Abstract
Background:
Acute respiratory distress syndrome is regarded as a formidable clinical challenge due to its high prevalence and mortality. The treatment of acute respiratory distress syndrome is very complex and difficult. As an adjuvant therapy, the antioxidant N-acetylcysteine has been investigated for several years but the benefit is controversial.
Objectives:
We performed the systematic review and meta-analysis of randomized controlled trials to evaluate the efficacy of N-acetylcysteine on patients with acute respiratory distress syndrome.
Methods:
We searched PubMed, CENTRAL, and CBM databases. Randomized controlled trials comparing the effects of N-acetylcysteine and control were included. Overall mortality was the primary outcome; length of intensive care unit stay, duration of mechanical ventilation, glutathione levels, and PaO2/FiO2 were the secondary outcomes.
Results:
Eight trials with a total of 289 patients were included. Compared to the control group, the N-acetylcysteine group did not lower the overall mortality (risk ratio: 0.83; 95% confidence interval: 0.62 to 1.11; P = 0.21; I2 = 0%). However, N-acetylcysteine significantly shortened intensive care unit stay in the random-effects model (mean difference: –4.47 days; 95% confidence interval: –8.79 to −0.14; P = 0.04; I2 = 46%). Due to substantial heterogeneity and limited number of studies, the data of duration of mechanical ventilation, glutathione levels, and PaO2/FiO2 could not be pooled in the meta-analysis.
Conclusion:
N-acetylcysteine is ineffective in reducing mortality but beneficial for intensive care unit stay. Nonetheless, the effectiveness of N-acetylcysteine for acute respiratory distress syndrome is limited and further research is required before strong recommendations can be made.
Background
The acute respiratory distress syndrome (ARDS), first described in 1967, 1 is a form of non-cardiogenic pulmonary edema, due to lung’s alveolar-capillary membrane injury secondary to an inflammatory in response to pulmonary or extrapulmonary insults. 2 An American-European Consensus Conference (AECC) formulated the first agreed and widely cited definition of ARDS in 1994. 3 However, considering its reliability and validity about the diagnostic criteria, the European Society of Intensive Care Medicine engaged in a consensus process to develop the Berlin definition for ARDS in 2012. 4 Based on the degree of hypoxemia, ARDS is classified as mild (200 < PaO2/FiO2 (partial pressure of arterial oxygen to fraction of inspired oxygen) ⩽ 300 mmHg), moderate (100 < PaO2/FiO2 ⩽ 200 mmHg), or severe (PaO2/FiO2 ⩽ 100 mmHg). 4
ARDS continues to experience high prevalence and mortality, despite major improvements in supportive care. 5 The LUNGSAFE study 6 showed that the period prevalence of ARDS was 10.4% of intensive care unit (ICU) admissions and the mortality was approximately 40% in hospital. What is more, survivors have a prolonged ICU stay 7 and demonstrate significant muscle wasting, limiting weakness, and neuropsychiatric illness that degrade the ability to work after ICU discharge. 2
The therapy for ARDS mainly includes mechanical ventilation supports and adjuvant pharmacological interventions.8–10 The pathogenesis of ARDS was reviewed elsewhere, 11 providing several potential targets for pharmacological interventions. Although the pathophysiology of ARDS is complex and has not been fully illuminated, it has been proposed that oxidative stress is an important pathophysiological mechanism in acute impairment of lung function. 12 Glutathione system (glutathione/oxidized glutathione (GSH/GSSG)), the most abundant antioxidant in the lung, decreases significantly in lung inflammatory conditions. 13 Thus, as a precursor of GSH, N-acetylcysteine (NAC) can be replenished to limit oxidative lung injury by augmenting the intracellular pool of GSH. 14 NAC acts as a powerful oxygen free radical scavenger and also restores the oxidant/antioxidant balance, enhancing the endogenous antioxidant capacity. 15 NAC has been used clinically for chronic bronchitis, 16 idiopathic pulmonary fibrosis, 17 and hepatic injury induced by acetaminophen intoxication 18 historically. In spite of the theoretical advantages, the effects of NAC in critically ill patients remain controversial. 19 Furthermore, results on the administration of NAC in ARDS are still conflicting and some trials have not demonstrated its efficacy. 20 Recently, a meta-analysis including five randomized controlled trials (RCTs) demonstrated that NAC is beneficial for the duration of ICU stay but ineffective in reducing mortality. 21 However, an important RCT conducted by Jepsen et al. 22 which comprised 66 patients has not been included. What is more, that meta-analysis did not elaborate the NAC efficacy for ARDS in regard to the genotyping, disease severity, and antioxidative mechanisms.
In the context of the situation, we reviewed all RCTs of NAC in patients with ARDS and conducted a meta-analysis to evaluate the effects of NAC compared to control on clinical outcomes and try to illustrate the underlying mechanisms.
Methods
Search strategy
We systematically conducted electronic searches of PubMed, Cochrane Central Register of Controlled Trials (CENTRAL), and Chinese Biomedical Literature Database (CBM, in Chinese) from inception to June 2017. Search terms included “acetylcysteine,” “N-acetylcysteine,” “NAC,” “respiratory distress syndrome, adult,” “acute respiratory distress syndrome,” “respiratory distress syndrome,” “adult respiratory distress syndrome,” “ARDS,” “acute lung injury,” “lung injury,” “sepsis,” “septic shock,” “infectious shock,” “systemic inflammatory response syndrome,” “SIRS,” “acute respiratory failure,” “respiratory failure,” and “multiple organ failure.” In addition, we searched the reference list in the included studies. There was no language restriction.
Inclusion criteria
The characteristics of the included trials were as follows:
Population. Adult patients with ARDS or acute lung injury (ALI; using previous nomenclature, equal to mild ARDS) 4 were eligible for inclusion. Adults were defined based on the definition of the trials included.
Intervention. The intervention had to be intravenous NAC, regardless of the dose and administration time and formula.
Control. The control group had to receive placebo therapy or usual care.
Outcomes. Our primary outcome measure was overall mortality, defined as the longest follow-up data regardless of the period of follow-up. Secondary outcomes included the length of ICU stay, duration of mechanical ventilation, GSH levels, PaO2/FiO2. The GSH levels in the red blood cell (RBC), granulocyte, epithelial lining fluid (ELF), and plasma were analyzed.
Type of study. We only included RCTs.
Data extraction and quality assessment
We used a standardized data extraction form to collect data. The following phases were extracted from each study: (1) general information—first author and year of publication; (2) study characteristics – study design and clinical setting; (3) participants—diagnostic criteria and number of patients (NAC and control); (4) interventions—dose, administration time, and formulation; (5) outcomes—overall mortality, length of ICU stay, duration of mechanical ventilation, GSH levels, and PaO2/FiO2. Discrepancies in all phases were ultimately resolved through team consensus.
We assessed the risk of bias using the Cochrane Collaboration tool to assign a value of “high,” “low,” or “unclear” for every study. The following domains were reviewed: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other bias. If one or more domains were assessed as high risk of bias, the trial was considered as high risk. If all domains were assessed as low risk of bias, the trial was considered as low risk. Otherwise, the trial was considered to be at unclear risk.
We used the Grading of Recommendations Assessment, Development and Evaluation (GRADE) methodology to evaluate the quality of evidence.
Statistical analysis
We used more conservative random-effect models throughout the analysis since the clinical heterogeneity could not be excluded. The relative risk ratios (RRs) for dichotomous data were pooled using the Mantel–Haenszel (M-H) method. The mean difference (MD) for continuous data was analyzed using the inverse variance method. RR and MD were provided with 95% confidence intervals (CIs). The heterogeneity was assessed in accordance with the guidelines of the Cochrane Handbook for Systematic Reviews of Interventions. Substantial heterogeneity was suggested when I2 value was >60%, and then meta-analysis cannot be done. When the I2 value was ⩽40%, we consider little about statistical heterogeneity. The two-sided P values < 0.05 were considered to be statistically significant. All statistical analyses were performed on RevMan 5.3 software.
Results
We identified 922 records in the initial search. After assessment for eligibility, eight RCTs14,20,22–27 met the inclusion criteria and six RCTs were included in the meta-analysis. The study flow diagram is shown in Figure 1.
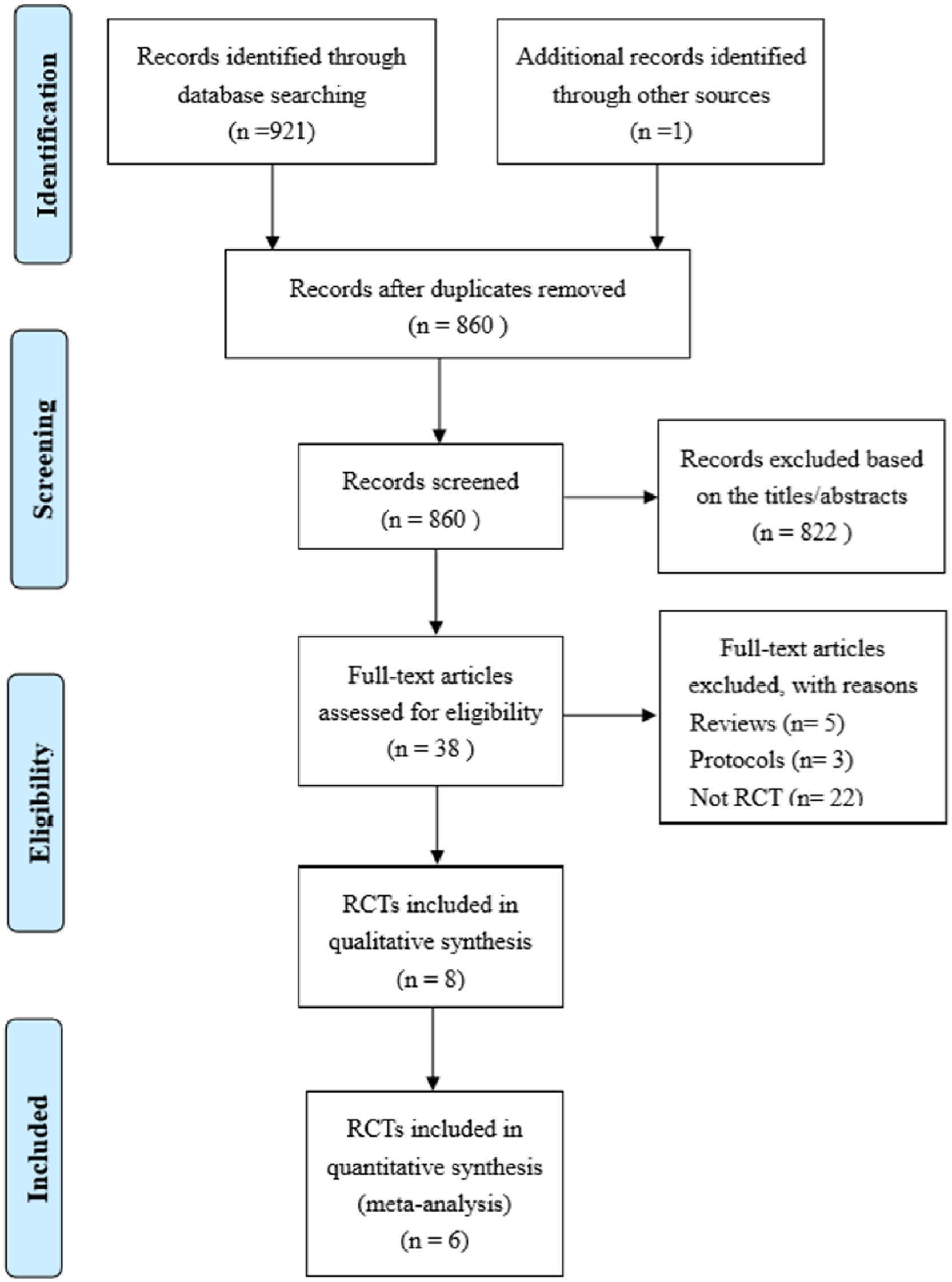
Flow diagram showing the results of the systematic database review and screening of articles, reasons of exclusion, and the number of articles included.
Study characteristics
The characteristics of the included studies are presented in Table 1. We included eight fully published studies in English including 300 patients, and 289 patients comprising 148 in the NAC group and 141 in the control group were analyzed finally. Sample size in each trial ranged from 16 to 66. All the trials were conducted in the ICU. All trials used intravenous NAC. There was a wide variety in the NAC dose used, which ranged from 40 to 190 mg/kg. In two trials,23,25 NAC was compared to rutin or L-2-oxothiazolidine-4-carboxylate (OTZ) and placebo, respectively; we only reported the data for the NAC and placebo groups from the two trials.
Characteristics of randomized controlled trials included in the systematic review.
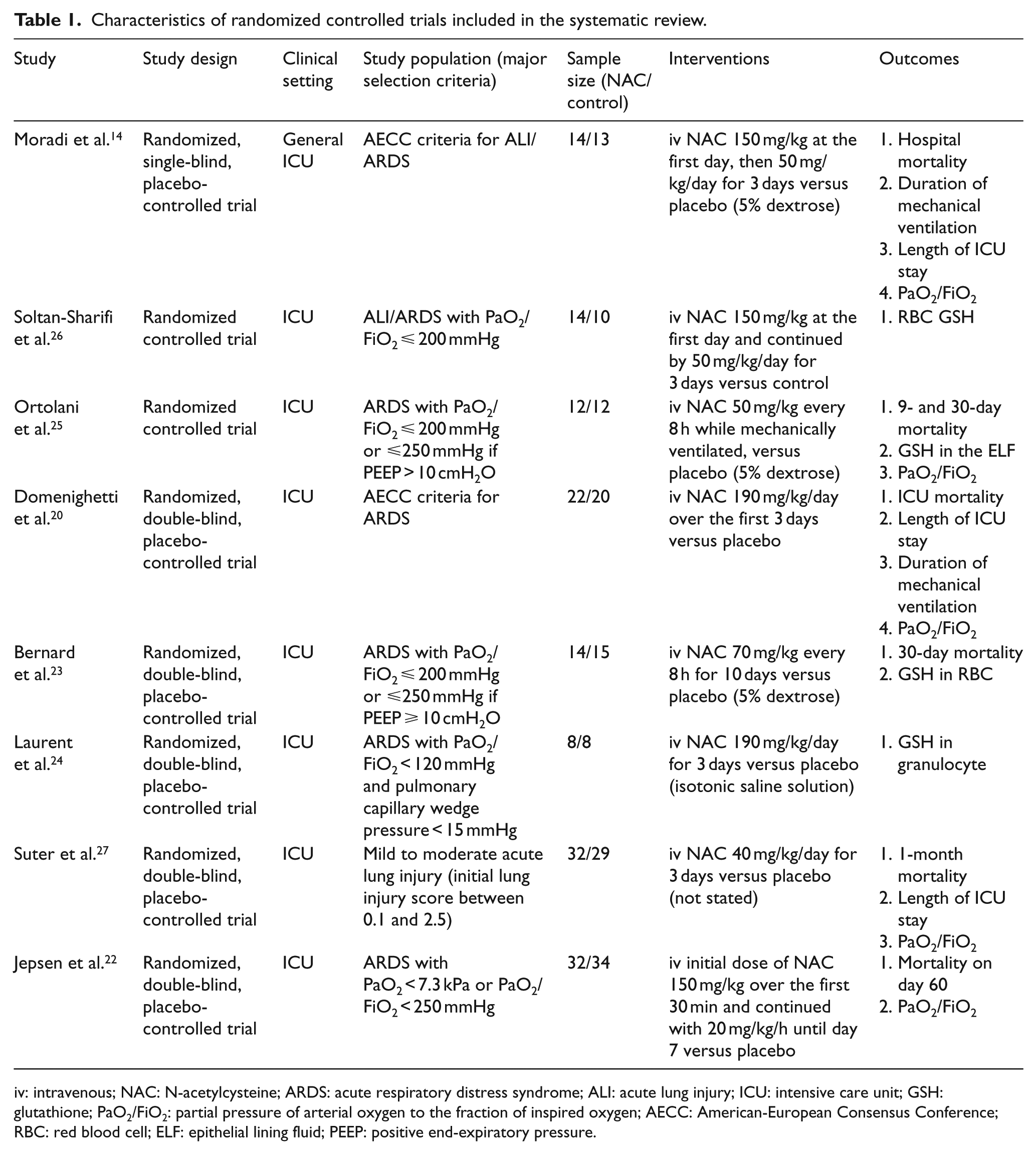
iv: intravenous; NAC: N-acetylcysteine; ARDS: acute respiratory distress syndrome; ALI: acute lung injury; ICU: intensive care unit; GSH: glutathione; PaO2/FiO2: partial pressure of arterial oxygen to the fraction of inspired oxygen; AECC: American-European Consensus Conference; RBC: red blood cell; ELF: epithelial lining fluid; PEEP: positive end-expiratory pressure.
Outcomes
Mortality data were available in six included trials.14,20,22,23,25,27 The overall mortality data in the NAC and control groups were 46 (36.2%) of 127 and 55 (43.3%) of 123, respectively. The pooled analysis showed no statistically significant effect of NAC (RR: 0.83; 95% CI: 0.62 to 1.11; P = 0.21; I2 = 0%; Figure 2).
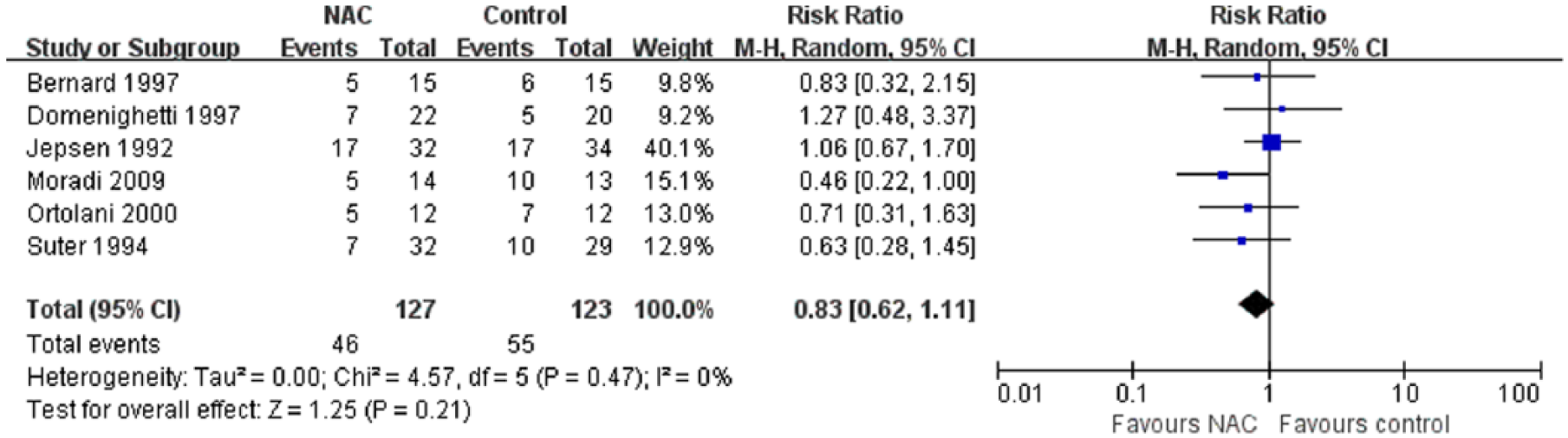
Forest plot of the effect of N-acetylcysteine (NAC) versus control on overall mortality.
Data on the length of ICU stay were available in three included trials.14,20,27 The pooled analysis revealed a statistically significant shortening of ICU stay in favor of NAC (MD: –4.47 days; 95% CI: –8.79 to −0.14; P = 0.04; I2 = 46%; Figure 3).
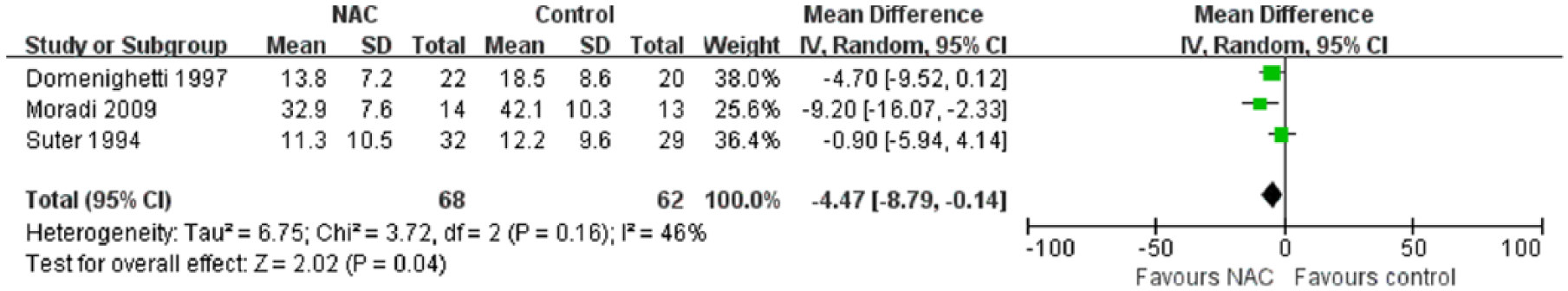
Forest plot of the effect of N-acetylcysteine (NAC) versus control on intensive care unit (ICU) stay.
The value of PaO2/FiO2 on day 3 was reported in three studies, but the data could not be pooled in the meta-analysis due to substantial heterogeneity (overall I2 statistic = 91%). One study 14 reported a statistically significant increase of PaO2/FiO2 on day 3 favoring NAC versus control (MD ± standard deviation (SD): 344.0 ± 38.3 vs 166.5 ± 119.0; P < 0.001). Another study 20 reported that the PaO2/FiO2 ratio increased significantly in both groups during the observation days, whereas the difference in PaO2/FiO2 between the two groups was never significant.
Duration of mechanical ventilation data were available in two studies, but the data could not be pooled due to substantial heterogeneity (overall I2 statistic = 65%). Both of the studies14,20 revealed no difference between the two groups.
The data of GSH levels could not be pooled in the meta-analysis because the determination of GSH content in the four included trials was different. But it was the same for the trend of increase of GSH level in RBC, granulocyte, or ELF by NAC treatment. The comparison of the NAC and control groups showed a significant increase in all four trials. Two studies23,26 reported that GSH in RBC had significant increases over the baseline values of 59% and 47%, respectively. Another study 25 reported that GSH increased 30% in the ELF of the NAC group compared with the baseline (P < 0.01). The rest 24 revealed that the total GSH in granulocyte was significantly higher in the NAC group during NAC or placebo treatment, when compared with the placebo group (P < 0.01). However, this difference was totally abolished after treatment interruption.
Study quality
We used the Cochrane Collaboration tool to evaluate the risk of bias of the included studies. The details of risk of bias were summarized in Appendix 1. One trial was classified as low risk of bias, 23 three trials were classified as unclear risk of bias,20,22,27 and four trials were classified as high risk of bias.14,24–26 Due to the limited number (below 10) of studies included in each analysis, publication bias was not assessed. We found that the grade of evidence was moderate for overall mortality and low for ICU stay, which was evaluated by GRADE methodology. The details of the results are shown in the summary of findings table (see Appendix 1).
Discussion
In this meta-analysis of eight RCTs with 289 ARDS patients, we found no benefits of NAC on overall mortality. The pooled analysis revealed a statistically significant shortening of ICU stay in favor of NAC. However, there were no differences in the duration of mechanical ventilation and oxygenation between the control and NAC groups.
In spite of these findings, the effect of NAC on ARDS still remains a matter of debate. We found that NAC cannot reduce the mortality in patients with ARDS, which was in agreement with previous reviews.15,21,28 Compared with the meta-analysis by Zhang et al., 21 three additional studies22,24,26 were added and the pooled results could provide more powerful evidence regarding the effects of NAC. What is more, we found that taking account of the genotyping of glutathione-S-transferase (GST) and disease severity, NAC may play an important role in ARDS treatment.14,27 NAC can provide a large amount of antioxidant GSH that may account for the efficacy partially. 26
The most recent study 14 in this meta-analysis showed a decrease in mortality and an improvement of oxygenation in the NAC group. Furthermore, the genotyping of GST might predict patients’ outcome of treatment, since NAC was effective and critical in patients with the genotypes of GST M1 null or GST M1 and T1 double null. 14 Likewise, Bernard 29 demonstrated that NAC exerted positive effects on oxygen delivery and oxygen consumption in a pilot study. In contrast to Moradi and Bernard’s findings, Domenighetti et al. 20 found that NAC made no difference to ICU mortality and systemic oxygenation. The GST genetic variations may account for the controversies, and the dose, administration time, severity of illness, and mortality endpoints were also the reasons. It has been suggested that NAC can decrease oxidative stress at high dose in patients with sepsis. 30
In terms of the duration of mechanical ventilation, both Moradi and Domenighetti demonstrated that there was no difference between the NAC and control groups in patients with established ARDS. The inconsistency between oxygenation and mechanical ventilation period may be due to complications, such as coma. 14 In another trial, Suter et al. 27 showed that early administration of NAC in patients with mild-to-moderate ALI (PaO2/FiO2 ⩽ 300 mmHg but ⩾200 mmHg) accelerated recovery with lower FiO2, lung injury scores, and less need for ventilator support than the placebo group. What is more, Suter et al. 27 demonstrated that the NAC group had the tendency to experience a shorter ICU stay, which was in accordance with the results of this meta-analysis. Taking the results into consideration, we speculated that early administration of NAC in less severe patients was beneficial. If the gas exchange abnormalities were too severe (PaO2/FiO2 ⩽ 200 mmHg), the efficacy may be overcome by disease severity. 20 Therefore, early recognition is essential for the therapy on ARDS.
Several studies have proven that oxidative injury plays a crucial role in the pathogenesis of ARDS 31 and patients with oxidant–antioxidant imbalance can get benefits when supplemented with NAC. 26 Bernard et al., 23 Laurent et al., 24 and Ortolani et al. 25 showed that the levels of the antioxidant GSH were decreased in RBC, granulocyte, plasma, and ELF in patients with ARDS and the depletion of GSH can be replenished with NAC.23,25 Furthermore, it has been suggested that NAC treatment may shorten the duration of ALI. 23 Besides, Soltan-Sharifi 26 found that treatment by NAC can successfully increase intracellular GSH and extracellular total antioxidant power and also enhance the outcome of patients. The antioxidant effect of NAC provided the theoretical basis for its efficacy on patients with ARDS.
Although NAC as an antioxidant for ARDS therapy was referred by reviews, 32 there was no recommendation grade since little evidence illustrated the benefits of NAC. However, NAC has been used clinically for over 40 years for chronic bronchitis, cystic fibrosis, and liver injury induced by acetaminophen intoxication. 15 As the recent meta-analysis showed that no adverse events were reported in all trials, which means that NAC was at least safe for use, 21 further research is required before definitive recommendations can be made regarding the benefits of NAC for ARDS. Thus, larger trials should await preliminary studies to identify GST genotypes, variations of dose, administration timing, and formulation that may be associated to clinical efficacy. Given that NAC’s effectiveness may be related to the severity of ARDS, patients can be divided into different groups according to the degree of severity to determine the efficacy of NAC.
Our meta-analysis has several strengths and limitations. A major strength of the analysis was conformity with the Cochrane handbook methodology and GRADE approach. And we followed the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) statement to report our meta-analysis. Besides, a thorough literature search was performed and Chinese articles also were reviewed. This is a systematic review to make a comprehensive evaluation about the effects of NAC on patients with ARDS. Nonetheless, there were several limitations to our meta-analysis. A major limitation was the high bias risk of the included studies, which may prevent a definitive conclusion. What is more, different diagnosis criteria of ARDS and formula of NAC administration and different mortality endpoints were used in each trial. All the factors should be considered when interpreting the results.
Conclusion
NAC is ineffective in reducing mortality but beneficial for ICU stay. Nonetheless, the effectiveness of NAC for ARDS is limited and further research is required before strong recommendations can be made.
Footnotes
Appendix 1
GRADE summary of findings.
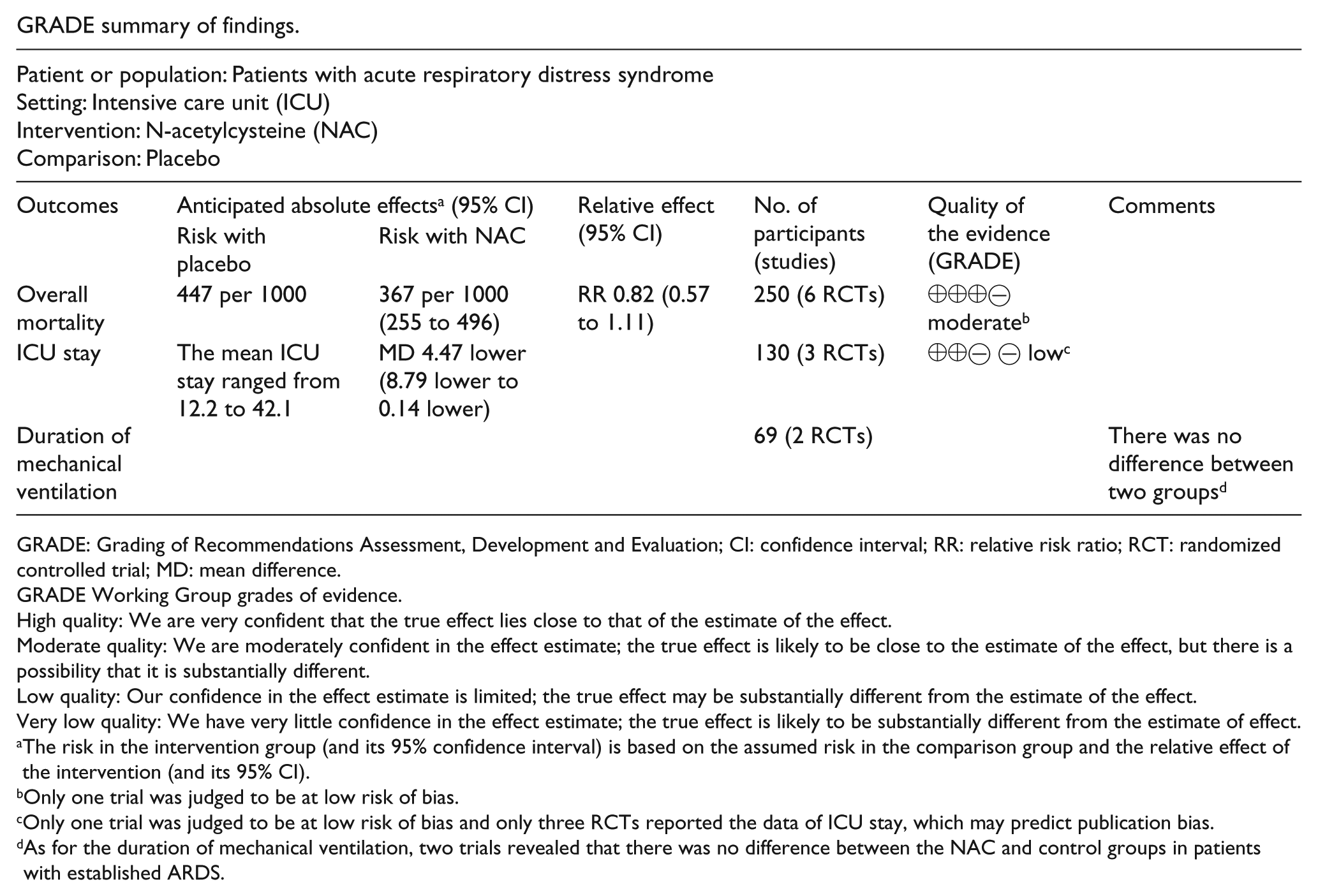
| Patient or population: Patients with acute respiratory distress syndrome |
||||||
|---|---|---|---|---|---|---|
| Outcomes | Anticipated absolute effects a (95% CI) | Relative effect (95% CI) | No. of participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Risk with placebo | Risk with NAC | |||||
| Overall mortality | 447 per 1000 | 367 per 1000 (255 to 496) | RR 0.82 (0.57 to 1.11) | 250 (6 RCTs) | ⊕⊕⊕⊝ moderate b | |
| ICU stay | The mean ICU stay ranged from 12.2 to 42.1 | MD 4.47 lower (8.79 lower to 0.14 lower) | 130 (3 RCTs) | ⊕⊕⊝ ⊝ low c | ||
| Duration of mechanical ventilation | 69 (2 RCTs) | There was no difference between two groups d | ||||
GRADE: Grading of Recommendations Assessment, Development and Evaluation; CI: confidence interval; RR: relative risk ratio; RCT: randomized controlled trial; MD: mean difference.
GRADE Working Group grades of evidence.
High quality: We are very confident that the true effect lies close to that of the estimate of the effect.
Moderate quality: We are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different.
Low quality: Our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect.
Very low quality: We have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of effect.
The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI).
Only one trial was judged to be at low risk of bias.
Only one trial was judged to be at low risk of bias and only three RCTs reported the data of ICU stay, which may predict publication bias.
As for the duration of mechanical ventilation, two trials revealed that there was no difference between the NAC and control groups in patients with established ARDS.
Acknowledgements
Drs Y.L. and X.L. set up the idea for writing the paper; Drs J.H. and Y.M. collected the data regarding the paper; Drs X.L., H.Z., and X.Y. analyzed the data; Drs Y.L. and X.L. wrote the original paper in English; and Drs Y.L. and X.Y. revised the paper, worked on the English, and prepared the final version of the manuscript. All authors reviewed the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the National Natural Science Foundation of China through Grant No. 81550034 to Y.L.
Availability of data and materials
All the data generated or analyzed during this study are included in this published article and its supplementary information files.
