Abstract
Background:
Utilization of renal biomarkers such as neutrophil gelatinase-associated lipocalin in the management of acute kidney injury may be useful as a diagnostic tool in the emergency department.
Objective:
The aim of this study is to determine the relationship between serum neutrophil gelatinase-associated lipocalin level and the severity of the acute kidney injury based on the Risk, Injury, Failure, Loss of kidney function, and End-stage kidney disease (RIFLE) classification, and to investigate the role of the serum neutrophil gelatinase-associated lipocalin level in differentiating the etiology and predicting the 30-day mortality rate and need for dialysis.
Methods:
This prospective, observational study was conducted from March 2015 to 2016. Adult patients with acute kidney injury in the emergency department were enrolled in the study. Demographic and clinical features such as hypovolemic state, nephrotoxic substance exposure, renal functions, and serum neutrophil gelatinase-associated lipocalin level were evaluated. After the etiology of the acute kidney injury was ascertained, the severity of the acute kidney injury was determined according to RIFLE criteria. Primary outcome was defined as the correlation between serum neutrophil gelatinase-associated lipocalin level and the severity of the acute kidney injury according to RIFLE classification. Secondary outcomes were defined as the relationship between the serum neutrophil gelatinase-associated lipocalin level and the etiology of the acute kidney injury; need for dialysis and 30-day mortality were defined as poor outcomes.
Results:
A total of 87 patients were included in the study. Mean serum neutrophil gelatinase-associated lipocalin levels were 380.14 ± 276.65 ng/mL in RIFLE-R, 425.80 ± 278.99 ng/mL in RIFLE-I, and 403.60 ± 293.15 ng/mL in RIFLE-F groups. There was no statistically significant relationship between the severity of acute kidney injuries and serum neutrophil gelatinase-associated lipocalin level. Initial serum neutrophil gelatinase-associated lipocalin levels in the emergency department did not indicate a statistically significant ability to predict the etiology of acute kidney injury, 30-day mortality rates, or need for dialysis.
Conclusion:
Initial serum neutrophil gelatinase-associated lipocalin level in the emergency department is not a determinant tool for predicting the severity, etiology, 30-day mortality rates, or need for dialysis in cases of acute kidney injuries.
Keywords
Introduction
Acute kidney injury (AKI) is a clinical syndrome that is characterized by impaired renal functions within hours to days of injury onset. It usually leads to accumulation of toxic substances and impaired internal hemostasis. 1 AKI is a frequently confronted condition in emergency departments (EDs) and the incidence of AKI has been increasing. 2 It is also an independent determinant of morbidity and mortality in many accompanied comorbidities. 3
Both Risk, Injury, Failure, Loss of renal function, and End-stage kidney disease (RIFLE) and Acute Kidney Injury Network (AKIN) classifications are described based on serum creatinine level and urine output. Both classifications have been shown to be effective in assessing risk and predicting poor clinical outcomes.4,5
Although serum creatinine is the gold standard in the diagnosis of AKI, serum creatinine levels may not always indicate the presence of renal damage. There may not be an increase in creatinine level until half of kidney functions have been impaired. Therefore, it is possible to see impaired renal function with a normal creatinine level. 6 Although glomerular filtration rate (GFR) may be greatly reduced in the early period of AKI, this may only manifest with a slight increase in creatinine. Serum creatinine level may also be elevated in chronic kidney failure; however, there may not be an AKI. 7 Therefore, interpreting the actual serum creatinine may be difficult in patients whose basal renal functions are not known in the ED.
Management strategies differ depending on the type of AKI. Therefore, early diagnosis and treatment initiation have significant effects on treatment success. 8 Conventional use of fractional sodium excretion, fractional urea excretion, and urea/creatinine ratio values have limited abilities to differentiate between AKI types in the ED. 9 Although renal functions cannot be specified by renal biomarkers, which are indicative of structural renal cell injury, they may be useful in differentiating AKI types. 10 Utilizing renal biomarkers in AKI diagnoses and assessing the intensity and etiology of the AKI may be useful for optimizing ED management. 11
Suggested biomarkers are neutrophil gelatinase-associated lipocalin (NGAL) and cystatin C in the plasma, and NGAL, kidney injury molecule-1 (KIM-1), interleukin-18 (IL-18), and cystatin C in the urine. 6 NGAL is shown to be produced from renal tubules in response to structural renal injury. 12 Previous studies have demonstrated that NGAL is elevated in the early period of AKI and is useful for predicting poor outcomes of AKI. 13 It was also shown that NGAL becomes elevated before creatinine in the early period of AKI. 14
The aim of this study is to determine the relationship between serum NGAL level and the severity of kidney injuries classified on the basis of RIFLE classification, and to investigate the role of the serum NGAL level in differentiating the etiology and predicting 30-day mortality rates and need for dialysis.
Method
Study design
This is a prospective, observational study that was conducted from March 2015 to 2016 in an academic urban ED with an annual census of approximately 50,000 patients per year. Institutional review board approval and informed consent from the patients were obtained for the study.
Study setting and population
Patients 18 years or older who were admitted to the ED with a detected AKI or AKI on the basis of chronic kidney disease were enrolled in the study. Patients under the age of 18 years, chronic dialysis patients, pregnant patients, and patients who refused to give consent were excluded from the study.
In both genders, creatinine levels <1.3 mg/dL were accepted as normal. A 1.5-fold or greater increase in baseline creatinine and urine output lower than 0.5 mL/kg in the previous 6 h were described as AKI. 15 Patients with an unknown baseline creatinine level were considered to have AKI if their measured creatinine level was higher than the normal baseline range with low Modification of Diet in Renal Disease (MDRD) study equation in the ED and if the patient was anuric or the urine output was lower than 0.5 mL/kg in the last 6 h. 16
The severity of the AKI was classified according to the RIFLE criteria as risk (R), injury (I), or failure (F). Because we did not follow patients who had long-term AKI, classes that describe loss of kidney function for longer than 4 weeks (L) and the need for renal replacement treatment for longer than 3 months (E) were excluded from the study.
Data collection
Age, gender, height, medication, past medical history, nephrotoxic drug use, history of contrast agent exposure, vital signs, baseline creatinine, and blood urea nitrogen (BUN) were recorded. Indicative factors for a hypovolemic state such as decreased oral intake, intense vomiting, or diarrhea were evaluated. A urinary ultrasound was performed to exclude postrenal etiology. Creatinine, BUN, MDRD, pH, and HCO3 levels were evaluated from the initial blood draw. The second blood sample for the NGAL measurement was obtained within the first 2 h of admission from the patients who were considered to have AKI. For the NGAL measurement (BioVendor Human Lipocalin-2/NGAL ELISA, Kit—Catalog No: RD191102200R), 10 mL of blood samples were obtained for the serum separating tubes, which were left to coagulate for 30 min and then centrifuged at 4000 r/min for 15 min. The obtained serums were stored in separate Eppendorf Tubes for each patient and were kept at −60°C until the analysis. A fully automated micro-ELISA (enzyme-linked immunosorbent assay) method was performed for the measurement.
AKIs were classified as prerenal, intrinsic, and postrenal by an independent nephrologist who was blind to the study. After the etiology of the AKI was ascertained, the severity of the AKI was determined according to RIFLE criteria. Patients who needed to undergo dialysis during the hospital stay were also recorded. The endpoint of the data collection period was defined as discharge, admission to the hospital, admission to the intensive care unit, or death. The follow-up BUN and creatinine levels were recorded once between 1 and 4 weeks after admission and 30-day mortality rates were determined. The follow-up information of the study patients were obtained either from the hospital electronic records or a follow-up appointment for the discharged patients.
Outcome measurements
Primary outcome was defined as the correlation between serum NGAL level and the severity of kidney injury according to RIFLE classification.
Secondary outcomes were defined as the relationship between the serum NGAL level and the etiology of the AKI; need for dialysis and 30-day mortality were defined as poor outcomes.
Data analysis
All statistical analyses were performed with SPSS for Windows (version. 21.0, SPSS Inc., Chicago, IL, USA). Variable normality was evaluated using visual (histogram and probability plots) and analytical methods (Kolmogorov–Smirnov/Shapiro–Wilk tests). For non-normal distributed data, those with skewness and kurtosis values between −1.5 and +1.5 were considered normal.
17
Mean and standard deviations were used for normally distributed data, and median and interquartile ranges were used for non-normally distributed data. Student’s
Results
Between March 2015 and March 2016, 137 patients with AKI were evaluated for the study. After excluding 50 patients, a total of 87 patients were selected (Figure 1). Patients’ characteristics, including laboratory test results, are shown in Table 1. Arranged by the severity of AKI, a total of 19 patients (21.8%) were RIFLE-R, 20 patients (23%) were RIFLE-I, and 48 patients (55.3%) were RIFLE-F. Mean serum NGAL levels were 380.14 ± 276.65 ng/mL (95% confidence interval (CI): 246.80 ± 513.48) in RIFLE-R, 425.80 ± 278.99 ng/mL (95% CI: 295.21 ± 556.436) in RIFLE-I, and 403.60 ± 293.15 ng/mL (95% CI: 318.44 ± 488.68) in RIFLE-F groups, respectively. There was no statistically significant relationship between the severity of AKI and serum NGAL level (p = 0.874). The relationship between serum NGAL level and AKI severity is shown in Figure 2.
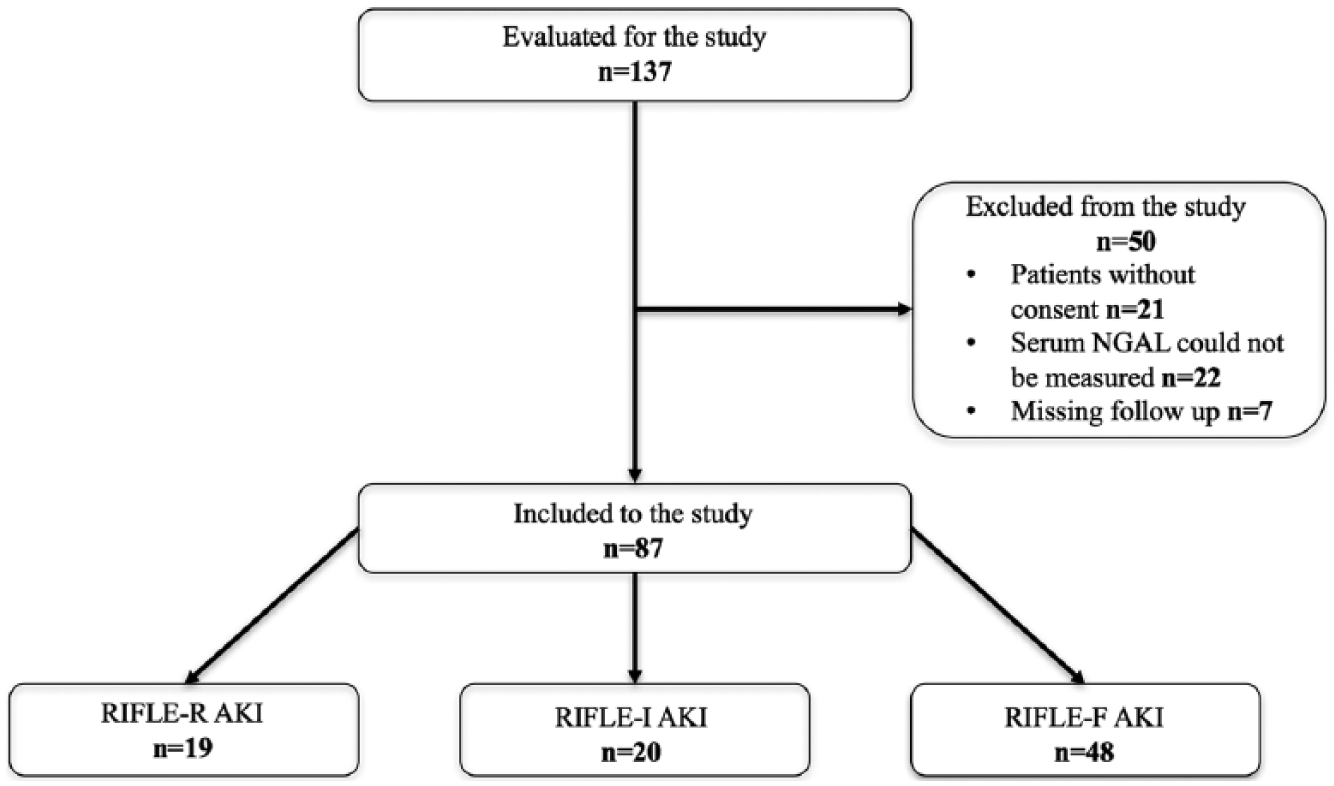
Patient flowchart.
Basic characteristics of the study population.
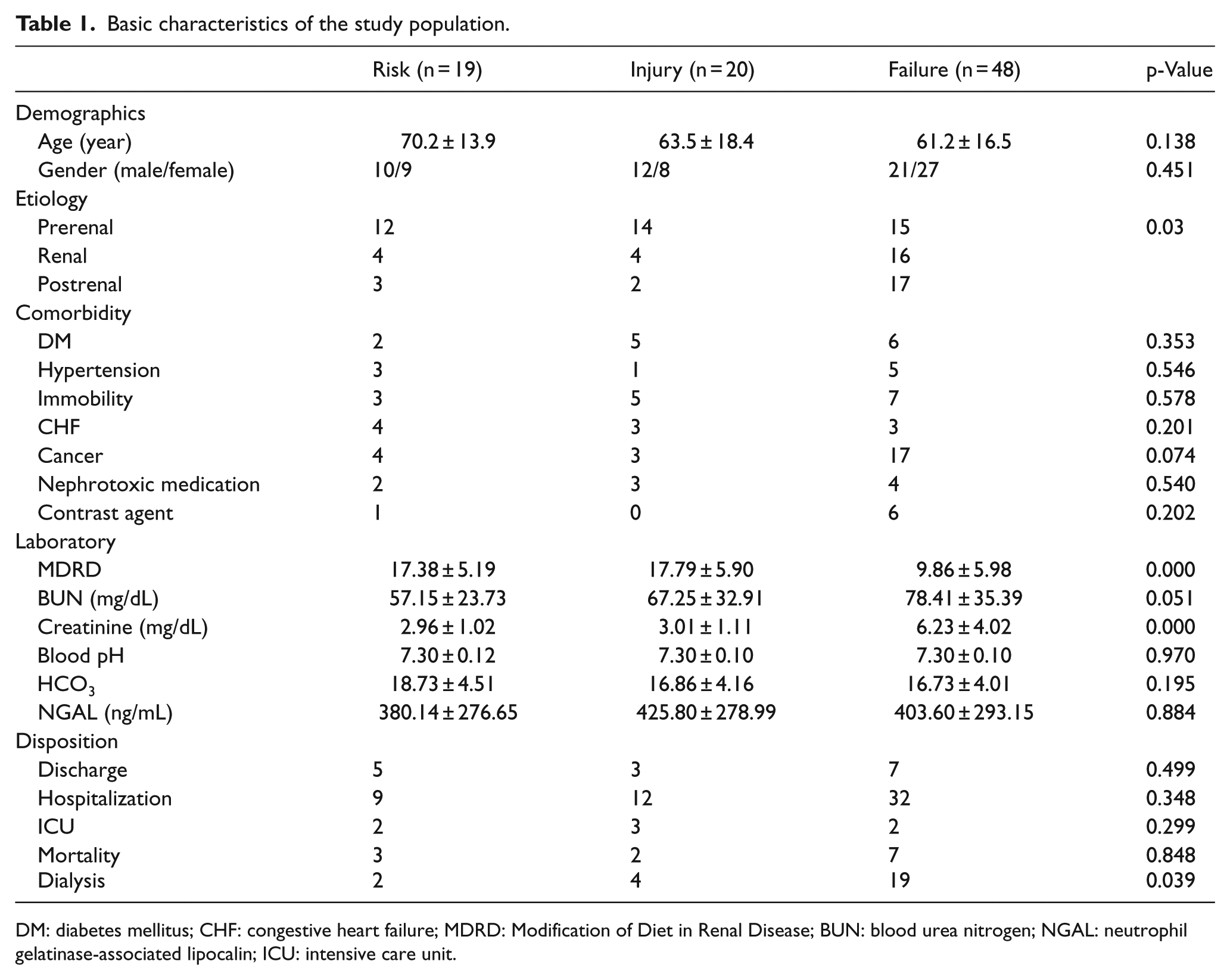
DM: diabetes mellitus; CHF: congestive heart failure; MDRD: Modification of Diet in Renal Disease; BUN: blood urea nitrogen; NGAL: neutrophil gelatinase-associated lipocalin; ICU: intensive care unit.
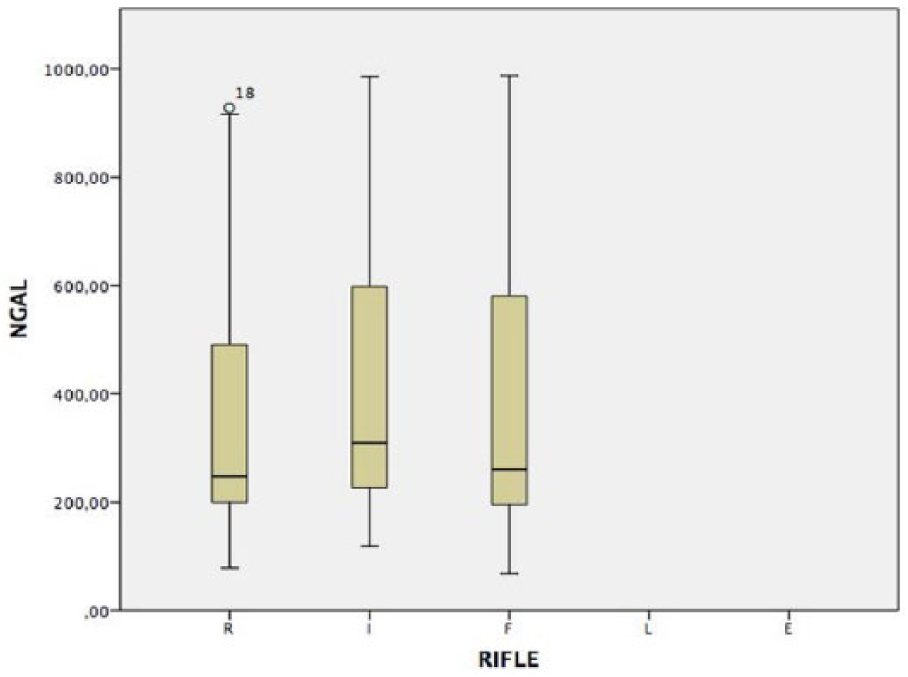
Relationship between serum NGAL level and AKI severity.
During the follow-up procedure, study patients’ results were evaluated by an independent nephrologist who was blind to the study. A total of 41 patients (47.1%) were diagnosed prerenal, 24 patients (27.6%) were intrinsic, and 22 patients (25.3%) were postrenal in their AKI status. Mean serum NGAL level was 406.18 ± 283.10 ng/mL (95% CI: 316.82 ± 495.53) in the prerenal group, 380.46 ± 287.25 ng/mL (95% CI: 259.17 ± 501.76) in the renal group, and 423.86 ± 291.96 ng/mL (95% CI: 294.41 ± 553.31) in the postrenal group (Table 2). There was no statistically significant relationship between the groups (p = 0.874).
Relation of the serum NGAL level and the etiology and outcome of the AKI.
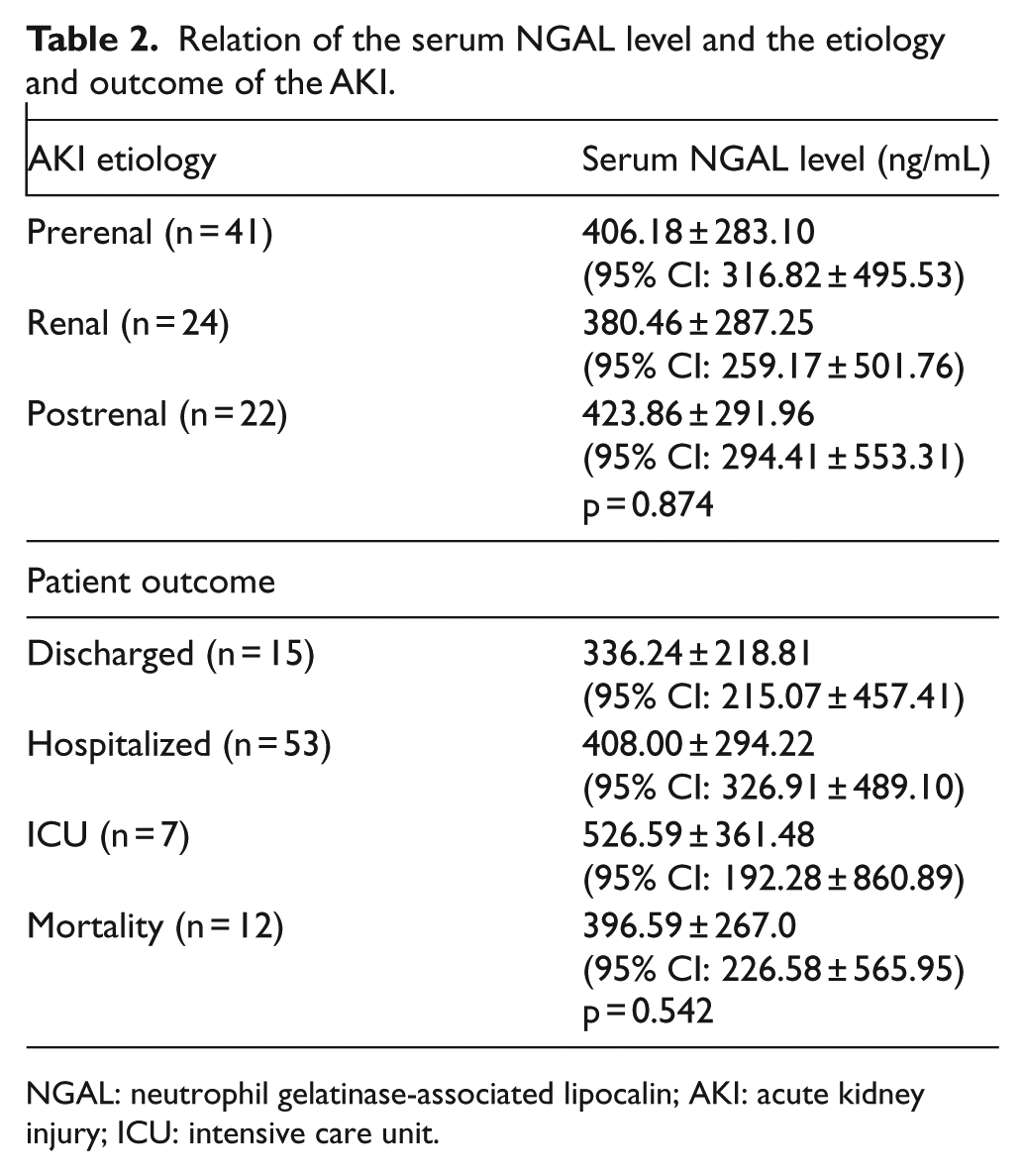
NGAL: neutrophil gelatinase-associated lipocalin; AKI: acute kidney injury; ICU: intensive care unit.
Among the 32 patients with poor outcomes, 25 underwent dialysis and 12 died within 30 days (Table 2). Five of the patients died after undergoing dialysis. Mean serum NGAL level was 440.78 ± 322.57 ng/mL in patients with poor outcomes and 381.89 ± 258.92 ng/mL in those without poor outcomes. The mean difference was −58.89 ng/mL (95% CI: −184.34 to 66.56). There was no statistically significant difference between patients with and without poor outcomes. Mean serum NGAL levels in patients with mortality within 30 days were not significantly different from those who survived (396.59 ± 267.06 ng/mL and 404.72 ± 287.83 ng/mL), respectively. Mean difference was 8.45 ng/mL (95% CI: –167.87 to 184.77). Mean serum NGAL levels in 25 patients who underwent dialysis were not significantly different from those who did not. (Mean serum NGAL levels were 471.92 ± 349.21 ng/mL and 375.99 ± 250.42 ng/mL, respectively. Mean difference was −95.93 ng/mL (95% CI: –228.69 to 36.83).)
There was no statistically significant difference between the patients who were discharged from the ED and those were not discharged. (Mean serum NGAL levels were 336.24 ± 218.35 ng/mL and 417.58 ± 294.63 ng/mL, respectively. Mean difference was 81.34 ng/mL (95% CI: –78.67 to 241.35).).
Discussion
AKI is generally diagnosed with serum creatinine, GFR, and urine output in the ED. However, changes in the serum creatinine and urine output may not be seen in the early phase of AKI. This may lead to delays in diagnosis 17 of AKI, which is associated with increased mortality, length of stay in the hospital, and healthcare costs. 18 Previous studies recommended the use of novel injury biomarkers including NGAL, KIM-1, and IL-8 in addition to renal functional markers such as creatinine, GFR, and urine output. 17 Our study results indicate that serum NGAL level is not a useful biomarker for determining the severity and etiology of AKI or prognosis of patients with AKI in the ED.
De Geus et al. 19 assessed serum NGAL’s ability to predict severe AKI in critically ill patients using RIFLE criteria. They reported a significant relationship between the serum NGAL level and the severity of the AKI. Soto et al. 20 made a three-grade risk classification according to patients’ serum NGAL levels and evaluated the accuracy of predicting AKI based on serum NGAL level in the patients admitted to hospital wards from the ED. They confirmed the predictive ability of serum NGAL levels regarding the severity of AKI. Our study, however, did not reveal a relationship between the serum NGAL level and the severity of AKI.
Nickolas et al. 13 reported that urinary NGAL level can be used in differentiating intrinsic AKI from the other AKI etiologies, such as prerenal etiologies or non-progressive chronic kidney disease, as well as from normal kidneys. Soto et al. 20 reported that increased serum NGAL level in the first 12 h of the AKI can discriminate intrinsic AKI from prerenal AKI and chronic kidney disease. Our study did not support the previous studies in terms of finding that serum NGAL level is useful in differentiating the etiology of AKI.
Several studies reported that an increased serum NGAL level is useful in predicting nephrology consultation, dialysis, and ICU admission.13,21,22 However, the study by Rewa et al. 23 did not show the significance of serum NGAL level for predicting 30-day mortality rates and the rates of dialysis in the ICU. Our study also did not confirm that serum NGAL level is useful for predicting 30-day mortality rates or need for dialysis. This result may be due to the nephrologists’ differing opinions about initiating dialysis.
Kavalci et al. 24 reported that mean serum NGAL level was 817.65 ± 334.76 ng/mL in the patients who underwent dialysis, whereas the level was 398.97 ± 202.42 ng/mL in those who did not. For the serum NGAL level of 615 ng/mL predicted the need for dialysis with a sensitivity of 82% and specificity of 80%. Although mean serum NGAL levels in our study were higher in the patients who underwent dialysis than in those who did not, the difference was not great enough to be of statistical significance.
Our study suggests that serum NGAL level is not a useful tool for deciding patients’ disposition from the ED. This result may have been caused by our particular study population, as they had other severe comorbidities such as cancer or congestive heart failure, and their physicians made different decisions regarding their care management.
Limitations
Our study has several limitations. First, it is a single-center study, which limits generalizability of the study’s results. Second, although we enrolled patients in the study over a 1-year period, only 87 of the patients were eligible for the study. Third, we did not make a serial NGAL measurement. Because of the short follow-up period with the patients in the ED, we could only analyze one serum NGAL measurement. This may have resulted in a high level of NGAL in some patients which may represent injuries over a large time-frame. Fourth, despite our meticulous efforts in drawing blood, centrifuging, and adhering to the cold chain, we could not elicit the serum NGAL levels of 22 patients. This was probably caused by hemolysis of their blood in the tubes or very high serum NGAL levels that were beyond the device’s ability to analyze which might lead it to miss RIFLE-F patients with a high level of NGAL. Fifth, because some patients were referred to our ED from other healthcare facilities, we might have detected more severe AKI than the initial healthcare facility admission accounted for.
Conclusion
Our study demonstrated that serum NGAL level is not a determinant biomarker in the ED for predicting the severity, etiology, and prognosis of AKIs.
Footnotes
Acknowledgements
A.I. and S.Y. contributed to conception; S.Y., E.Y., N.O.D., and M.P. contributed to design; S.Y. contributed to supervision; S.Y., I.U.O., H.G., and T.K. contributed to data collection and processing; S.Y., A.I., and I.U.O. contributed to analysis and interpretation; A.I. and I.U.O. contributed to literature review; A.I. and I.U.O. contributed to writing; and S.Y., I.U.O., N.O.D., E.Y., and M.P. contributed to critical review.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was obtained from the institutional ethical committee before the study.
Human rights
The study was conducted in accordance with the tenets of the Declaration of Helsinki.
Informed consent
All patients signed the informed consent prior to the study enrollment.
