Abstract
Introduction:
Hyperammonemia can represent organic dysfunction of the brain, kidney, or liver. Evaluation of serum ammonia concentrations as a parameter for organ dysfunction may be justified.
Objective:
To evaluate the performance of serum ammonia as an additional or substitute variable for organ systems in the Sequential Organ Failure Assessment (SOFA) score.
Methods:
A prospective cohort study including 173 patients admitted to the intensive care unit between March 2015 and February 2016. SOFAMONIA scores were defined as follows: SOFAMONIA1 (Glasgow coma scale replaced by serum ammonia), SOFAMONIA2 (serum bilirubin replaced by serum ammonia), SOFAMONIA3 (renal system score replaced by serum ammonia), and SOFAMONIA4 (addition of serum ammonia to the original SOFA as the seventh variable, changing the maximum score from 24 to 28).
Results:
The original SOFA presented an area under the curve–receiver operating characteristic of 0.697 to predict hospital mortality. There was a positive correlation between SOFA and SOFAMONIA scores. SOFAMONIA1 presented a cut-off point of 5 for area under the curve 0.684 (0.610–0.753, 95% confidence interval), SOFAMONIA2 presented a cut-off point of 9 for area under the curve 0.701 (0.626–0.768, 95% confidence interval), SOFAMONIA3 presented a cut-off point of 8 for area under the curve 0.674 (0.598–0.743, 95% confidence interval), and SOFAMONIA4 presented a cut-off point of 8 for area under the curve 0.702 (0.628–0.769, 95% confidence interval).
Conclusions:
The addition of ammonia as the seventh parameter of the SOFA score showed the best performance to predict hospital mortality. The addition of ammonia as a representative of metabolic dysfunction may be useful in the follow-up of critically ill patients.
Introduction
The Sequential Organ Failure Assessment (SOFA) is one of the most commonly used evaluation scales in intensive care, being useful for the evaluation of organ dysfunction. The score is associated with the risk of death of critically ill patients.1–3 The SOFA evaluation of organic dysfunction in the respiratory, cardiovascular, neurological, renal, hepatic, and coagulation systems is performed through clinical and laboratory parameters.3,4 Adaptations of this score have been proposed for better prognostic evaluation in different populations, such as patients with acute or acute-on-chronic liver failure.5–7 Baradari et al. 1 analyzed 250 critically ill patients and compared the performance of a modified SOFA score, in which laboratory parameters were replaced by clinical parameters, showing a positive correlation between the original and the modified score. More recently, Singer et al. introduced a simplified SOFA proposal called the quickSOFA (qSOFA) as a bedside screening criterion, without the need for laboratory tests. Thus, a proposal was raised for rapid and early identification of patients with suspected sepsis. In this tool, only respiratory rate, systolic blood pressure, and level of consciousness were taken into account. 8 A retrospective cohort study involving more than 180,000 critically ill patients admitted to intensive care units (ICU) over 15 years compared the prognostic accuracy between the original SOFA and qSOFA, in addition to the criteria of the Systemic Inflammatory Response Syndrome (SIRS), concluding that the SOFA score demonstrated better prognostic accuracy in relation to the hospital outcome. 9 These observations show that the SOFA score can be modified to improve performance, especially in certain patient groups.
High blood ammonia concentration (hyperammonemia) is observed because of several reasons, particularly liver damage, innate errors of urea metabolism, use of drugs such as valproic acid and chemotherapy, specific bacterial infections (Proteus sp., Corynebacterium, Helicobacter pylori, Klebsiela sp., and Morganella sp.), postoperative period after bariatric surgery, transplants, portosystemic shunt, and hyperalimentation syndrome.10–14 Hepatic dysfunction is the main diagnosis to consider in the presence of hyperammonemia in adults and, excluding acute or chronic liver injury, other causes should be investigated. In this case, it is denominated nonhepatic hyperammonemia (HNH).10,15–17 Ammonia concentrations may not correlate with encephalopathy symptoms, and although it may be asymptomatic, the main signs and symptoms of hyperammonemia are neurological in nature, the most severe being herniated cerebral edema and death.10,12,18 Hyperammonemia may be caused by acute kidney injury (AKI), in which there is insufficient excretion of nitrogen load, 10 but AKI may also be due to the same causes of ammonia elevation, such as systemic inflammatory response with organ dysfunction and other causes of concurrent hepatic and renal aggression. 12 Maldonado et al. 19 defined hyperammonemia as values above 94 µg/dL. Prado et al. defined hyperammonemia as serum concentrations above 35 µmol/L and classified it as mild at concentrations between 36 and 99 µmol/L and severe at concentrations ⩾100 µmol/L, since at these concentrations, there was a significant association with mortality. Butterworth demonstrated that elevated blood ammonia concentrations in patients with liver failure predict the risk of cerebral herniation. 20 Larangeira et al. 15 demonstrated an association between HNH and intracranial hypertension in critically ill patients through ultrasonography of the optic nerve sheath and transcranial doppler.
Considering the possible derivations of the SOFA score already published in the medical literature, besides demonstrating that hyperammonemia can represent organic dysfunction of at least three organs (brain, kidney, and liver), evaluation of the use of serum ammonia concentrations as an additional parameter or substitute for other organic dysfunction parameters of the SOFA score is justified. The aim of this study was to evaluate the performance of serum ammonia as an additional or substitute variable for the hepatic, neurological, or renal organ systems in the SOFA score for intensive care patients.
Materials and methods
Design and location of the study
A prospective cohort study was conducted at a University Hospital with three ICUs for adults. ICUs 1 and 2 each contained 10 beds for clinical and surgical patients over 12 years of age, and the burn ICU contained 6 beds, totaling 26 intensive care beds.
Collection of data and variables
The study population consisted of critically ill patients admitted to the ICUs from March 2015 to February 2016, totaling 12 months. Sampling was performed including, consecutively, all patients with the following inclusion criteria: over 18 years and expected ICU admission for 48 hours or more. Patients without ammonia measurement at ICU admission and those previously diagnosed with chronic liver disease or acute hepatitis were excluded. Patients were followed from ICU admission. For each patient, age, sex, length of ICU stay, length of hospital stay, and diagnosis that led to ICU admission were recorded. The following ICU admission data were recorded: SOFA score, including all necessary data for the six evaluated parameters: serum creatinine, diuresis, serum bilirubin, Glasgow coma scale (GCS), blood pressure, vasopressor and inotropic drug use, number of platelets, and respiratory/ventilatory parameters, as well as serum ammonia measurement, antibiotic therapy, need for hemodialysis, mechanical ventilation (MV), arterial lactate measurement, and Acute Physiology and Chronic Health Evaluation (APACHE) II.
Ammonia measurement
For ammonia measurement, blood samples were collected in a vial containing ethylenediaminetetraacetic acid as an anticoagulant, centrifuged to obtain plasma, and the device, Dimension® model RxL Max Siemens-Camberly, UK, was used with normal reference values of 11 to 35 µmol/L.
SOFAMONIA scores
The results of serum ammonia concentration found in our patients were divided into five strata (interquintile intervals). The serum ammonia concentration was scored from 0 to 4, and each score level was determined by the proportional distribution of the values found in the sample, divided into interquintile intervals.
The SOFAMONIA scores were defined as follows:
SOFAMONIA1: the GCS classification of the original SOFA score was replaced by serum ammonia;
SOFAMONIA2: the serum bilirubin concentrations from the original SOFA score were replaced by serum ammonia;
SOFAMONIA3: the renal system score (serum creatinine and/or diuresis) from the original SOFA score was replaced by serum ammonia;
SOFAMONIA4: in this fourth variation of the score, ammonia levels did not replace another SOFA variable but was added to them. The addition of 4 points to the original SOFA score for serum ammonia concentrations changed the maximum value to 28.
The other parameters of SOFA (cardiovascular, coagulation, and respiratory) were not replaced, as there is no basis in the pathophysiology of these organs in the participation of ammonia metabolism.
Ethical aspects
The study was approved by the local Research Ethics Committee (Approval Number: 1,000,088, date: 27/03/2015). Written informed consent was obtained from the patients for their anonymized information to be published in this article.
Variables and statistical analysis
The primary outcome variable was hospital mortality. Predictor variables were as follows: SOFA score and SOFAMONIA scores, in addition to age, sex, type of hospitalization, MV, acute renal failure (AKI), use of vasopressor, serum creatinine, total serum bilirubin, serum ammonia, and GCS. The data were related to the moment of admission to the ICU. Sample size was calculated to compare two means. To calculate sample size, we adopted an expect 2 points difference in mean SOFA between groups with a standard deviation (SD) of 50% the mean value in each group, with a 2:1 ratio between survivors and no survivors, 95% confidence interval (CI) and 80% power. Sample size was calculated as 168 participants (112 in survivors’ group and 56 in non-survivors’ group).
Continuous variables are presented as means and SDs or medians and interquartile intervals (ITQs), as dictated by the normality or non-normality of the data. The Student’s t-test was used for normal distribution variables and the nonparametric Mann–Whitney test for variables with non-normal distribution. The qualitative variables are presented as absolute and percentage frequencies and evaluated by the Chi-square test. Spearman’s coefficient (r) was used to evaluate the correlation between the original SOFA score and SOFAMONIA scores. The area under curve–receiver operating characteristic (AUC–ROC) was used to compare both scores in relation to hospital mortality, and sensitivity and specificity were calculated. The pairwise comparison of the AUC–ROC was performed using the DeLong method. Logistic regression analysis was performed to evaluate variables independently associated with the hospital outcome. Results were presented as unadjusted and adjusted odds ratios (ORs), with 95%CI. Variables that reached P < 0.05 were considered significant. The program used was MedCalc Statistical Software version 18.9 (MedCalc Software, Ostend, Belgium).
Results
In total, 655 patients were admitted to the ICU during the study period, of which 207 were not included, since they did not have an ammonia measurement at ICU admission, 182 for an ICU stay less than 48 hours, 11 because they were under 18, 47 because they did not sign the ICF, and 9 because they were diagnosed with chronic liver disease or acute hepatitis. Of the 199 patients included in the study, there were 26 losses due to incomplete data, resulting in 173 patients analyzed.
The median age was 59 years (ITQ 40.75–72.00), and there was a higher frequency of men (60.7%). Medical hospitalizations were more frequent (41%), followed by hospitalizations for emergency surgeries (28.9%), elective surgeries (15.6%), and major burns (13.9%). The median SOFA score was 8 (ITQ 5–12), the mean APACHE II score was 22.9 (SD 10.1), and the median scores of the SOFAMONIA 1 to 4 are shown in Table 1. At admission, 46.2% of the patients had a diagnosis of AKI, 19% were on hemodialysis, 52% were on vasopressors, and 75% were on MV. The median ICU stay was 10 days (ITQ 5–17), and the median length of hospital stay was 24 days (ITQ 13–36).
Clinical and outcome characteristics among survivors and non-survivors.
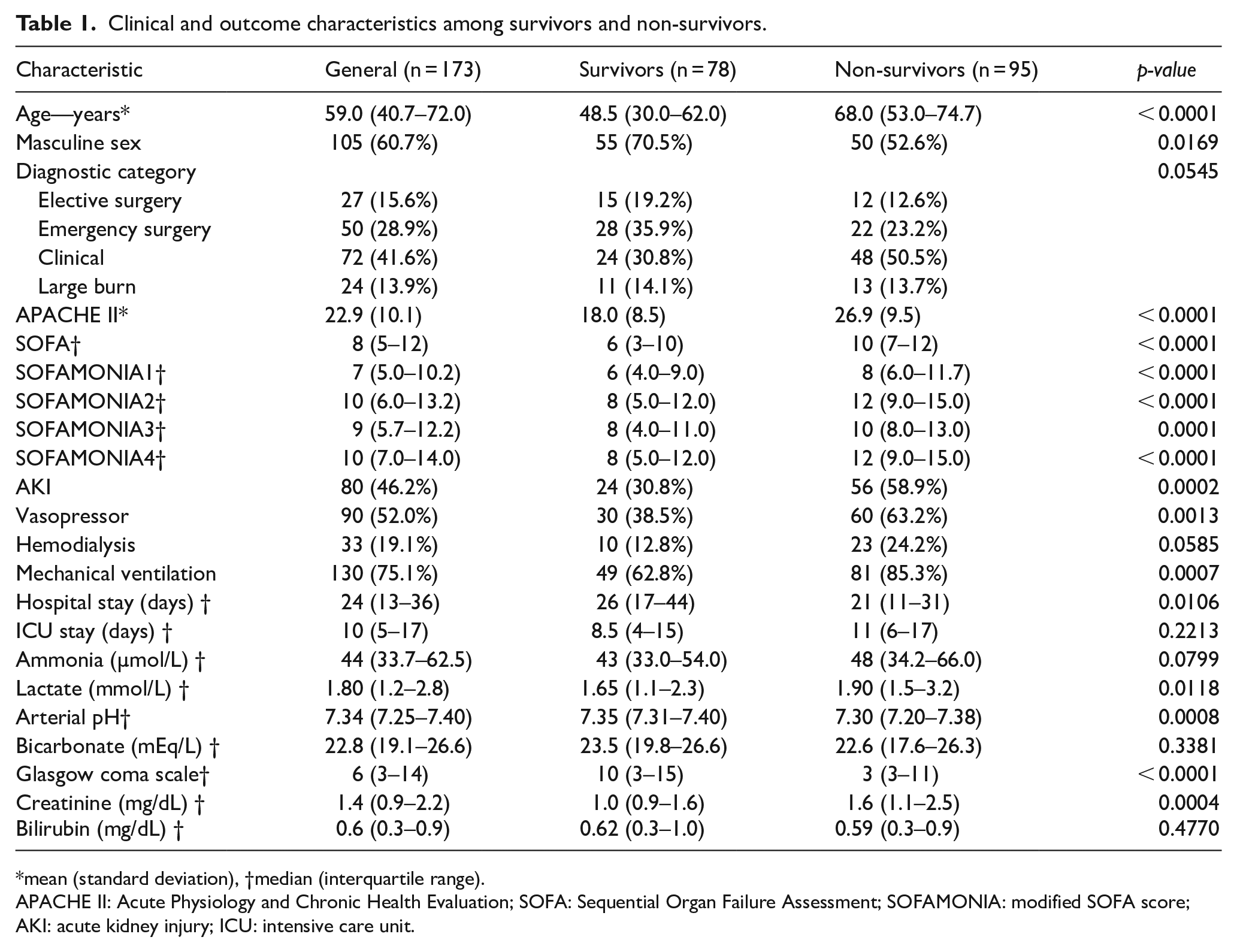
mean (standard deviation), †median (interquartile range).
APACHE II: Acute Physiology and Chronic Health Evaluation; SOFA: Sequential Organ Failure Assessment; SOFAMONIA: modified SOFA score; AKI: acute kidney injury; ICU: intensive care unit.
Hospital mortality was 54.9%. Patients who died were older, less frequently male (P = 0.0169), had a higher need for vasopressors and MV on admission, as well as a lower GCS and higher prevalence of a diagnosis of AKI. In addition, in this group, arterial lactate was higher and pH was lower. Among the group of survivors, hospital stay was longer (26 vs 21 days, P = 0.0106). The APACHE II and SOFA scores, as well as the SOFAMONIA 1, 2, 3, and 4 scores, were higher among patients who died.
Figure 1 shows the score of ammonia as a criterion in the SOFAMONIA score. In the sample, the patients with the lowest scores (0, 1, and 2) for ammonia in the SOFAMONIA score presented 49.0% in-hospital mortality, while the patients with the highest scores (3 and 4) presented 63.7% in-hospital mortality (P = 0.0573). Regarding the prediction of in-hospital mortality, the original SOFA presented an AUC–ROC of 0.697. There was a positive correlation between SOFA and SOFAMONIA1 (r = 0.862, P < 0.0001), SOFAMONIA2 (r = 0.944, P < 0.0001), SOFAMONIA3 (r = 0.905, P < 0.0001), and SOFAMONIA4 (r = 0.947, P < 0.0001). The discriminatory power analysis of SOFA and SOFAMONIA 1 to 4 was evaluated using AUC–ROCs, as shown in Figure 2. The SOFAMONIA1 presented a cut-off point of 5 for AUC 0.684 (0.610–0.753, 95%CI), with sensitivity and specificity in predicting hospital mortality of 80.0% and 48.72%, respectively. The SOFAMONIA2 presented a cut-off point of 9 for AUC 0.701 (0.626–0.768, 95%CI), with sensitivity and specificity in predicting hospital mortality of 69.47% and 61.54%, respectively. The SOFAMONIA3 presented a cut-off point of 8 for AUC 0.674 (0.598–0.743, 95%CI), with sensitivity and specificity in predicting hospital mortality of 70.53% and 62.82%, respectively. The SOFAMONIA4 presented a cut-off point of 8 for AUC 0.702 (0.628–0.769, 95%CI), with sensitivity and specificity in predicting hospital mortality of 70.53% and 61.54%, respectively.
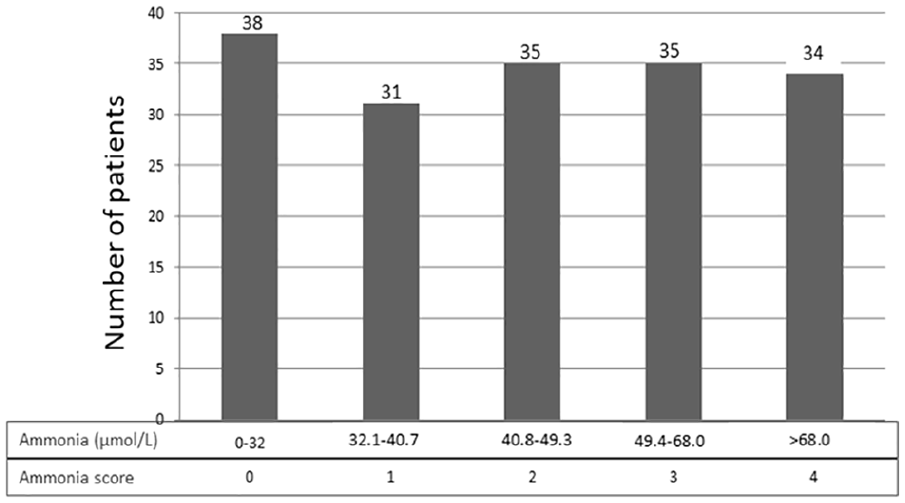
Ammonia criterion points in the SOFAMONIA score.
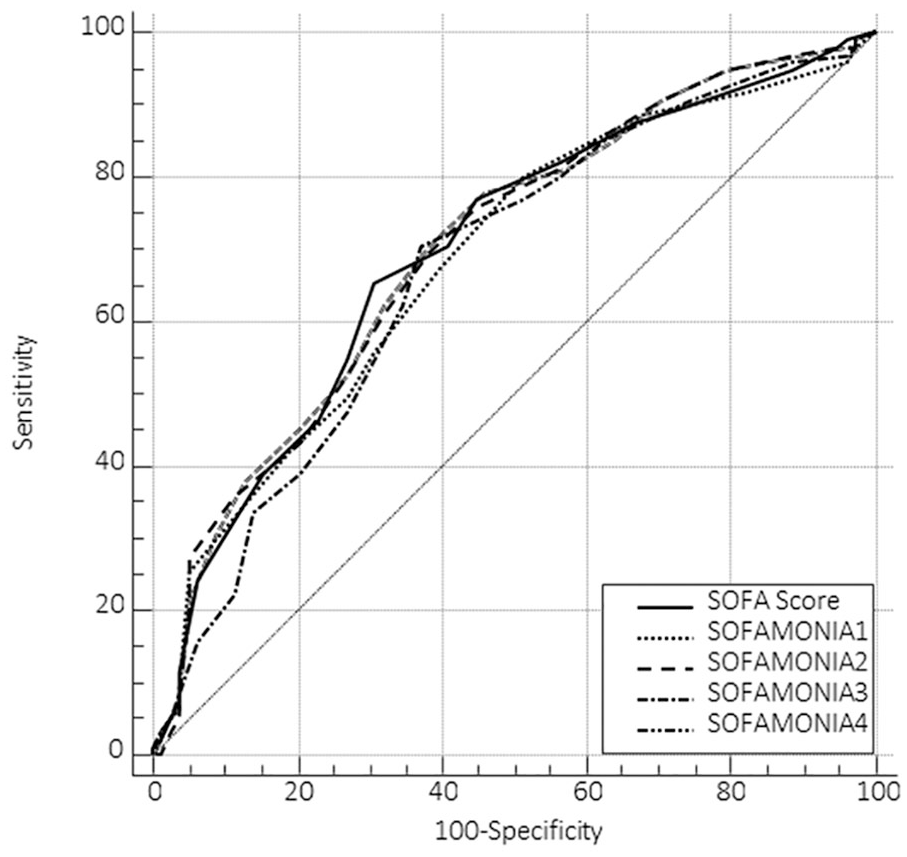
ROC curve for SOFA and SOFAMONIA.
In the first logistic regression analysis for variables independently associated with the hospital outcome, which excluded the SOFA, SOFAMONIA, and APACHE II scores due to the potential for collinearity, there was an association of hospital mortality with advanced age, female sex, burn diagnosis, presence of AKI on admission, need for a vasopressor on admission, and arterial pH. For each point increase in GCS, there was a 16% decrease in the chance of death (Table 2). Although the value of ammonia alone was not significantly associated, in additional logistic regression analyses, there was an increased risk of death with an increased APACHE II score (22% increase in risk of death for each one point elevation in the APACHE II), SOFA score (14% increase in risk of death for each one point elevation in the standard SOFA), and SOFAMONIA2 and SOFAMONIA4 scores (both with 13% increase in risk of death for each one point elevation) (Supplementary Tables 1, 2, and 3).
Logistic regression for variables independently associated with hospital death.
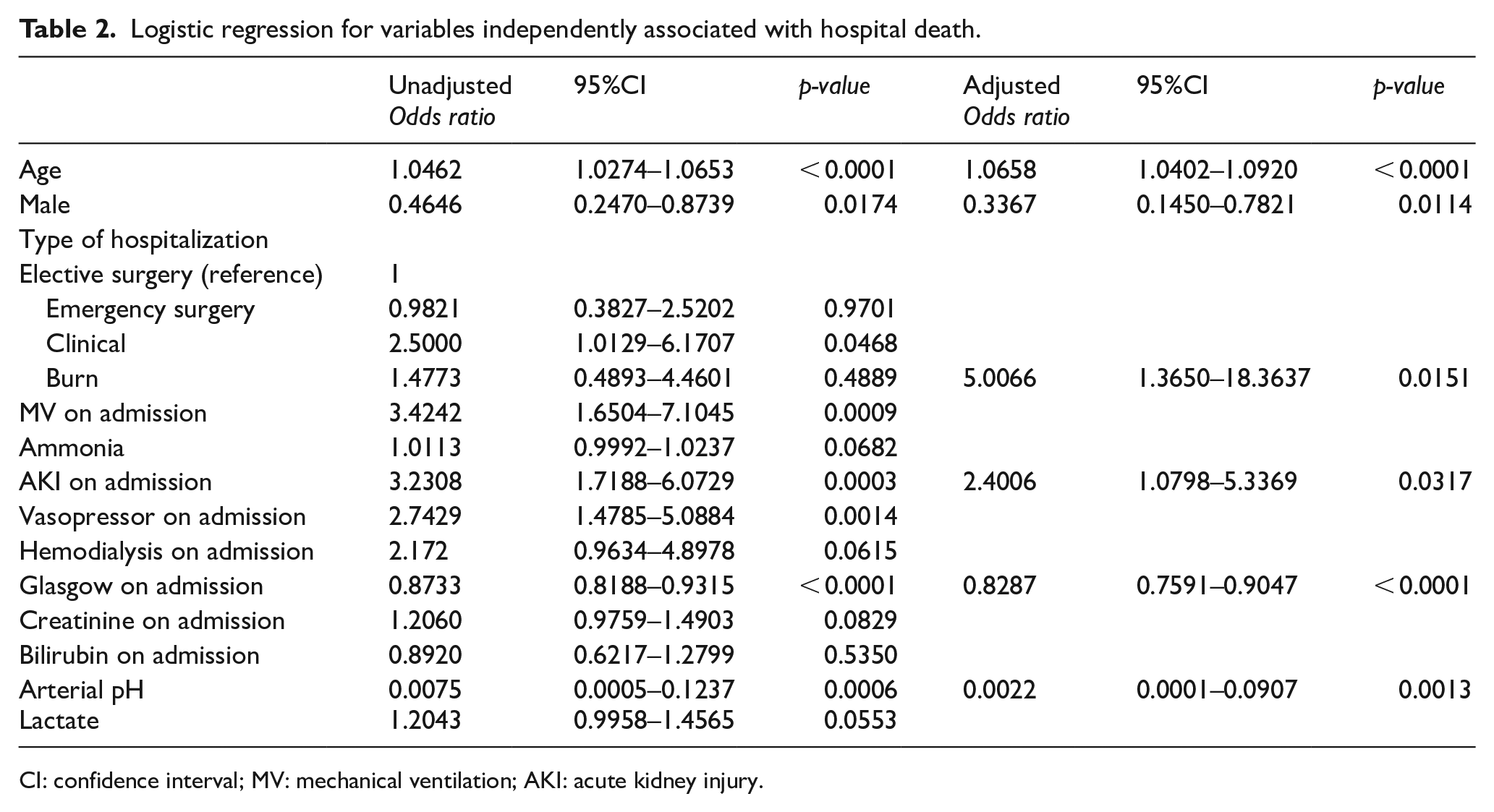
CI: confidence interval; MV: mechanical ventilation; AKI: acute kidney injury.
Discussion
The main purpose of the study was to evaluate the performance of ammonia as an additional or substitute variable for the hepatic, neurological, and renal systems in the original SOFA score in patients admitted to the ICU. The results showed that all the four ways to use ammonia in the evaluation of organ dysfunction by modifying the SOFA (SOFAMONIA 1 to 4) presented a positive correlation with the original score. We highlight the SOFAMONIA4, in which ammonia was added as a seventh parameter, as the best way to insert ammonia in this evaluation of organ dysfunction, since there was a better balance between sensitivity and specificity. In this case, this would be a seventh dysfunction, considered metabolic. Although this suggests that hyperammonemia may be a marker of organ dysfunction, it was not possible to prove the association of ammonia alone with mortality, as there was no difference between survivors and those who died. This differs from two other markers of organ dysfunction evaluated in the same sample (GCS and creatinine), as they were associated with mortality, even when analyzed in isolation.
These results for ammonia could be explained in two ways. First, ammonia was not included in the SOFAMONIA as a pre-established value, as there is no consensus on the values that determine severe hyperammonemia, but was classified according to the distribution in quintiles within the sample. The other variables were awarded points within the same score according to their reference values previously considered significant for the original SOFA score and not divided into quintiles within the sample. Another factor was the limited sample size, since the isolated value of ammonia has been associated with mortality in previous studies with more robust samples. Sakusik et al. 17 evaluated 3,908 patients with hyperammonemia, finding only 4.5% HNH cases. In this group, there was a difference between survivors and deaths even with relatively close ammonia values (74 vs 67 μg/dL, P = 0.05). Although in our sample serum ammonia concentration alone tended to be associated with increased hospital mortality, this difference probably did not reach statistical difference because of the small number of observations. This result is close to that reported by Prado et al. 16 in their study, when evaluating HNH in critically ill patients, showing higher mortality (P < 0.01 for comparison between groups) among patients with serum ammonia above 100 μmol/L.
In the present study, the original SOFA score demonstrated a moderate ability to predict hospital mortality, with an AUC of 0.697. This differs from studies with more robust samples (n = 1,034), in which the AUC–ROC was higher, but still did not reach a value of 0.80, which is considered an appropriate AUC–ROC for this type of evaluation. 21 Based on studies that showed elevated ammonia as neurotoxic,15,18,20,22–25 it was expected that replacing the SOFA neurological parameter score with ammonia would result in a better correlation and perhaps a better prediction of mortality. This was considered because many patients with a high number of points in the standard SOFA neurological parameter are under a sedative effect and, even if a daily ICU awakening is performed, the GCS might not adequately reflect the severity of neurological dysfunction. However, the AUC–ROC of the SOFAMONIA1 demonstrated a lower correlation with the original score, not confirming the hypothesis. This could be explained by the fact that the GCS better reflects neurological impairment not only of metabolic origin, but also structural. The latter is probably not well represented by a laboratory alteration such as hyperammonemia.
Even though the admission criteria excluded patients with previously known liver disease, replacement of total bilirubin with ammonia in the SOFAMONIA2 score was performed to identify whether possible liver dysfunction could be detected by hyperammonemia. As expected, this substitution demonstrated the second-best correlation compared to the original score, since elevated ammonia is a well-defined marker of liver dysfunction. 26 We consider that this could be explained by new liver dysfunctions or previous undiagnosed dysfunctions on ICU admission. The SOFAMONIA3 score, in which the renal parameter (creatinine/diuresis) was replaced by ammonia, presented the worst performance compared to the other variations. This result corroborates the concept that the isolated measurement of a nitrogen load, whether creatinine or ammonia, does not replace the association of this marker with the urinary output as a parameter to evaluate renal dysfunction. 27
Despite the positive correlation between the original score and modified scores, the data indicate that the parameters are not considered interchangeable. On the other hand, replacing traditional parameters with ammonia did not change the ability of the SOFA to evaluate organ dysfunction in critically ill patients. The addition of ammonia to the SOFAMONIA4 organ dysfunction evaluation score provides an indication that adding this parameter could be a way to improve evaluation of this dysfunction. The fact that lactate was associated with mortality indicates that this marker stands out as a parameter of metabolic dysfunction and corroborates its association with hospital mortality, especially due to sepsis, already described in the medical literature.28,29 It is worth noting that organ dysfunction in critically ill patients is not static and, as shown by Vincent et al., 4 scores for this type of assessment should be developed to allow a dynamic perception of these alterations.
The main limitation of the study is the fact that it is taken from a single center study with reduced sampling, making a multicenter study with more robust sampling necessary for external validation of the results. Another significant limitation is the non-inclusion of approximately 30% of admitted patients due to lack of measurement of the ammonia level. This event happened because the ammonia level is not a routine measurement in the study center, and it requires a specific technique of maintaining the samples refrigerated immediately after the blood sample has been drawn. Therefore, all inadequate samples were excluded from analysis.
Conclusion
There was a positive correlation between the four ways to use ammonia in the evaluation of organ dysfunction by modifying the original SOFA. Adding ammonia as the seventh parameter representing metabolic dysfunction stood out as the best correlation. The increase in ammonia appears to be multifactorial and cannot be explained by an organ dysfunction alone. Thus, replacing the parameters already classically used in the original SOFA score does not seem to be justified; however, the addition of ammonia as a representative of metabolic dysfunction may be useful in monitoring critically ill patients.
Supplemental Material
Supplement_material – Supplemental material for SOFAMONIA: Comparison of the original SOFA score with the proposed new score including serum ammonia
Supplemental material, Supplement_material for SOFAMONIA: Comparison of the original SOFA score with the proposed new score including serum ammonia by Amanda Carolina Damasceno Zanuto, Alexandre Sanches Larangeira, Marcos Toshiyuki Tanita, Hugo Kenzo Ishioka, Cintia Magalhães Carvalho Grion and Vinicius Daher Alvares Delfino in Hong Kong Journal of Emergency Medicine
Footnotes
Author contributions
All authors listed meet the criteria for authorship and have approved the final version of the article. All those entitled to authorship are listed as authors. Authors’ contributions are as follows: ACDZ and VDAD researched literature and conceived the study. ACDZ and HKI was involved in protocol development, gaining ethical approval, patient recruitment, and data acquisition. ASL and MTT was involved in data analysis. ACDZ and CMCG wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Human rights
The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a priori approval by the institution’s human research committee.
Availability of data and materials
The datasets generated and/or analyzed during the current study are available from the corresponding author by reasonable request.
Informed consent
Written informed consent was obtained from the patients or legal representative for their anonymized information to be published in this article.
Ethical approval
The study was approved by the local Research Ethics Committee (Comitê de ética em pesquisa envolvendo seres humanos Universidade Estadual de Londrina. Approval Number: 1,000,088, date: 27/03/2015).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
