Abstract
Background:
Gelsemium elegans is an extremely toxic plant, but gelsemium poisoning is seldom reported in the English literature.
Objectives:
To evaluate the clinical manifestations and causes of gelsemium poisoning in Hong Kong.
Methods:
A retrospective review of gelsemium poisoning recorded by the Hong Kong Poison Information Centre from 2005 to 2017.
Results:
In total, 33 cases (55% female, median age 44 (interquartile range: 30–56)) were identified in 14 incidences. Consumption of contaminated Ficus hirta (五指毛桃) soup is the commonest cause (52%). Other causes include misidentification of herbs (12%), consumption of parasitic plant Cassytha filiformis (無根藤) (15%) and suicidal ingestion of Gelsemium elegans (斷腸草) (3%). Most patients (94%) had mild to moderate toxicity, with one fatal case and one severe case presented with coma and respiratory depression. All patients complained of dizziness (100%), followed by visual blurring (34%) and nausea (28%). More than half (53%) had ocular manifestations (e.g. visual blurring, ptosis, nystagmus, diplopia) which are not commonly reported in other herbal poisoning. The time of symptom onset was early (median: 50 min (interquartile range: 30–60)) and all occurred within 2 h after oral intake. Most patients (94%) recovered uneventfully with conservative treatment.
Conclusion:
Most gelsemium poisoning in Hong Kong was due to contamination or misidentification. Early-onset dizziness (<2 h) with ocular symptoms after herbs consumption highly suggests Gelsemium poisoning. Good supportive care, particularly respiratory support, is the mainstay of management. Early recognition and the corresponding preventive measures would be useful.
Introduction
Gelsemium, a small genus of flowering plants, which belongs to the Gelsemiaceae family, is extremely poisonous. It consists of three species only, in which Gelsemium elegans Benth. is indigenous to Southeast Asia, while Gelsemium sempervirens Ait. and Gelsemium rankinii Small. are native to North America. G. elegans is an evergreen shrub with long stems up to 12 m, grows as a twining vine and may interweave with surrounding edible plants. It may also be mistaken for various lookalike benign herbs, leading to inadvertent consumption and poisoning. It is highly poisonous, which is mainly attributed to its strong respiratory and neurological inhibitory effects.1,2 Among more than 100 alkaloids isolated from the plant, koumine (LD50 ~ 100 mg/kg mice intraperitoneally) is the most abundant but a less toxic alkaloid in G. elegans than gelsemine (LD50 ~ 56 mg/kg mice intraperitoneally), which is the principal alkaloid in G. semperviren 3 and the only alkaloid common to both G. elegans and G. semperviren, 4 while gelsenicine (humantenmine) is the most toxic alkaloid (LD50 ~ 0.2 mg/kg mice, intraperitoneally).5–7 Due to its potent toxicity, the use of G. elegans as traditional medicine, also known as 鈎吻 (‘Gou-wen’, meaning lethal kiss) in China to treat pain, spasticity and skin ulcer, is limited to topical application only. 8
Although G. elegans poisoning is quite commonly found in the Chinese literature, large case series are seldom reported in English journals. 9 The objective of this study is to present the local situation of gelsemium poisoning in Hong Kong over the past decades since the establishment of Hong Kong Poison Information Centre (HKPIC) and to evaluate the clinical manifestation and causes.
Materials and methods
HKPIC is the only regional poison control centre in Hong Kong. It currently provides 24-h toxicology consultation service to all health professionals in the region. It also maintains records of poisoning cases consultation and reporting by 43 local public hospitals. A retrospective chart review of the HKPIC database was performed from July 2005 to December 2017. All laboratory-confirmed clusters and poisoning cases due to Gelsemium plant in Hong Kong were included, and patients treated in overseas centres were excluded. Laboratory-confirmed gelsemium poisoning is defined as either gelsemium alkaloid being detected in the food sample consumed by patient, or gelsemium alkaloid detected in patient’s biological sample (e.g. urine). In case of clustering of patients, urine sample should be positive for gelsemium alkaloid in at least one patient in the cluster involved together with compatible clinical symptoms in other patients in order to be considered as laboratory-confirmed cluster of cases.
The data extracted include patient’s demographic information (age and gender), the time of ingestion, symptom onset and presentation time, the culprit involved in poisoning and the source of culprit, symptoms and signs developed, investigations results, treatment given, time of discharge and outcome. All data were abstracted using a standardized spreadsheet. Data abstraction was done by the principal investigator, and the second investigator was responsible for checking the accuracy of the data abstracted. Apart from the HKPIC database, patient records within the electronic patient record (ePR) were also reviewed and relevant information was abstracted.
Continuous data were presented as medians with interquartile ranges (IQRs), while categorical data were presented as percentage frequencies. Information used in this study was considered as part of the usual clinical data obtained by HKPIC and was passed to the researchers in a fully anonymized format.
Results
In the study period of 12 and a half years, there were 14 incidences and a total of 33 cases of laboratory-confirmed gelsemium poisoning identified with no case excluded, averaging around one incidence per year. Of them, 15 (45%) were male and 18 (55%) were female, with age ranging from 11 to 71 years (median age: 44, IQR: 30–56; Table 1).
Summary of all 14 incidences of Gelsemium elegans poisoning cases in Hong Kong.
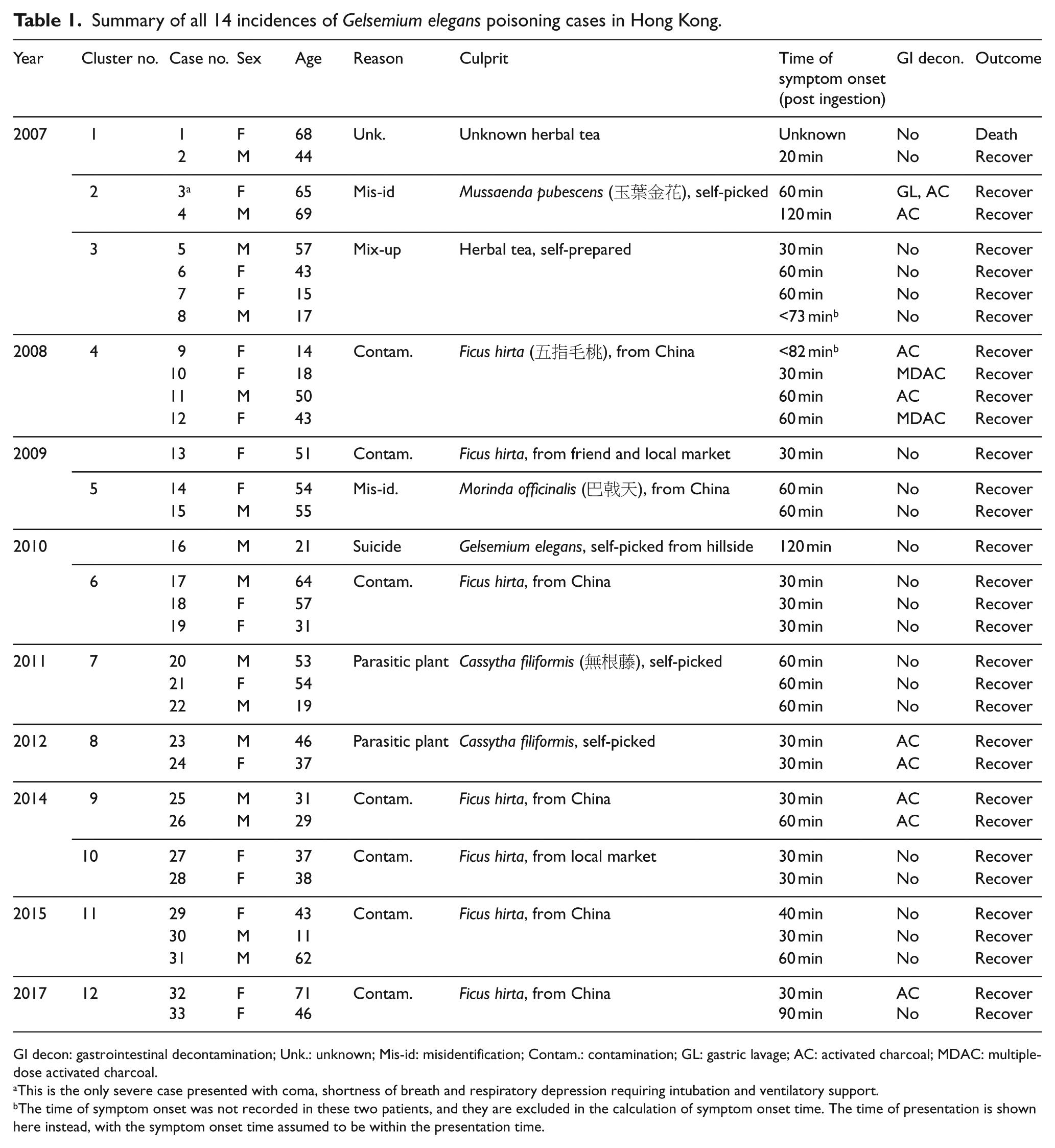
GI decon: gastrointestinal decontamination; Unk.: unknown; Mis-id: misidentification; Contam.: contamination; GL: gastric lavage; AC: activated charcoal; MDAC: multiple-dose activated charcoal.
This is the only severe case presented with coma, shortness of breath and respiratory depression requiring intubation and ventilatory support.
The time of symptom onset was not recorded in these two patients, and they are excluded in the calculation of symptom onset time. The time of presentation is shown here instead, with the symptom onset time assumed to be within the presentation time.
Among the 33 identified cases, 2 incidences involved a single patient. The remaining 31 patients were involved in 12 clusters. A total of 10 incidences involving 25 patients (76%) were poisoned due to the mixing-up of another benign herb by contamination or misidentification. The commonest benign herbs involved (Table 1) were Ficus hirta Vahl. (五指毛桃) from seven incidences (68%), followed by Folium Mussaenda pubescentis (玉葉金花) in a cluster of two patients (8%) and Morindae officinalis Radix (巴戟天) in another cluster of two patients (8%). The involved herbs cannot be identified in a cluster of four patients (16%) who suffered from gelsemium poisoning after intake of an herbal tea. Five other patients in two clusters consumed a parasitic plant Cassytha filiformis Linn (無根藤). Another cluster involved one fatal case who was found collapsed at home and a moderately symptomatic case, both of whom took an herbal tea. In the fatal case, gelsemine was detected in the urine, blood and the broth remnant. The remaining patient intentionally consumed self-picked G. elegans Benth. (斷腸草) for suicidal attempt (Table 1).
In our case series, majority of patients (94%) suffered from only mild to moderate gelsemium toxicity, with one (3%) death and one (3%) severe case with coma and respiratory depression requiring mechanical ventilation and intensive care unit (ICU) support for 2 days before discharging to general ward. The clinical presentations are summarized in Table 2. Since the death case was found collapsed at home by relative with unknown initial presentation, it is not included in the following analysis.
Clinical features of 33 cases a of gelsemium poisoning.
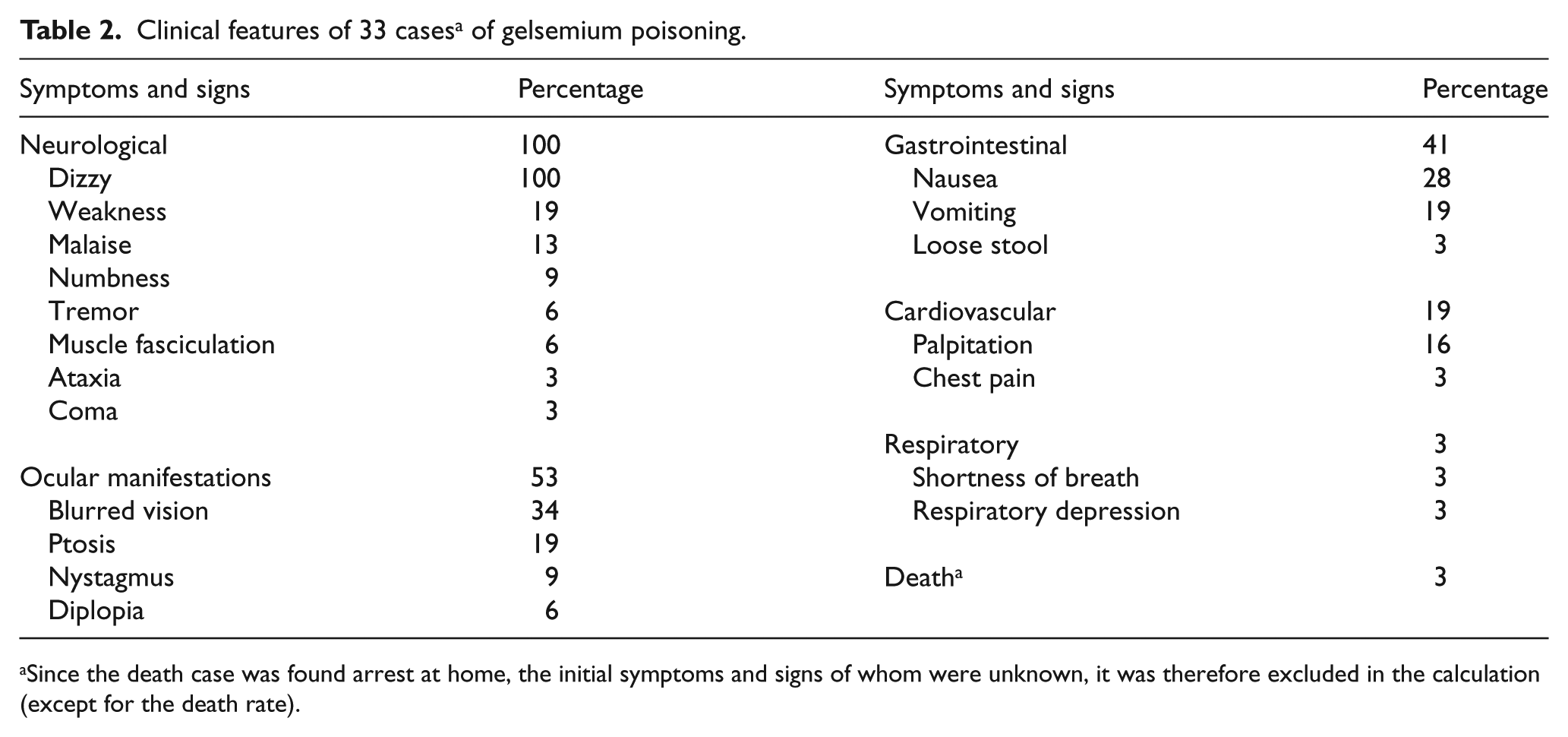
Since the death case was found arrest at home, the initial symptoms and signs of whom were unknown, it was therefore excluded in the calculation (except for the death rate).
The most commonly encountered symptom was dizziness, which was reported in all patients (100%), followed by visual blurring (34%), nausea (28%), vomiting (19%), weakness (19%) and ptosis (19%). Interestingly, about half of the patients (53%) complained of one or more ocular symptoms which were not commonly reported in other herbs or plants poisoning. The ocular signs and symptoms include visual blurring, ptosis, nystagmus and diplopia (Table 2). Onset of symptoms was early (median time: 50 min after oral intake (IQR: 30–60)). The earliest time of symptom onset reported was 20 min post ingestion of suspected culprit and all patients complained of at least one symptom within 2 h, with most cases (90%; 27/30) having had their symptom onset within 1 h and 93% (28/30) within 1.5 h. Note that the time of onset of symptoms in two patients (cases 8 and 9) was not recorded who were excluded in the calculation of symptom onset time. However, they all presented within 1.5 h. Therefore, we can still conclude that all gelsemium poisoning patients had symptoms occurring within 2 h (Table 1).
Treatment of gelsemium poisoning is mainly supportive. In total, 34% of patients were given gastrointestinal decontamination, in which gastric lavage was performed in one patient (3%) and activated charcoal was given in 11 patients (34%) with 2 of them (6%) receiving multiple doses of charcoal (Table 1). One patient (3%) required intubation and mechanical ventilation followed by gastric lavage. Nearly one-third (31%) of the patients in our case series recovered without the need of any treatment and one-third (33%) recovered on intravenous fluid and/or antiemetics only. All survivors recovered uneventfully.
Discussion
Although G. elegans is extremely poisonous, it is seldom reported in the English literature. 9 This is by far the largest case series in the English literature with the involvement of 33 patients. Similar to a previous review of 28 Chinese academic papers, 9 the most common reason for G. elegans poisoning in Hong Kong is mixing-up of benign herbs (76%), either due to misidentification by self or contamination of benign herbs bought from market.
F. hirta Vahl. (五指毛桃) is the most common benign herb found to be mixed up with G. elegans in our study and was found in seven incidences for making soup. 五指毛桃 is the root of F. hirta and can be used as a food ingredient in soups. It is also used as traditional Chinese medicine for treating cough, abscess and relieving skin pain and swelling. In the past, the origin of 五指毛桃 used in Chinese medicine had been mistaken as from F. simplicissima Lour. However, recent studies confirmed that it should be the root of F. hirta.10–12 Due to morphological similarities, F. hirta may be mixed up with the root of G. elegans, which is also known as ‘大茶藥根’ in traditional Chinese medicine. For patient suffering from G. elegans poisoning due to mixing-up with F. hirta, symptoms were usually mild in our case series, which is contradictory with the fact that the most toxic part of G. elegans is the root. 6 One possible explanation is that commercially available F. hirta as a soup ingredient may be contaminated with a small amount of G. elegans during the harvesting process of the closely located root. On the other hand, misidentification of self-picked herbs usually involved the whole toxic plant. Another explanation is that, in China, the process of making soup with food ingredients produces a much more diluted content in contrast to the process of making herbal tea, which aims at extracting much more concentrated herbal ingredients for therapeutic purpose. These might also be the reasons why the only severe poisoning case in our series involved misidentification of self-picked M. pubescentis (玉葉金花) by G. elegans in the making of herbal tea for consumption. Interestingly, a recent study showed a protective effect of M. pubescentis when used together with G. elegans in a 40:1 extract ratio in mice without affecting the therapeutic effect as analgesia. 13 The protective effect of M. pubescentis was not observed in our case likely because of the inadequate dosage of M. pubescentis used. There are two reasons for the misidentification. Although the flowers of M. pubescentis are readily distinguished from that of G. elegans, they may be mixed up by non-experts outside the flowering period. 14 Another reason is that G. elegans tends to accompany the growth of M. pubescentis in nature, 13 favouring its contamination.
The second common cause of gelsemium poisoning involved the consumption of C. filiformis L. (無根藤) in two clusters of a total of five patients. C. filiformis is a climbing, twinning, vine-like, non-toxic parasitic plant. Besides the close proximity of the growth of C. filiformis with G. elegans and their morphological similarities causing easy misidentification and contamination, 15 it was observed that C. filiformis parasitized on G. elegans could accumulate enough toxic gelsemium alkaloid from the latter to cause human poisoning upon consumption. 16
Besides those mentioned in our study, other benign herbs reported to be mixed up or contaminated with G. elegans include Sargentodoxa cuneata (大血藤), 2 Spatholobus suberectus Dunn. (雞血藤) 17 and Lonicerae japonicae Flos. (金銀花).18,19 Risk assessment and preventive measures should also be considered by the health authorities, and public can be warned against the high-risk herbs and plants in order to increase their awareness of gelsemium poisoning.
From our study, all patients complained of early onset (<2 h) dizziness after herbs consumption. Together with the unique ocular symptoms, gelsemium poisoning should be highly suspected. Close monitoring of respiratory effort and timely respiratory support in case of deterioration is the key to prevent fatality. More severe patients will also present with muscle weakness, respiratory depression and coma. Other clinical features uncommonly reported include liver and renal impairment, and cardiovascular symptoms9,14, 15 were essentially not seen in our case series, apart from high blood pressure and tachycardia, which was likely secondary to anxiety, and mild renal impairment (creatinine: 115 µmol/L) in only one patent.
Different alkaloids have different actions, and many of them appear to have inhibitory effects on the respiratory centre in the medulla oblongata and anterior horn cells of the spinal cord, resulting in respiratory depression and muscle paralysis. 5 Although the toxic mechanism of gelsemium is not clearly defined, recent studies showed that gelsemine, the second abundant alkaloid in G. elegans, 20 has an agonistic effect on glycine receptor in the central nervous system which, upon its stimulation, causes influx of chloride ions and hyperpolarizes the post-synaptic cell, inhibiting neuronal firing.3,4,20–22 Other gelsemium alkaloids may have different mechanisms of action. Since the chemical composition of G. elegans is complex, and more and more different alkaloids have been identified with potential therapeutic value, further researches in this area are ongoing.
For the management, high suspicion to gelsemium poisoning after herbs consumption and recognition of severe cases with prompt intubation and mechanical ventilation to provide respiratory support is of utmost importance. There is so far no antidote and supportive treatment is the mainstay of management. For cases presented early, gastrointestinal decontamination can be considered. The mortality in our study is 3%, which is comparable to other Chinese case series. 15
Our study shares similar limitations as those in other retrospective chart reviews. As information abstracted was based on reviewing medical records and our database retrospectively, it was not possible for the authors to confirm or evaluate any aspect of the history or clinical findings. Any information obtained was dependent on the detail and accuracy of the documentation by the treating clinician, and missing data is unavoidable.
Conclusion
G. elegans is an extremely poisonous plant. Most poisoning results from contamination or misidentification of benign herbs. Early-onset dizziness (<2 h) with ocular symptoms after herbs consumption is typical for gelsemium poisoning. Early recognition of its toxidrome by medical professionals and good supportive care, particularly respiratory support, are the most important elements in managing gelsemium poisoning patients. Since gelsemium poisoning is recurring in Hong Kong as a result of mixing-up of herbs, health agencies should consider preventive measures to further lower the incidence of local gelsemium poisoning.
Footnotes
Acknowledgements
We certify that we have each made substantial contribution so as to qualify for authorship and that we have approved the contents.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Availability of data and materials
Primary data used were submitted to the research articles.
Informed consent
Since this is a retrospective case series with no patient contact and intervention, written informed consent was considered not necessary because no patient identifiable data have been included in the manuscript.
Ethical approval
Our institute considered ethical approval not necessary as the study involved no patient identifier and intervention. Information used in this study was considered as part of the usual clinical data obtained by our institute and was passed to the researchers in a fully anonymized format.
