Abstract
Ischemic stroke following snakebite is rare. We report a case of a 55-year-old male who developed headache with expressive aphasia following a Russell’s viper bite. Computed tomography scan of the brain revealed infarcts in bilateral frontal lobes. The possible mechanisms for cerebral infarction in this scenario are discussed, which include disseminated intravascular coagulation, toxin-induced vasculitis, and endothelial damage. In viper bites, whenever there is any central nervous system manifestations or deficits, immediate computed tomography scan of the brain should be taken to delineate the underlying pathology, whether hemorrhagic or ischemic, as treatments differ in both situations. Although ischemic stroke is rare in viper bites, if the treatment is started early, the neurological deficits can be minimized.
Introduction
In India, more than 2,000,000 snakebites are reported annually, of which 35,000–50,000 people die. 1 Russell’s viper is the leading cause of fatal snakebite in India. 2 The clinical manifestations include local reactions and necrosis, renal failure, and coagulopathy complicated by hemorrhagic manifestations such as pituitary and intracranial haemorrhage. 3 There are only few case reports of ischemic stroke associated with snakebite. 4 In this case report, we are presenting an unusual complication, cerebral infarction following Russell’s viper bite. Ethical approval was obtained from the Institutional Ethical Committee of the institute.
Case report
A middle-aged previously healthy male was admitted for a snakebite on his left foot which he sustained in his paddy fields. The snake was caught and identified as Russell’s viper by snake expert. He complained of severe pain over the left foot. Local examination showed two deep fang marks, with local reactions of erythema and edema. No other hemorrhagic or neurological manifestations were observed clinically on admission. Twenty-minute whole blood clotting test (WBCT20) was positive. He was immediately treated with 3 vials (30 mL) loading dose of equine polyvalent anti-snake venom (ASV-ASIA, Bharat Serum and Vaccines Ltd), followed by continuous intravenous administration of another 3 vials of anti-snake venom in normal saline. While receiving ASV infusion, patient developed ptosis. As soon as the ptosis developed during admission to ward, 10 vials of ASV was given as infusion followed by again 10 vials of ASV as infusion after 1 h along with atropine and neostigmine by the attending physician. An injection of adsorbed tetanus toxoid was given and a course of antibiotic injection of ceftriaxone was started. Initial laboratory investigations on day 1 revealed that blood sugar was 100 mg/dL (5.6 mmol/L), blood urea was 60 mg/dL (9.96 mmol/L) (normal range, 14–44 mg/dL), serum creatinine was 1.6 mg/dL (141.4 µmol/L) (normal range, 0.8–1.4 mg/dL), serum bilirubin was 2.3 mg/dL (39.3 µmol/L) (normal range <1.5 mg/dL), alanine transaminase was 38 IU/L (normal range, 3–36 IU/L), aspartate transaminase was 89 IU/L (normal range, 3–36 IU/L), and alkaline phosphatase was 69 IU/L (normal range, 35–100 U/L).
On the next day (day 2), further 5 vials of ASV was given as infusion along with tapering doses of atropine and neostigmine as WBCT20 was still positive, which was normalized on day 3. Another dose of 10 vials of ASV was given on day 3. On the seventh day (day 7), patient developed left-side weakness and speech disturbances. Neurological examination revealed left hemiparesis (muscle power 3/5) and expressive aphasia while he remained hemodynamically stable. Computed tomography (CT) scan of the brain showed acute ischemic infarcts in bilateral frontal lobes (Figure 1). Lipid profile, electrocardiogram, and echocardiogram were normal. WBCT20 was negative. Patient was treated with aspirin and clopidogrel. Patient showed improvement in the motor power (4+/5) and speech at the time of discharge on day 10.
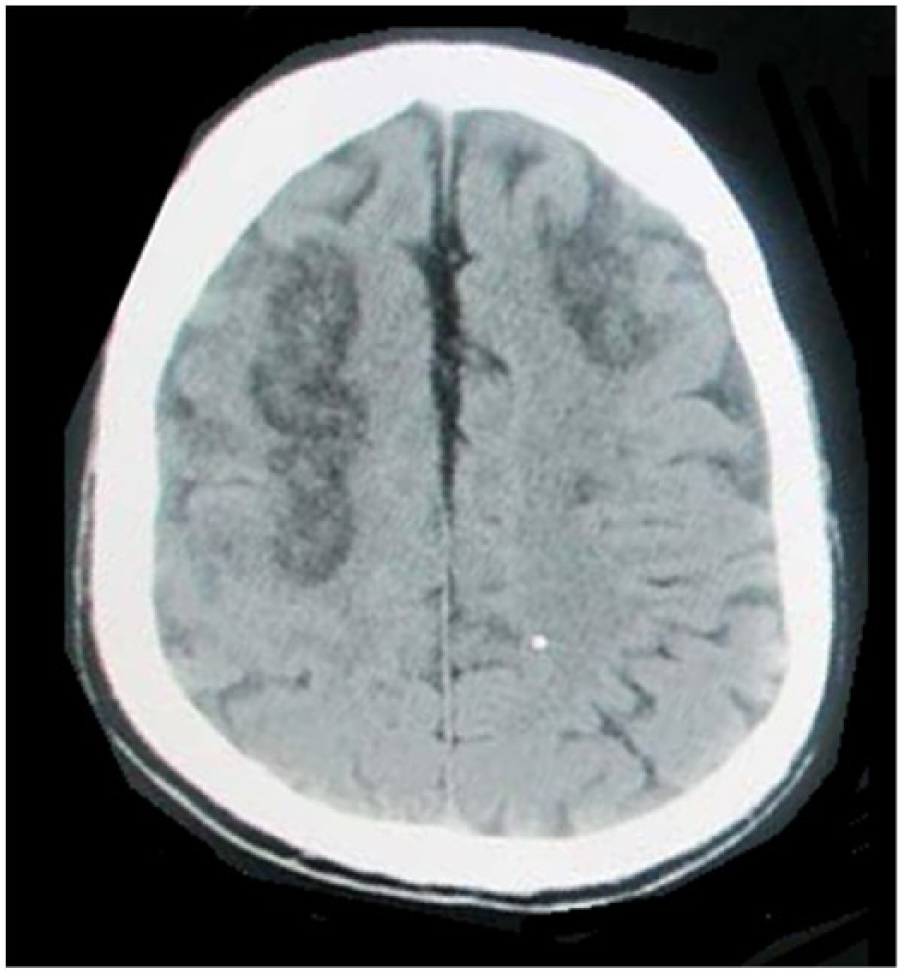
CT scan of the brain showing acute infarcts in bilateral frontal lobes.
Discussion
Cerebral complications, particularly ischemic complications, after snakebite are rare. 3 Very few cases of cerebral infarction resulting from a viper bite have been reported.5–7 In a study of 309 patients with Bothrops snakebite, Mosquera et al. 5 reported cerebrovascular complications in eight patients (2.6%), seven hemorrhagic strokes, and one ischemic stroke, while laboratory bleeding disorder was present in all hemorrhagic cases but absent in the ischemic case. Bashir and Jinkins 6 reported a patient in whom envenomation with Russell’s viper resulted in hemiplegia and aphasia, consistent with bilateral frontal lobe infarction, and normal clotting profile. Murthy et al. 7 reported a case of cerebral infarction and diffuse encephalopathy with normal coagulation and platelet count following a Echis carinatus viper bite. In addition to the above cases developing ischemic stroke in the presence of normal clotting, there was a reported case of posterior circulation ischemic infarction occurring very early following viper bite in a previously healthy woman along with deranged coagulation profile—prolonged prothrombin time, prolonged activated thromboplastin time along with positive D-Dimer test. 8
Viper venom is a complex toxin with rich components principally affecting hemostatic mechanisms. 9 In high doses, it can cause massive intravascular coagulation leading to small and even large vessel occlusions resulting in cerebral infarction. 10 Other mechanisms include toxic vasculitis resulting in thrombosis; 7 direct action of the venom on vascular endothelial cells; 6 hypercoagulation state due to procoagulants in the venom, such as arginine, esterase, and hydrolase;6,7 hemorrhagins that result in severe vascular spasm, endothelial damage, and increased vascular permeability; 6 and hyperviscosity caused by hypovolemia and hypoperfusion secondary to hypotension. 3
Our patient developed pain, erythema, and edema following the snakebite. As WBCT20 was positive, he was given 3 vials of ASV as bolus and another 3 vials of ASV as infusion. As the patient developed ptosis (presynaptic curare-like effect of snake venom), he was given additional doses of ASV along with neostigmine and atropine till the neurological and hematological manifestations become normal and WBCT20 was normal on the third day. On the seventh day, patient developed left-side weakness and speech disturbances due to cerebral infarction. Vale et al. 11 reported a case similar to our case—a case of an adolescent who developed bilateral posterior circulation stroke after 1 week of Crotalus durissus terrificus snakebite. The authors concluded that stroke was probably due to toxic vasculitis or toxin-induced vascular spasm and endothelial damage as the clotting profile was normal when the stroke evolved. 11 In our case, there was no documented hypotension, and there was no evidence of disseminated intravascular coagulation around the time of onset of stroke.
Early administration of ASV is essential to neutralize the maximum circulating venom before it is fixed in tissue. Therefore, it should be given to cases with evidence of systemic envenomation as early as possible.11,12 In a study which included patients with Bothrops snakebite, no thrombotic complications or stroke was observed in the group with early antivenom therapy within 6 h. 13 Early administration of antivenom may have a role in prevention of thrombotic complications including ischemic stroke.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Written informed consent was obtained from the patient(s) for their anonymised information to be published in this article.
