Abstract
The density of a hematoma on a non-contrast computed tomography of the brain is determined by the degree of clot retraction, hematocrit and globin content. Classically, the appearance of an intracranial haemorrhage on a computed tomography correlates with the temporal evolution of hematoma. However, this case illustrates there are circumstances that this classical temporal appearance may not hold true.
A man in his 80s, who walked unaided and enjoyed independence in his activities of daily living, was bumped by a pedestrian from behind and suffered a fall the day before. He was on apixaban for his atrial fibrillation. He complained of increasing occipital swelling and pain. There was no loss of consciousness, vomiting, dizziness or any other injury. He was also certain there was no minor head injury in the past few months. On physical examination, he was alert and his left occiput was tender and swollen. Otherwise, his Glasgow Coma Scale was 15, blood pressure was 142/59 mm Hg and pulse was 65 per minute. Neurological examination was unremarkable. His haemoglobin, platelet and international normalized ratio (INR) values were 12.1 g/dL, 190 × 109/L and 1.1, respectively. A computed tomography (CT) of the brain was arranged (Figure 1).
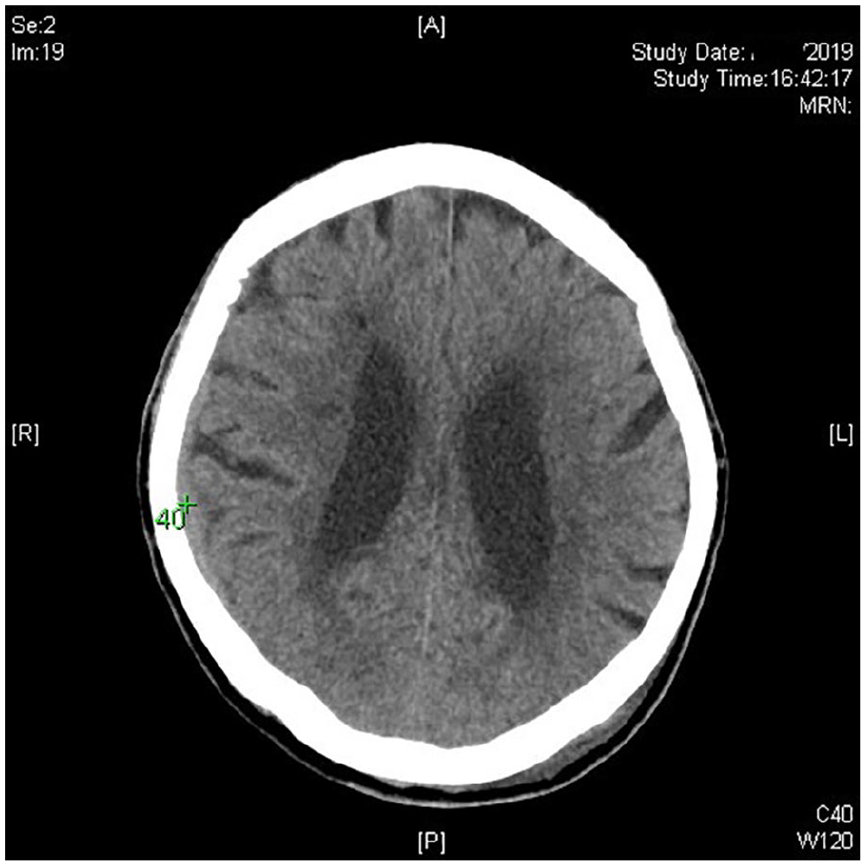
Non-contrast computed tomography of the brain. The Hounsfield unit value was 40 over the marked collection.
Questions
What is the diagnosis?
Name two conditions where such phenomenon can exist.
What specific antidote can be prescribed if the patient develops life-threatening rise in intracranial pressure?
Answers
There is a 4-mm isodense apixaban-associated acute subdural haemorrhage over the right frontoparietal lobe. Isodense acute subdural haemorrhage is exceedingly uncommon, which only accounts for about 1% of the acute subdural haemorrhage. 1
In coagulopathies and severe anaemia, acute haemorrhage can appear isodense with the adjacent cerebral cortex.
Andexanet alfa is the antidote for direct factor Xa inhibitors such as apixaban and rivaroxaban.
Discussion
In classical medical school teaching, the appearance of subdural haemorrhage on CT can simply be correlated with its age. Hyperacute collection (<12 h) can appear isodense as the adjacent parenchyma is equally dense with the matrix mass of white/red blood cells, platelets and serum. In the acute phase (12 h to 2 days), the density of the hematoma increases due to progressive blood clot retraction and serum extrusion. Specifically, the hyperdensity on CT largely results from the increasing concentration of the protein component of haemoglobin, not from the iron. 2 During the subacute phase (2 days to 1 month), the hematoma becomes isodense with progressive red blood cell lysis and globin proteolysis. After about a month, it becomes hypodense as the chronic hematoma is progressively phagocytized. 3
However, there are caveats to this classical hyperdense appearance of acute subdural haemorrhage on CT. In those with coagulopathies or on anticoagulants, the rapid clot dissolution or inhibition of clot formation can lead to the appearance of isodense hematoma despite the acuity. 4 Furthermore, in severe anaemia with haemoglobin concentrations of 8–10 g/dL, an isodense collection can also appear in the acute setting because of the reduced globin concentration. 5
The CT image in this case illustrates that radiodensity can be misleading in the estimation of the age of the haemorrhage especially in those with coagulopathies/on anticoagulants or severe anaemia. Therefore, disposal plan in the emergency department should not be based solely on the classical temporal evolution of hematoma on CT imaging.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of data and materials
Not applicable as no collection is involved.
Informed consent
Written informed consent was not necessary because no patient data have been included in the manuscript.
