Abstract
Background:
Gastrointestinal bleeding is a commonly seen multidisciplinary clinical condition in emergency departments which has high treatment cost and mortality in company with hospital admission. Risk evaluation before endoscopy is based on clinical and laboratory findings at patient’s emergency visit.
Objective:
The purpose of this study is to investigate the efficacy of “Glasgow-Blatchford scale + lactate levels” to predict the mortality of patients detected with gastrointestinal bleeding in the emergency department.
Methods:
A total of 107 patients with preliminary diagnosis of upper gastrointestinal bleeding included in the study after approval of the ethics committee were prospectively evaluated. Glasgow-Blatchford scale scores were calculated and venous blood lactate levels were assessed. Need for blood transfusion in the follow-up, the amount of transfusion, and mortality in the next 6 months were evaluated.
Results:
A statistically significant difference was found in mortality rates between the lactate and Glasgow-Blatchford scale cohorts in our study (p = 0.001 and p < 0.01, respectively). The mortality rate was significantly higher in the lactate(+) GBS(+) cases compared to the lactate(–) GBS(+), lactate(+) GBS(–), and lactate(–) GBS(–) cases compared to the bilateral comparisons (p = 0.004, p = 0.001, p = 0.001, and p < 0.01, respectively). There was a statistically significant relationship between the rate of erythrocyte suspension replacement in the cases according to Glasgow-Blatchford scale levels (p = 0.001 and p < 0.01, respectively). The incidence of erythrocyte suspension replacement was 7.393 times greater in patients with Glasgow-Blatchford scale score of 12 and above.
Conclusion:
Glasgow-Blatchford scale is highly sensitive to the determination of mortality risk and the need for blood transfusion in upper gastrointestinal bleeding. Glasgow-Blatchford scale with lactate evaluation is more sensitive and more significant than Glasgow-Blatchford scale alone. This significance provides us to establish “modified Glasgow-Blatchford scale.” In the future, studies which will use Glasgow-Blatchford scale supported by lactate could be increased and the results should be supported more.
Keywords
Introduction
Gastrointestinal bleeding (GIB) is a commonly seen multidisciplinary clinical condition in emergency departments (EDs) which incurs high treatment cost and mortality together with hospital admission. Upper GIB is a common medical emergency with significant mortality and morbidity rates. The annual incidence of upper GIB is 5–10 out of 10,000 and the mortality rate ranges between 6% and 10%. 1
Lactic acidosis and base excess are the valuable parameters that indicate tissue perfusion disorders beforetime and used for sepsis follow-up. Lactic acidosis together with GIB can cause poor clinical outcomes. An increase in lactate levels is caused by hypovolemia due to bleeding or dehydration, severe anemia, septic shock, and severe hypoxemia.
There are various risk scoring methods to evaluate upper GIB. Risk evaluation before endoscopy is based on clinical and laboratory findings at patient’s emergency visit. Fast evaluation of these parameters is an advantage to managing the situation and provides results. In emergency settings, patients were classified using the Glasgow-Blatchford scale (GBS) by risk and ease to identify high/low-risk patients. For earlier determination of the need for blood transfusion, recurrent bleeding risk, and mortality, patients should be evaluated for blood urea nitrogen (BUN), heart rate, blood pressure, existence of syncope, melena, chronic liver disease, or heart failure at the initial ED visit (Table 1). 2 Regarding GBS, a score of 0 indicates that patients can be treated as outpatients, and the score of 6 or higher indicates that the patients have 50% chance to need medical intervention.
Glasgow-Blatchford scoring system.
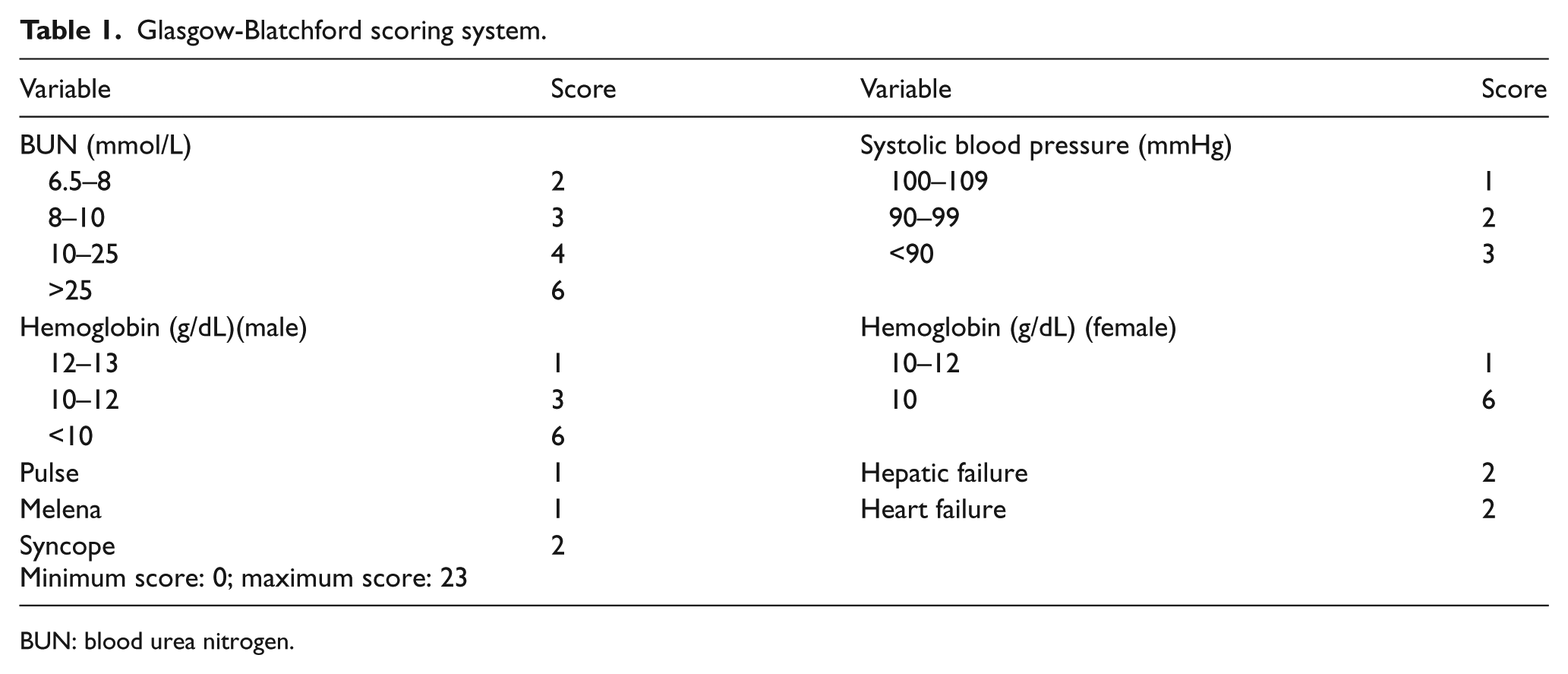
BUN: blood urea nitrogen.
The purpose of this study is to investigate the efficacy of “GBS + lactate levels” to predict the mortality of patients detected with GIB in the ED.
Methods
Our study was conducted prospectively between 1 July 2015 and 31 June 2016 at Kartal Dr Lutfi Kirdar Training and Research Hospital with approval of the scientific research evaluation committee. All patients above the age of 18 with early diagnosis of GIB are included. Patients who had diagnosed with esophageal variceal bleeding, malignancies, pregnancy, or metformin usage were excluded from the study. Written informed consent was obtained from the patients for their anonymized information to be published in this article.
Medical histories were taken from patients who were asymptomatic but suspected for upper GIB and those with complaints like fatigue, dizziness, syncope, black stool, and vomiting blood. Initial heart rate and blood pressure were measured. A nasogastric tube was placed, the gastric content was obtained, and rectal examination was performed in ED. History of liver or heart failure was questioned. According to the obtained data and physical examination, GBS score for each patient was calculated. Complete blood count, renal and liver function, and lactate levels in venous blood samples were measured. Need for blood transfusion in the follow-up, the amount of transfusion, and mortality in the next 6 months were evaluated. All the obtained data were recorded on study data forms during the study (Figure 1).
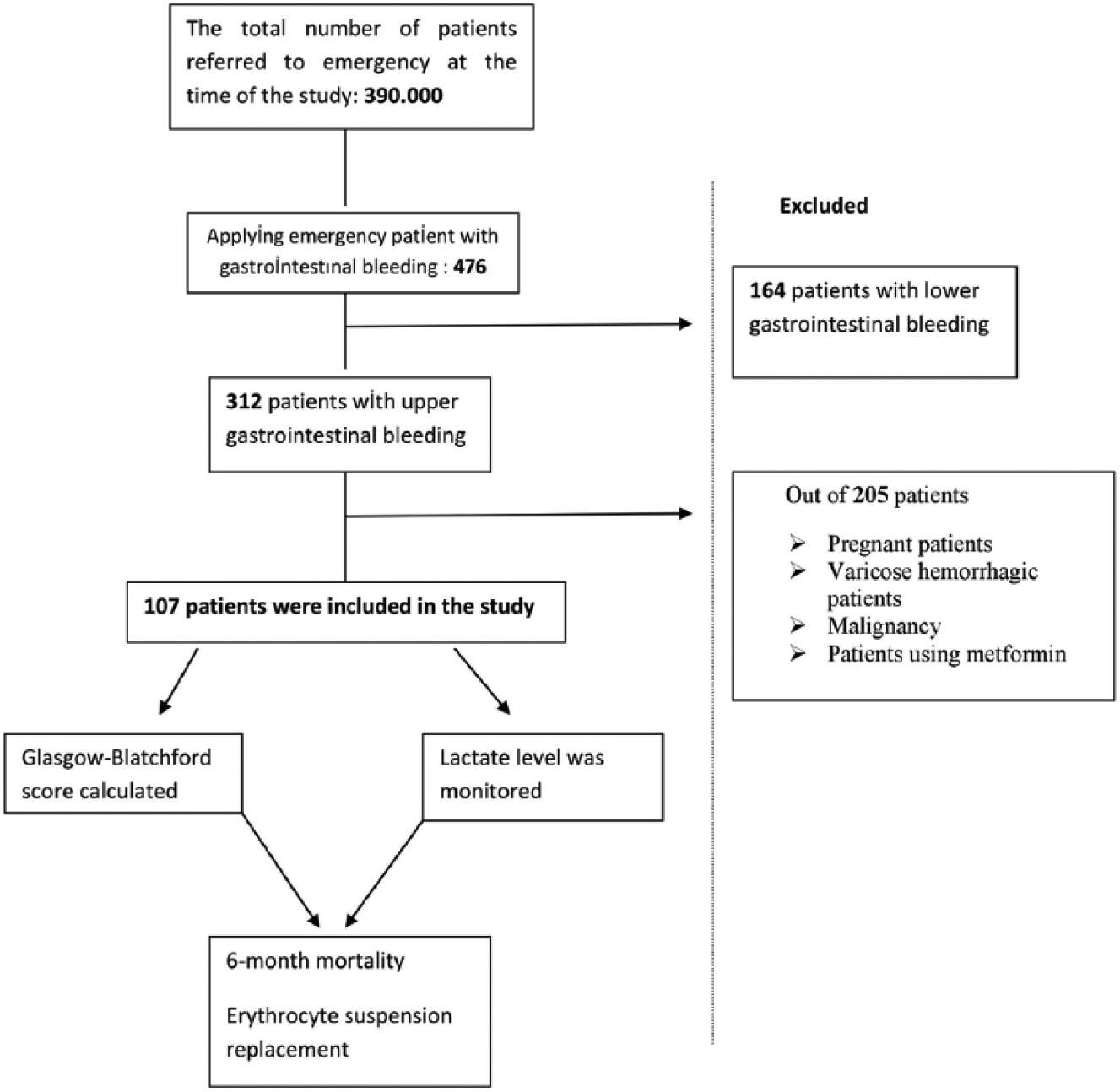
Flow-chart of the study.
Statistics
Statistical analyses of the study were performed with NCSS (Number Cruncher Statistical System) 2007 (Kaysville, UT, USA) software. While assessing the study data, definitive statistical methods such as mean, standard deviation, median, frequency, ratio, minimum, and maximum were calculated, and the comparison of the two data groups of abnormal distributions of quantitative data was performed with Mann–Whitney U test. Comparison of qualitative data was performed with Pearson Chi-square test. Cut-off value was determined with receiver operating characteristic (ROC) curve analysis and classification functions (sensitivity and specificity) were used. Level of significance was set and evaluated at p < 0.01 and p < 0.05, respectively.
Results
The study was conducted with a total of 107 patients who were eligible for study and 71.0% (n = 76) were male. Mean age was 57.42 ± 18.76 years and ranged between 20 and 92.
Vomiting blood was identified as the most common complaint and dizziness was the rarest. In all cases, while 5.6% (n = 6) of them were discharged, 84.1% (n = 90) were admitted to the internal medicine ward, 8.4% (n = 9) to intensive care unit (ICU), and 1.9% (n = 2) to general surgery ward.
The measured lactate levels ranged between 4 and 71 mg/dL with a mean value of 18.26 ± 12.58 mg/dL. In all patients, 8.4% (n = 9) had syncope, 99.1% (n = 106) had BUN elevation, 83.2% (n = 89) had hemoglobin decrease, 8.4% (n = 9) had liver failure, and 15.9% (n = 17) had heart failure. Based on this information, GBS score ranged between 1 and 19, and the mean GBS score was 10.59 ± 3.80 (Table 2).
Distribution of descriptive properties.
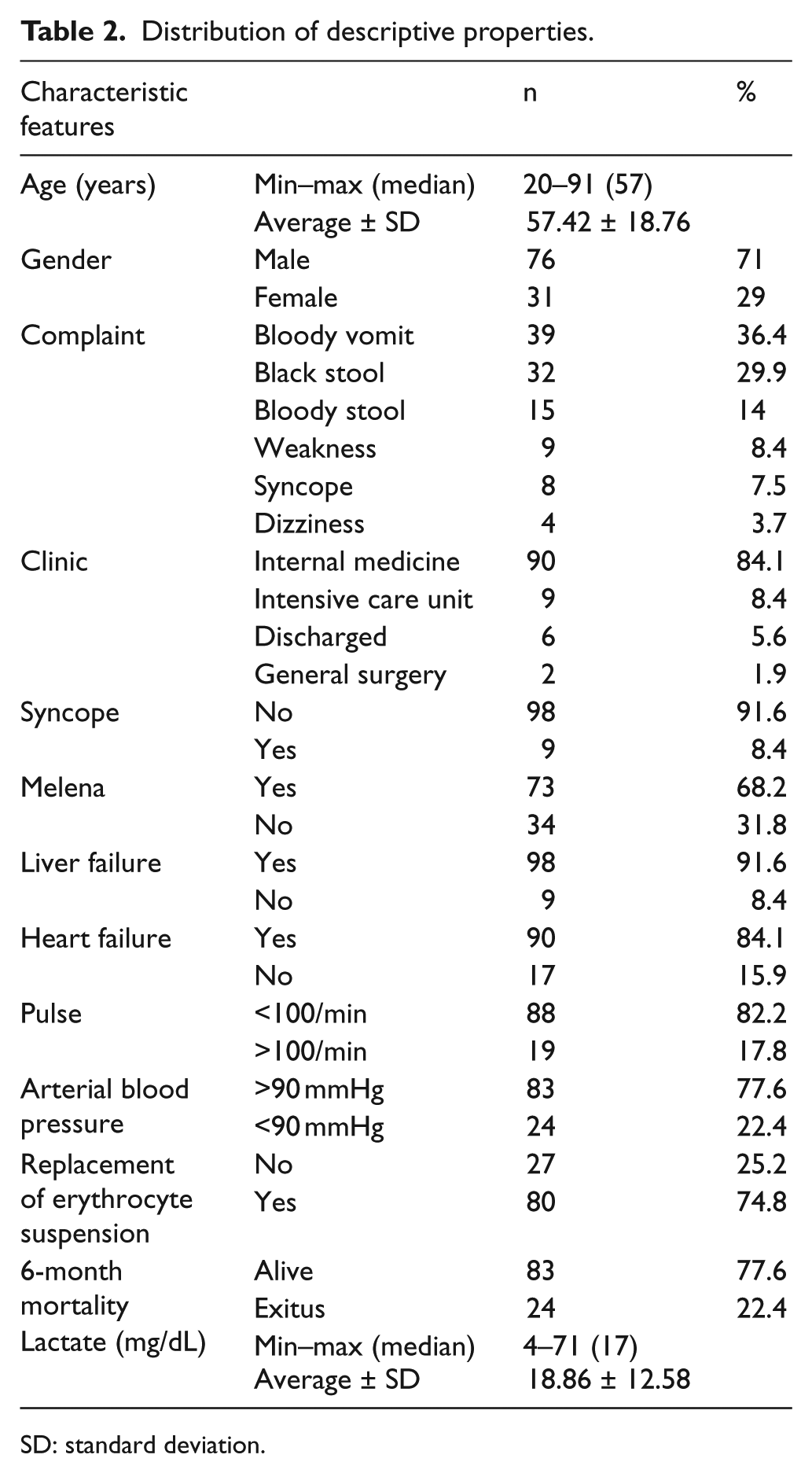
SD: standard deviation.
Evaluations regarding mortality
The mean GBS scores for patients with and without mortality were 13.04 ± 2.63 and 9.88 ± 3.80, respectively. In GBS score regarding mortality, statistically significant difference was identified (p = 0.001 and p < 0.01); GBS scores were determined to be significantly higher in patients with mortality (Figure 2).
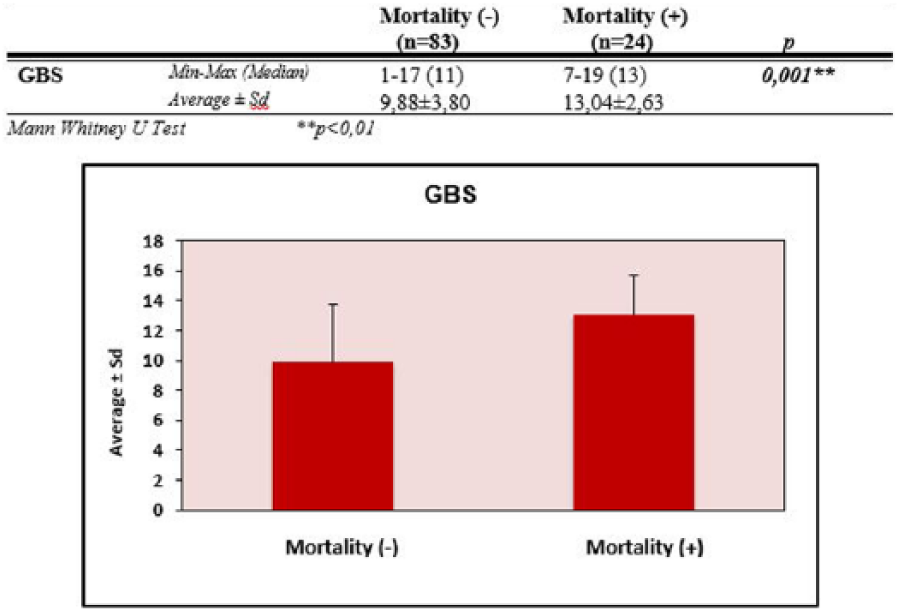
Distribution of GBS measurements by mortality.
Determining cut-off value for GBS score regarding mortality
Cut-off value was determined by ROC curve analysis. According to the presence of mortality, the GBS score cut-off value was determined as 12 and above. For this cut-off value, sensitivity was 70.83%, specificity was 61.45%, positive predictive value was 34%, and negative predictive value was 87.93%. Area under the obtained ROC curve was 74.0% and standard error was 5.2% (Figure 3).
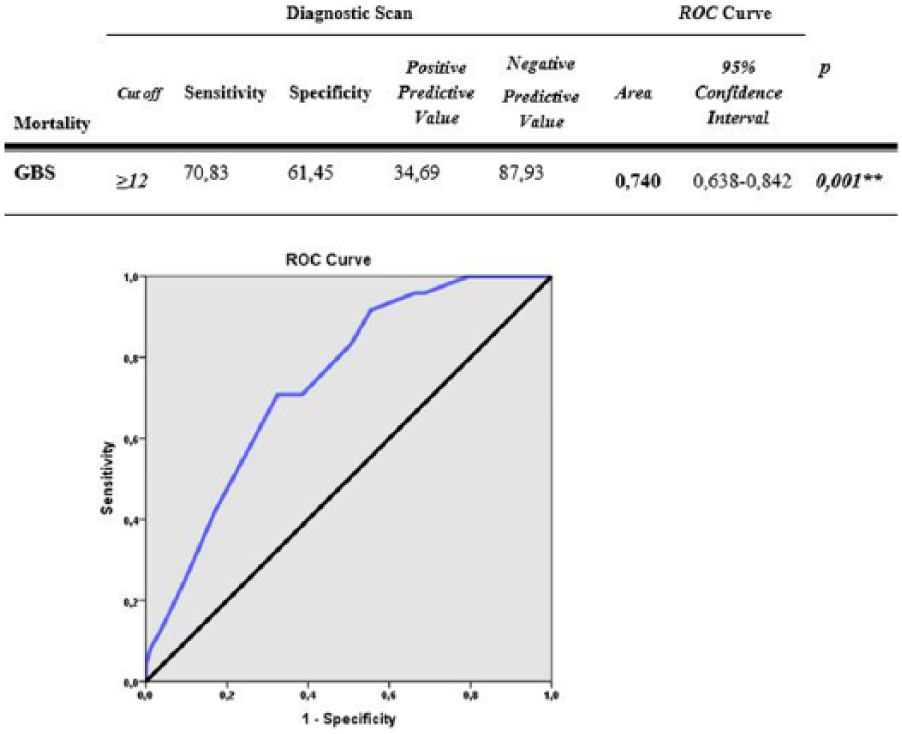
Diagnostic tests and ROC curve results by mortality. **p < 0.01.
A statistically significant relation was identified between a GBS score cut-off value of 12 and mortality (p = 0.001 and p < 0.01, respectively). Patients who had a GBS score of 12 or higher had 3.871 times more mortality risk compared to those with a GBS score lower than 12. Odds ratio for GBS was 3.871 (95% confidence interval (CI): 1.445–10.364; Figure 4).
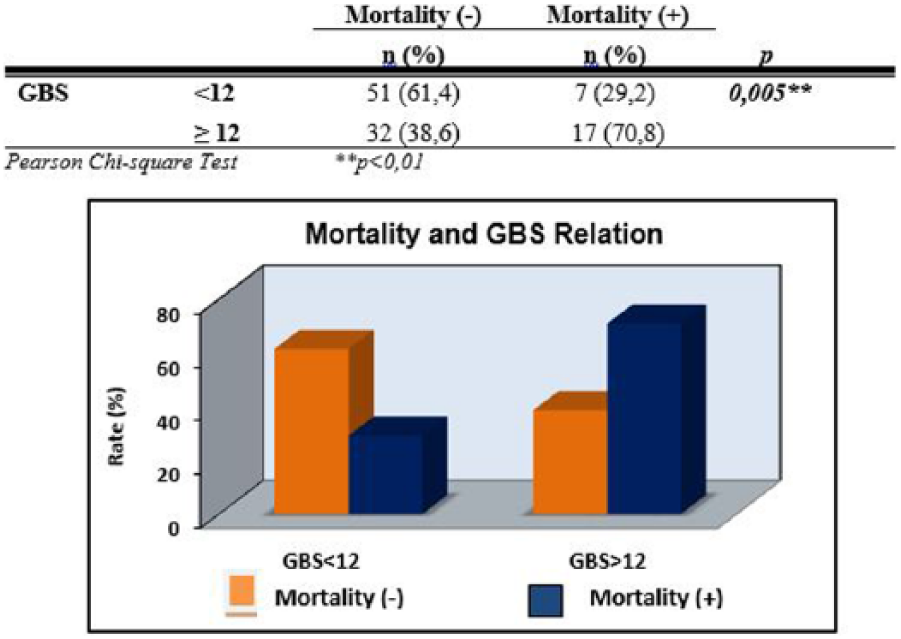
Relationship between mortality and GBS. **p < 0.01.
Mean lactate levels of patients with and without mortality were 25.79 ± 20.10 and 16.85 ± 5.56 mg/dL, respectively. Although there was not any statistically significant difference in lactate levels and regarding mortality close to significant difference was determined (p = 0.083 and p > 0.05, respectively), but elevation of lactate levels in patients with mortality was remarkable (Figure 5).
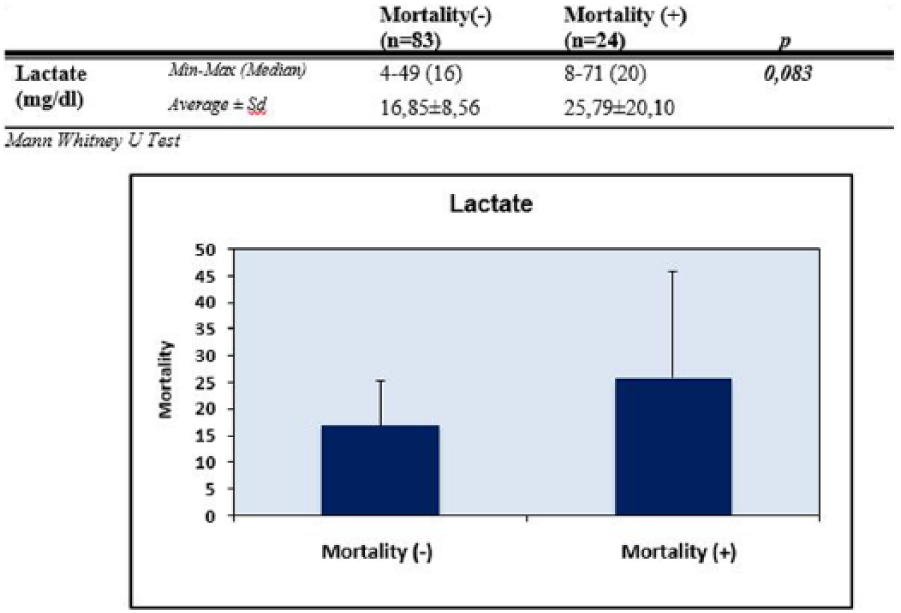
Distribution of lactate measurements by mortality.
According to the presence of mortality, the lactate level cut-off value was determined as 23 and above. For this cut-off value, sensitivity was 37.50%, specificity was 80.72%, positive predictive value was 36.0%, and negative predictive value was 81.71%. Area under the obtained ROC curve was 61.6% and standard error was 6.9% (Figure 6).
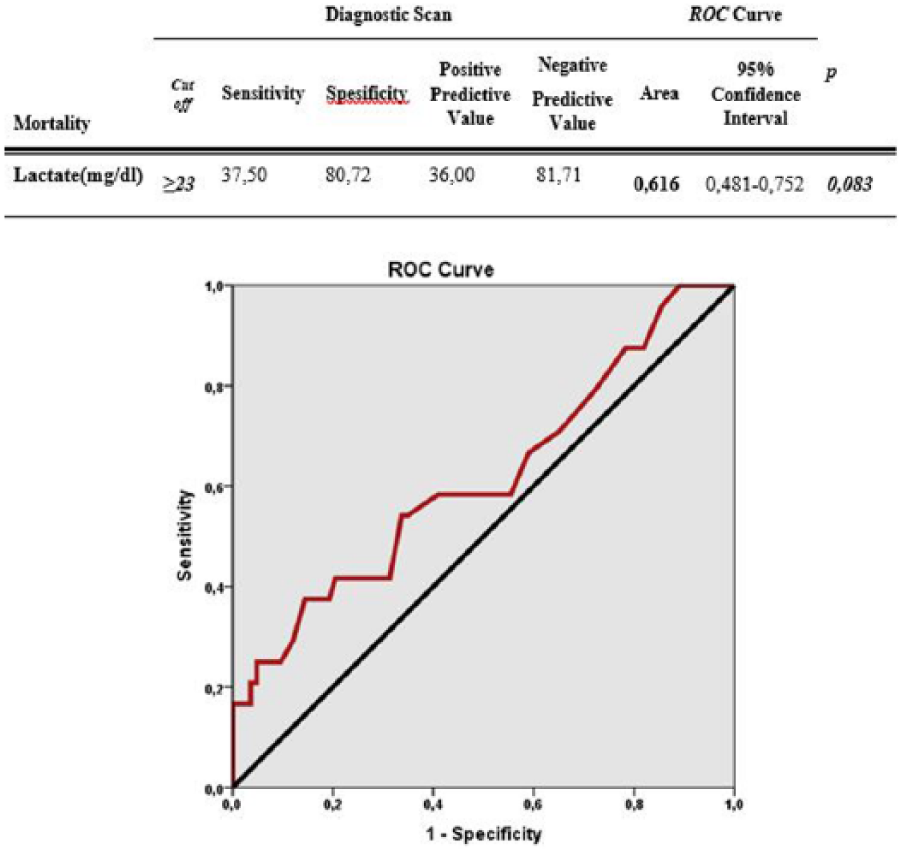
Diagnostic screening tests and ROC curve results for lactate measurements by mortality.
Evaluations regarding blood transfusion
A statistically significant difference was identified between GBS scores regarding blood transfusion (p = 0.001 and p < 0.01, respectively); GBS scores were determined to be significantly higher in patients who had blood transfusion.
A statistically significant relation was identified between the GBS score cut-off value of 12 and blood transfusion (p = 0.001 and p < 0.01, respectively). Patients who had a GBS score of 12 or higher had 7.393 times more need for blood transfusion compared to those with a GBS score lower than 12 (odds ratio: 7.393; 95% CI: 2.341–23.348; Table 3).
Evaluation of GBS measurements according to ES replacement.
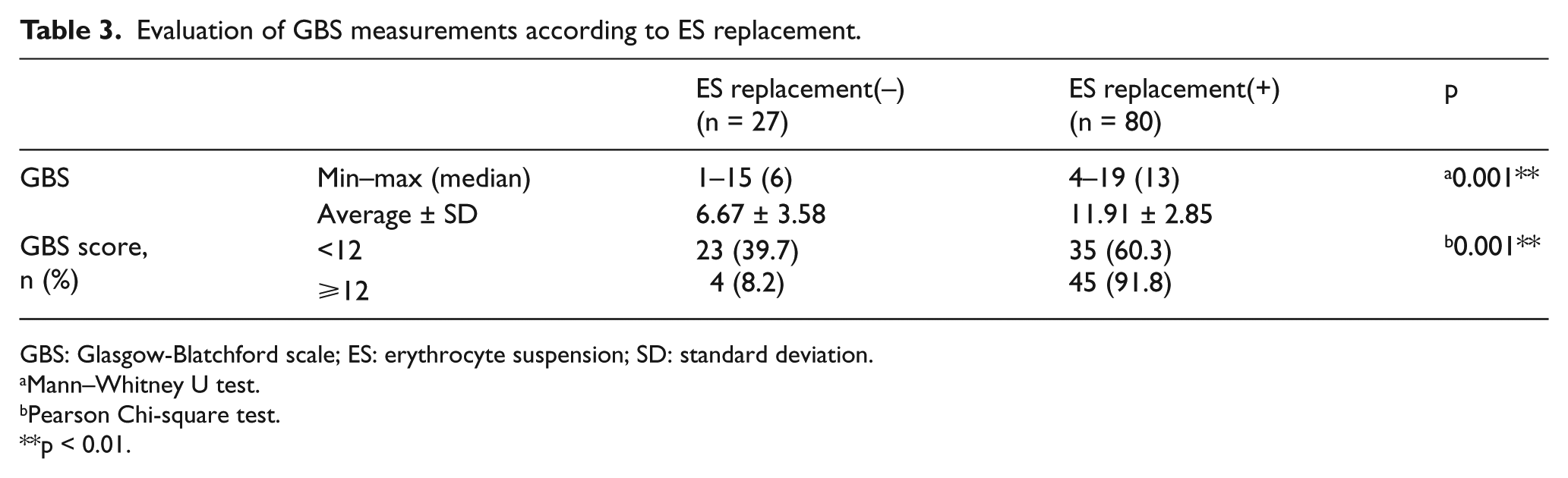
GBS: Glasgow-Blatchford scale; ES: erythrocyte suspension; SD: standard deviation.
Mann–Whitney U test.
Pearson Chi-square test.
p < 0.01.
Between lactate levels and lactate cut-off value (i.e. 23 mg/dL) regarding need for blood transfusion, no statistically significant difference was determined (p > 0.05; Table 4).
Evaluation of lactate measurements by ES replacement.

ES: erythrocyte suspension; SD: standard deviation.
Mann–Whitney U test.
Pearson Chi-square test.
Evaluation of co-occurrence of lactate level and GBS score elevation
When the calculated cut-off values for lactate level (23 mg/dL) and GBS score (12) were taken as the basis, 15% (n = 16) of patients were both lactate(+) and GBS(+), 30.8% (n = 33) were lactate(–) GBS(+), 24.3% (n = 26) were lactate(+) GBS(–), and 29.9% (n = 32) were lactate(–) GBS(–). Between lactate level and GBS score co-occurrence regarding mortality, a statistically significant difference was identified (p = 0.001 and p < 0.01, respectively). According to the analyzed paired comparisons, mortality rate was found to be significantly higher in lactate(+) GBS(+) patients than in the lactate(–) GBS(+), lactate(+) GBS(–), and lactate(–) GBS(–) patients (p = 0.004, p = 0.001, p = 0.001, and p < 0.01, respectively). There was not any statistically significant difference in mortality rates between the other groups (p > 0.05; Figure 7).
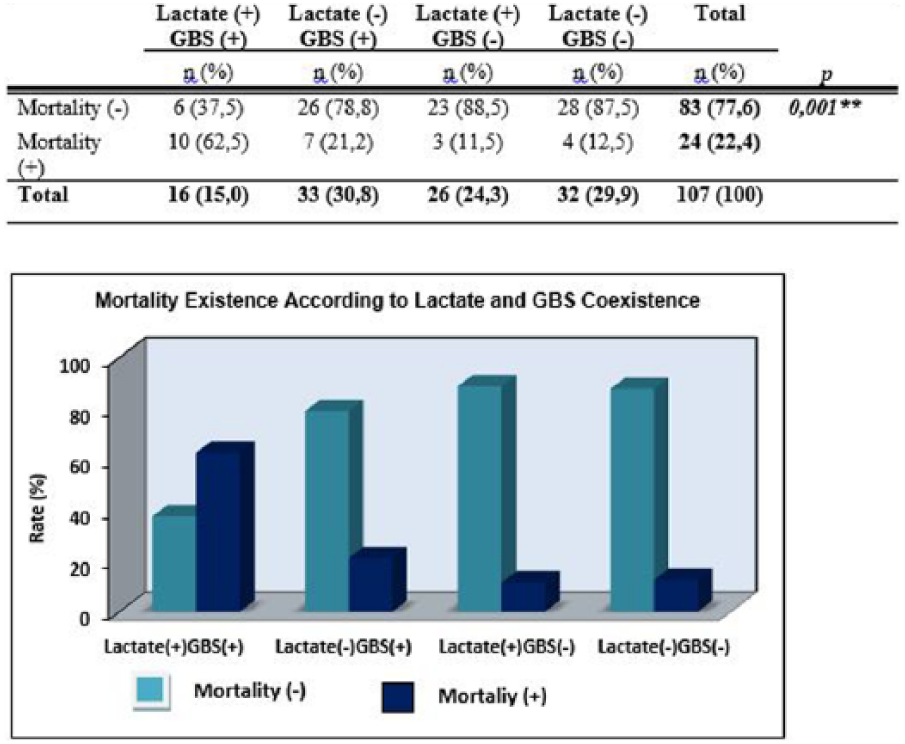
Distribution of mortality existence by lactate and GBS association. **p < 0.01.
Discussion
Upper GIB is an important condition because it is a commonly seen multidisciplinary clinical condition in ED patients which incurs high treatment cost and mortality together with hospital admission. Especially, risk determination of patient at initial ED visit is critical for progression of clinical condition. Although many risk scoring scales are available, emergency medicine physicians need objective and accurate data for evaluation. In this study, GBS scores and venous lactate levels were assessed together to determine the risk of mortality and need for blood transfusion and compared with only GBS evaluation; as a result, it was observed that the combination of GBS score and lactate level was more specific and sensitive against only GBS.
As observed in the literature, while melena had been reported as the most common initial complaint, it is remarkable that in our study hematemesis was the most common initial complaint.3–5 Excessive number of patients who had hematemesis as the initial complaint could suggest that patients were late to visit emergency room (ER) or had more severe clinical condition. In Rockall et al.’s 6 and AIM65 7 scales, melena or hematemesis does not take part in the evaluation.
Blood pressure is one of the most important vital parameters that show hemodynamic instability and it was reported that a decrease in blood pressure is related to an increase in mortality.8,9 In our study, patients with low blood pressure had high GBS and high GBS was found to be related to a high mortality rate. Blood loss due to upper GIB causes hypotension and results from hemodynamic instability caused by ineffective tissue perfusion. Blood pressure is one of the most important markers to evaluate, begin treatment, and to stabilize the patient. Fortunately, among GBS, hypovolemic shock and mortality risk can also be evaluated, and thus patients with ineffective tissue perfusion could be treated earlier.
Hemoglobin level has major importance in monitoring and treatment of patients with upper GIB and is one of the first laboratory samples to be taken. Hearnshaw et al. 10 reported that more than half of the patients with upper GIB had hemoglobin level under 10 mg/dL. We found similar results to Hearnshaw and more than half of our patients had low hemoglobin levels. Low hemoglobin level is a guide for physicians to determine the severity and beginning of hemorrhage. Low hemoglobin level increases the GBS and therefore the mortality rate.
Blatchford et al. 2 evaluated patient’s initial symptoms, vital and laboratory parameters to determine, especially the need for blood transfusion, risk of recurrent hemorrhage, and mortality risk beforehand. Martinez-Cara and Jimenez-Rosales 4 compared the AIM65, Rockall, and GBS scores on hospital admission and mortality and reported that patients with a GBS score of 6 and below do not need to undergo endoscopy and those with a GBS score of 3 and below do not need blood transfusion. Sheifer and Aquarius 11 evaluated the increase in GBS score on admission, discharge, need for treatment, and blood transfusion and stated that 43% of patients who had higher GBS scores needed blood transfusion. In all three studies, higher GBS was related to higher mortality rates and increased need for blood transfusion.2,4,11 In our study, low GBS scores revealed that the mortality rate and need for blood transfusion were significantly lower. Thus, GBS is an effective tool to guide and manage the treatment process of upper GIB. Establishment of this scale in emergency setting may affect the clinical progress and would help the clinician to decide the course of action.
Rockall et al.’s 6 score is one of the commonly used scoring systems to determine the mortality rate in upper GIB. This scoring system uses comorbid diseases, age, existence of shock, and endoscopic findings as the evaluation data. If the patient’s score is less than 3 after the endoscopy, mortality and recurrent hemorrhage risk are scarcely any, so these patients can be discharged earlier from the ED. If the score is higher than 3, hospital admission and monitoring are necessary. A score of 8 or higher shows a high risk of recurrent hemorrhage. While Işık et al. 3 and Martinez-Cara and Jimenez-Rosales 4 used GBS and Rockall scores together, Sheifer and Aquarius 11 only used GBS. Ali et al. 12 reported that GBS has high sensitivity in upper GIB and is advantageous because of no need of endoscopy. In our study, GBS was used rather than Rockall score because GBS does not require endoscopic findings and this enabled faster evaluation and decision making. Thus, risk scores were quickly determined after initial ED visit and mortality risk and need of blood transfusion could be predicted early.
There is only one study present in the literature that assessed the effects of additional parameters with scoring systems. Işık et al. compared the Rockall and GBS scores in patients with upper GIB and researched the relation of mean platelet volume (MPV) and mortality as an additional feature in the aforementioned study. And they reported that MPV was higher in patients with mortality. 3 Similar to this study, we used venous lactate level with GBS. The purpose of both studies was to support the scoring systems used for upper GIB for a long time with another parameter. Many vital and laboratory parameters can be affected in upper GIB and these can be used for determining risk in addition.
Correlation of venous and arterial lactate levels was studied by Kruse et al. 13 Mortality risk was determined to be higher in patients whose venous lactate levels were 20 mmol/L. Puscarich et al. 14 showed that a lactate level more than 20 mmol/L was related to increased mortality risk. They studied lactate levels in patients with infectious diseases but we studied in GIB. In either case, increased lactate level, which shows tissue perfusion disorder, was found to be related to increased mortality rate.
There are few studies that investigate the effects of high lactate levels in upper GIB to mortality and need of blood transfusion. As an example, El-Kersh et al. 15 found that lactate levels were highly sensitive when assessing the mortality rate. In this study, lactate level was added to Rockall score for evaluation. No other study that takes into account the lactate level and GBS together in the evaluation exists. Our study shows that GBS and lactate level could be more valuable than only GBS or lactate level alone to determine the need of blood transfusion and mortality risk.
Limitations
This is a single-center study. Further and similar studies with multi-center settings would be more reliable and accurate. Although no data sufficiency has occurred while performing statistical analysis, endorsing the results with more patients and new studies are needed.
Conclusion
GBS is highly sensitive to determine the mortality risk and need for blood transfusion in upper GIB. GBS with lactate evaluation is more sensitive and more significant than only GBS. This significance allows us to establish “modified GBS.” In the future, studies which will use GBS supported by lactate could be increased and the results should be supported more.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Ethical approval
University of Health Sciences Kartal Dr Lutfi Kirdar Education and Research Hospital Scientific Research Evaluation Committee approval was obtained for this study (No. 89513307/1009/465).
Human rights
This study is subjected to ethical standards that promote and ensure respect for all human subjects and protect their health and rights (Declaration of Helsinki).
