Abstract
Background:
Although altered mental status is a term commonly used for older patients, the limits of the definition are unclear.
Objectives:
We aimed to determine the predictive factors of altered mental status and to standardize this broad-spectrum definition.
Methods:
The level and content of consciousness were assessed both in the basal status and in the emergency department presentation status of patients who were aged 65 years and above. The Richmond Agitation–Sedation Scale (RASS) for level and five features for content of consciousness were used to determine the actual change in consciousness.
Results:
Among 1250 patients, the rate of true altered mental status was 7.7% (n = 96), transient altered mental status was 3.5% (n = 44), and non-altered mental status was 88.8% (n = 1110). The 1- and 3-month mortality rates were higher for patients with true altered mental status (32.3% and 40.6%, respectively) than for other patients (3.3% and 8.4%, respectively; p < .001). The following criteria predicted true altered mental status with a high specificity: a change in RASS score ⩾2 points, newly disorganized thoughts, a perception disorder, inattention and disorientation, and an inability to communicate.
Conclusion:
True altered mental status criteria can be used to determine alterations in consciousness that lead to high mortality, and they can also be used as a common language for current and further investigations.
Introduction
Altered mental status (AMS) is not only a frequent complaint but also a life-threatening clinical condition consisting of a complex of symptoms. For this reason, AMS can manifest in different forms such as coma, agitation, hallucinations, or the inability to communicate.
In theory, the deterioration of one or both of the components of consciousness (level and content) is defined as AMS.1–3 However, not every change in these components indicates AMS in clinical situation. There are some gray areas in the definition; for example, if a patient with a fever is slightly drowsy, she may or may not have AMS. If a patient has temporary memory impairment during a vertigo attack, AMS may be present.
Difficulties like these and the lack of a standardized definition for AMS have led to different understandings and interpretations of the condition. 2 Therefore, AMS frequency (1%–15%)4,5 and mortality rates (8%–35%)4,6 are distinctly different among studies involving emergency department (ED) patients. In addition to the lack of standardization, it is not known which findings are specific to AMS.
AMS leads to higher mortality rates in elderly patients than in younger ones. 6 Elderly patients also have a greater risk of developing AMS because they have more predisposing factors. Moreover, the mortality rate in elderly patients increases with a prolonged duration of AMS. 7 Therefore, determining the presence of AMS in these patients is important for patient survival.
In this study, we aimed to standardize the definition of AMS in order to develop a common language for practitioners. To do so, we attempted to determine the diagnostic value of the predictive factors that define AMS by examining changes in consciousness content and level.
Methods
Study design
This prospective observational study was performed from January to June 2017 in a secondary-care ED of a hospital in Turkey (210,000 ED visits annually). Ethics committee approval and informed consent from patients or their relatives were obtained. The study protocol was previously published on ClinicalTrials.gov (NCT03040843).
Selection of participants
All patients who were 65 years and above were evaluated for the study. Patients who presented with trauma, needed emergency intervention due to acute clinical conditions (e.g. severe chest pain or severe breathlessness), had an unknown basal mental status (no relatives or incognizant relatives), were unable to communicate due to the basal condition (e.g. hearing-impaired or tracheostomized patients), or did not want to participate in the study were excluded.
Study protocol
Three emergency medicine attending physicians consecutively assessed the consciousness state of the patients within 30 min of the ED presentation. The purpose of the time constraint was to determine the actual change in consciousness before the patients received treatment. These physicians were involved in the follow-up of patients during their 24-h clinical shifts. Therefore, three physicians were minimum required number to follow up patients consecutively as well as to maintain interphysician reliability.
There were two methods of detecting the presence of AMS in patients upon admission. One was the patients’ relatives’ direct mention of AMS findings (meaningless speech, hallucinations, aggression, newly developed memory impairment, newly developed inability to communicate, and other abnormal behaviors) and the acceptance of these findings by the physicians. The other was that, if AMS findings were not mentioned as the chief complaint, the physicians questioned the relatives about the presence of the findings and the relatives’ acceptance of them. In both cases, AMS was considered to be present and was classified under three headings:
True AMS (Tr-AMS): Change in level and/or content of consciousness with ongoing clinical status at the presentation.
Transient AMS (T-AMS): Change in level and/or content of consciousness characterized by rapid onset, short duration, and spontaneous complete recovery before arrival at the hospital.
Normal mental status (non-AMS): No change in level and content of consciousness.
In this study, each patient was assessed for the level and content of consciousness separately. In addition, both the basal consciousness status and the ED presentation consciousness status of the patient were noted (Figure 1). The patient’s basal state of consciousness was obtained from the relatives or surrogates through questions asked by the physicians. The ED presentation status was determined by the physicians’ interviews with the patient (Appendix 1). The consciousness assessment lasted less than 5 min for each patient.
The level of consciousness was questioned and determined both in the basal status and in the ED presentation status through the Richmond Agitation–Sedation Scale (RASS; Appendix 2). 8 By comparing the previous status with the final, it was noted whether there was a change in the RASS score.
For the content of consciousness, five features were assessed both in the basal status and in the ED presentation status, and each content item was investigated separately by comparing the previous and final statuses to see whether there was any deterioration. Flow of thought (newly disorganized thoughts), perception (new perceptual impairment), orientation (new disorientation), attention (new inattention), and memory (new memory impairment) were assessed for content of consciousness. Plum and Posner 1 reported that these components change if a patient has AMS. Also, these content items are components of cognitive tests such as the Mini Mental State Examination 9 and the Six-Item Screener. 10 Moreover, inattention is the main feature of delirium, according to the fifth edition of the Diagnostic and Statistical Manual. 3 If the patient was unable to communicate because of an acute clinical condition (stroke, coma, etc.), this was noted as “newly unable to communicate.”
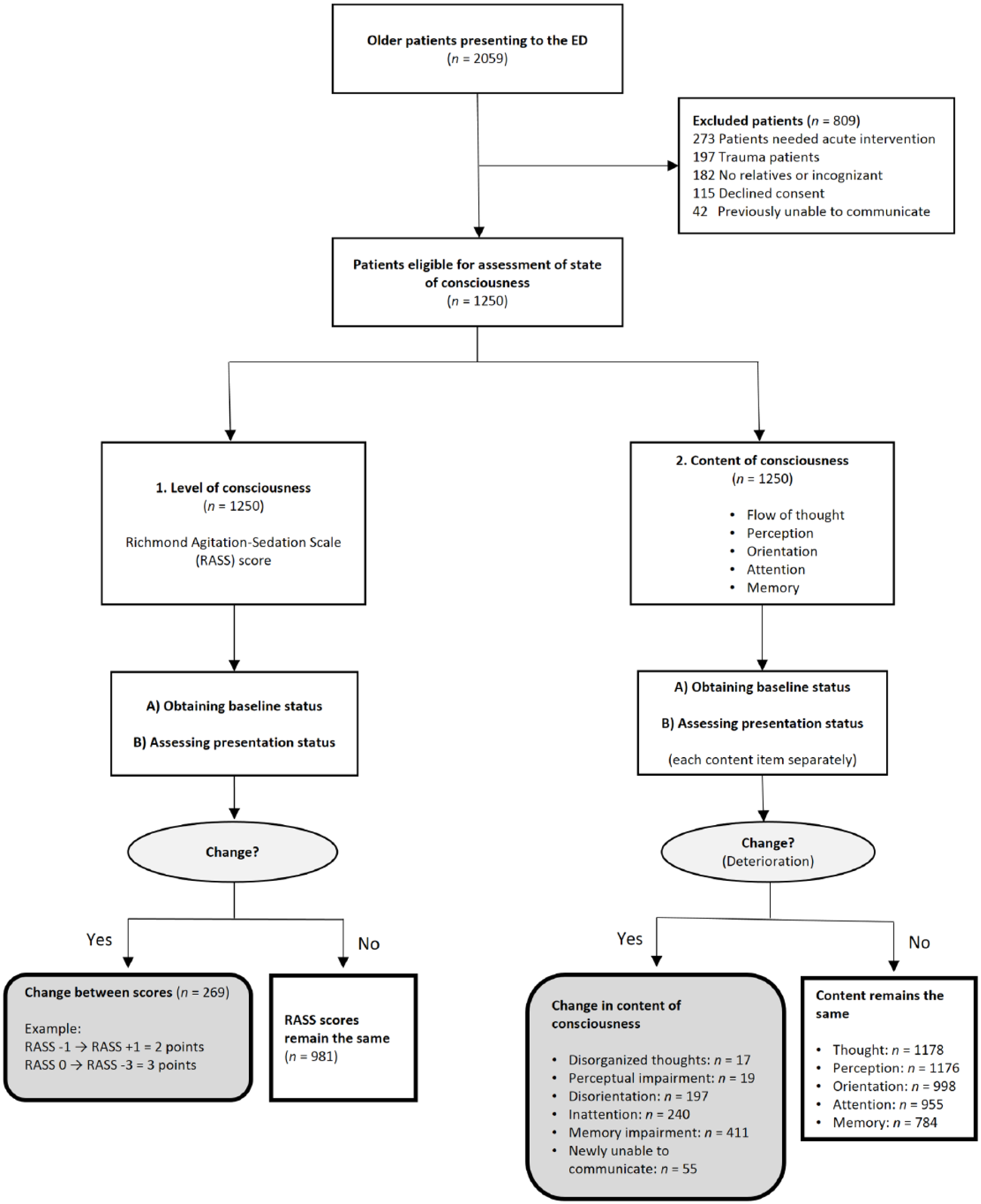
Study design and flow diagram of older patients evaluated for altered mental status.
The applicability of these five consciousness content items, the RASS, and the Glasgow Coma Scale (GCS) was tested with the previous 1 month pilot study. The GCS was removed from the study due to the difficulties of using it to assess the basal status. Five content items and the RASS score were used to evaluate level and content consciousness separately; these methods made it easy to learn the basal status. In addition, the investigators were trained about how to evaluate patients both theoretically for 4 h and practically for 8 h at the bedside after the initial pilot study to ensure interphysician reliability. A standardized script consisting of annotated RASS score (Appendix 2) and detailed content items (Appendix 1) was used to minimize the potential effects of any confounding variables. In the following process, these collected scripts were regularly checked by the same physician. In the presence of suspicion of consciousness level/content or AMS, the final decision was made by at least two physicians.
In addition, the patients’ characteristics, chief complaints, comorbidities, and 1- and 3 month mortality were noted in the study. Mortality information was tracked through the Central Civil Registration System.
Outcome measures
The primary outcome measure was the diagnostic value of predictive factors for AMS in older patients. The secondary outcome measure was mortality status of patients with three different groups named as Tr-AMS, T-AMS, and non-AMS.
Data analysis
Statistical analyses were performed using IBM SPSS Statistics for Windows, Version 21 (Armonk, NY: IBM Corp.) and MedCalc® Version 15.8 (MedCalc Software bvba, Ostend, Belgium). Continuous variables were presented as median values and interquartile ranges (IQRs). Categorical variables were summarized as frequencies and percentages. The differences between two groups of continuous variables were determined by the Mann–Whitney U test. Categorical variables were compared using Pearson’s χ2 or Fisher’s exact test. Odds ratios (ORs) were presented with 95% confidence intervals (95% CI). A critical α value of 0.05 was accepted as statistically significant.
A binary logistic regression model was constructed to define factors predicting the Tr-AMS. The univariate model considered demographic data that included age, chief compliants, comorbidities, RASS, and consciousness contents variables. Each variable was tested in the univariate model, and those comparisons that have a loose p value of less than .1 were then tested in the multivariate model. The area under the receiver operating characteristic (ROC) curve (AUC) was calculated to determine the relationship between RASS change and the presence of Tr-AMS.
Results
Cohort characteristics
During a 6-month period, 1250 eligible patients were screened for the presence of AMS. The rate of Tr-AMS was 7.7% (n = 96), T-AMS was 3.5% (n = 44), and non-AMS was 88.8% (n = 1110). The 1- and 3-month mortality rates were 32.3% and 40.6% for patients with Tr-AMS, 4.5% and 6.8% for T-AMS, and 3.2% and 8.5% for non-AMS, respectively. The mortality rates were similar in the T-AMS and non-AMS groups but significantly higher in the Tr-AMS group (p < .001). Final diagnoses of patients with 3-month mortality at the ED are shown in Appendix 3.
Characteristics and the changes in the consciousness status of the patients with Tr-AMS and the other patients (non-AMS and those with T-AMS) according to the mortality results are given in Tables 1 and 2. While a change in the RASS score between 0 and 1 points was seen in 99.9% of the other patients, the proportion was 55.2% in Tr-AMS patients. A change in the RASS score of ⩾2 points was significantly more common in the Tr-AMS group (44.8%, n = 43) than in the other group (0.1%, n = 1; χ2 test: p < .001). RASS change and the presence of Tr-AMS were moderately correlated (AUC: 0.75; 95% CI: 0.72–0.77; Figure 2).
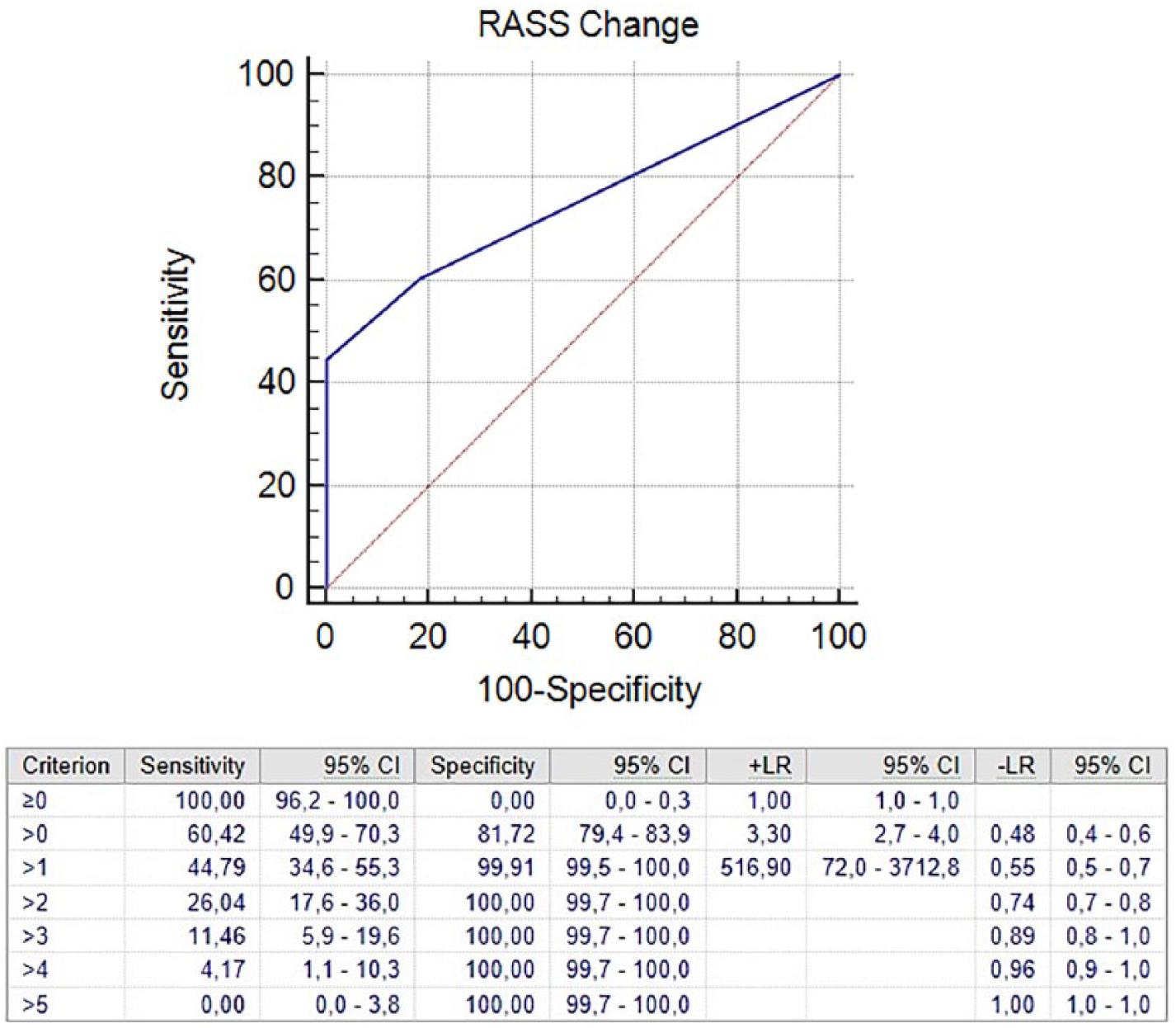
ROC curve of RASS change for predicting Tr-AMS.
Characteristics of older patients assessed for mental status in the ED.
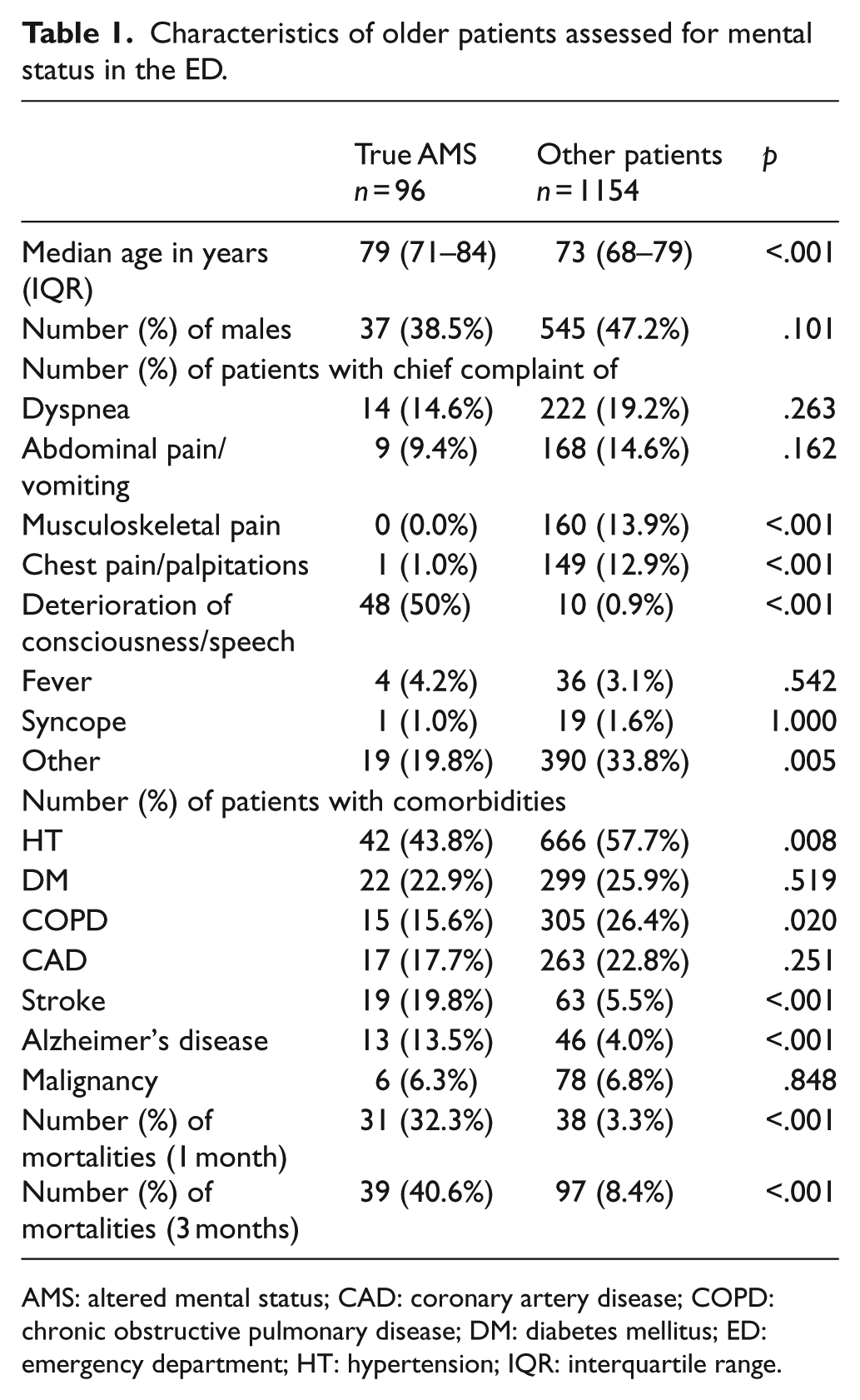
AMS: altered mental status; CAD: coronary artery disease; COPD: chronic obstructive pulmonary disease; DM: diabetes mellitus; ED: emergency department; HT: hypertension; IQR: interquartile range.
Changes in predictive factors between true AMS patients and other patients.
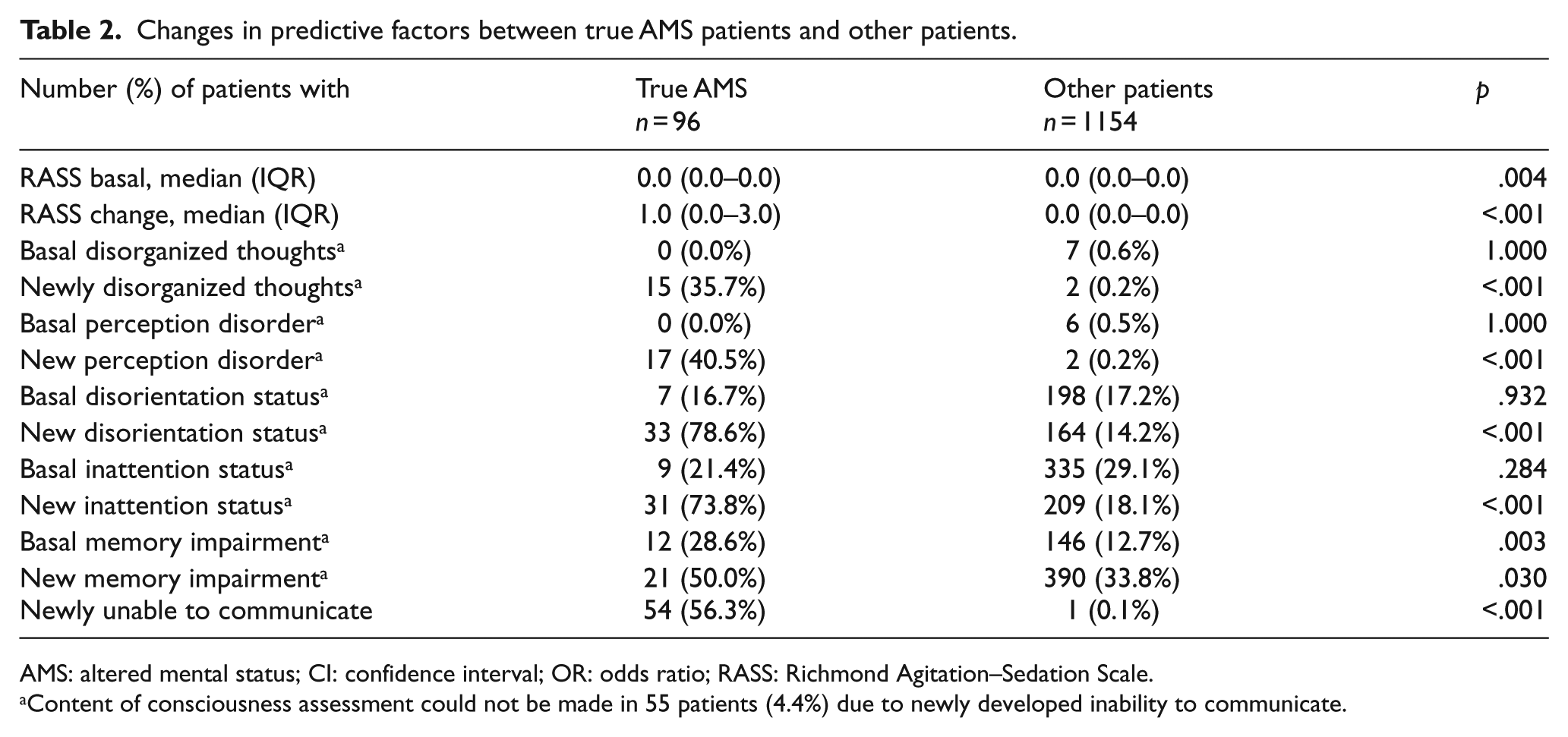
AMS: altered mental status; CI: confidence interval; OR: odds ratio; RASS: Richmond Agitation–Sedation Scale.
Content of consciousness assessment could not be made in 55 patients (4.4%) due to newly developed inability to communicate.
Basal disorganized thoughts, perception disorders, disorientation status, and inattention status rates were similar in both groups. The rate of deterioration in the five consciousness content items was significantly higher in the Tr-AMS group than in the other group. However, an assessment of the consciousness content was not possible in 56.3% of the patients with Tr-AMS (n = 54) due to their new inability to communicate; this condition was found in 0.1% of the other group (n = 1; χ2 test: p < .001). New inability to communicate status was present in 43 non-comatose patients in total.
Derivation of Tr-AMS criteria
The diagnostic accuracy of the changes in the content and level of consciousness for Tr-AMS is shown in Table 3. The specificity of the predictive factors was 99.9% (95% CI = 99.5–100.0) for a change in the RASS score of ⩾2 points, 99.8% (95% CI = 99.4–99.9) for newly disorganized thoughts, 99.8% (95% CI = 99.4–99.9) for new perception disorders, and 99.9 (95% CI = 99.5–100) for a new inability to communicate. In addition, the specificity was 99.3% (95% CI = 98.6–99.7) in combination with a newly developing inattention and disorientation status (28 patients in the Tr-AMS group). Memory impairment was found to have low diagnostic value for Tr-AMS both alone and in combination. In the logistic regression analyses, however, only new memory impairment (OR = 98.136 (95% CI = 1.594–6043.474); p < .029) was found to be significant in the predicting of Tr-AMS (Table 4).
Diagnostic accuracy of predictive factors for true AMS.
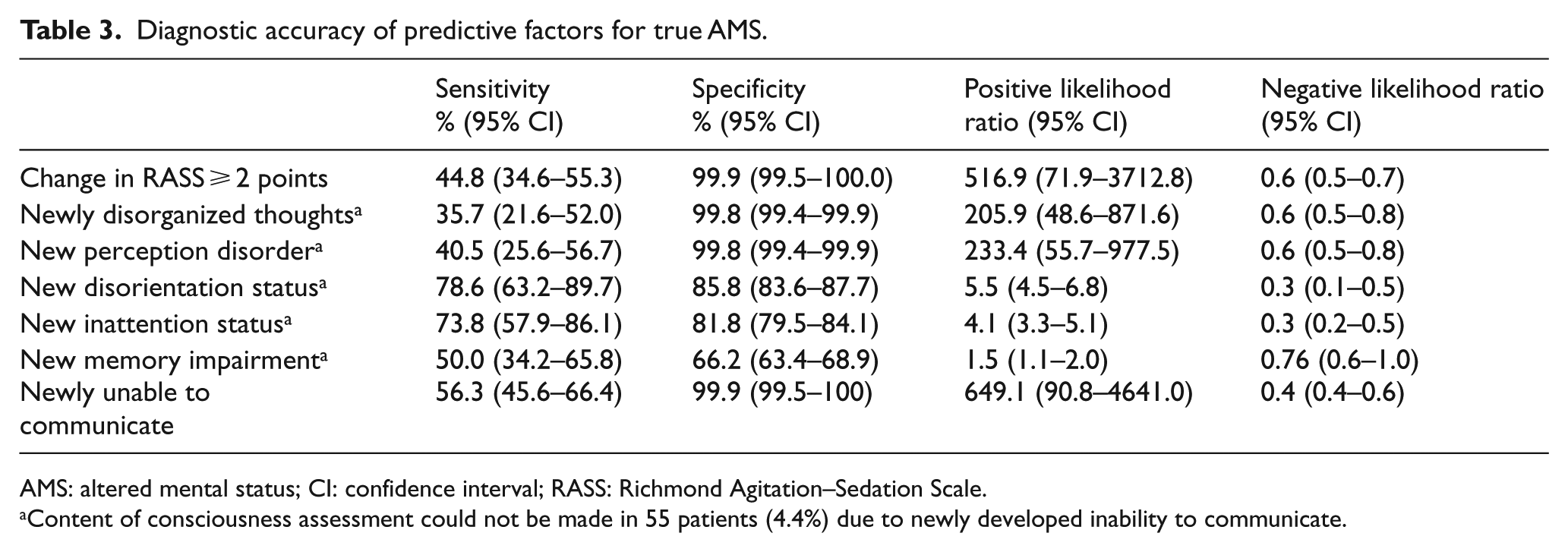
AMS: altered mental status; CI: confidence interval; RASS: Richmond Agitation–Sedation Scale.
Content of consciousness assessment could not be made in 55 patients (4.4%) due to newly developed inability to communicate.
Multivariate logistic regression model to define factors predicting the Tr-AMS a .
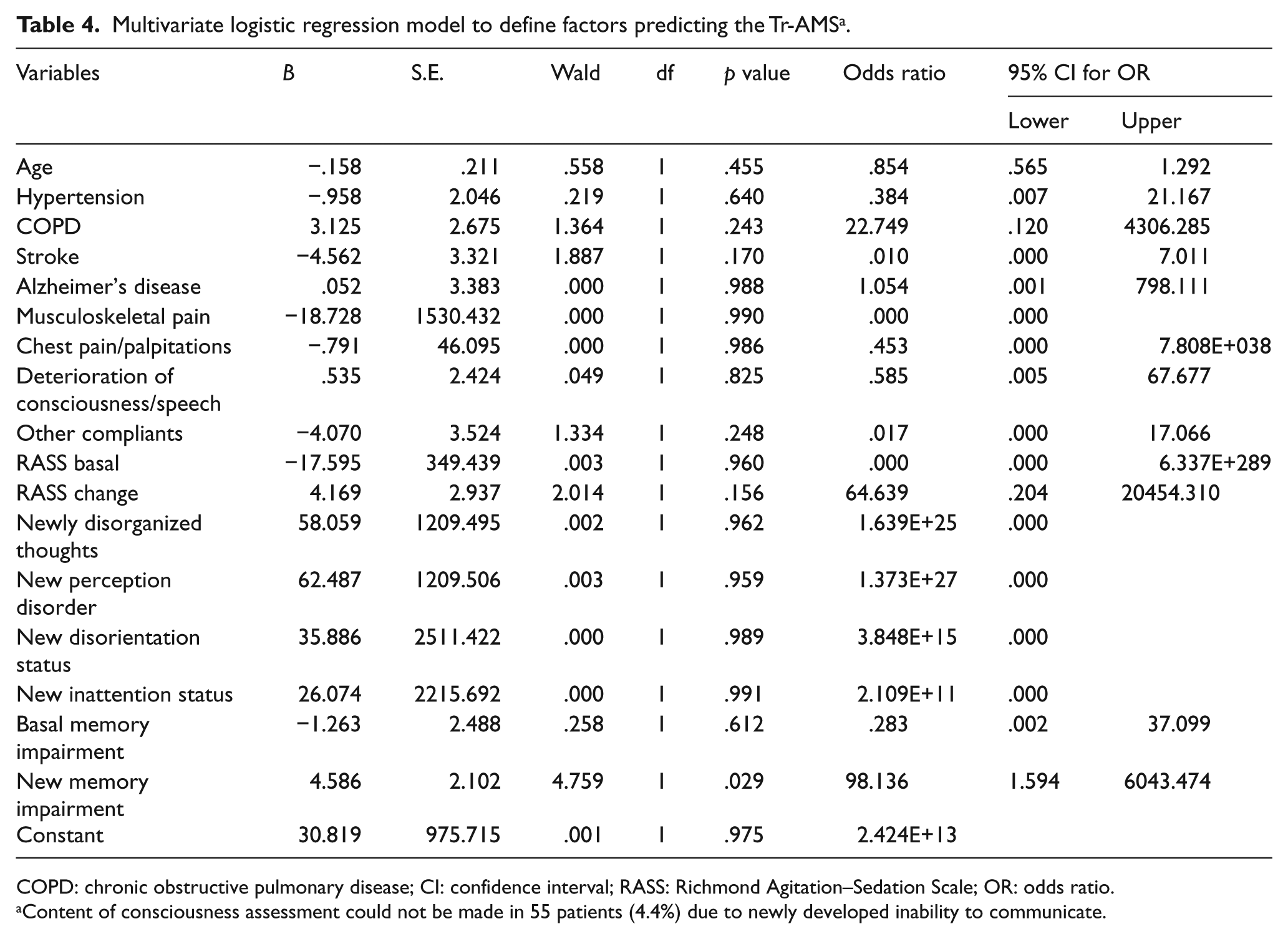
COPD: chronic obstructive pulmonary disease; CI: confidence interval; RASS: Richmond Agitation–Sedation Scale; OR: odds ratio.
Content of consciousness assessment could not be made in 55 patients (4.4%) due to newly developed inability to communicate.
At the end of the study, the first two non-AMS readmissions of patients were reviewed in order to determine the accuracy of the basal consciousness status obtained from the patients’ relatives. A total of 86 readmissions occurred, and the RASS basal score was the same for 97.7% of the readmissions. The same basal status was 98.8% for disorganized thoughts and perception disorders, 82.6% for disorientation, 79.1% for inattention, and 77.9% for memory impairment.
Discussion
In addition to being an urgent complaint, AMS can also be an important indicator of a life-threatening clinical scenario. In the majority of diseases, an impaired consciousness is associated with a poor prognosis, and almost all mortality-related scoring systems have a consciousness factor. 11 It would be correct to take the consciousness status as the fifth vital sign when evaluating patients. 12 For this reason, the recognition and comprehension of this sign are necessary to demonstrate the seriousness of the underlying cause. In the literature, there have been no studies regarding the standardization of AMS.
A various etiological factors cause AMS to appear in different forms. When looking at AMS studies, it is clear that many different criteria are used. In some studies, even T-AMS (transient) has been used as a criterion. However, large population studies with syncope presentations have shown that the 1 month mortality rate in elderly patients is around 1%–2%.13,14 In hospitalized patients with a transient loss of consciousness (known as a T-LOC), it was reported that the 1 month mortality rate was 5.6%.15,16 In our study, of the patients with T-AMS, the mortality rate was 4.5%. This mortality rate was similar to that of non-AMS patients. For this reason, these two groups of patients were accepted as other groups in the study. The difference between T-AMS and T-LOC is that the coverage of T-AMS is wider.
Delirium is the other commonly misunderstood term in the definition of AMS. Though delirium is one of the causes of AMS, not every patient with AMS has delirium. A study investigating the coexistence of delirium and AMS showed that delirium was seen in only half the patients with acute AMS. The same study reported that delirium was not assessable in nearly 40% of all AMS patients with different level of consciousness. 7 The ability to diagnose delirium depends on the patient having a sufficient level of arousal; also, delirium should not be diagnosed in the context of coma. 17 Distinctly lethargic, agitated, and dysphasic patients are other challenging groups in the diagnosis of delirium. In this study, an assessment of the consciousness content was not possible in 56.3% of the patients with Tr-AMS, due to their new inability to communicate; also, causes of new inability to communicate status were related to non-comatose reasons as 78.2%.
The prevalence of AMS has been reported as around 2%–15% in studies conducted in elderly patients.4,18,19 This rate was 7.7% for Tr-AMS in our study. AMS was the primary complaint in half of the patients, but it was questioned about and gleaned from the history taken from the other half of the patients. The hospital mortality rates range from 10% to 40% in elderly patients with AMS.4,6,7 In our study, the 1- and 3-month mortality rates were 32% and 40% for Tr-AMS, respectively.
To detect the presence of AMS in a patient, there must be a change in the patient’s basal state of consciousness, and it should be recognized by the patient’s relatives. This was an important part of our study because while the patients’ presentation state of consciousness was measured objectively, the basal state of consciousness was questioned subjectively from the history. So, it was important to ask the same questions and to understand the same meanings of these questions. When we looked at non-AMS readmissions, the rate of receiving similar responses for the basal consciousness status of patients was between 77% and 98%.
A change in the RASS score between 0 and 1 points was seen in 99.9% of the T-AMS + non-AMS group of patients; only one person in that group had a greater RASS score change. Therefore, a change of ⩾2 points in the RASS score predicted Tr-AMS with a high specificity, and the change was diagnostic in about 44% of the patients with Tr-AMS. In addition, changes of ⩾2 points can be more easily recognized and expressed by the patient’s relatives.
Of the patients with Tr-AMS, 56% were unable to communicate due to a new condition or disease. The “new inability to communicate” status was highly specific and diagnostic for Tr-AMS. When five consciousness content items were evaluated in the available patients, newly developed disorganized thoughts and perception disorders were highly specific and diagnostic, and these findings were detected in 35% and 40% of patients, respectively. The newly developed inattention and disorientation were not diagnostic alone, but in combination, they were diagnostic with a high specificity in 66% of the patients. Five predictive factors (change in RASS =2 points, newly disorganized thoughts, new perception disorder, new inattention and disorientation combination, and newly unable to communicate) were able to catch 100% of the Tr-AMS patients in the study.
The Tr-AMS model we have developed could not be confirmed by regression analysis, because content of consciousness assessment was not evaluable in 55 patients (4.4%) due to newly developed inability to communicate. And also, these patients were half of the Tr-AMS patients. Therefore, when analyzing by logistic regression, half of the Tr-AMS patients had to be removed from the analysis. This situation caused the analysis to be inadequate.
Limitations
Although there are studies and standardized tests for determining the status of consciousness (content and/or level), there are no accepted studies for the determination of altered consciousness. For this reason, there is no gold standard for AMS diagnosis. This is the greatest limitation of this study.
Another issue is that it is necessary to determine the previous and final state of consciousness in order to say that a change in consciousness has occurred. When patients presented to the hospital, we had the chance to assess their final state of consciousness. However, we tried to assess each patient’s basal state with some simple, easy-to-answer questions to relatives who knew the patient best. So, we tried to determine the most appropriate factors after the pilot study. Although the RASS is a commonly used method for the level of consciousness, there are many components that constitute the content of consciousness. There are even many content evaluations in cognitive tests. However, to determine the basal condition of the patient, not all consciousness content items can be obtained easily and simply from the history of the patient.
Conclusion
AMS is a clinical condition with different definitions, variable manifestations, and a high mortality rate. We investigated the diagnostic success of various factors in predicting alterations in the consciousness content and level. We found that some of the factors we referred to as “Tr-AMS criteria” were highly specific, while others were not. Future investigations should be structured to include combinations of these factors and other clinical variables in the definition of AMS.
Footnotes
Appendix
Final diagnosis of patients with 3-month mortality in each category.
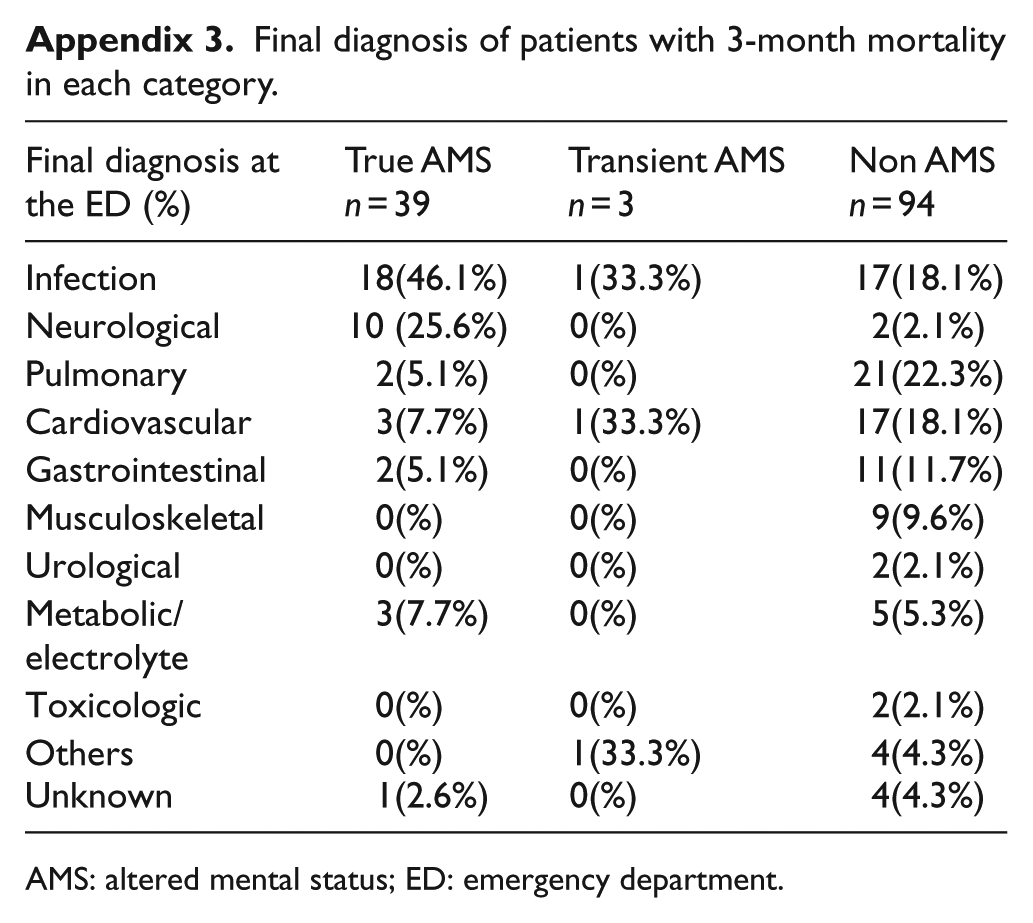
| Final diagnosis at the ED (%) | True AMS |
Transient AMS |
Non AMS |
|---|---|---|---|
| Infection | 18(46.1%) | 1(33.3%) | 17(18.1%) |
| Neurological | 10 (25.6%) | 0(%) | 2(2.1%) |
| Pulmonary | 2(5.1%) | 0(%) | 21(22.3%) |
| Cardiovascular | 3(7.7%) | 1(33.3%) | 17(18.1%) |
| Gastrointestinal | 2(5.1%) | 0(%) | 11(11.7%) |
| Musculoskeletal | 0(%) | 0(%) | 9(9.6%) |
| Urological | 0(%) | 0(%) | 2(2.1%) |
| Metabolic/electrolyte | 3(7.7%) | 0(%) | 5(5.3%) |
| Toxicologic | 0(%) | 0(%) | 2(2.1%) |
| Others | 0(%) | 1(33.3%) | 4(4.3%) |
| Unknown | 1(2.6%) | 0(%) | 4(4.3%) |
AMS: altered mental status; ED: emergency department.
Acknowledgements
M.A.A., N.B., N.Ö.D., and M.Z. participated in the design of the study; M.A.A., N.B., and M.Z. in data collection; M.A.A. in the literature search; and M.A.A. and N.Ö.D. in writing of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent and Ethical Approval
Ethics committee approval and informed consent from patients or their relatives were obtained.
