Abstract
This article analyzes the interaction between lead firms and Chinese and Indian contract research and development (R&D) firms in integrated circuit (IC) and chemistry-based pharmaceutical global value chains (GVCs) in order to delineate what factors influenced the upgrading and value capture outcomes of these contract R&D firms in the two sectors. There are five main findings. First, the two sectors differed significantly in terms of the upgrading of contract R&D firms’ activities and capabilities. More upgrading was found in chemistry-based pharmaceuticals than in ICs. Second, in both industries, lead firms utilized a range of strategies to control and limit upgrading and value capture by contract R&D firms. Third, level of R&D uncertainty (risk) and lead firms’ level of market access control informed the strategies of lead firms. Lead firms generally adopted strategies to limit upgrading by contract R&D firms except in two situations: where R&D risk was high so that lead firms wished to offload costly risks onto contract R&D firms and where lead firms had significant market access control to limit value capture by upgrading contract R&D firms. Fourth, the strategies to limit upgrading included minimizing the flow of information possible in modular linkages and discouraging IP development by contract R&D firms. Fifth, the lead firm strategies to limit value capture included limiting upgrading and thus any associated potential value capture, enhancing lead firm bargaining leverage, and controlling market access via established market channels and continual engagement with regulatory agencies in relevant markets.
Introduction
This article addresses a question neglected by the burgeoning global value chain (GVC) literature: how contract R&D firms 1 in the developing world upgrade and capture value within GVCs. 2 Focusing on two R&D-intensive GVCs, integrated circuit (IC) design and chemistry-based pharmaceuticals, that make extensive use of external contract R&D firms (referred to as suppliers in our discussion of the wider GVC literature), our article examines contract R&D firms from China and India as these two countries represent the majority of the developing world’s contract R&D activity in these two sectors.
Our findings challenge two important arguments made in GVC research. First, the article provides evidence contesting the long-standing structural arguments that modular and relational linkages (Gereffi et al., 2005; Gereffi and Güler, 2008, 2010; Pietrobelli and Rabellotti, 2011; Sako and Zylberberg, 2019) and more multipolarity of GVC governance (Ponte and Sturgeon 2014) are particularly conducive to supplier upgrading and value capture. Instead, we find that lead firms can use strategies, such as minimizing the flow of knowledge in modular linkages, to limit upgrading (and any concomitant value capture) in modular networks (the IC case and certain segments in the pharmaceutical case) and limit the value capture in relational linkages through utilizing lead firms’ control of market access (the pharmaceutical case). Similarly, lead firms can limit the opportunities multipolarity offers to suppliers (in our case, contract R&D firms) for upgrading and value capture through increasing bargaining leverage (the IC case) and limiting knowledge flow through modularity (both cases) and market access control via established market channels and engagement with regulators (the pharmaceutical case). Second, the article flips on its head the claims of more recent research that supplier initiative and strategy lead to greater supplier value capture (Ma et al., 2022;Sako and Zylberberg, 2019). In our cases, the lead firms use the various strategies described above (increasing bargaining leverage, minimizing flow of knowledge in modular linkages, and controlling market access) to counter strategies by the contract R&D firms to upgrade and capture more value.
In sum, our findings are as follows. First, the two sectors differed significantly in terms of the upgrading of contract R&D firms’ activities and capabilities. More upgrading was found in chemistry-based pharmaceuticals than in ICs. Second, in both industries, lead firms utilized a range of strategies to control and limit upgrading and value capture by contract R&D firms. Third, level of R&D uncertainty (risk) and lead firms’ level of market access control informed the strategies of lead firms. Essentially, lead firms generally adopted strategies to limit upgrading by contract R&D firms (the IC case and some of the pharmaceutical case) except in those situations where R&D risk was high so that they wished to offload costly risks onto contract R&D firms and where the lead firms had significant market access control to limit value capture by upgrading contract R&D firms (certain segments of the pharmaceutical case). Fourth, the strategies to limit upgrading included minimizing the low flow of information possible in modular linkages (the IC case and part of the pharmaceutical case) and discouraging IP development by contract R&D firms (the IC case). Fifth, the lead firm strategies to limit value capture included limiting upgrading and thus any associated potential value capture (the IC case and part of the pharmaceutical case), enhancing lead firm bargaining leverage (the IC case) and controlling market access via established market channels and continual engagement with regulatory agencies in relevant markets (the pharmaceutical case).
The paper proceeds as follows. The next section concerns methodology. The third section is the literature review. The fourth and fifth sections discuss the pharmaceutical and IC design industries, respectively. The sixth section compares R&D contracting in the two sectors in detail. The conclusion highlights the limits of GVC structural characteristics and contrastingly wide space for lead firms’ strategies to shape GVC characteristics in order to control the upgrading and value capture outcomes of their suppliers.
Methodology
In order to explore the dynamics between lead firms, which are the firms contracting out R&D for their own products, and R&D contract firms in the developing world in these two GVCs, this study employed semi-structured interviews with mid-level to senior R&D management (supplemented by some documentary data) in the vein of the grounded theory approach (Strauss 1987; Strauss and Corbin 1990). In pharmaceuticals, we interviewed seven of the 2016 global top 10 (by revenue) lead firms (collectively, these firms account for approximately 30% of global market share and have a revenue of US$274.31 billion), three lead firms that are not part of the top 10, and six Indian and Chinese contract R&D firms and seven non-Chinese/Indian contract R&D firms. The purpose of interviewing non-Chinese/Indian contract R&D firm was to see if the contract R&D firms outside of China and India had different relationships with the lead firms. The interviews revealed that the relationships with lead firms were similar across contract R&D firms. Interviews took place between 2012 and 2017. For the largest Chinese contract R&D firm, we obtained additional detailed documentary information on the status of the firm’s contract R&D activities in 2021. Thus, for contract R&D firms in India and China, we collected direct interview and documentary data from four large firms (among top 10 contract R&D firms in each country) and two other contract R&D firms in order to have some diversity of firm size in our data. Additional data on contract R&D activities in India and China were collected from lead firms and non-Indian/Chinese contract R&D firms.
In IC design, we interviewed nine contract R&D firms representing the majority of design service work across India and China in 2014–2015. We also interviewed one Chinese and four Indian contract R&D firms in 2007. We found strong continuities between the lead firm-contract R&D firm relationships in 2007 and the later research period. As for lead firms in the IC design industry, we interviewed 11 of 2016's top 20 global (by revenue) lead firms as well as 14 other lead firms from 2012 to 2015. In 2022 in order to check that the patterns we had uncovered still held in this industry, we conducted a telephone interview with an Indian entrepreneur previously involved in founding several contract R&D firms we interviewed and who is also very well connected as a senior figure in the India Electronics and Semiconductor Association, and a telephone interview with a serial entrepreneur who founded one of the most active design service firms in China and follows the IC design industry in China closely.
Direct data collection from lead firms and R&D contract firms.

aIncludes two firms (one each in top 10 and non-top 10) which are Chinese-Taiwanese hybrid firms.
bIncludes one Indian firm that mainly does EDA tool development but also does design service.
Literature review
From early on in the development of the GVC literature, the structural approach positing that the structures of the GVCs heavily shape upgrading and value capture outcomes has been a major focus (Gereffi et al., 2005). Two types of structures have been especially prominent: linkages and value chain governance (polarity).
Linkages refer to the nature of the interactions between firms at specific nodes (e.g., interaction node between lead firms and first-tier suppliers or the node where first-tier and second-tier suppliers interact). Following the seminal work of Gereffi et al. (2005), there are five modal types of value chains that feature market, modular, relational, captive, or hierarchy linkages. Transaction costs and transaction codifiability are the primary determinants of GVC structures. As one moves from market linkages to hierarchical ones, transaction costs grow due to increasing complexity and intolerance of distance between the two actors at a specific node, and at the same time, the codifiability of information at the linkage decreases. Gereffi et al. (2005) acknowledge that capabilities of the suppliers differ across the linkage types with low supplier capabilities in captive and hierarchical linkages, but supplier capability seems less determinant than transaction costs and codifiability in shaping GVC structures in their account.
GVC linkage structure matters because many have argued that these structures heavily influence the upgrading and value capture outcomes. The near consensus view on linkage structure and upgrading and value capture is that modular and, especially, relational linkages are the most conducive to positive upgrading and value capture outcomes for supplier firms (Gereffi et al., 2005; Pietrobelli and Rabellotti 2011; Raj-Reichert, 2019; Ramirez 2014; Ramirez and Rainbird, 2010; Sako and Zylberberg, 2019; Thun, 2007). Development practitioners have also picked up this mantra that modular and relational linkages promise the best upgrading outcomes for the developing world (ABD WTO, 2021; Engel and Taglioni, 2017). Our article will demonstrate, that lead firm strategies, often utilizing the external environment in the form of increasing the population of suppliers, regulation or market channels, can
Of course, GVC structures are not solely defined by these technical characteristics but have dominant actors governing the GVC. Value chain governance refers to the polarity and drivenness of value chains with unipolar chains being highly driven by one actor (typically a lead firm but occasionally a state actor, e.g., biofuels—see Ponte, 2014), bipolar chains are heavily influenced by two lead firms/actors, and multipolar value chains have relatively weaker drivenness (influence) with multiple major actors involved including governments, standard-making organizations, and other non-corporate actors participating in value chain governance. Thus far, the GVC literature observes polarity in a given GVC rather than offering a theory or framework to explain what factors increase and decrease polarity in GVCs generally (Ponte, 2014; Ponte and Sturgeon, 2014).
Both the pharmaceutical and IC GVCs are multipolar GVCs. For example, in pharmaceuticals, the powerful actors include lead firms and government regulatory bodies such as the Federal Drug Administration of the United States. In the IC industry, the powerful actors are lead firms in the IC value chain, such as chipmaking foundries (e.g., TSMC) as well as lead firms doing their own design (e.g., Nvidia)—the focal lead firms for our study because they are the firms contracting out R&D to contract R&D firms—and major buyers of chips (e.g., Apple).
Like linkages, GVC governance is assumed in the literature to be an important structure shaping upgrading and value capture outcomes for supplier firms. Ponte and Sturgeon (2014) have suggested that the further along a given GVC is from unipolarity toward multipolarity, the more opportunities for supplier firms to upgrade. Similarly, Dindial et al. (2020) suggests that unipolarity, or in their terms, low redeployability of the developing world suppliers, leads to low value capture. Multipolarity, or high redeployability in their terms, leads to high value capture. Documenting the rise of transnational first-tier suppliers, Raj-Reichert (2019) also demonstrates that their rise took place not coincidentally within multipolar GVCs. In contrast to these promising cases of greater multipolarity leading to greater upgrading and value capture, this article provides a case where lead firms, reacting to somewhat unfavorable, multipolar value chain governance systems, devise other means to limit supplier’s value capture. Thus, this article serves to bring in lead firm strategy to counter the dangers (for lead firms) of greater multipolarity and surrendering more value capture to suppliers.
Recently, scholars have become very interested in strategic factors shaping upgrading and value capture outcomes in GVCs. These scholars have moved the focus away from structures and lead firms to examine supplier strategies to upgrade and capture more value in GVCs. Lechner et al. (2020) show that large suppliers can hollow out of lead firms’ activities along the sports shoe GVC due to lack of focus of branded lead firms on upstream activities to their own detriment. Ma et al. (2022) finds similar outcomes in suppliers integrating important parts of the GVC in lower tier manufacturing suppliers from various industries. Borrowing most explicitly from the strategic management literature, Sako and Zylberberg (2019) argue that appropriability regimes (regimes for IP protection) and specialized complementary assets (assets that create unilateral or mutual dependence between the firm holding the asset and another party) are two key preconditions for value capture from upgrading.
The key contribution Sako and Zylberberg make is to emphasize strategic choice in engaging with which type of GVC. They argue that suppliers can manipulate the GVC to upgrade by moving from captive to relational or modular linkages. Our article reverses the directionality of their strategy argument by documenting how lead firms strategically choose linkages to constrain supplier upgrading and value capture given the wider technological constraints of a particular GVC context. Here the nature of linkages themselves is somewhat malleable to strategic purpose of powerful firms.
At the same time, our paper concurs with the insights of Sako and Zylberberg (2019) that appropriability regimes and specialized complementary assets matter for value capture by suppliers. They argue that where appropriability regimes are strong, suppliers have a greater opportunity to capture some value from upgrading. However, suppliers can also use specialized complementary assets to force the other party into dependence and thus the surrender of some value. Our article points out that lead firms can strategically use specialized complementary assets or the structure of a particular GVC itself to prevent suppliers from capturing value. In the pharmaceuticals case, lead firms use specialized complementary assets in terms of knowledge of how to navigate regulatory regimes for pharmaceuticals to prevent suppliers from using intellectual property rights (IPR) (an appropriability regime) to capture more value. In ICs, lead firms, facing strong IPR regimes and no strong regulatory barriers to market entry by erstwhile suppliers, double down on using modular GVCs to restrict knowledge flows and learning to suppliers so the latter simply cannot upgrade into segments where the value creation resides. In sum, while suppliers can utilize moving into different linkages, appropriability regimes and complementary assets to upgrade and capture value as Sako and Zylberberg (2019) posit, our article shows that lead firms can utilize similar methods to prevent surrendering more value to suppliers.
Having discussed which aspects of the GVC literature we are engaging, we now need to discuss how we define upgrading, value capture, and risk. In the GVC literature, upgrading is typical conceived in four different ways: process upgrading, product upgrading, functional upgrading, and chain (inter-sectoral) upgrading. Within each task, there are task of greater or lesser technical and organizational complexity. Upgrading is the act of moving into those tasks of greater technical and organizational complexity (Kaplinsky and Morris, 2001). For this study, the relevant types of upgrading are product and functional upgrading. However, in contrast to much of the literature, we recognize that R&D is not one monolithic functional block, but a series of functions requiring various levels of technical skills. Product upgrading is movement into more sophisticated product lines. Functional R&D upgrading is moving into R&D functions requiring higher skills. To provide examples from our research, product R&D upgrading in IC design could be moving from digital product designs to mixed-signal ones as the latter require greater technical competency and thus command greater premiums. Functional upgrading would be moving from back-end design activities to architecture-level design (see details below in case section).
Upgrading does not necessarily lead to value capture as GVC scholars have long recognized (Arnold, 2010; Kaplinsky and Morris, 2001). Implicit in much of the work on value capture—and explicit in Kaplinsky and Morris—is the notion that value capture is the capture of economic rents or profits. We operationalize value in this paper by defining value capture as profits (inclusive of reinvested profits) plus wages for those engaged in the respective activity. This wage-inclusive definition is not an expanded definition vis-à-vis Kaplinsky and Morris because any accurate measurement of economic rents includes wages (Khan, 2000). Moreover, other scholars have included wages as a key metric of value capture (Schrank, 2004).
We understand risk as the risk of R&D not leading to useable product in the sense of something that can be used in the next segment of the value chain rather than the narrow definition of product as the physical end good sold. Risk is critical because firms calculate the estimated value of activities based upon risk, that is, estimated profit from the activity multiplied by the likelihood of success of that activity. 3
Pharmaceuticals
Organizational changes in the industry
This article focuses on lead firms undertaking research, development, and production activities leading to patented, chemistry-based pharmaceuticals as the dynamics in this sub-sector of the industry are different from those of the off-patent sector (Haakonsson, 2009). Despite major advances in scientific understanding of the pathways of disease over the last 40 years, the pharmaceutical industry’s R&D productivity has experienced a prolonged period of decline. Under pressure to make their R&D more productive, lead firms have responded with a range of strategies and organizational changes including the integration of new areas of science and technology into the knowledge base of lead firms as well as the outsourcing of R&D activities to contract R&D firms.
In the pharmaceutical sector under study, the main aim of the R&D process is to find new chemical entities with therapeutic effects. Drug R&D consists of two fundamental sets of activities: (1) the identification of biological targets that play a role in disease and can be a point of intervention for drug therapy, and (2) the discovery and design of molecules that can be turned into medicines. The industry’s R&D function can be represented as a R&D value chain as in Figure 1. The discovery-research stage of the R&D value chain is made up of all activities associated with target identification up to pre-clinical development. This stage is critical as it is here that the intellectual property (IP) of firms is created. Pharmaceutical R&D global value chain.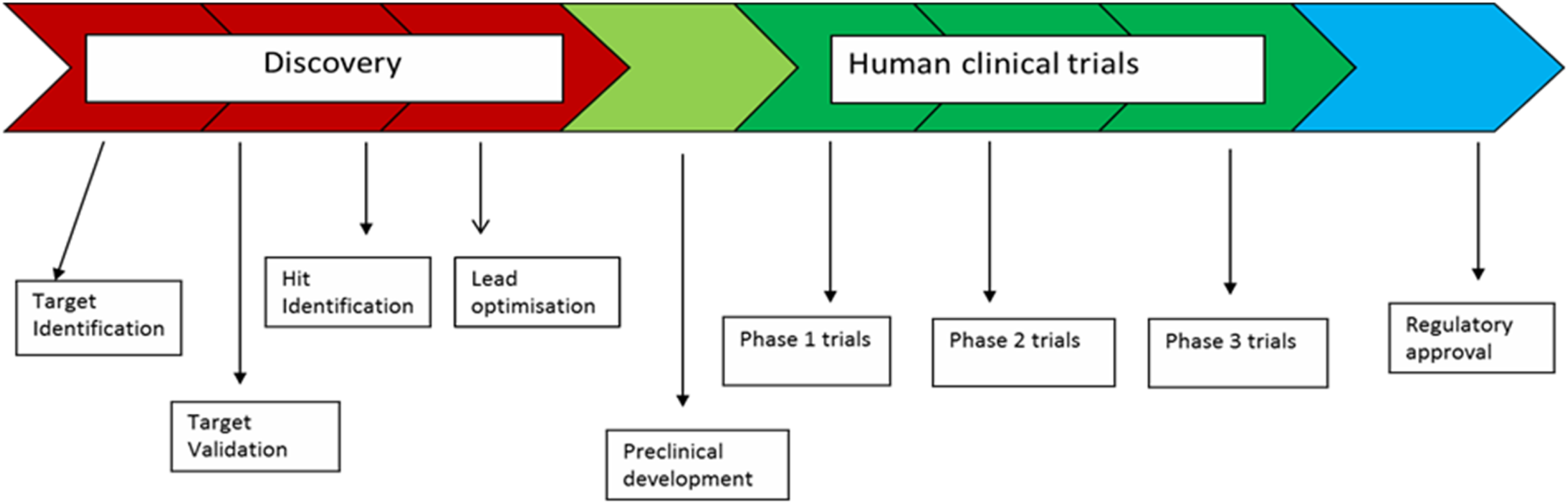
Given the complexity of human biology, today much of the research on the identification of biological targets for intervention takes place in academic research centers and spin-outs firms. Within lead firms, much of the activities of the R&D function involve the design and synthesis of thousands of molecules that could become potential drugs, followed by multiple tests that generate information about the chosen molecules’ safety and efficacy (Pisano, 2006). These tests, which include standardized as well as very complex tasks, are critical to the R&D process as they generate the data that determines the selection of innovative projects. The tests are regulated by strict government standards that need to be complied with for drugs to be given the regulatory approval needed for marketing.
A key characteristic of the discovery-research stage of the R&D value chain of chemistry-based pharmaceuticals is the integrated nature of the problem-solving process and the high degree of interdependence across different scientific and technical domains as well as the various phases of the R&D process (Pisano, 2006). Therefore, while it is possible to conceptualize the different stages of the R&D value chain as distinct (as in Figure 1), they are closely integrated to enable information to flow fluently between the different steps of the research process. In pharmaceuticals, therefore, it is very difficult (though not impossible) to modularize problem-solving processes in the early stages of R&D, which is why the industry has been characterized as integral rather than modular (Gassmann et al., 2004; Pisano, 2006). Integrality is much less important in later-stage clinical trials where work packages can be allocated to organizationally distinct and internationally dispersed clinical management organizations (CMOs) (Gassmann et al., 2004). From the point of view of the organizational and geographical configuration of the R&D function, the integral character of the R&D process is important as the less modular an activity the more difficult it is to outsource due to the difficulties and costs of coordination (Pisano, 2006).
Another important characteristic of the R&D process in pharmaceuticals is the high degree of uncertainty and failure (Wong, 2011). Despite major advances in genetics and molecular biology since the mid-1970s, it is still extremely difficult to predict how a particular molecule will work in humans (Cowlrick et al., 2011; Pisano, 2006). Moreover, drug R&D can fail for many reasons which existing scientific knowledge cannot elucidate. Test results are often inconclusive with the interpretation of results depending on the judgment of scientific teams. This means that the formal decision-making processes involves an important element of judgment based on experience (Cowlrick et al., 2011). This has important implications for outsourcing as it introduces uncertainty about the causes of failures (whether something is wrong with the molecule, or the tests have not been done appropriately).
The allocation of investment within pharmaceutical R&D is as follows. Just over 20% of total R&D costs are accounted for by discovery-research activities up to pre-clinical development whilst over 48% of total costs are represented by clinical trials from phases I to III (EFPIA, 2017). The costs of Phase III clinical trials (which represent nearly 30% of total R&D costs), marketing and sales have acted as barriers to entry that prevent contract R&D firms from becoming fully fledged, independent, pharmaceutical companies as is widely recognized in the literature (McKelvey et al., 2004; Tait, 2007; Wield et al., 2017). The costs of clinical trials Phase III, the knowledge and networks needed to navigate the regulatory process in large, advanced markets (e.g., USA and EU), and control of market channels give incumbent lead firms effective control of GVCs. However, no one lead firm can be said to be the one true lead firm governing the GVC. Instead, this is a classic multipolar GVC with these pharmaceutical giants each serving as a powerful actor in the GVC but simultaneously counterbalanced by the existence of these other pharmaceutical giants (cf. Pisano 2006; Wong 2011).
Organizational configuration of the global R&D function: Rise of outsourcing
Up to the 1980s pharmaceutical firms were fully integrated organizations with all stages of R&D value chain carried out in-house and, in most cases, in the home country. Technological advances in both biotechnology and ICT as well as global changes in regulation governing intellectual property rights—namely, extending patent protection to products as well process innovation—introduced by the World Trade Organization under the Trade-Related Aspects of Intellectual Property Rights (TRIPS) during the 1990s enabled processes of organizational and geographical decentralization. By the 1990s, for example, the automation of many routine aspects of data management was leading to the outsourcing of Phase III clinical trial activities (Howells et al., 2008; Jones, 2000).
In what represented a significant change for the industry, from the early 2000s, American and European pharmaceutical lead firms begun partitioning the discovery-research stages of the R&D value chain into smaller tasks which they then outsourced to independently owned, internationally dispersed contract R&D firms (Ramirez, 2013). Given the integral nature of the early stages of the R&D value chain, this partitioning and outsourcing of discovery-research tasks represented a significant change in the organization of the global R&D function leading to the emergence of a new global division of labor in knowledge production.
The outsourcing of discovery-research activities begun in the late 1990s when developments in ICT and biotechnology allowed the creation and testing of vast chemical libraries to be automated. Our interviews indicate that toward the end of the 1990s, pharmaceutical lead firms had begun to outsource the synthesis of basic chemicals to European-based contract R&D firms. By early 2000, these activities were increasingly being outsourced to contract R&D firms located in India and China. By 2012 (the year in which the first interviews for this study took place), routine and complex activities requiring knowledge of chemistry and biology throughout the discovery-research process were being outsourced to European, American, Chinese, and Indian contract R&D firms. Though cost reductions were important reasons for outsourcing, the main motivation for outsourcing discovery-research tasks was the flexibility this arrangement gave lead firms to undertake high-risk projects without having to invest in internal capacity. As one of the interviewees explained:
“…. [what we] want is flexibility in the budget. So, if you outsource you can turn it on or off, whereas if you have a big infrastructure, it’s a burden….”
The early 2000s saw the emergence of a number of contract R&D firms with the technological capabilities to undertake research contracts, creating a market for discovery-research outsourcing. Today the contract R&D market is composed of firms with proprietary technologies that sell very specialized services based on their own IP (their revenue is mainly the selling of services rather than the IP itself); contract R&D firms that perform a wide range of routine and complex tasks—including “problem-solving” work—but do not sell IP; and contract R&D firms that mainly perform routine-type work. A key characteristic of contract R&D firms is that, as service-providers, they will not claim ownership on any of the IP they create or help create as part of their contract with a client. On the contrary, the contract R&D firms interviewed gave several examples of organizational and management practices designed specifically to protect client’s IP. Protecting the IP of clients has been a critical element of the contract R&D business model that has enabled the outsourcing of discovery-research tasks. As the contract R&D model has evolved, a number of these firms have developed their own IP that they offer for sale to lead firms along with the services needed to turn IP into products. Other contract R&D firms, however, had consciously decided against the sale of their own IP so as not to create potential conflicts of interest with their powerful pharmaceutical clients.
The growth of discovery-research outsourcing has led to the growth of a global contract research industry where contract R&D firms from the developing world have become significant players. For example, WuXi AppTec, established in Shanghai in 2000, by 2016 had 23 R&D facilities with 10,000 employees and was performing R&D services for the top global pharmaceutical lead firms (Chemical and Engineering News, 2016). Indian supplier, Jubilant Life Sciences, employs over 988 people in R&D (Company Report, 2018).
The organizational and geographical dispersal of the discovery-research value chain represents a significant change in the global configuration the R&D function of pharmaceutical lead firms. However, although the outsourcing of discovery-research tasks is now an established practice, none of the lead firms interviewed had fully outsourced any stage of discovery-research and all firms retained in-house a fully integrated discovery-research value chain. Different therapeutic areas, however, had discretion to outsource specific tasks and/or specific projects depending on the degree of risk associated with individual product development projects, internal capacity constraints and costs. Essentially, the lead firms were manipulating GVC segmentation to retain critical, high value segments.
Outsourcing to contract R&D firms in the developing world: Capabilities upgrading
Contract R&D firms from India and China started participating in the discovery-research value chains of pharmaceutical lead firms by synthesizing their large chemical libraries. Outsourcing these tasks to contract R&D firms was possible because the knowledge underpinning these activities can be codified and it is possible to easily monitor the quality of the work produced. The early business models adopted by Indian and Chinese contract R&D firms were based on the performance of research tasks for a fee, a modular form of relationship. As the outsourcing of standardized, high-volume, low value work became more prevalent and lead firms became more experienced at managing contracts, price competition between contract R&D firms increased. In response to intensified price pressure, a number of Indian and Chinese contract R&D firms upgraded their scientific and technological capabilities in order to provide higher-value services, including problem-solving activities. As discussed above, a number of these firms have also developed their own proprietary compounds which they offer for licensing to lead firms. What we see here is the emergence of more relational forms of GVC governance: “… when we started out with the discovery business model we said “Okay, these are the capabilities we have, and this is what we can do for you”. That used to work very well … but now with so many players around it’s become a price sensitive issue and we do not want to play in a price sensitive market …. We find that when we go with our own pipeline it’s easier to attract potential partners for our programs… So that has kind of led us to build our own pipeline.” (Indian CRO 1)
As the capabilities of Chinese and Indian contract R&D firms have developed and performance pressures on pharmaceutical lead firms have intensified, several lead firms have opted to outsource specific product development projects in their high-risk therapeutic areas to these contract R&D firms. For example, one of the European lead firms interviewed had outsourced all tasks in early discovery for high-risk central nervous diseases (a therapeutic area with a high failure rate) to an Indian contract R&D firm. The Indian contract R&D firm explained the deal in the following terms:
“…there was a need by big pharma to explore some of their assets that were sitting in their cupboards and were not seeing the light of day…they found partners to look at those assets and see if we could develop candidates that were worth taking to the clinic. It’s been a process, an evolution. …there has been learning from both sides. I think pharmaceutical companies have learnt how they could extract value by partnering with CROs. The CRO model has also evolved over the years and there is now a lot of risk-sharing...” (Indian CRO2)
Indian and Chinese contract R&D firms have been able to upgrade their expertise in a number of ways including knowledge transfer within GVCs, mergers and acquisitions with companies with technological expertise, and the recruitment of scientific and managerial employees with previous experience of work in lead firms. Our interviews with pharmaceutical lead firms and contract R&D firms agree that in the initial period there was a significant amount of technical and managerial knowledge transferred from lead firms to contract R&D firms.
“Most of the folks in the synthesis space who join these companies are pretty good, so you don’t need to train them from scratch. In other areas where we’re doing more outsourcing…there’s less expertise and we’ve even helped them build that expertise so they can provide that good quality service… it’s in our interests as well as theirs.” (European pharmaceutical MNC 8).
Indian contract R&D firms interviewed acknowledge that they have developed much of their expertise working in the discovery-research GVCs of pharmaceutical lead firms, but they have only been able to benefit from this knowledge transfer because they already had strong internal scientific teams. Indian contract R&D firms have also been able to build on the strong traditions of chemistry and pharmaceutical production existing in the country.
Thus, our interviews indicate a significant upgrade in the activities performed by contract R&D firms from India and China in the discovery-research value chain of pharmaceutical lead firms in terms of the knowledge intensity and complexity of the technological and managerial tasks undertaken. Despite these significant achievements, contract R&D firms still have a dependent position within GVCs so that their paths of development are still strongly determined by the strategies of leading lead firms.
According to our interviews, pharmaceutical lead firms have retained the competencies and complementary assets to develop new drugs and bring them to market. Moreover, the considerable resources required to fund clinical trials Phase III, achieve regulatory approval and market a new drug are prohibitive for most firms. These competencies and complementary assets allow lead firms to constrain value capture by suppliers even as the latter upgrade as we will explain in greater detail in the subsequent section comparing value chains.
Integrated circuit design
Industry evolution
The IC value chain is broken down into three large blocks of activities (excluding marketing and distribution): design, fabrication, and assembly and testing. The design stage is the execution of a design idea into code that serves as the blueprint for the IC in the fabrication stage. The design stage itself consists of a number of discrete steps that will be discussed in greater detail below. The fabrication stage takes the code and, according to the code, inscribes circuitry onto physical material (typically a type of silicon) using lithography and treats the physical material with chemicals. The result of this fabrication process is the IC, but the IC is not yet complete. In the final stage, the IC undergoes: (1) assembly of its packaging that protects it and allows it to connect to other electronic components and devices and (2) testing to see if it works properly.
Since the late 1980s, the IC industry has seen a radical re-organization moving from integrated device manufacturers (IDMs) that did all the functions in-house to an industry with a classic modular structure. Most firms concentrate on one of the three main activities: design (fabless design firms), fabrication (foundries), or assembly and test (Fuller et al., 2003). Within the design function, the design flow has been segmented into discrete segments that can also be outsourced on their own (Hurtarte et al., 2007).
R&D outsourcing in China and India
In recent decades, the complexity of IC designs has increased exponentially and required commensurate engineering resources to match. To acquire sufficient engineering resources without spiraling costs, lead firms in IC design have turned to the developing world, principally China and India, to find lower wage engineers (Brown and Linden, 2009; Ernst, 2004). Some of this work has been conducted in-house by lead firms in China and India just like in the pharmaceutical industry, but the lead firms have also engaged outside contractors. In contrast, the lead firms involved in R&D for fabrication have not outsourced their R&D to contractors (Yeung, 2022) so IC design outsourcing is the best comparator to pharmaceuticals outsourcing.
From this description of the IC industry, one can see critical differences with pharmaceuticals. Although the cost barriers to designing a chip are rising (Hurtarte et al., 2007), the barriers to entry for start-ups aiming to design their own chips (as opposed to chip design for others) is much lower than pharmaceuticals where contract R&D firms face tremendous knowledge barriers to becoming full-fledged pharmaceutical companies of their own. These lower barriers have little to do with any standardization of knowledge bases in ICs. Instead, the wider GVC provides easy access to external advanced software design tools, fabrication, and assembly and testing of ICs. These various functions are not fully standardized either, but the modularity along the GVC allows start-ups to access external expertise, manufacturing, and services at relatively low cost compared to pharmaceuticals (Hurtarte et al. 2007).
In order to evaluate the upgrading of contract R&D firms, the article will first delineate the specific activities involved in designing an IC. The “design flow,” Figure 2 as these activities in total are often referred to, generally runs along the following stages. The path goes from a conception of how the chip will operate within a larger electronics system (architecture stage) through to the use of various design languages to define the circuitry moving from greater to lesser levels of abstraction [behavioral to register transfer level (RTL) to gate level design] in the process. These processes along with the architectural level are commonly referred to as front-end design. The back-end of design consists of the processes of implementation of these abstract designs into a design for real physical components and connectors embedded in silicon. Generally, the front-end is considered more technically sophisticated than the back-end (Fuller et al. 2017). As Hurtarte and his colleagues (2007: 50) put it in reference to fabless design firms, “In fact, the architecture and RTL implementation of the device is usually the core differentiating aspect of the company’s design". IC design R&D flow.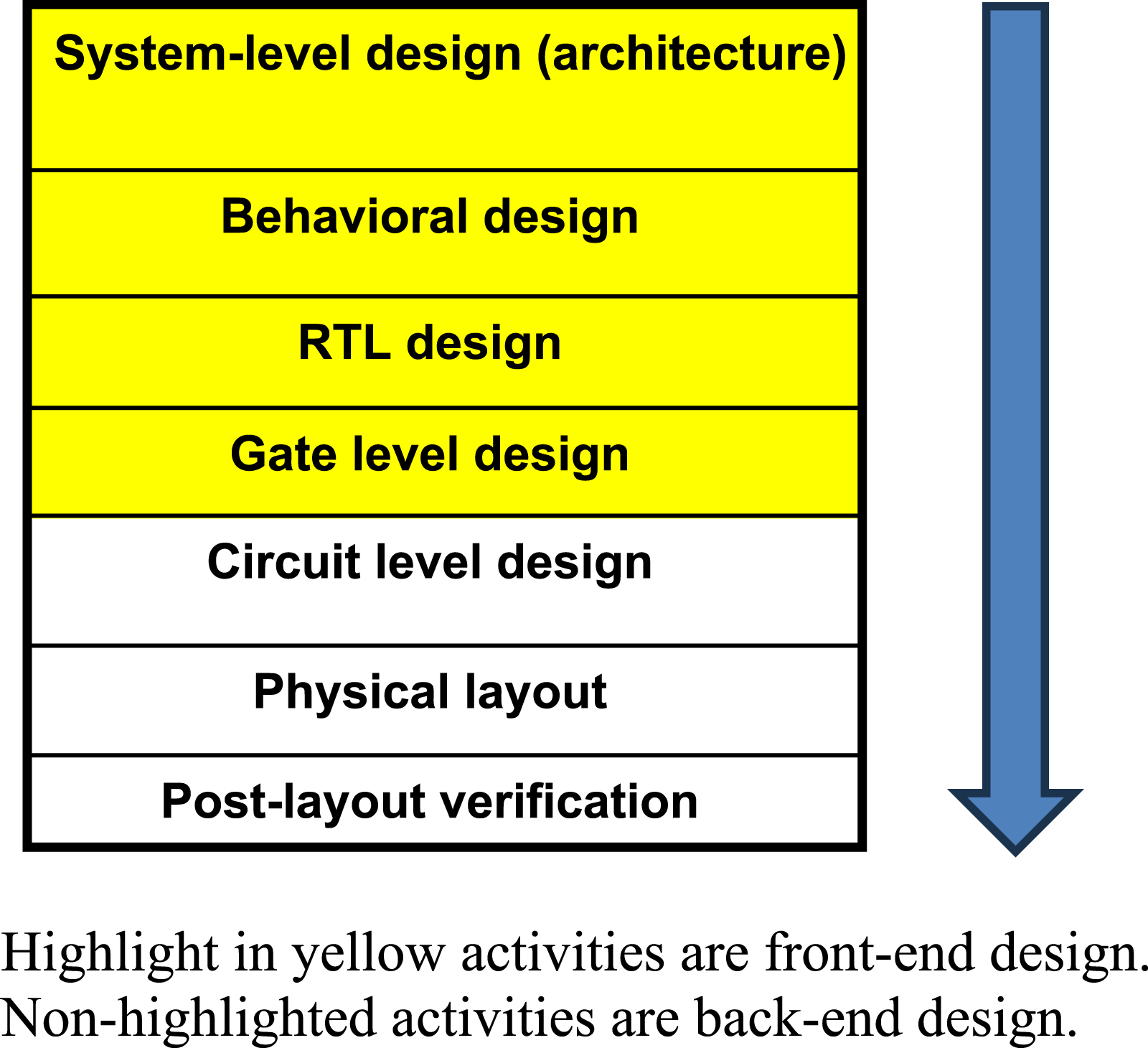
There is not just the front-end and back-end distinction. From our lead firm and design service R&D contract firms, we learned that for firms to really add and capture value from design service, they needed to create their own intellectual property (IP) that could be incorporated into various designs. Typically, such IP would be in the form of what IC designers called IP blocks, which are silicon designs representing certain functions needed for the final IC product that can fit into a larger system-on-a-chip (SOC) design, that is, a chip incorporating multiple functions.
While contract R&D firms in the initial period of 2007–2008 were hopeful that they could be able to use their contracting work to move up into more sophisticated segments of design (e.g., the front end) or even take over turnkey projects where they were given specs and had to do end-to-end design, these ambitions were largely thwarted by the lead firms. As one of the more successful contract R&D firms in that it had achieved some turnkey cases put it, “The turnkey is where the real value addition comes in. We take ownership and initiative on a milestone basis” (Design Service Firm No. 5). But the standard lament was that this milestone basis where contract R&D firms could get a cut of profits is rare, as another firm put it, “The idea that design service firms could move to a cut of revenue based on product performance has not materialized” (Design Service Firm No. 8). The same firm pointed out that turnkey alone would not really lead to higher profits because of higher costs, “Turnkey is more technically satisfying because doing [a] complete job but the money comes out similar. In some cases, could be more profitable if had IP, but if just doing turnkey, then not really more profitable.”
The fundamental issue was that in this modular value chain the lead firms could strategically outsource segments or even whole but not very profitable products that they were easing out while maintaining control over intellectual property. With this modular value chain, switching costs for the lead firms were low. This situation combined with the low barriers to entry for new design service start-ups meant that lead firms could resist profit-sharing and punish design service firms that attempted to build up their own internal intellectual property (in essence setting up a situation where they might potentially compete with their lead firm customers) by withholding contracts. As Design Service Firm Number 5 bluntly stated,” Very clear [we] do not want to get into IP, do not want to get into competition with customers. [We] Consciously stayed away from IP development and concentrated on service.” The problem with this strategy is the contract R&D firms remained dependent on the lead firms for markets and even IP to complete the designs.
All the large design service firms originally aspired to build up capabilities in IP so as to have more robust capabilities to design products, but they were all stymied. Design Firm Number 5 eventually sold itself to a lead firm. The others simply gave up trying to escape their dependence. Another firm (Design Service Firm No. 2) admitted that it too avoided developing IP because of the reaction such a move would incite in lead firm customers and complained that as the firm developed engineers, such engineers were then hired away by the lead firms, which offer higher pay. As another Design Service Firm Number 7 pointed out these IC industry lead firms were not keen to develop the IC design capabilities, especially IP creation, of their contract R&D firms because it threatened the lead firms’ core competencies and resources, “The service providers don’t capture value. Still caught in research augmentation mode. Probably because lots of semiconductor companies view IC design as their core competency.” Ultimately, none of the contract R&D firms were aiming to build their own IP and even milestone payments for full design projects were a small fraction (interviewees’ estimate of 10 percent) of the revenues for the largest, most sophisticated design service firms.
From the lead firm-supplier relationships delineated above, we can see that there are several mechanisms for lead firms to be able to contain technological upgrading and, more importantly, value capture by contract R&D firms. First, lead firms helped to encourage entry into the design service segment by providing electronic design automation (EDA—the software to design ICs) tools and computing equipment to the design service contract R&D firms. EDA tools are the most expensive input for these contract R&D firms, so the lead firms effectively lowered the barriers to entry into design service. Second, the lead firms always kept some design operations in-house, particularly the valuable architecture-level design, and when they wanted to expand design operations, they often simply acquired contract R&D firms. Acquisition of contract R&D firms encouraged more entrepreneurs to enter this market because the lead firms provided a clear exit path for investors in these contract R&D start-ups. These lead firm actions created a large pool of contract R&D firms struggling to obtain lead firm customers’ orders (i.e., dependency on the lead firms) at the same time that the lead firms kept in-house design operations, so they had very little dependency on the design service firms. In this situation, lead firms could easily dissuade design service firms from bargaining too hard even when they did upgrade to turnkey solutions and effectively discourage them from developing their own IP.
The issue of product upgrading in the context of IC design (switching from the design of one product to the design of another product) was a non-issue for these suppliers. They took part in the design of many different products of theoretically more or less value. However, these contract R&D firms, no matter the product they were helping to design, were confronted with lead firms that would either not let them do the more technically sophisticated front-end tasks/IP creation or, when they did, would not actually allow them to share more in the profit because the lead firms had alternative suppliers. The IC lead firms only turned over turnkey, complete design flow projects to contract R&D firms in lower profit, mature product areas as both lead firms and design service firms acknowledged.
Dindial et al. (2020) argue that suppliers from the developing world can escape dependency from lead firms and thereby capture value by clustering and through government support. In the case of IC design, the industry heavily clustered in two cities, Bangalore and Shanghai, in India and China, respectively, and received very proactive industrial policy in China. Despite this clustering and proactive policy, design service contract R&D firms in both India and China were unable to capture much value.
Comparing value chains: Pharmaceuticals versus integrated circuits
Much of the GVC literature assumes that relational and modular linkages provide better opportunities for upgrading and value creation for supplier firms (e.g., Gereffi et al., 2005; Pietrobelli and Rabellotti 2011; Lechner et al., 2020) although scholars like Schrank (2004) and Sako and Zylberberg (2019) have pointed out that value creation does not automatically mean value capture for suppliers. Turning to the two cases of contract R&D firms’ role in GVCs, for pharmaceuticals, we find that the relational linkages represent higher upgrading/technical sophistication than modular ones which are technically more sophisticated than market linkages. Nevertheless, lead firms first control upgrading and value capture for some lower risk activities by purposely choosing to engage in modular and market linkages in order to ensure valuable knowledge is not unnecessarily leaked to the supplier firms. For these suppliers, upgrading and thus consequently value creation and capture are constrained. In high-risk areas, lead firms acquiesce to relational linkages, which embody more upgrading, and thereby open up opportunities for some concomitant value creation and capture by suppliers. However, lead firms via their control of market channels still constrain the upside of suppliers’ value capture in spite of upgrading in these high-risk areas.
In contrast to the significant role of relational linkages in pharmaceuticals, modular linkages are the dominant mode for ICs. Suppliers may be able to combine several steps in the design flow together, but, contrary to the findings in other studies (Dindial et al., 2020; Lechner et al., 2020), such integration of segments together does not provide value capture. Lead firms are able to dictate what functions they allow suppliers to enter, and they generally choose to keep technically sophisticated, high value design segments in-house. Even when they allow contract R&D firms to do the most sophisticated, whole design flow “turnkey” projects, that is, integration of the segments, lead firms are able to reject any requests for profit sharing. Lead firms have cultivated many capable design service suppliers so they can use this surfeit of suppliers to dissuade design service firms from hard bargaining and forays into IP creation.
Navas-Aleman (2011) suggests that supplier firms that engage in multiple GVCs can have better opportunities for upgrading. However, in this paper’s IC case, there are a number of design service firms that are part of large business-processing and knowledge-processing firms spanning several GVCs. Unfortunately, these multi-chain firms have the same outcomes in the IC design space in terms of upgrading and value capture as the other single GVC suppliers. Lead firms have strategically chosen which segments to outsource and encouraged a large pool of design service contract R&D firms in order to gain so much leverage over suppliers that even potential advantages such as being multi-chain suppliers does not yield better upgrading and value capture results.
In terms of overall value chain governance, the pharmaceutical GVC is characterized by multipolar governance with the major lead firms exercising significant power over their suppliers, and government regulators in major markets are also very powerful actors in this industry. The typical entities driving the pharmaceutical GVC are government regulatory bodies and multiple major lead firms (Pisano, 2006; Pisano and Teece, 2007; Wong, 2011). For the IC industry, the value chain governance is also multipolar as multiple corporate actors (lead firms designing and marketing ICs, lead firms as foundries manufacturing ICs and major buyers of ICs such as Apple) exercise significant influence over the IC GVC (Fuller et al., 2003; Hurtarte et al., 2007; Yeung, 2022). From the vantage point of the IC design contract R&D firms, their customers might be chip-only businesses (e.g., Intel, Qualcomm, and Micron) or end-product firms beginning to expand into chip design (e.g., Apple). In either case, the suppliers are still in a GVC with a number of powerful actors. For example, Apple, as a powerful firm engaging many suppliers to design numerous ICs for its products, still has to deal with other powerful firms in the IC GVC, such as TSMC and Samsung as foundries and rival end-product makers that design and consume large quantities of ICs (e.g., Samsung and Xiaomi).
Bipolar or multipolar governance mechanisms are posited as the environments in which suppliers can best upgrade and capture value (Raj-Reichert, 2019; Sako and Zylberberg, 2019). Our cases suggest that even in multipolar value chains, contract R&D service suppliers may find it difficult to capture value or even technical upgrading because of lead firms’ own strategies. In pharmaceuticals, suppliers can upgrade technically but benefit much less so in terms of value capture. In ICs, the lead firms carefully manage the modular linkages and their own bargaining leverage either to prevent suppliers from upgrading into higher stages of design or to suppress their value capture when they do upgrade.
While the nature value chain linkages and value chain governance mechanisms provided opportunities to transfer technology and knowledge, lead firms’ strategies loomed large in determining the constraints and opportunities for developing world contractors to upgrade their capabilities and increase value capture in these value chains. The strategies of the lead firms are informed by the riskiness and uncertainty of product development, and the regulatory and marketing barriers for market entry. The pharmaceutical industry compared to ICs faces much greater uncertainty in terms of whether R&D will actually yield a successful commercial product (Wong, 2011) and thus faces much greater risks in R&D. Thus, even though in both technology-intensive industries, R&D can be considered a core capability and resource of participating lead firms, risk management by lead firms in pharmaceuticals incentivized lead firms to outsource even some potentially high value-added capabilities. In pharmaceuticals, the high-risk disease areas would be unlikely to be funded internally so lead firms outsource these tasks which would otherwise go unfunded due to the high risk leading to lower expected value for these projects (expected value being the likelihood of success multiplied by the estimated value of success). However, due to the high regulatory and marketing barriers to bring products to markets, the lead firms utilize their superior knowledge of how to deal with the other major value chain governance actor, the national market regulators. It is well recognized in the literature that the very high costs of clinical trials Phase III, the regulatory stages that requires the networks and political contacts with regulators and the huge marketing budget (which is bigger than the R&D budget), are effective barriers to market for contract R&D firms (McKelvey et al., 2004; Pisano, 2006; Tait, 2007; Wield et al., 2017). Since lead firms control the path to market, the danger of transferring too much knowledge and capabilities to the suppliers is reduced. This relationship between the lead firms and the regulators in major markets and the knowledge the lead firms have of how to navigate these regulatory environments represent what Sako and Zylberberg (2019) invoking Pisano (2006) call co-specialized complimentary assets with mutual dependence. Sako and Zylberberg (2019) recommend that suppliers develop such complimentary assets, but in this case, the lead firms control them for the lucrative developed world markets. Given the enormous potential size of their respective home markets, Chinese and Indian firms may be able to develop such valuable complimentary assets for their home markets.
To elaborate on the market regulations in pharmaceuticals, extensive product regulation in advanced economies gave lead firms with their superior knowledge of how to navigate these regulatory systems leverage over the contract R&D firms, which remain dependent on the lead firms to gain access to these advanced markets. Thus, even while transferring technical and managerial knowledge and encouraging capability development among the developing world contractors, the pharmaceutical lead firms are able to block the contractors from the critical Phase III R&D due to their superior knowledge of the regulations, marketing channels and the considerable upfront investment costs for both marketing and Phase III clinical trials (McKelvey et al., 2004; Pisano, 2006; Tait, 2007; Wield et al., 2017).
In contrast, for lead firms in IC design with its more certain R&D outcomes, lead firms managed their outsourcing strategies to minimize knowledge transfer to prevent relative erosion of their core competitive capabilities. Moreover, the regulatory barriers to market entry are much lower in ICs so lead firms cannot hope to utilize these regulations to prevent highly capable design service suppliers from bringing their own products to market as direct competitors to the lead firms. Thus, the lead firms tried to limit the upgrading of their suppliers. They did so by discouraging contract R&D firms from developing their own IP, lowering the barriers to entry for design service and fostering dependence of those same contract R&D firms by sharing EDA tools and computing equipment with their suppliers, and cherry-picking the best talent that the contract R&D firms had trained to incorporate into their own internal operations.
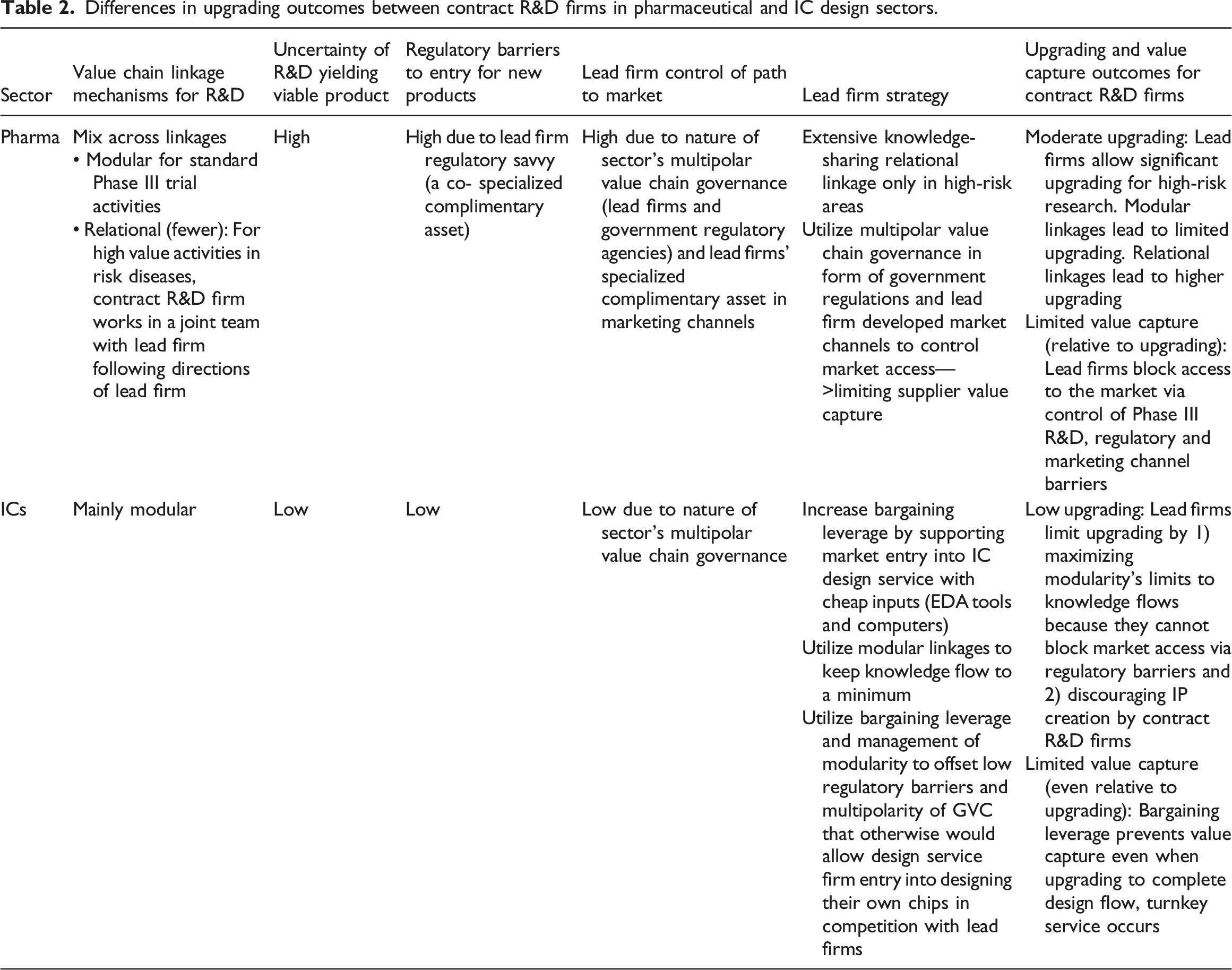
Differences in upgrading outcomes between contract R&D firms in pharmaceutical and IC design sectors.
Conclusion: Value chain characteristics and R&D upgrading
Comparing the role of external R&D suppliers in the R&D processes of two technology- and knowledge-intensive value chains, this paper demonstrates how GVC linkages and GVC governance (multipolarity) by themselves exhibit a fair amount of indeterminacy in predicting both upgrading and value capture outcomes. While past scholarship has emphasized the role of government policies and national systems of innovation (Dindial et al., 2020; Pietrobelli and Rabellotti 2011) in order to explain the differential outcomes for GVC characteristics or GVC engagement, this article builds upon the turn toward firm strategy as the focus of GVC analysis pioneered by Sako and Zylberberg (2019). Our article goes further than their model in emphasizing the indeterminacy of linkage characteristics even in determining upgrading outcomes (Sako and Zylberberg accept strong determinacy between linkage characteristics and upgrading if not value capture). Furthermore, while Sako and Zylberberg (2019) join others (Lechner et al., 2020; Ma et al., 2022; Raj-Reichert, 2019) in placing powerful suppliers at the forefront of GVC analysis and thus emphasize suppliers’ strategies, this article considers how lead firms can employ strategies to counteract unfavorable (from their point of view) linkage characteristics and multipolarity in order to ensure they retain most of the value capture. This is not simply a contribution based on considering lead firm rather than supplier strategy. This article considers how strategy counteracts GVC structures whereas recent strategy-oriented GVC literature examines how strategy helps suppliers choose or switch to more favorable GVC structures (Sako and Zylberberg 2019) or change the GVC structures through capability-building (Lechner et al. 2020; Ma et al. 2022).
Going forward, building upon both the pessimists about supplier upgrading (e.g., Dallas, 2015; Mahutga, 2012; Naseemullah, 2017; Schrank, 2004) and the scholars emphasizing rising suppliers (Lechner et al., 2020; Ma et al., 2022; Raj-Reichert, 2019; Sako and Zylberberg, 2019) in the developing world, GVC analysis needs to embrace a more dynamic model of firm strategizing incorporating both the strategies of lead firms and supplier firms in the context of the structural issues each type of firm faces in a given GVC. Sako and Zylberberg have been very helpful in proposing additional metrics to consider in the attractiveness of a given GVC structure (appropriability, technology stage, and complementarity channels). This article adds a strategic dimension on how lead firms maneuver to limit upgrading and/or value capture within a given GVC structure. Future work should look at counter-strategies by suppliers to counteract these lead firm tactics, and examine the interactions of public policy and strategies of both lead firms and developing world suppliers.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the FP7 Socio-Economic Sciences and Humanities; Grant agreement ID: 244552.
Correction (October 2023):
Article updated to add yellow highlights in Figure 2.
