality between two groups (43.8% v/s 36.3%, p value: 0.418). In a single-center, pragmatic, multiple-crossover trial involving 13,347 patients (Balanced Crystalloids, n = 6708; Saline, n = 6639) conducted in emergency services, no difference was found in hospital-free days between two treatment groups; however, a lower incidence of secondary endpoints like a composite of death, new RRT, and persistent renal dysfunction was observed in the balanced crystalloid group. 13 Another similar clustered trial was conducted in ICU setup among 15,802 critically ill patients. A total of 7942 patients were randomly assigned to a balanced crystalloids group, while 7860 patients received saline. The primary outcome assessed in the form of new RRT or persistent renal failure was significantly lower in the balanced crystalloid group. 14
The major advantage of our study lies in the way it is conducted, that is, patients were randomized immediately at the first point of contact to the emergency and not after being admitted to the ICU. This made sure that patients randomized received only the fluid of interest and did not receive any other fluid before recruitment in the study.
The major limitations were first, as our study was a single-center study carried out at emergency medical services, results, therefore, cannot be extrapolated to the ICU population and the patients presenting to the surgical/trauma care setup. The number of patients recruited was small. Our study was an open-label randomized controlled study and this might have introduced bias though the majority of primary and secondary outcomes assessed were objective parameters. We also restricted the assignment of fluids for the initial 24 h only because of logistics issues.
As seen in the index study, the incidence of hyperchloremia in the 0.9% saline group was much higher. A significant difference in the incidence of AKI at 24 and 48 h after study fluid introduction was noted, though no significant difference in hospital/30-day mortality was observed. This study reinforces the serum biochemical changes observed post intravenous fluid resuscitation with 0.9% saline in patients in emergency settings. Multicenter randomized controlled trial recruiting a fairly large number of sepsis patients would provide definitive answers regarding the effects of different resuscitation fluids on hard end points like hospital and 30-day mortality.
Footnotes
Author note
This research work was presented in Association of Physicians of India conference (APICON)-2020, held at Agra, Uttar Pradesh, from 6 January to 9 January 2020.
Author contributions
Rithvik Golla and Susheel Kumar contributed to concepts, design, definition of intellectual content, literature search, clinical studies, and data acquisition. Deba Prasad Dhibhar, Ashish Bhalla and Navneet Sharma contributed to definition of intellectual content, clinical studies and data acquisition.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Informed consent was obtained from the patient or next of kin.
Ethical approval
This trial conforms to standards of the code of ethics mentioned in the Declaration of Helsinki. The study was approved by the institutional ethics committee (NK/3727/MD/699, Dated 10 October 2017). It was retrospectively registered at Clinical Trials Registry of India (Registration number, CTRI/2018/03/012372).
