Abstract
Background:
Cross-reactivity between penicillin and cephalosporin is of crucial concern among patients who have had a previous allergic reaction to penicillin, and cephalosporin is the first choice for their diseases. There is very little evidence concerning the prevalence of cross-reactivity in the Asian population.
Objectives:
This study aims to explore the cross-reactivity between penicillin and cephalosporin in the Asian population.
Methods:
This study included patients who were registered at Lampang Hospital from January 2011 to January 2018. Only patients who were prescribed penicillins and cephalosporins at least once as recorded in the electronic medical records (EMR), were included. Patients having penicillin and cephalosporin allergies were defined as those who were diagnosed in the EMR as allergic to drugs in the penicillin/ cephalosporin groups, in either the probable or definite categories with respect to the Naranjo’s algorithm. Cross-reactivity is defined as patients who were diagnosed as being allergic to both penicillin and cephalosporin. Data about characteristics and types of allergic reactions (type I & IV) were obtained from the EMR.
Results:
A total of 13,959 patients were included in the study. Of these, 99 penicillin-allergic and 13,860 non-penicillin-allergic patients were identified. The prevalence of cross-reactivity among patients with a history of penicillin allergy was 8.1%. Results showed high cross-reactivity rates among patients with type 1 (16.7%) and type 4 (13.3%) hypersensitivity, in relation to other allergic manifestations (3.4%).
Conclusions:
This study raises the possibility that cross-reactivity reported in previous research may differ from that in the Asian population. More Asian-based studies are needed to evaluate this cross-reaction so that this information can further assist medical professionals in clinical decision-making.
Introduction
Cross-reactivity between penicillin and cephalosporin is of crucial concern among patients who have had a previous allergic reaction to penicillin, and cephalosporin is the first choice for their diseases. There were studies in the 1960s and 1970s which reported high cross-reactivity rates of 8%–18%.1,2 They explained that this cross-reaction occurred because penicillins and cephalosporins both have a four-member β-lactam ring (BLR). Penicillins have a five-member thiazolidine (THIAZ) ring, while the cephalosporin structure contains a six-member dihydrothiazine (DHT) ring. R1 side chains may bind to the BLR, THIAZ, or DHT rings, and they are the main causes of cross-reaction between penicillin and cephalosporin. 3
Hence, physicians who are confronted with these patients have to make a decision and weigh up risks and benefits. Owing to the fear of cross-reactivity, some doctors choose another antibiotic that doesn’t have a BLR, such as quinolones or macrolides. However, as cephalosporins are among the first-line treatments in some diseases,4,5 dropping this group of drugs from the choice might cause some adverse effects such as reduced effectiveness, increased antimicrobial resistance, or higher costs.2,6
However, evidence from a literature review has demonstrated that the incidence of cross-reaction is approximately 1%, and not as high as previously reported. 7 This high rate of cross-reaction was explained by the contamination of the preservative. 7
Moreover, the prevalence of cross-reaction may depend on the generations of cephalosporin:3,7 the prevalence of cross-reaction between penicillin and first-generation cephalosporins is 10.9%, while only 1.1% is reported in third-generation cephalosporins. This reactivity was explained by the presence of the R1 side chain. This side chain exists among cephalexin and cefaclor, and these first- and second-generation cephalosporins have an α-amino side chain as found in aminopenicillins (ampicillin and amoxicillin). This R1 side chain is also present in the second-generation cephalosporin cefuroxime, cefixime, and the third-generation ceftibuten, cefotaxime, and ceftriaxone. 7
In addition, recent literature has pointed out that the occurrence of cross-reaction may be associated with some types of allergic reaction such as immediate hypersensitivity (or type I) and delayed-type (or type IV) hypersensitivity. 3 A study found the cross-reactivity to be as high as 27.14% among those with penicillin-allergic manifestations of immediate hypersensitivity, in comparison with only 14.57% among those with other allergic presentations to penicillins. 8
Despite some existing studies about penicillin and cephalosporin cross-reactivity, there remain differences in terms of study designs used and small number of samples. Moreover, all studies were based on the Caucasian population.
There is very little evidence concerning the prevalence of cross-reactivity in the Asian population. This information would benefit doctors by assisting their decision-making about whether cephalosporins could be prescribed with a low risk in penicillin-allergic patients.
Method
This study included patients who were registered at Lampang Hospital, Thailand, from January 2011 to January 2018. Only patients who were prescribed penicillins and cephalosporins at least once as recorded in the electronic medical records (EMR) were included.
Drug codes of penicillin and cephalosporin were used to identify the samples of interest. Drugs in the penicillin group include amoxicillin, amoxicillin with clavulanic acid, ampicillin, benzathine benzylpenicillin, dicloxacillin, penicillin, piperacillin with tazobactam, and piperacillin.
The group of cephalosporins included in this study was cephalexin, cefazolin, cefuroxime, cefdinir, cefixime, cefoperazone with sulbactam, cefotaxime, ceftazidime, ceftriaxone, and cefditoren.
Lampang Hospital records the history of drug allergy connected to the drug code in the EMR. This includes penicillins and cephalosporins. Therefore, the history of drug allergy was retrieved from the EMR alongside the drug code. Data about characteristics and types of allergic reactions (type I and IV), which were diagnosed by clinicians using the International Classification of Diseases (ICD) 10 codes, 9 were also obtained from the EMR.
Diagnosis of drug allergy conforms to the standard of Naranjo’s algorithm. 10 Information about the characteristics of the manifestations of allergic reactions to penicillins was also retrieved from the EMR.
Patients having penicillin and cephalosporin allergies were defined as those who were diagnosed in the EMR as allergic to drugs in the penicillin/cephalosporin groups, in either the probable or definite categories with respect to Naranjo’s algorithm. 10 Cross-reactivity is defined as patients who were diagnosed as being allergic to both penicillin and cephalosporin.
The prevalence of cross-reactivity was calculated by the number of patients with cross-reactivity divided by the number of penicillin-allergic cases. The prevalence of cephalosporin allergy in non-penicillin-allergic patients was also approximated and compared with those with a history of penicillin allergy.
Subgroup analyses were performed with respect to the penicillin-allergic manifestation. A chi-square test, Fisher’s exact test, and
Results
A total of 13,959 patients were included in the study. Of these, 99 penicillin-allergic and 13,860 non-penicillin-allergic patients were identified, as illustrated in Figure 1.
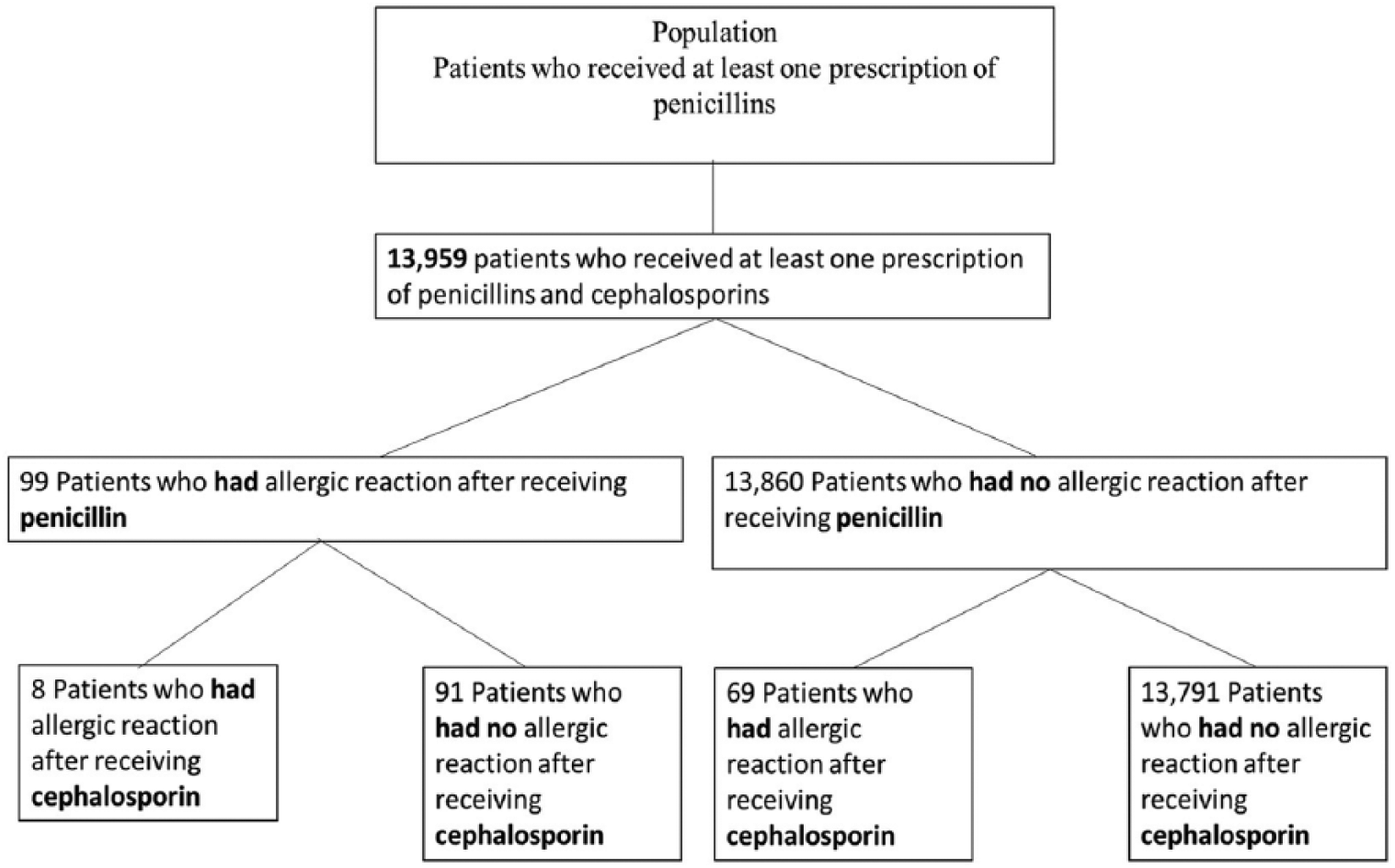
Study protocol.
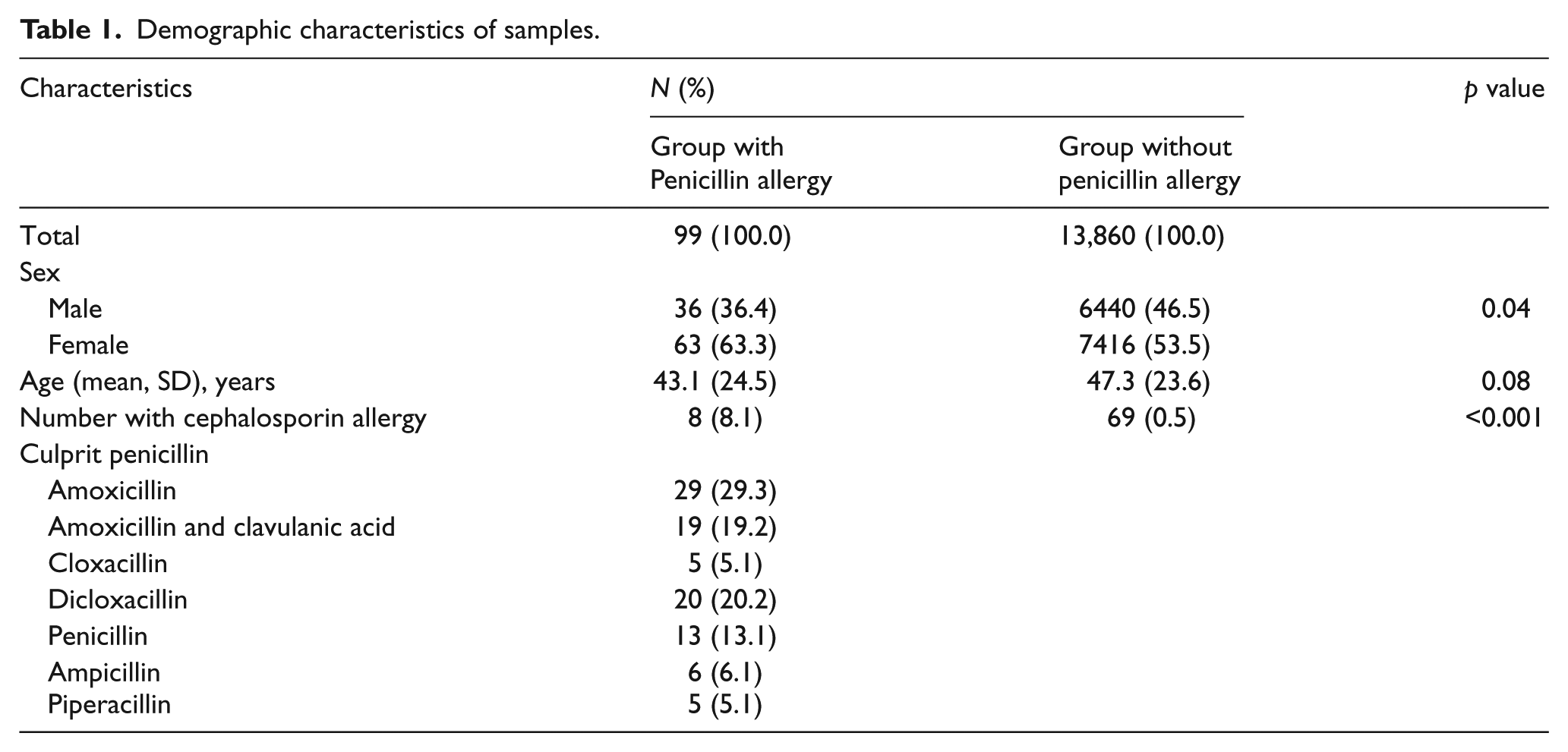
The prevalence of cross-reactivity among patients with a history of penicillin allergy was 8.1%, which is higher than the prevalence of cephalosporin allergy among those without a history of penicillin allergy, 0.5%. The most prevalent penicillins prescribed were amoxicillin (29.3%), followed by dicloxacillin (20.2%), as can be seen in Table 1.
Demographic characteristics of samples.
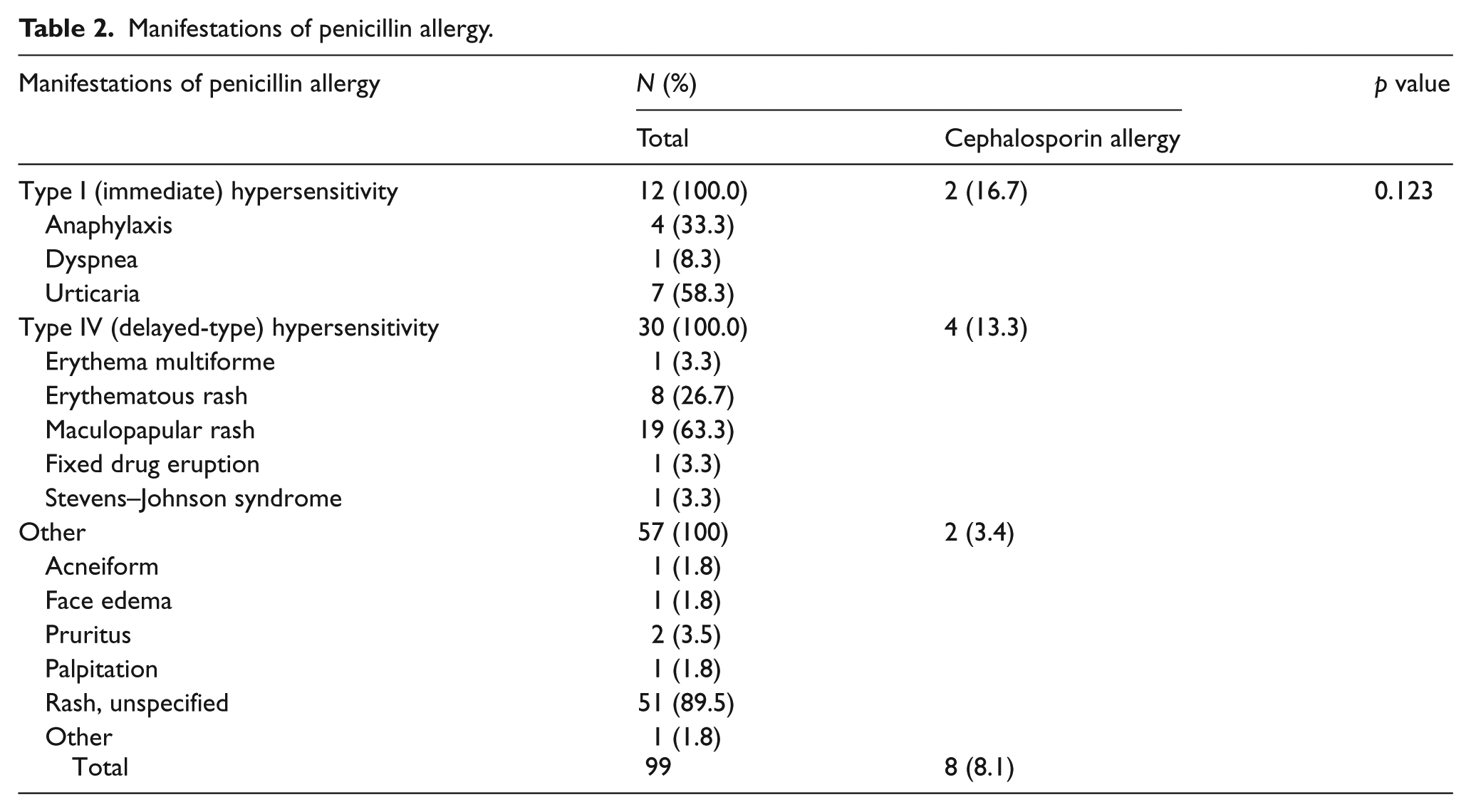
Among 99 penicillin-allergic patients, type I (immediate) hypersensitivity was found in 12, 30 patients presented the delayed hypersensitivity type, and 57 in other forms. While the most common allergic manifestation is an unspecific rash, anaphylaxis was found in four patients. Results showed high cross-reactivity rates among patients with type I (16.7%) and type IV (13.3%) hypersensitivity, compared with other allergic manifestations (3.4%); however, no statistically significant differences were found, as can be seen in Table 2.
Manifestations of penicillin allergy.
Discussion
As all previously published research pertaining to cross-reaction between penicillins and cephalosporins has been conducted in western countries, the authors regard this study as the first attempt to report about cross-reactivity in the Asian population. Results from this study showed a surprisingly high cross-reactivity rate of 8.1%, compared with a low prevalence of cephalosporin allergy in patients without penicillin allergy of only 0.5%. This is much higher than the rate of approximately 1% reported in a recent literature review 7 of the cross-reactivity of cephalosporins in penicillin-allergic patients. The cross-reactivity rate from this study should alarm health scientists and prompt them to investigate further the prevalence of and factors influencing the cross-reactivity among the Asian population.
Regarding patients with immediate-type hypersensitivity, this study revealed a cross-reactivity of 16.7%, higher than that in other allergic presentations. These findings resonate with the results from a previous study, which identified immediate-type hypersensitivity as a high risk for cross-reactivity. This information supports the association between this specific type of reaction and an increased risk of cross-reactivity, which results in the administration of cephalosporin not being recommended in patients with this type of allergic presentation. 12 , 13 Hence, the elevated risk of cross-reactivity should be brought to the attention of both physicians and patients, with the aim of preventing possible serious and undesirable health effects.
Results of this study also demonstrated an unexpectedly high rate of cross-reactivity among the delayed-type hypersensitivity of 13.3%, which is consistent with the findings from an existing publication. 3 Confirmation of this high possibility of cross-reaction suggests careful consideration when using cephalosporins in penicillin-allergic patients who present with delayed-type reaction to avoid severe adverse consequences from drug allergy.
This study demonstrated a low cross-reactivity of 3.4% for other allergic manifestations apart from type I and IV hypersensitivity. This prevalence echoed the low probability of allergy when using cephalosporin that was presented in a review, 7 with a recommendation that cephalosporin can be used with these other allergic manifestations. Therefore, results of this study confirm that cephalosporin could be available as a treatment choice in such patients. It is noted that once the decision to use cephalosporin is made in these patients, despite the low prevalence of cross-reaction, prompt management for severe allergic reactions still needs to be available to ensure the patients’ safety. 14
Existing evidence has pointed out a high risk of cross-reactivity between particular drugs in the penicillin and cephalosporin groups, because of the same R1 side chain in each group. 7 For example, patients who are allergic to amoxicillin and ampicillin may experience an increased probability of cross-reaction to cephalexin, cefaclor, and cefadroxil. However, exploring this association is beyond the scope of this study. Future research could incorporate these elements into the analysis to examine these associations.
Research to identify predictive factors of cross-reactivity is still of interest because there is currently no unanimously agreed method for predicting the occurrence of cross-reaction. Some evidence has suggested the use of patch and prick tests as a worthwhile screening tool when the use of cephalosporin is considered.3,15 However, doing so was not recommended by recent studies due to their low sensitivity and specificity. 3 On one hand, negative tests do not always mean patients can take both penicillin and cephalosporin safely. On the other hand, patients would unnecessarily lose the opportunity to use cephalosporin if false positive results occur. Information about the diagnostic accuracy of each method to predict cross-reactivity would thus be required by clinicians to incorporate into clinical decision-making.
A limitation of this study pertains to the data source, which came from only one provincial hospital. This potentially affects the external validity as the prevalence of cross-reaction might vary across different types of patients. In addition, results of this study may also contain uncertainty due to the small sample size. Future research, using national data from different levels of healthcare settings, is recommended to fill this gap.
An issue worth noting concerns the accuracy of the allergy diagnosis as, in reality, physicians can seldom witness the allergic presentation themselves. Diagnosis of allergy was thus mostly based on self-reported information, which may impact the diagnosis validity, including drug misidentification and misclassification of the allergic reaction type. This potential uncertainty is acknowledged as a limitation of the study.
While most arguments about allergic cross-reactivity are made to facilitate decision-making by physicians, very little attention is paid to the allergic patients who must suffer all the risks of drug allergy. Concerning this, the concept of patient-centered care 16 could play a role in helping to prevent the risks of cross-reactivity by encouraging the penicillin-allergic patients to be informed about their conditions and types of medicines that they should be aware of. Moreover, when considering the administration of cephalosporin in such patients, a shared decision between doctors and patients should be undertaken, with information about the risks of cross-reactivity being provided. Doing so would not only promote patient rights when embarking on medical treatment, but this may also help to resolve the disagreement about the existence of allergic cross-reaction.
While most currently available research concerning cross-reactivity between penicillin and cephalosporin has been conducted in Caucasians, results from this study raise the possibility that cross-reactivity reported in previous research may differ from that in the Asian population. To ascertain the prevalence of and factors affecting the cross-reactivity rate in Asian people, more Asian-based studies are needed so that this information can further assist medical professionals in clinical decision-making.
Footnotes
Acknowledgements
The authors thank Andrew Sherratt for comments on the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of data and materials
This study used the data from electronic medical records at Lampang Hospital, Thailand.
Informed consent
Written informed consent was not necessary because no patient data have been included in the manuscript.
Ethical approval
This research was approved by the Ethics Committee at Lampang Hospital.
Human rights
Not applicable, as this study used the secondary data.
