Abstract
Gastropericardial fistula is an abnormal communication between the stomach and the pericardium. It is a rare, life-threatening condition that has numerous etiologies. We report the case of a 53-year-old male patient, with a history of wedge resection for gastrointestinal stromal tumor, who presented to the emergency department for epigastric and chest pain along with lethargy leading to the uncommon diagnosis of gastropericardial fistula with pneumopericardium. Through this case, we would like to draw clinicians’ attention to gastropericardial fistula as a differential diagnosis in patients presenting for epigastric and/or chest pain with a history of esophagogastric surgery, emphasizing on the key role of computed tomography in this regard, and underscore the management basics of this unusual condition.
Introduction
First reported in 1947, gastropericardial fistula (GPF) is a rare and life-threatening condition in which abnormal communication exists between the stomach and the pericardium. 1 It is an uncommon etiology of chest and epigastric pain that should be considered in patients with a history of gastroesophageal surgery. We report the case of a 53-year-old patient, with a history of wedge resection for gastrointestinal stromal tumor, admitted for epigastric and chest pain in whom diagnostic work-up revealed a significant GPF with pneumopericardium.
Case study
A middle-aged male with a history of wedge resection for gastrointestinal stromal tumor several years before presented to the emergency department for new, sudden onset epigastric and chest pain as well as lethargy. Evaluation of vital signs revealed pyrexia (temperature: 38.8°C), tachycardia with a pulse of 128 beats/min, hypotension of 80/57 mmHg, pulse oximetry 95% on ambient air, tachypnea of 28 breaths/min, and normal urine output. Examination revealed pericardial rub, diffuse abdominal guarding, and an immobile epigastric mass measuring 10 × 6 cm2.
On admission, the patient’s hemoglobin, white blood cell count, and platelet count were 7 g/dL, 22.000/mm3, and 276.000/mm3, respectively. Serum level of alanine aminotransferase (ALAT) was 290 U/L (normal: <40 U/L), aspartate aminotransferase (ASAT) was 350 U/L (normal: <40 U/L), lipase was 26 U/L (normal: <60 U/L), C-reactive protein was 480 mg/L (normal: <6 mg/L), sodium was 129 mmol/L (normal: 135–145 mmol/L), and potassium was 5.9 mmol/L (normal: 3.5–5 mmol/L). High-sensitive cardiac troponin test was positive (450 µg/L; normal: <26 µg/L). Laboratory tests also showed renal function impairment with urea and creatinine plasmatic levels of 1.2 g/L and 20 mg/L, respectively. Electrocardiogram showed widespread ST-segment elevation (Figure 1). Chest X-ray revealed a pneumopericardium (Figure 2). Subsequent thoracic and abdominal computed tomography (CT) displayed a large, irregularly shaped, exophytic, gastric tumor measuring 28 × 12 mm2, invading adjacent structures, and a GPF with pneumopericardium (Figures 3–5). Bedside transthoracic echocardiography showed a moderate pericardial effusion without any signs of hemodynamic compromise.
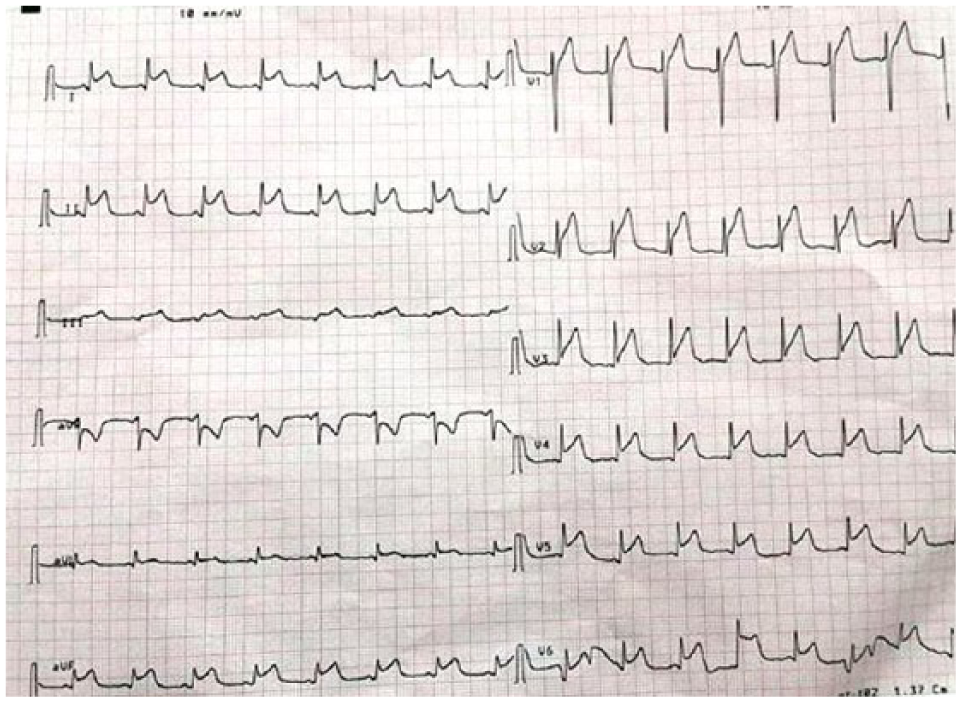
ECG showing widespread ST-segment elevation.
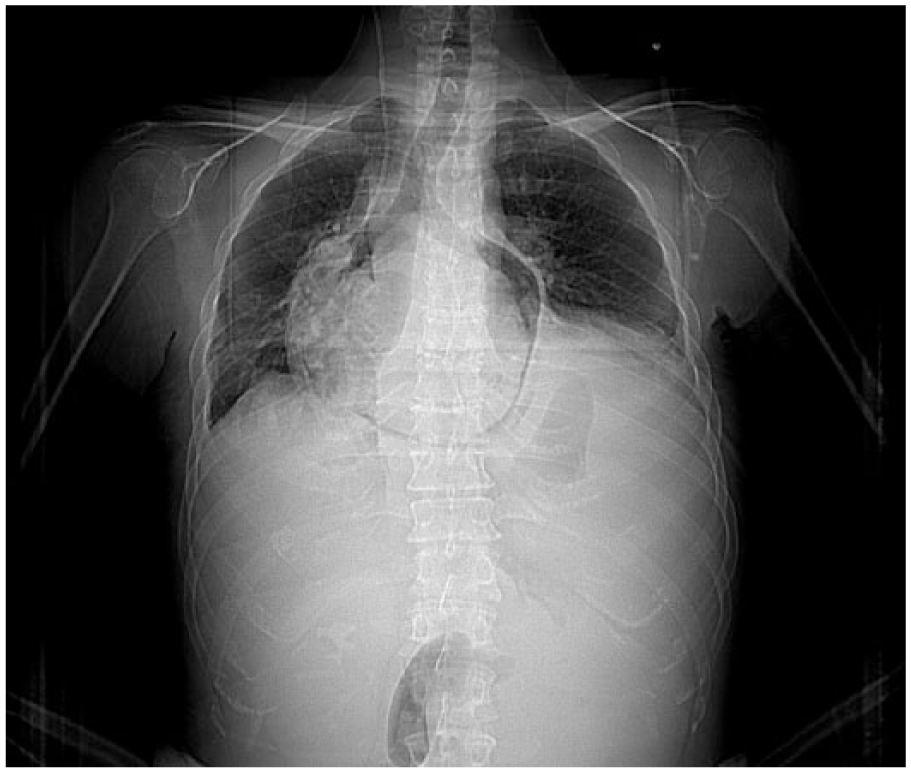
Chest X-ray showing pneumopericardium and left diaphragmatic cupola ascension.
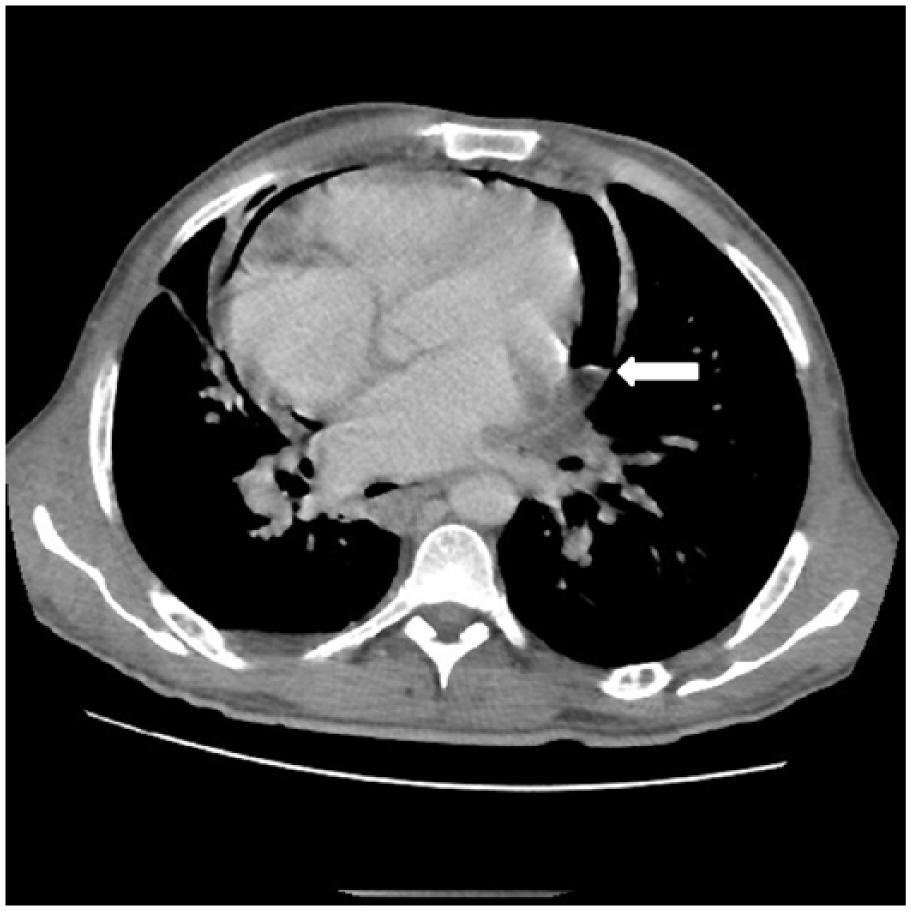
Axial chest CT displays hydropneumopericardium with an air-fluid level (white arrow).
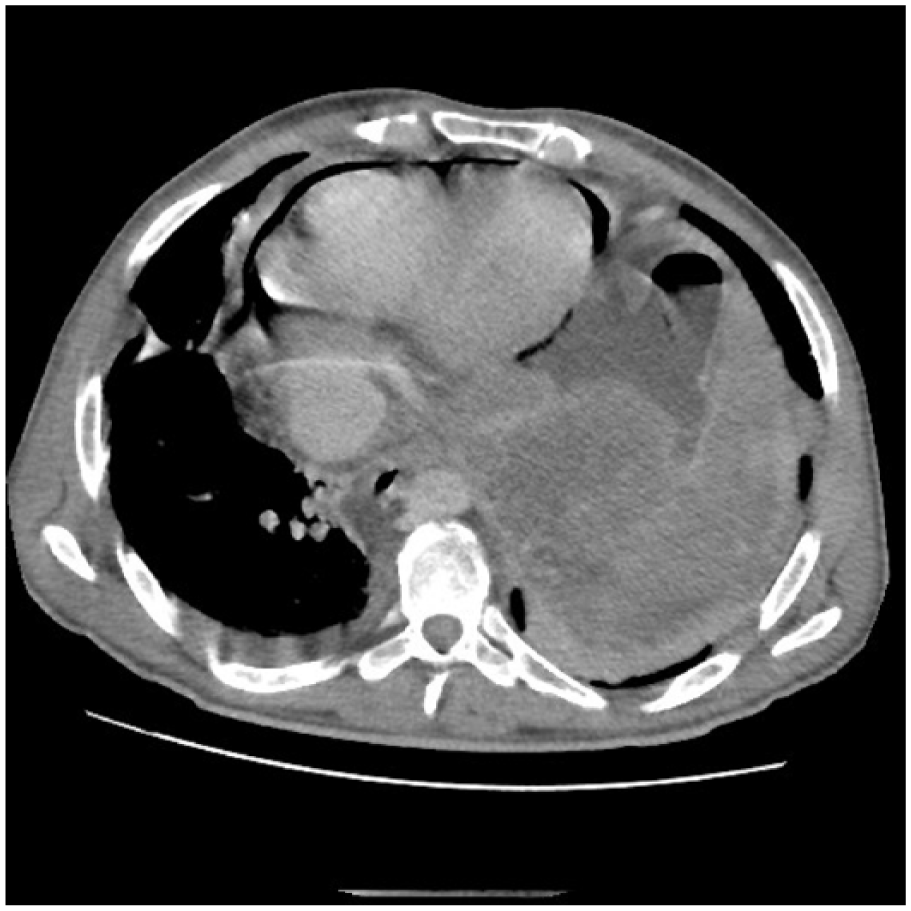
Axial chest CT demonstrating a large, locally advanced gastric tumor more marked at the level of the greater gastric curvature with a fundic parietal defect (thin white arrow) and contact between the digestive content and the pericardium responsible of pneumopericardium (thick white arrow).
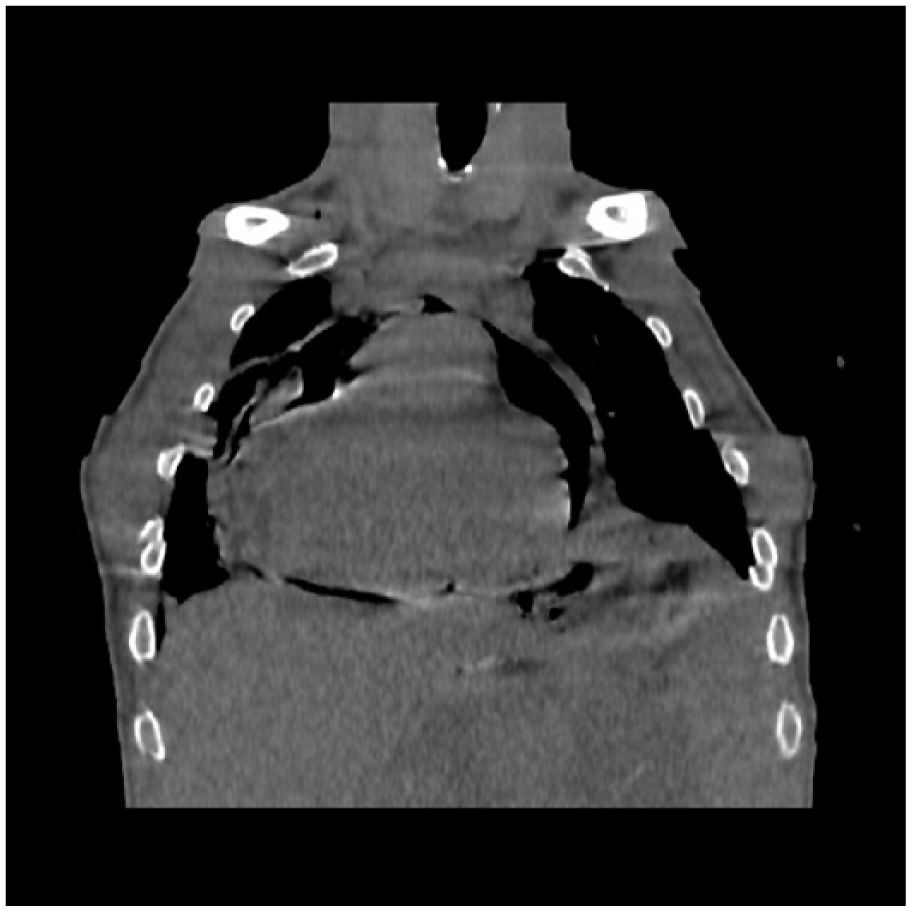
Coronal CT without IV contrast reveals gastropericardial fistula with pneumopericardium (white arrow).
Initial treatment comprised transfusion of 4 units of packed red blood cells, intravenous fluid resuscitation, and intravenous broad-spectrum antibiotics (piperacillin-tazobactam, amikacin, and fluconazole). After discussion within a multidisciplinary team, and with the family, the patient was taken promptly to surgery where he underwent, through a midline vertical laparotomy, a monobloc resection of the tumor with splenectomy, left adrenalectomy, and diaphragmatic resection. Pus coming from the pericardium was noted. Unfortunately, during the procedure, the patient presented ventricular fibrillation followed by cardiorespiratory arrest refractory to resuscitation measures despite our best efforts.
Discussion
GPF was reported for the first time in 1947 by Harp and Peeke. 1 It is a rare and life-threatening condition defined as an abnormal communication between the stomach and the pericardium. Etiologies include previous gastroesophageal surgery, peptic ulceration usually associated with hiatal hernia, trauma, infection, malignancy, presence of a foreign body both ingested and implanted, caustic agent ingestion, or a combination of the above. In our case, the patient had previous esophagogastric surgery for neoplasia with subsequent cancer recurrence, only reported in 5% of published GPF cases.2–4
Clinical presentation includes chest, shoulder, or epigastric pain, dyspnea, fever, dysphagia, vomiting, hematemesis, or melena. Often, signs and symptoms of sepsis are present.3,5 An electrocardiogram may show repolarization disorders such as widespread ST-segment elevation probably due to pericardial irritation. Chest radiography classically shows pneumopericardium, which is commonly seen in chest trauma, asthma, perforation of lung or tuberculosis abscess, and medical procedures. Prior gastroesophageal surgery, with a reported average duration between presentation and surgery of 7.3 ± 6.2 years (SD), should raise suspicion of GPF as an etiology, although exceptional (<5%), of pneumopericardium.2,5,6 Transthoracic echocardiography may show two pathognomic signs, albeit inconstant, of pneumopericardium: “the air gap sign” traducing a cyclic disappearance of the cardiac shape during systole coinciding with a cycling appearance of air within the pericardium during this phase and “The swirling bubbles sign” revealed in echocardiography by several tiny bright echogenic spots in the pericardial sac evoking micro air bubbles. 7 The primary work-up test in case of suspicion of GPF is CT. Hydropneumopericardium is a classic finding highly suggestive of GPF. The fistulous tract is seldom seen, and multiplanar reconstruction should be used to increase CT sensitivity. In cases of negative CT with high GPF likelihood, the use of oral contrast agents can demonstrate contrast in the pericardium. However, the effects of contrast on the pericardium have not been well studied and should be used with precaution.2,7 Esophagogastroduodenoscopy, although controversial due to the potential risk of tension pneumopericardium after gastric insufflation, may be used to increase CT’s sensitivity and specificity. Fluoroscopic contrast study, using water soluble rather than barium and effervescent contrast agents, may be discussed in case of negative CT.2,3,8 We readily established the diagnosis of GPF with pneumopericardium by CT without the use of contrast agent in our patient.
Previously heralding poor prognosis, advances in surgical procedures and intensive care have allowed GPF mortality rates to fall from 85% to 11.8% in the most recent literature review.2,5,9 Management should be performed in a timely manner. Most authors recommend antibacterial/antifungal therapy, fluid resuscitation, nutritional support, and surgery, even in multi-morbid patients as most cases fare better with surgery than without. Pericardial drainage is recommended in almost all cases. Diversion of gastrointestinal from the pericardium is also indicated. A most definitive surgical management may be performed simultaneously or planned for a later time.3,8 In our case, despite the patient’s very poor prognosis, it has been decided to intervene surgically. The patient did not survive the surgery.
Conclusion
GPF is a rare and probably under-reported condition. Clinicians should keep it in mind in the differential diagnosis of chest and epigastric pain, a fortiori in cases of prior gastroesophageal surgery or locally advanced gastric tumors. Chest X-ray, readily accessible, usually displays pneumopericardium. The gold standard imaging technique is CT with, eventually, oral contrast agents. In equivocal cases, esophagogastroduodenoscopy or fluoroscopic contrast study may be of valuable help. Prompt surgery is the cornerstone of treatment, along with antibacterial/antifungal therapy, and supportive measures.
Footnotes
Informed consent
Every effort was made to obtain informed consent from the patient with no success so the case report has therefore been anonymised as far as possible.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
