Abstract
Background:
Community-acquired pneumonia is an important cause of mortality and morbidity in all age groups. Oxidant and antioxidant mechanisms play an important role in the pathogenesis and mortality of community-acquired pneumonia.
Objectives:
In this study, the role of thiol/disulfide homeostasis in the diagnosis and prognosis of community-acquired pneumonia was investigated.
Methods:
This was a prospective, controlled, observational study involving 73 community-acquired pneumonia patients and 68 healthy volunteers.
Results:
The native thiol and total thiol, which are thiol/disulfide homeostasis components, were significantly lower in the community-acquired pneumonia group. It was also found that the native thiol was lower in the high-risk community-acquired pneumonia group and that the native thiol and total thiol were associated with the Pneumonia Severity Index, CRB65 (confusion, respiratory rate, blood pressure, ⩾65 years old), and CURB65 (confusion, uremia, respiratory rate, blood pressure, ⩾65 years old) scores. The thiol compound levels were also associated with the C-reactive protein and procalcitonin levels. However, there was no significant difference between the survivors and non-survivors in terms of the thiol/disulfide homeostasis parameters.
Conclusion:
This study demonstrated the important role that oxidative stress plays in the pathogenesis of community-acquired pneumonia. The thiol/disulfide homeostasis biomarkers especially the native thiol and index-1 levels were significantly lower in patients with community-acquired pneumonia. Further studies are needed to investigate the diagnostic and prognostic value of thiol/disulfide homeostasis parameters in community-acquired pneumonia.
Introduction
Community-acquired pneumonia (CAP) cases constitute a significant number of intensive care hospitalizations. 1 Moreover, CAP is an important cause of mortality and morbidity in all age groups. 2 In the 2009 update of the British Thoracic Society guidelines, CAP was defined as:
(a) Symptoms of acute lower respiratory tract disease (cough and at least one of the other symptoms);
(b) Newly detected focal pulmonary findings in the physical examination;
(c) Signs of systemic disease (fever > 38°C and/or sweating, fever, chills, and pain);
(d) There is no other explanation for the illness. 3
The Pneumonia Severity Index (PSI), 4 CURB65 (confusion, uremia, respiratory rate, blood pressure, ⩾65 years old), 5 and CRB65 (confusion, respiratory rate, blood pressure, ⩾65 years old) 6 scores are used to predict the CAP severity and mortality. However, clinicians often present a subjective clinical opinion based on the general condition of the patient, and on the basis of this opinion, they choose the treatment. 4 The subjective and difficult assessment of certain parameters, such as consciousness, in these scoring systems suggests that certain biomarkers may be used for similar purposes. In relation to this, it has been stated that the current use of the serum procalcitonin (PCT) level can be useful for the early prediction of mortality. 7 Similarly, the absence of a decrease in the C-reactive protein (CRP) level with antibiotic therapy has been associated with mortality. 8 Additionally, it was stated that the total antioxidant status (TAS) levels can be used as an auxiliary criterion for the pneumonia severity in the study done by Katsoulis et al. 9 In a similar study, Castillo et al. 10 also reported that the ferric reducing ability of the plasma and lipid peroxidation biomarkers could be used for this purpose. Moreover, proadrenomedullin, provasopressin, neutrophil-to-lymphocyte ratio, platelet-to-lymphocyte ratio, atrial natriuretic peptide, cortisol, copeptin, lactate, and carbon monoxide are some of the biomarkers evaluated in previous researches.11–15
Several recent studies have evaluated the associations between CAP and the oxidant and antioxidant statuses.9,10,16,17 Reactive oxygen species (ROS) occur normally in physiological conditions, and they are counterbalanced by oxidant-antioxidant mechanisms. In CAP, these mechanisms are destroyed by phagocytes acting via the primary defense mechanisms. Excessive ROS amounts are released with respiratory destruction due to the massive phagocytic flow. 16 Some of the antioxidants that attempt to remove ROSs are superoxide dismutase (SOD), catalase (CAT), and the glutathione redox system, which includes reduced glutathione (GSH), glutathione disulfide (GSSG), glutathione reductase (GR), and glutathione peroxidase (GPx). 16
ROSs play a role in the pathogenesis of many pulmonary diseases, such as acute respiratory distress syndrome, bronchopneumonic dysplasia, emphysema, pneumoconiosis, hyperoxia, bleomycin toxicity, cystic fibrosis, bronchial asthma, and pneumonia. In one recent study, the serum TAS values in patients with severe asthma or CAP were significantly lower than those of healthy volunteers. It was indicated that this result probably points to an oxidant/antioxidant imbalance due to an increase in the oxidative load. 18 In another study, increased oxidative stress and the associated enzymatic and non-enzymatic antioxidant activity reductions were demonstrated in children with acute pneumonia. 19
ROSs are inactivated by antioxidant factors, such as SOD, CAT, GPx, albumin, uric acid, lactoferrin, β-carotene, and vitamins C and E. 18 Moreover, the plasma thiol pool is also an antioxidant mechanism. Thiols are oxidized by oxidants and form disulfide bridges, and these bridges can also be reduced to thiol groups. This dynamic thiol/disulfide homeostasis (TDH) mechanism plays a very important role in antioxidant defense. 20 Erel and Neşelioğlu 20 measured the TDH for the first time in the literature via the automatic colorimetric method, and ultimately, the plasma disulfide levels were shown to decrease in smoking, diabetes, some types of cancer, obesity, and pneumonia patients.
There have been limited CAP studies investigating the diagnostic values of these oxidant and antioxidant system parameters and their predictive value on mortality.9,10,16,17 Therefore, we investigated the value of dynamic TDH, which is part of the oxidant-antioxidant system, in the diagnosis of CAP and the prediction of mortality for the first time in the literature.
Methods
A total of 141 subjects, 73 patients and 68 healthy volunteers, were included in this open, prospective, controlled study. The study was carried out in the Ankara Atatürk Training and Research Hospital Emergency Department which is a district hospital in its region in a 6-month period, and approval was obtained from the Ethics Committee of the Yildirim Beyazit University Faculty of Medicine. Written informed consent was obtained from the patients for their anonymized information to be published in this article. Those patients >18 years old who presented to the emergency unit of a training and research hospital which is in the center of the capital and were diagnosed with CAP were included in this study consecutively. The diagnosis of CAP was made by hematological and biochemical studies, chest X-ray, thorax tomography, and emergency medicine and chest diseases specialists’ joint opinion consensus. Patients with acute symptoms, pneumonic infiltration in thorax tomography, and no history of hospitalization in the last 1 month were recorded as CAP. Patients who were pregnant, refused to participate in the study, and immunocompromised (immunosuppressive treatment, hematologic malignancy, congenital or acquired immunodeficiency such as HIV infection), with a history of hospitalization last 1 month or in nursing homes (health-care-associated pneumonia), were excluded from the study. Healthy volunteers were selected from those who were over 18 years of age, were not in pregnancy or lactation, did not have acute or chronic disease, did not have chronic drug use history, and agreed to participate in the study.
Blood samples were taken within the first few minutes of presenting to the emergency services, before any medication such as antibiotics, acetyl cysteine, antipyretics, anti-inflammatory drugs (except oxygen) was started. The levels of the parameters used for the TDH assessment were measured using an automatic analyzer via the spectrophotometric method previously described by Erel and Neşelioğlu. 20 The native thiol (NT), total thiol (TT), and disulfide (D) levels were determined using this method, and the ratios of these three parameters were calculated (index 1: D/NT, index 2: D/TT, index 3: NT/TT). Thus, we used this new test cluster concept containing these ratios which was introduced by Erel and Neşelioğlu. 20
The PSI, CURB65, and CRB65 scores were calculated in each of the patients, and the CAP cases were evaluated as high risk (classes 4 and 5) and low risk (classes 1, 2, and 3) according to the PSI scores; mutual analyses were performed with these oxidative stress parameters. In addition, the correlation between the TDH parameters and the prognostic biomarkers was evaluated.
Statistical analysis
The Statistical Package for the Social Sciences for Windows, version 16.0 (SPSS Inc., Chicago, IL, USA) was used for the statistical analyses in this study. The normality analyses of the data were conducted using the Kolmogorov–Smirnov test. The data showing normal distributions were expressed as the mean ± standard deviation, while the data without normal distributions were expressed as the median and min-max. The Mann–Whitney U test was used for the parameters without normal distributions, and the independent-samples t-test was used for the normally distributed parameters when comparing two independent groups. The Spearman correlation test was used for correlating the biochemical parameters, and the Pearson test was used for the normally distributed data. Multiple logistic regression analysis was used to investigate the parameters influencing the 28-day mortality. A receiver-operating characteristic curve (ROC) analysis was used to estimate the effects of the oxidative stress parameters on the mortality, and the area under the curve (AUC), sensitivity, and specificity were calculated. Thorax tomography and emergency medicine and chest diseases specialists’ joint opinion were jointly used as the gold standard for CAP. p < 0.05 was used for the statistical significance level.
Sample size
In the literature, there is no similar study that has been conducted on TDH in pneumonia cases. For this study, we calculated the sample size using the mean and standard deviation of the control group (NT: 261 ± 44.5 µmol/L; D: 15.4 ± 4.9 µmol/L) of Topuz and colleagues’ 21 study in pulmonary thromboembolism. We were inspired by studies on thiol-disulfide homeostasis and predicted that an approximately 30 and 3 µmol/L difference in NT and D parameters, respectively, between the control and the patient groups would be valuable for this study. Accordingly, the minimum sample size required for each group was calculated as 30 for the NT parameter and 33 for the D parameter with 80% power and 5% Type-1 error.
Results
A total of 73 patients with CAP and 68 healthy volunteers were included in this study. There were no statistically significant differences in terms of the age and gender distributions of the groups (Table 1).
Demographic features, risk scores, mortality rate, and thiol/disulfide homeostasis parameters (control vs CAP groups).
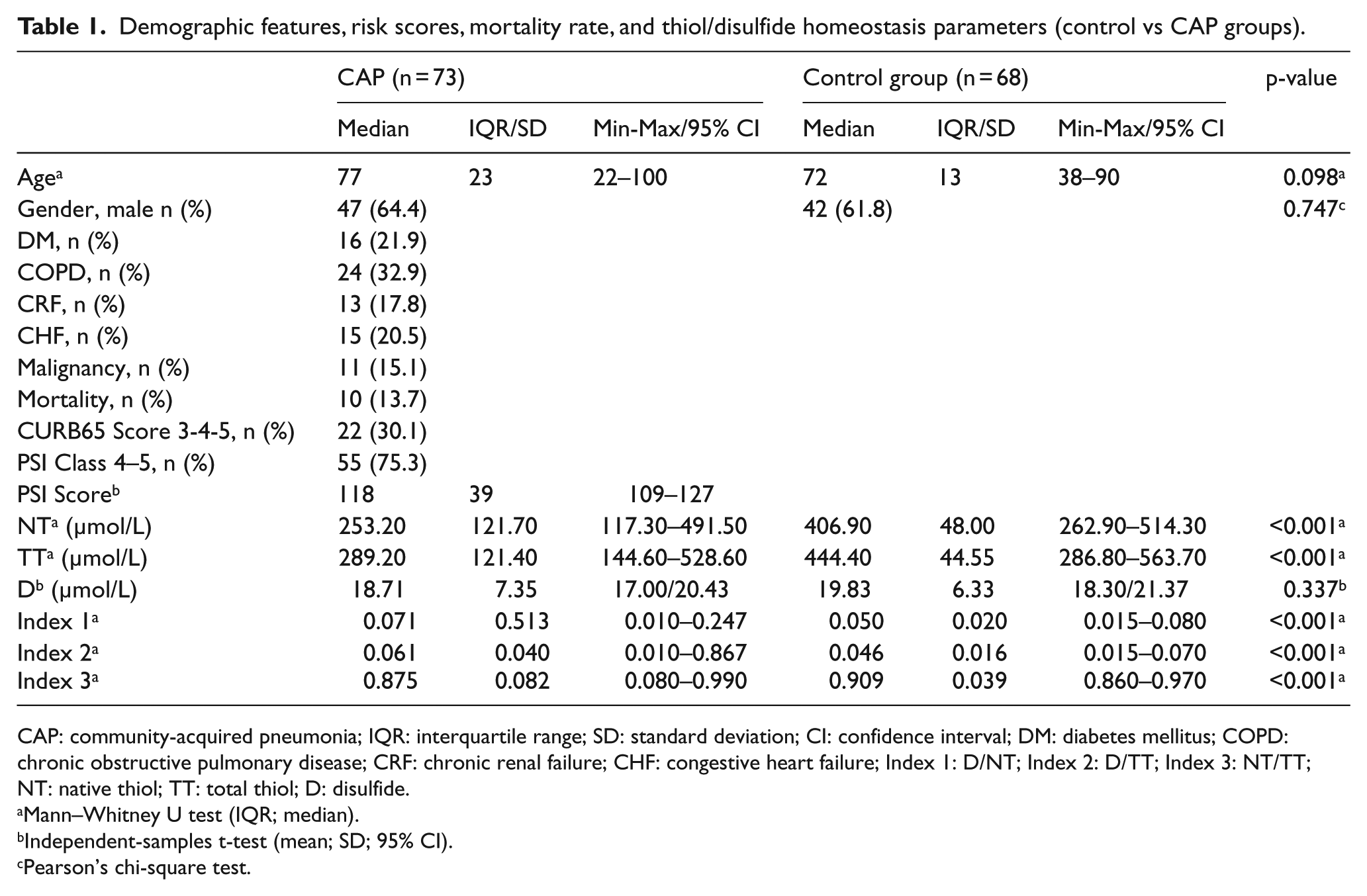
CAP: community-acquired pneumonia; IQR: interquartile range; SD: standard deviation; CI: confidence interval; DM: diabetes mellitus; COPD: chronic obstructive pulmonary disease; CRF: chronic renal failure; CHF: congestive heart failure; Index 1: D/NT; Index 2: D/TT; Index 3: NT/TT; NT: native thiol; TT: total thiol; D: disulfide.
Mann–Whitney U test (IQR; median).
Independent-samples t-test (mean; SD; 95% CI).
Pearson’s chi-square test.
There were significant differences between the two groups with regard to the NT, TT, and index 1-2-3 (p < 0.001; Table 1). In the patient group, the NT, TT, and index 3 were lower, while indexes 1 and 2 were higher. There was no significant difference between the groups according to the D (p = 0.171). The ROC analysis of the control group and patient group, the AUC, and the sensitivity and specificity values for the NT/TT are presented in Figure 1 and Table 2.
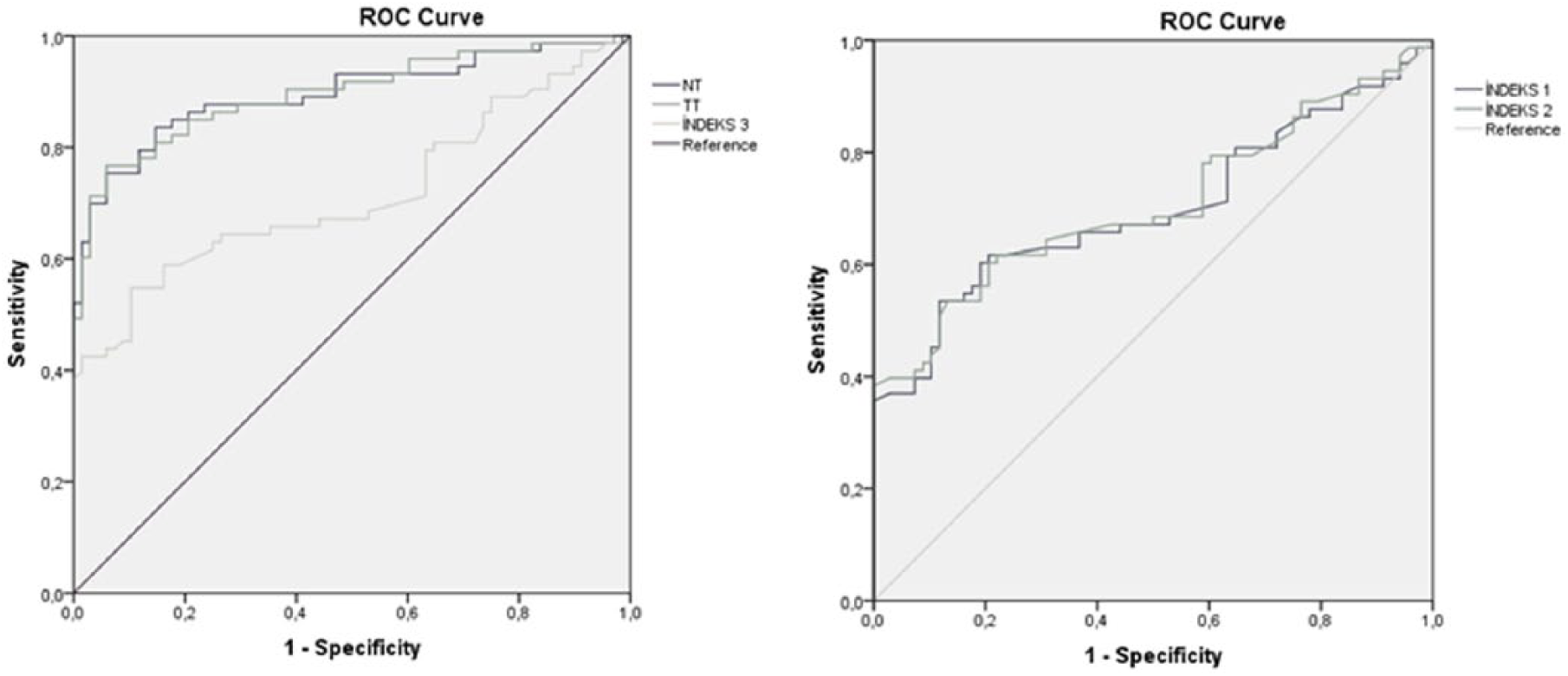
ROC analysis of TDH parameters (CAP diagnosis).
ROC analysis for TDH parameters.
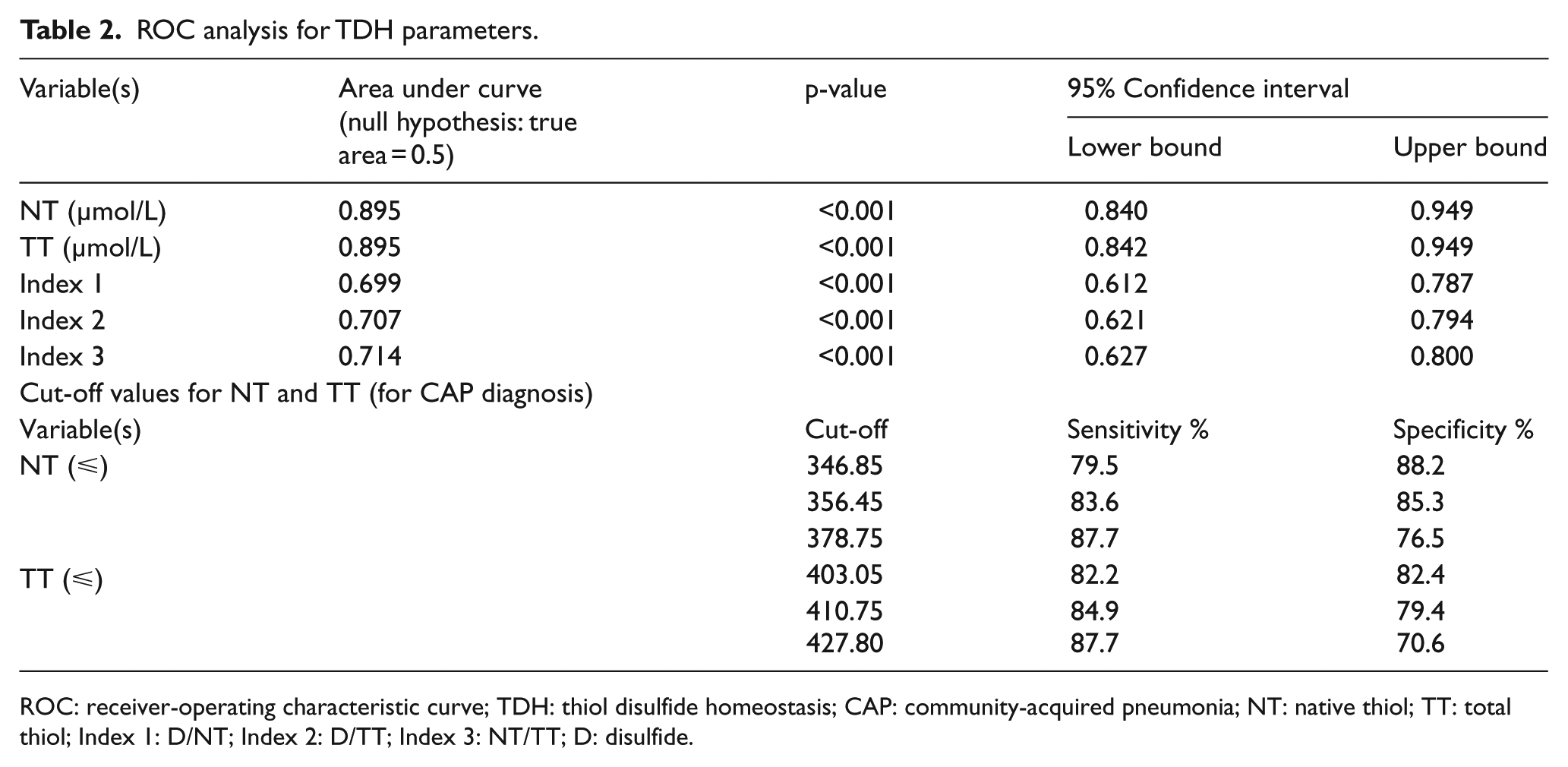
ROC: receiver-operating characteristic curve; TDH: thiol disulfide homeostasis; CAP: community-acquired pneumonia; NT: native thiol; TT: total thiol; Index 1: D/NT; Index 2: D/TT; Index 3: NT/TT; D: disulfide.
The patients with and without 28-day mortality were compared in terms of the NT, TT, and D and indexes 1, 2, and 3. However, the differences were not statistically significant in any of the parameters (Table 3).
Thiol/disulfide homeostasis parameters by groups according to mortality.
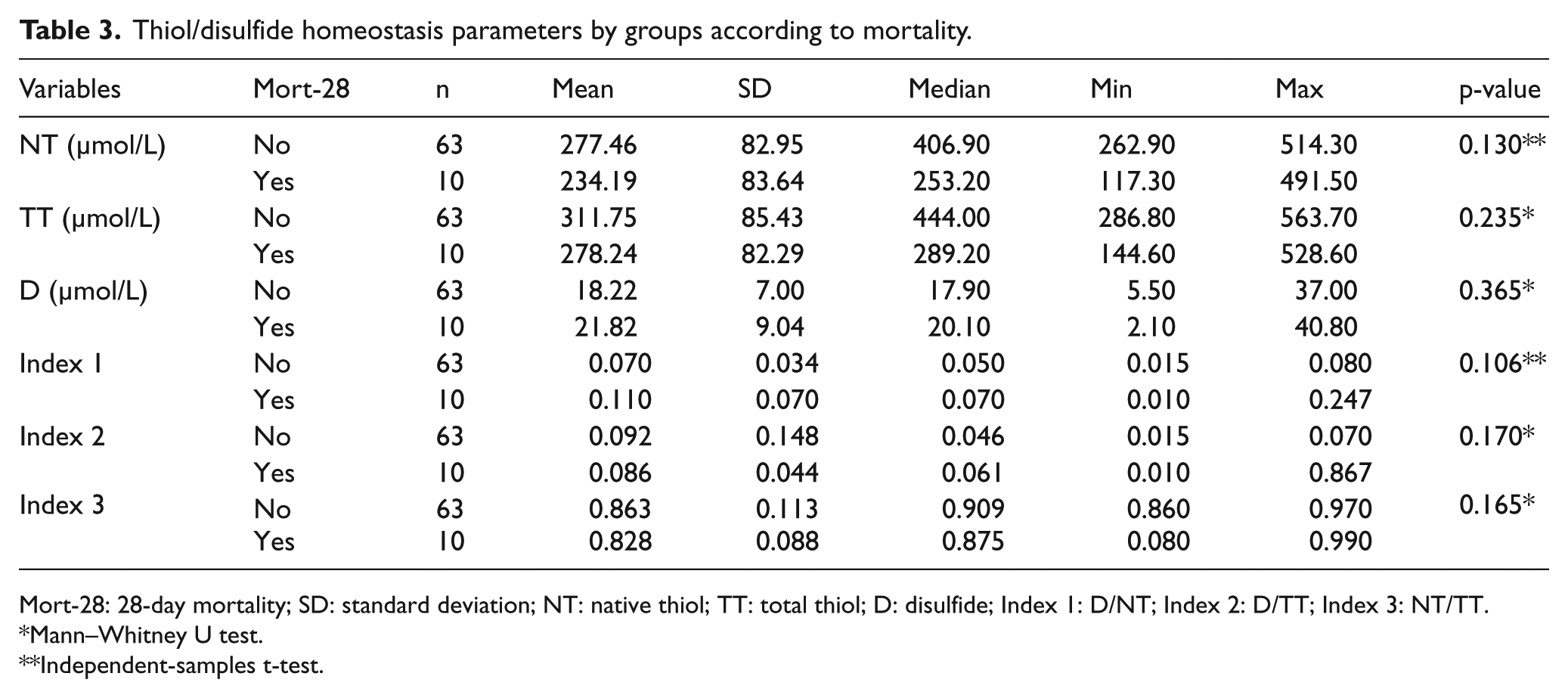
Mort-28: 28-day mortality; SD: standard deviation; NT: native thiol; TT: total thiol; D: disulfide; Index 1: D/NT; Index 2: D/TT; Index 3: NT/TT.
Mann–Whitney U test.
Independent-samples t-test.
When the factors affecting the mortality were evaluated using a multiple regression analysis, it was found that the malignancy history, oxygen saturation, blood urea nitrogen, and hematocrit were related to the mortality; however, relationships with the other parameters were not found (Table 4).
Multiple logistic regression for 28-day mortality.
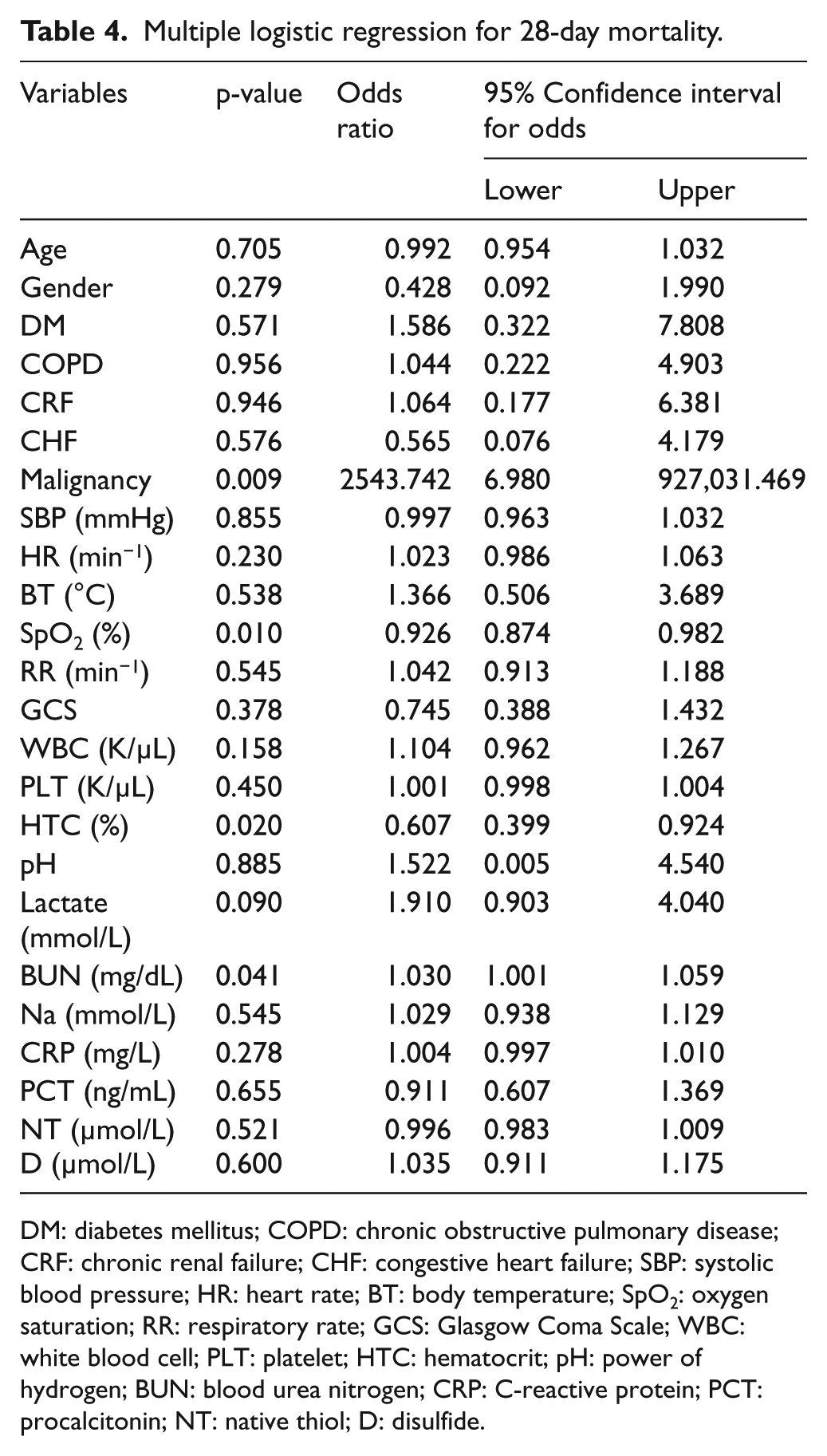
DM: diabetes mellitus; COPD: chronic obstructive pulmonary disease; CRF: chronic renal failure; CHF: congestive heart failure; SBP: systolic blood pressure; HR: heart rate; BT: body temperature; SpO2: oxygen saturation; RR: respiratory rate; GCS: Glasgow Coma Scale; WBC: white blood cell; PLT: platelet; HTC: hematocrit; pH: power of hydrogen; BUN: blood urea nitrogen; CRP: C-reactive protein; PCT: procalcitonin; NT: native thiol; D: disulfide.
The PSI results were classified as low risk (1, 2, and 3) and high risk (4 and 5); and then, the NT, TT, and D and indexes 1, 2, and 3 were compared between these two groups. The NT parameters were found to be significantly lower in the high-risk group (260 ± 79/305 ± 90; p = 0.047; independent-samples t-test). However, there was no significant difference between the low- and high-risk groups in the other parameters. When the ROC analysis was performed for the NT, the AUC was not statistically significant (AUC = 0.634; p = 0.082).
The correlations between the prognostic parameters, staging systems, and TDH parameters were also evaluated (Table 5).
Correlation between biomarkers and prognostic scoring tools.
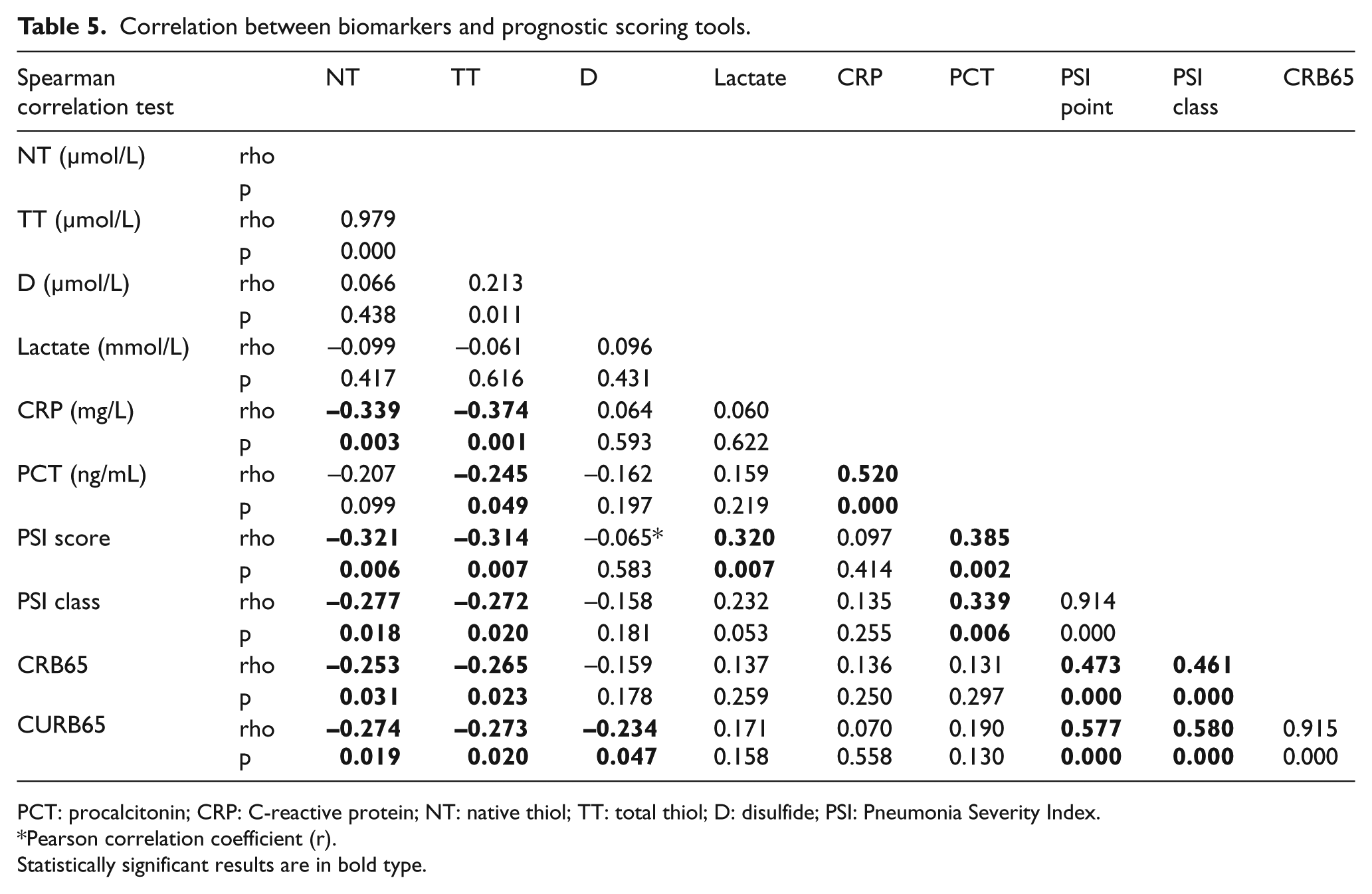
PCT: procalcitonin; CRP: C-reactive protein; NT: native thiol; TT: total thiol; D: disulfide; PSI: Pneumonia Severity Index.
Pearson correlation coefficient (r).
Statistically significant results are in bold type.
Discussion
In this study, it was shown that the TDH parameters were statistically significantly different between the CAP and healthy volunteer groups. In the patient group, the NT, TT, and index 3 were found to be significantly lower, whereas significant elevations were found in indexes 1 and 2 (the decrease in D was not statistically significant). We believe that the significant drop in the NT and TT levels was the end result of an increased oxidative burden; however, there was no increase in the D (bridges) foreseen by the reduction of the thiols. Although there have been no previous studies regarding TDH in adult pneumonia cases, the antioxidant parameters, such as the GSH, have been found to decrease in studies conducted with pediatric pneumonia patients. 19 In the literature, in the TDH evaluations performed in non-pneumonic diagnoses, there are both parallel22,23 and contradictory21,24 results when compared to ours. However, if the NT and TT levels are discussed on their own, they appear to be low in all diagnoses.21–24 Moreover, different results and interpretations arise between studies in terms of the D levels. At this stage, it is not wrong to say that different equations or mechanisms work in the oxidation processes in two different lung pathologies, such as lung cancer and pulmonary thromboembolism.21,23 It should also be considered that different antioxidant defense systems may affect these outcomes.
In one lung cancer study, low NT and D levels were associated with poor mortality. 23 According to our results, the NT and TT were low, while the D was high in the mortality group, but no statistical significance was found. However, according to these data, it can be said that foreseen mechanisms work for NT and TT. For D, there were inverse results between the two studies. In our study, there was no result that could predict mortality (Tables 3 and 4); nevertheless, the lower NT and TT levels in the mortality group suggested that the oxidant burden was of a more serious dimension.
In previous studies, the relationship between the oxidative and antioxidant mechanisms and the severity of pneumonia has been evaluated.9,10 In our study, when the CAP cases were evaluated as high risk and low risk according to the PSI scores, significantly lower values were detected in the high-risk group in terms of the NT (p = 0.047). However, the AUC did not show statistical significance in the ROC analysis (AUC = 0.634; p = 0.082). Therefore, it is not possible to determine a cut-off value that would distinguish the high risk. There were no statistically significant differences in the TT, D, and indexes 1, 2, and 3. However, the NT was lower in the patient group than in the control group, in the survivors group than in the mortality group, and in the high-risk group when compared to the low-risk group. This suggested that the NT may be a promising parameter both as a diagnostic and a prognostic marker in CAP patients.
When the correlations between the staging systems used as prognostic indicators in the clinical approach (PSI, CURB65, and CRB65), the prognostic biomarkers (PCT, CRP, and lactate), and the TDH parameters discussed here were evaluated, it was seen that the NT and TT correlated with the staging systems and the CRP (Table 5). It was also seen that the TT correlated with the PCT and the D correlated with the CURB65, although weakly (Table 5). It is difficult to say whether the TDH parameters alone can be used in place of these staging systems. However, new studies with larger patient numbers may provide an approach to this area.
Limitations
The main limitation of our work is the low number of patients. In addition, the low number of patients in the low-risk group also limited the interpretations of the CAP diagnosis. The fact that the cause of pneumonia has not been evaluated was also a limitation of this study. In the patient group although the cause of pneumonia is thought to be infectious, there are no data about bacterial, viral, or fungal pathogens. Comorbid conditions such as smoking, diabetes, cancer, chronic renal failure, and congestive heart failure were not excluded. While the effects have not been clearly demonstrated, this should also be considered as a limitation.
Conclusion
In this study, the oxidative stress in CAP was quantified using biomarkers of TDH, whose levels can be obtained by easy, inexpensive, and automated techniques. According to the control group, the differences in the patient group suggest that these biomarkers will contribute to the diagnosis of CAP. However, studies with patient groups in which the severity classes are more homogeneous may produce stronger results. In addition, we believe that the thiol compound may be a guide for the CAP severity, due to the low levels of NT in the high-risk group and the correlations between the NT and TT and the PSI, CRB65, and CURB65. Finally, we think that index 1 (D/NT) may be a good indicator of TDH. In particular, we think that studies with higher series should be performed on index 1 and pneumonia severity relation.
Footnotes
Acknowledgements
We sincerely thank all the authors who helped in data collection. A.Ş., Ş.G., A.Ö., G.K.Ç., and F.T. contributed to the literature searches, study screen, data extraction, quality assessment, data analysis, and manuscript writing. S.K. contributed to study screen, data extraction, quality assessment, and manuscript writing. S.N. contributed to study screen, data extraction, data analysis, biochemical work, and manuscript writing. S.N. contributed to study screen, data extraction, biochemical work, and manuscript writing. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of data and materials
The datasets analyzed during this study are not publicly available due to patient privacy and laws but are available from the corresponding author on reasonable request.
Informed consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
Ethical approval
The study was carried out in the Ankara Atatürk Training and Research Hospital Emergency Department, and approval was obtained from the Ethics Committee of the Yildirim Beyazit University Faculty of Medicine on 1 December 2014.
Human rights
The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a priori approval by the institution’s human research committee.
