Abstract
Introduction:
Severe hypophosphataemia may occur in long distance runners presenting to Accident & Emergency (A&E) department with exertional heat illness.
Case presentation:
A 46-year-old man who collapsed in half marathon race was found to have raised body temperature (38.8°C) and confused with memory loss in the Accident & Emergency department. His amnesia was persistent even after his body temperature was normalized. He was found to have severe hypophosphataemia (0.21 mmol/L; reference range: 0.74 - 1.4 mmol/L). He had a neurological recovery after phosphate replacement. Another 45-year-old female half marathon runner was found to have moderate hypophosphataemia (0.5 mmol/L) co-presenting with exertional heat illness.
Discussion:
Moderate to severe hypophosphataemia complicated the clinical picture of the two cases presented with exertional heat illness. Causes of hypophosphataemia in runners are multifactorial.
Conclusion:
Emergency physician should be alert of profound hypophosphataemia as a complication in heat exertional illness.
Emergency physicians rarely encounter hypophosphataemia. Even among all hospitalized patient its incidence rate is only 1%–5%. 1 It is even less common in general populations without predisposing chronic conditions such as chronic kidney injury, alcoholism, chronic obstructive pulmonary disease and malignancy.
Still, hypophosphataemia can happen in healthy individuals. Exertional heat illness is among the leading causes of death in young athletes, 2 and it is actually one of the less recognized conditions associated with hypophosphataemia.3,4
Here, we described two cases of runners who joined half marathon event of Hong Kong Marathon 2019, presented with exertional heat illness complicated with hypophosphataemia. We will discuss the pathophysiology and mechanism of hypophosphataemia in exertional heat illness. Recommendations of phosphate correction will be given to emergency physicians who may encounter patients with similar presentation in their daily practices.
Case 1
In January 2019, 74,000 runners joined the Hong Kong Marathon. The measured outdoor air temperature was 18°C and relative humidity was 90% according to records from Hong Kong Observatory on the day of Hong Kong Marathon 2019.
A previously healthy 46-year-old Chinese man from Nanjing, China, who joined the half marathon collapsed and vomited after running 16 km. He was found to be unconscious by paramedics. His initial body temperature was 39°C. After cold pads and water spray were given, his conscious level improved gradually, but he remained confused and struggled on the way of transfer to hospital.
He was triaged as category 2 case in the Accident & Emergency (A&E) department. His tympanic temperature was 38.8°C. His initial blood pressure was 102/58 mmHg and his pulse was 125 per minute. He was breathing at a rate of 22 per minute. He could not recall that he had collapsed in the race, but he could tell that he only drank sips of water after the race had started. He complained of dizziness and nausea. He had no chest pain all along.
On physical examination, he was fully conscious and oriented to time, place and person. However, on further check, he failed to recall his wife’s name. He could not unlock his mobile phone as he forgot the password. Clinically he was moderately dehydrated. His chest, cardiovascular and abdominal examinations were normal. Neurological examination revealed no focal signs and limb powers were full.
Rehydration by a bolus of 1500 mL Normal Saline was given in A&E department. Intravenous metoclopramide 10 mg was given for nausea. External cooling was continued with ice pack and upright fan. His body temperature was further lowered down to 37.1°C half an hour after initial assessment, but he still remained amnestic, confused and agitated. He forgot all conversations with doctors and nurses during his brief stay in A&E department.
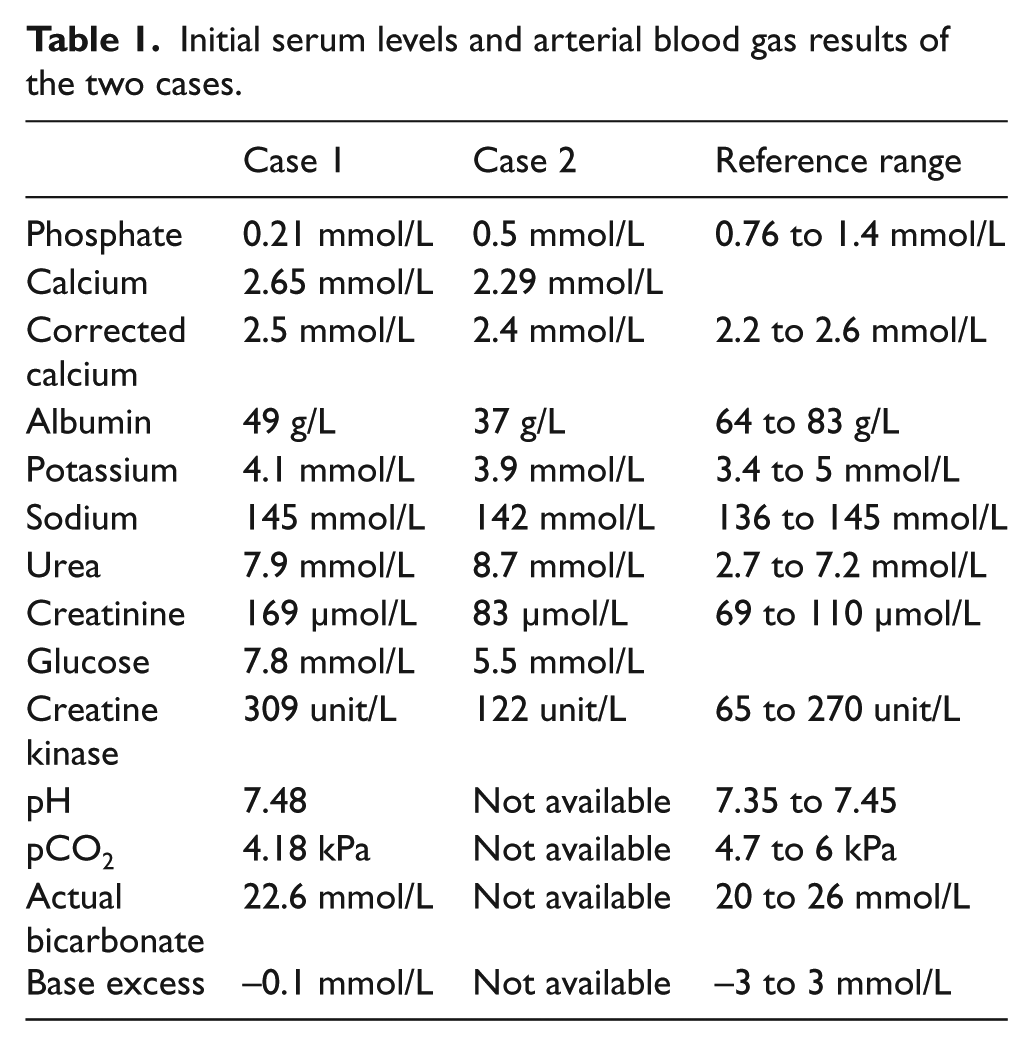
He was found to have severe hypophosphataemia with serum phosphate level at 0.21 mmol/L (reference range: 0.76–1.4 mmol/L). No other electrolyte disturbances was found. Blood gas showed respiratory alkalosis (see Table 1).
Initial serum levels and arterial blood gas results of the two cases.
Computed tomography (CT) of his brain was normal. Electrocardiogram (ECG) showed sinus rhythm without acute ischaemic change. Chest X-ray (CXR) was clear.
He was diagnosed to have heat stroke in view of his persistent confused state. He was admitted to medical ward. Intravenous potassium dihydrogen phosphate and dipotassium hydrogen phosphate (KH2PO4/K2HPO4) 10 mL infusion, containing 14.5 mmol phosphate ion, was given over 4 h. Phosphate level was raised to 0.76 mmol/L on recheck 6 h later. He was noted to be fully alert without amnesia afterwards.
However, he left hospital with his family against medical advice on the same day of admission.
Case 2
A 45-year-old female half marathon runner from Hong Kong just collapsed 100 m from the finishing line. She was noted to be confused on approach by the paramedics. She had no chest pain, vomiting, seizure or head injury. Her only past medical history was laparoscopic resection of ovarian cyst 2 years ago. Her initial body temperature was 39°C. Intravenous fluid replacement by normal saline was started by paramedics.
On arrival at A&E, she became fully conscious with body temperature of 38.3°C. Her blood pressure was 108/51 mmHg, pulse was 103 per minute. She was pale looking and dehydrated. She had no focal neurological deficit. Physical examinations of her cardiovascular and respiratory systems were normal.
Her first plasma phosphate level was 0.5 mmol/L. There was no other electrolyte abnormality (see Table 1).
ECG showed sinus rhythm with no ischaemic change. CXR was normal.
She was admitted to our emergency medicine ward with the diagnosis of heat exhaustion. Intravenous rehydration was continued. Her plasma phosphate level 9 h later was 0.75 mmol/L. She had an uneventful recovery.
Discussion
Phosphate is involved in many critical human physiological pathways. By phosphorylation of protein, phosphate plays key roles in homeostatic regulation, enzyme activation and cell-signalling cascade. 5 Adenosine triphosphate is an important component in cellular energy storage and metabolism. 6 2,3-Diphosphoglycerate is a well-known regulator of oxygen release by haemoglobin. 7
Hypophosphataemia can arbitrarily be graded into moderate hypophosphataemia (serum phosphate 0.32–0.65 mmol/L) and severe hypophosphataemia (serum phosphate <0.32 mmol/L). 8 Mechanisms of hypophosphataemia include inadequate dietary intake, decreased intestinal absorption, excessive urinary excretion, removal by renal replacement therapy or a shift of phosphate from the extracellular compartment into cells. Among critically ill patients, typical causes of hypophosphataemia include sepsis, trauma, fluid therapy, refeeding, acid-base disorder, medication (e.g. diuretics, glucose/insulin) and renal replacement therapy. 5
Our cases illustrated moderate to severe hypophosphataemia in patients presenting with exertional heat illness. Heat stroke, the severe form of heat illness, is well known to emergency physicians for the severe neurological dysfunction and multi-organ damage, which if cooling is delayed, may lead to fatal outcome. However, in our two cases, in contrast to the classical presentation of exertional heat stroke, the confusion and amnesia could not be explained by the hyperthermia alone. The initial body temperature at scene of our patients was not very high, just 39°C, and the fact that the persistent confusion and memory loss even after normalization of body temperature in case 1 suggested that the other factor was actually causing the neurological symptoms and complicating the clinical picture. In addition, when comparing the two cases, it seems that the level of phosphate may correlate with the severity of symptoms, at least the confusion and memory loss. This may give insight to the clinical management of patients with exertional heat illness who had persistent confusion or memory loss even after cooling. To our knowledge, there was only one case report of patient with similar clinical presentation associated with hypophosphataemia in the literature. 9
The cause of acute hypophosphataemia in runners is probably multifactorial. Although prolonged exercise itself stimulates muscle glycogenolysis, increases cellular uptake of phosphate and theoretically lower serum phosphate level, severe hypophosphataemia in prolonged exercise is rarely mentioned in literature.
Hyperventilation and consequent primary respiratory alkalosis were often cited cause of hypophosphataemia in exertional heat illness.10,11 Respiratory alkalosis increases intracellular pH and activity of intracellular phosphofructokinase for glycolytic pathway. Phosphate shifts from extracellular space into cells. 12 Experiment with healthy subjects showed that extreme hyperventilation can lower phosphate level to a mean of 0.29 mmol/L with a sharp reduction of phosphate excretion in urine. 13
In an observational study of heat stroke, respiratory alkalosis was present in only about half of all patients. 14 Case series of heat stroke related hypophosphataemia found that most patients did not have respiratory alkalosis. 15 Instead of an absent of phosphate excretion, hyperphosphaturia is observed in most cases. 16 Therefore, respiratory acidosis itself cannot fully explain why people in heat stroke have hypophosphataemia. In case 1, the severe hypophosphataemia (0.21 mmol) and relatively mild respiratory alkalosis (pH 7.48, pCO2 4.18 kPa) may suggest other factors in addition to hyperventilation are contributing to the low phosphate level.
On the contrary, metabolic acidosis is the commonest acid–base change in heat stroke. 14 It is secondary to increased lactate content and was significantly associated with degree of hyperthermia. 15 Metabolic acidosis causes hypophosphataemia through increased urinary phosphate excretion. 5 Phosphaturia and increased fractional excretion of phosphate (24%–57%; normal 5%–20%) were observed in severe cases of heat stroke–related hypophosphataemia. 16 However, in patients with exertional heat stroke, with a reduced amount of urine output, it is unclear that such degree of change in fractional excretion of phosphorus can account for the magnitude and speed of changes in serum phosphate levels.
Fever and hyperthermia are shown to correlate inversely with hypophosphataemia in observation studies of therapeutic hyperthermia, malaria and bacterial pneumonia without respiratory alkalosis.17–19 It was postulated that similar to rise in pH, elevation of body temperature itself increases utilization of phosphate in glycolysis and increases cellular uptake of phosphorus. Induced hyperthermia increases oxygen consumption by 19%–26% per 2°C rise in body temperature.20,21 An increase in cellular metabolism increases transcellular shift of phosphorus into cells. Again, the degree of hyperthermia cannot fully explain the relatively low level of phosphate in our case.
Recognition of severe hypophosphataemia is important as the condition itself can be life threatening. 22 Severe hypophosphataemia complicates the clinical picture of heat stroke by causing seizure, confusion, rhabdomyolysis, arrhythmia and myocardial depression. 8 Severe hypophosphataemia is a known marker of poor prognosis among patients with sepsis in intensive care unit. 23 It has been shown that among patients with heat stroke, there is a strong correlation between Glasgow coma scale and serum phosphate level. 16 For a critically ill and deteriorating patient, rapid correction of hypophosphataemia is warranted as in our case 1.
Emergency physician should treat severe hypophosphataemia in heat stroke with intravenous phosphate therapy with a goal of normalizing phosphate level. A commonly accepted formulation is 4.5 mmol/h for 3 h (13.5 mmol in total), up to 90 mmol in 24 h. Potassium-based formulation, such as KH2PO4/K2HPO4 as in case 1, is often used because hypokalaemia is more frequently associated with hypophosphataemia. Sodium-based formulation is an available option if patient has hyperkalaemia. Adverse effects associated with intravenous phosphate therapy include overshoot hyperphosphataemia, hypocalcaemia, hypernatraemia, dehydration from osmotic diuresis and metastatic calcification.24,25
Concurrent electrolyte levels, renal function test, arterial blood gas and lactate level, and clinical parameters would help to guide further management. Rapid cooling to a target of 38°C is important not only for heat stroke management but also for hyperthermia-induced hypophosphataemia as discussed above. 26 Fluid rehydration should be offered according to patient’s deficit, maintenance and ongoing loss, further guided by electrolyte levels and renal function. These patients should be managed and monitored in intensive care unit if there is no rapid recovery.
Conclusion
In conclusion, we presented two cases who presented with exertional heat illness and hypophosphataemia to our A&E. We found that the phosphate levels correlate with the neurological dysfunction, in particular, confusion and memory loss in our cases. Those neurological symptoms could not be fully explained by the degree of hyperthermia and hyperventilation. Emergency physician should be alert of profound hypophosphataemia as a complication in heat exertional illness. Therefore, we suggest inclusion of serum phosphate level as part of initial assessment of patients with exertional heat illness, especially for those who have persistent neurological dysfunction after normalization of body temperature. Correction of phosphate level by means of intravenous phosphate infusion is clinically indicated in this group of patient.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This case report does not require ethical approval as it is not a human or animal research.
Human rights
This case report does not require human right permissions
Informed consent
Patient 1 did not leave any phone or address during his stay in our department or admission and we were unable to contact him. Informed consent signed by patient 2 is obtained. The article is anonymized.
