Abstract
Background
Duloxetine is a potent analgesic that reduces pain associated with knee osteoarthritis. This study aimed to evaluate the analgesic effects of regular postoperative duloxetine after high tibial osteotomy (HTO) and its influence on knee function, pain severity, and daily life.
Methods
This open-label randomised controlled trial was designed as a single-centre, parallel-group study. Patients undergoing HTO were randomly allocated to the control and duloxetine groups. Patients allocated to the duloxetine group were administered oral duloxetine for the first 12 weeks postoperatively and were followed up for 6 months following surgery. The primary outcome was pain assessed by the visual analogue scale (VAS), and secondary outcomes included evaluations using the Brief Pain Inventory, the Knee Society Score, and the 2011 Knee Society Score.
Results
A total of 38 patients in the duloxetine group and 37 patients in the control group were analyzed. The duloxetine group demonstrated significantly lower pain VAS scores at rest 6 months postoperatively compared with the control group (0.5 ± 1.0 vs 1.3 ± 2.0, respectively; p = 0.029). There were no significant differences between the groups in the other scores. However, patients in the duloxetine group showed significantly lower consumption of acetaminophen in the third postoperative week (0.0 ± 0.0 vs 0.3 ± 0.8 tablets/day, respectively).
Conclusions
Duloxetine demonstrated a modest analgesic benefit following HTO, with reduced pain at rest at 6 months and decreased rescue medication use during the third postoperative week. Routine use after HTO may therefore not be warranted in unselected patients. Due to the open-label design, performance and detection bias cannot be excluded.
Trial registration
The study protocol was registered in the UMIN clinical trial registration before starting recruitments: UMIN000040026 (registration date: June 1, 2020) https://center6.umin.ac.jp/cgi-open-bin/ctr_e/ctr_view.cgi?recptno=R000045630.
Keywords
Introduction
Osteoarthritis of the knee (KOA) presents with a complex pain profile, including neuropathic pain (NP), nociceptive pain, and nociplastic pain. 1 Among these, NP and nociceptive pain have been the most extensively studied. 2 Although the underlying mechanisms of NP in KOA remain unclear, it is thought to be caused by repeated mechanical stimulation and chronic inflammation affecting nerve endings in the subchondral bone, synovium, and meniscus.3–7 NP not only imposes physical and psychological distress in patients with KOA but also impairs functional recovery and quality of life following total knee arthroplasty (TKA). 8
Duloxetine is a potent selective serotonin and norepinephrine reuptake inhibitor that increases the synaptic concentrations of these neurotransmitters by inhibiting their reuptake in the presynaptic neuron and amplifying the transmission of descending inhibitory pain pathways in the central nervous system.9–11 Several studies have reported that duloxetine reduces NP and improves quality of life in patients with KOA, diabetic neuropathy, and spinal disorders.12,13
High tibial osteotomy (HTO) is an established joint-preserving surgical alternative to TKA in KOA.14,15 In contrast to TKA, which removes the lesion and places an implant in the knee joint, HTO preserves the knee joint, including the lesion, and corrects lower limb alignment outside the joint.14,15 Therefore, it is possible that preexisting NP is involved in postoperative pain following HTO and HTO itself may induce NP postoperatively. Duloxetine may have potential benefits in managing NP following HTO, and investigating its effect could enhance our understanding of pain mechanisms in this context and help optimise postoperative pain management.16,17
The aim of this study was to determine the efficacy of regular postoperative administration of duloxetine for 12 weeks following HTO to reduce postoperative pain. The secondary outcomes included improvements in objective and subjective knee function, pain severity, and daily life. This study was conducted as a randomised controlled trial.
Materials and methods
Study design and patient recruitment
This open-label, randomised controlled trial was designed as a single-centre, parallel-group study, enrolling 82 patients scheduled for HTO between July 2020 and September 2022. The study was approved by the institutional review board and registered with the UMIN Clinical Trials Registry (UMIN000040026, https://www.umin.ac.jp/ctr/index.htm). Written informed consent was obtained from all patients before participation, and documentation explaining the effects of duloxetine, including its analgesic effects on the central nervous system and side effects related to the central nervous system and gastrointestinal tract, was provided.
Eligible patients were aged ≥40 years and scheduled to undergo primary unilateral HTO for KOA, osteonecrosis, or subchondral insufficiency fracture of the knee. 15 Exclusion criteria were patients with prior duloxetine use within 1 month of consent for this clinical trial, American Society of Anesthesiologists class IV or higher, myocardial infarction, psychiatric or organic brain disorders, a history of coronary artery disease, unstable angina, heart failure, kidney failure, hepatic failure, pregnancy, breastfeeding, poorly controlled angle-closure glaucoma, intolerance or allergy to study drugs, administration of monoamine oxidase inhibitors within 2 weeks before enrolment, aspirin-induced asthma, or patients who would be unable to undergo 3 weeks of post-operative rehabilitation and take medication while hospitalized.
Patients were randomised into either the control or duloxetine group using a computerised, random allocation by a clinician not involved in the clinical analysis.
The basic examination items included age, sex, body mass index, the presence or absence of NP and central sensitisation (CS), and radiographic examination (Kellgren–Lawrance grade and Hip-knee-ankle angle). 16 Possible NP was defined as PainDETECT ≥14 points and CS was defined as Central sensitization index ≥40 points.16,17
Power analysis
The primary outcome measure was the pain visual analogue scale (VAS) scores at rest (rVAS) and on motion (mVAS), and the sample size calculation was based on detecting a mean difference of 2 points in the pain VAS between groups. 18 Using a two-sided hypothesis test with an alpha level of 0.05 and a power of 80%, 37 patients per group were required. Each group ultimately included 41 patients to allow for both dropouts and exclusions.
Surgical procedure and pain management
A femoral nerve block with 40 mL of 0.25% ropivacaine was performed prior to surgery. During surgery, pain was managed with intravenous administration of fentanyl 1–2 μg kg−1. A tourniquet was inflated to 100 mmHg above systolic blood pressure during the surgery. HTO with distal tibial tubercle osteotomy was performed by a single experienced surgeon using a standard technique.14,15 Before skin closure, a modified version of previously reported periarticular cocktail (0.5% ropivacaine 100 mg, ketoprofen 50 mg, methylprednisolone 40 mg, and tranexamic acid 1 g diluted with saline to 80 mL) was administered into the soft tissue around the capsule for perioperative pain control.19,20 No suction drains were used. Postoperatively, all patients received the standard pain management protocol in our institution, including oral celecoxib: 200 mg for the first week, and 100 mg for the second and third weeks, every 12 h, and oral acetaminophen (500 mg) was used as a rescue dose for unbearable pain. No patient-controlled intravenous analgesia was used. In addition, patients allocated to the duloxetine group received oral duloxetine: 20 mg once daily after breakfast for the first week, 40 mg for the second week, and 60 mg from the third to the twelfth weeks. Regarding compliance monitoring for duloxetine, nurses managed the number of tablets during hospitalization. After discharge, patients themselves managed their medication, which was monitored monthly by outpatient physicians. The use of acetaminophen as rescue medication was recorded in nursing records during hospitalization. However, after discharge, rescue medication use was not recorded. There were no restrictions on the range of knee motion after surgery, and patients were allowed partial and full weight-bearing in the second and third weeks after surgery, respectively.14,15 All patients were discharged on postoperative day 21.
Outcome measurements
The primary outcome was pain at rest and during motion, assessed by two independent physiotherapists preoperatively and at 1, 3, and 5 days; 1, 2, and 3 weeks; and 2, 3, and 6 months postoperatively, using rVAS and mVAS (scores 0–10, with 0 indicating no pain and 10 indicating the most severe pain). Secondary pain outcomes included the Brief Pain Inventory (BPI; pain severity and interference scores) assessed at the same time points. 21 Secondary outcomes measures included the Knee Society Score (KSS; knee and function scores) and the 2011 Knee Society Score (KSS2011) recorded preoperatively and at 6 months postoperatively, 22 acetaminophen consumption, and adverse events related to duloxetine, as reported by the drug manufacturer.
Statistical analysis
All analyses were performed using R software R (version 4.5.1; R Foundation for Statistical Computing, Vienna, Austria) and GraphPad Prism version 9.0 (GraphPad Software, La Jolla, CA, USA). Student’s t-test or the Wilcoxon signed-rank test was used to analyse continuous variables, and the chi-squared test or Fisher’s exact test was used to determine differences in categorical variables. The primary analysis was conducted according to the intention-to-treat (ITT) principle, including all randomized patients in their originally assigned groups. A per-protocol (PP) analysis excluding patients who discontinued the study or were deemed ineligible after randomization was performed as a sensitivity analysis.
Longitudinal changes in rVAS and mVAS were analyzed using a mixed-effects model for repeated measures (MMRM). The model included fixed effects for treatment group, time point, and the group-by-time interaction, with baseline VAS as a covariate. A random intercept was specified for each participant to account for within-subject correlation. Missing data due to dropout were handled under the missing-at-random assumption without ad hoc imputation. Two-sided p values <0.05 were considered statistically significant.
The minimum clinically important difference (MCID) was calculated for rVAS and mVAS, BPI subscales, and KSS2011 subscales at 6 months following surgery. The distribution-based method was used, and MCID was defined half the standard deviation of the difference between the preoperative and postoperative scores for each questionnaire.23–25
Results
Patients
Of the 88 patients enrolled, two did not meet the inclusion criteria in the study. Thus, 88 patients were randomly assigned to either the duloxetine group (n = 42) or the control group (n = 44) using computer-generated randomisation. There were seven patients excluded in the control group due to unrelated disease (n = 1) and deviation from the protocol (n = 6), and 4 excluded in the duloxetine group due to discontinued intervention due to adverse event (n = 1), unrelated disease (n = 1), and deviation from the protocol (n = 2). As a result, 37 patients in the control group and 38 in the duloxetine group completed the trial (Figure 1). Patient characteristics are summarised in Table 1. Flow diagram depicting study protocol and patient selection. Baseline characteristics of patients in the control and duloxetine groups. Values are presented as mean ± standard deviation or percentage (number), unless otherwise specified.
Pain VAS (ITT analysis)
Mixed-effects model for repeated measures (MMRM) analysis of rVAS and mVAS under the intention-to-treat principle.

The model included fixed effects for group, time, and group-by-time interaction, with baseline VAS as a covariate and a random intercept for each participant.
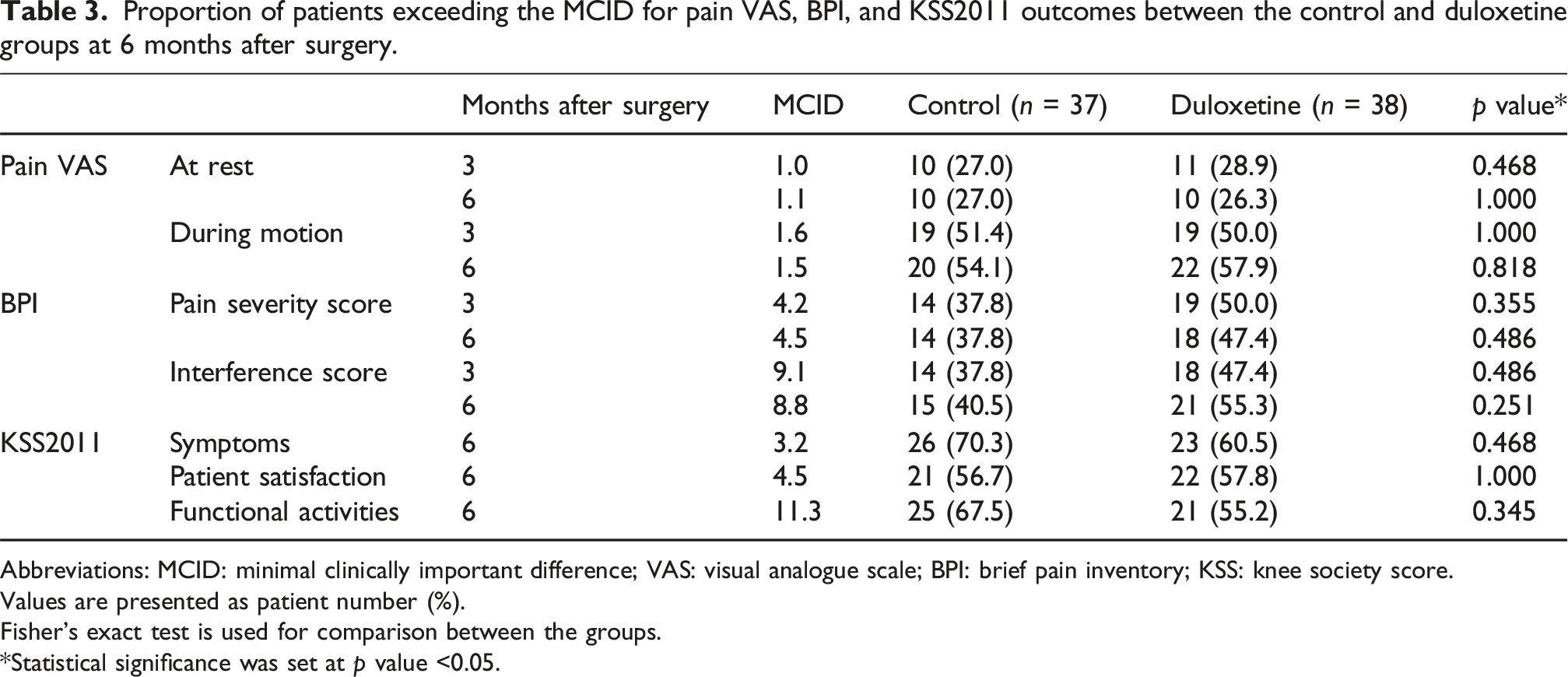
At the 6-months time point, patients in the duloxetine group reported significantly lower rVAS scores compared with the control group (mean 0.5 ± 1.0 vs 1.3 ± 2.0, respectively; p = 0.029, Figure 2). However, the proportion of patients achieving the MCID for rVAS and mVAS did not significantly differ between groups at either 3 (p = 0.468 and 1.000, respectively) or 6 months (p = 1.000 and 0.818, respectively) postoperatively (Table 3). Comparison of pain VAS scores at rest (rVAS) and during motion (mVAS) between the control (CTL) and duloxetine (DLX) groups. Abbreviation: VAS: visual analogue scale; Pre-op: pre-operative; d: days; w: weeks; m: months. Proportion of patients exceeding the MCID for pain VAS, BPI, and KSS2011 outcomes between the control and duloxetine groups at 6 months after surgery. Abbreviations: MCID: minimal clinically important difference; VAS: visual analogue scale; BPI: brief pain inventory; KSS: knee society score. Values are presented as patient number (%). Fisher’s exact test is used for comparison between the groups. *Statistical significance was set at p value <0.05.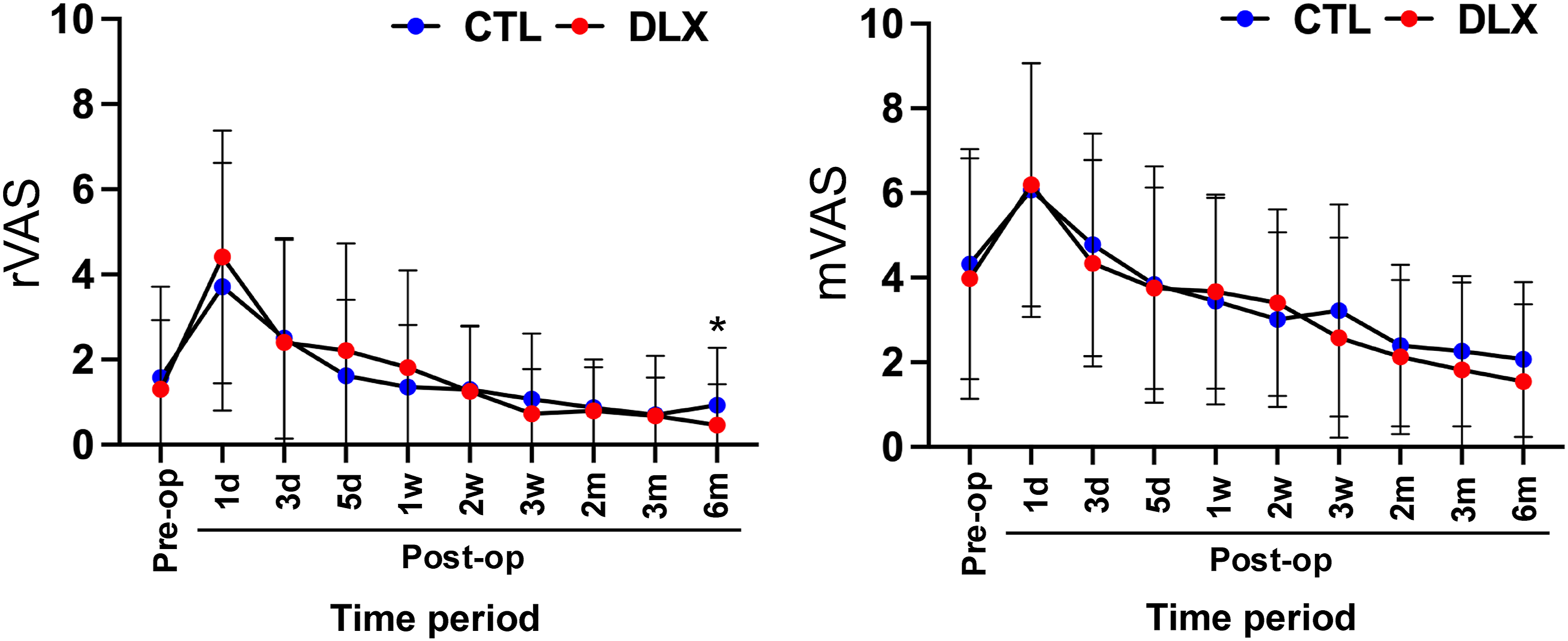
Pain VAS (PP analysis)
The PP analysis, excluding patients who discontinued the study or were deemed ineligible after randomization, yielded results consistent with the ITT analysis. No significant group-by-time interaction was observed for either rVAS or mVAS, confirming the robustness of the primary findings.
BPI
There were no significant between-group differences in BPI scores, including both pain severity and interference scores, although both groups showed improvement from baseline (Figure 3). The percentages beyond the MCID for the BPI (pain severity and interference scores) did not significantly differ between the groups at 3 (p = 0.355 and 0.486, respectively) or 6 months (p = 0.486 and 0.251, respectively) following HTO (Table 3). Comparison of BPI pain severity and interference scores between the control (CTL) and duloxetine (DLX) groups. Abbreviations: BPI: brief pain inventory; Pre-op: pre-operative; d: days; w: weeks; m: months.
KSS and KSS2011
Comparison of KSS and KSS2011 between the control and duloxetine groups.
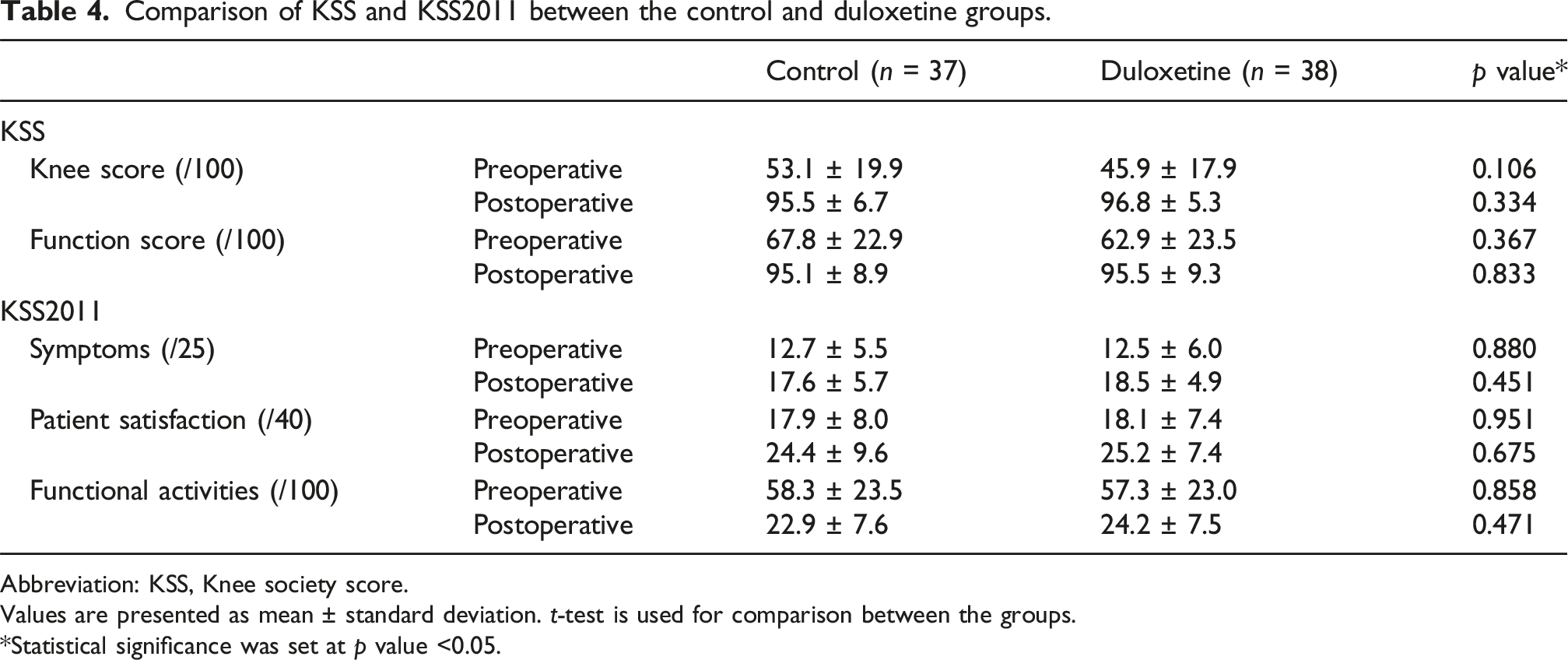
Abbreviation: KSS, Knee society score.
Values are presented as mean ± standard deviation. t-test is used for comparison between the groups.
*Statistical significance was set at p value <0.05.
Rescue dose (acetaminophen 500-mg tablets)
Acetaminophen tablet (500 mg) intake frequency (times) during the postoperative period in the control and duloxetine group.

Abbreviation: h: hours.
Values are presented as mean ± standard deviation. t-test is used for comparison between the groups.
*Statistical significance was set at p value <0.05.
Complication
There was one case of nausea in the duloxetine group, but it spontaneously resolved after discontinuation of duloxetine. No other serious adverse events occurred.
Discussion
One of the key findings of this randomized controlled trial is that duloxetine did not significantly alter the overall longitudinal trajectory of postoperative pain. Although patients in the duloxetine group reported lower rVAS at 6 months postoperatively, this difference was not accompanied by a higher proportion of patients achieving the MCID. Additionally, duloxetine was associated with reduced rescue medication use during the third postoperative week in patients undergoing HTO compared with usual treatment.
Exercise-based therapy in combination with pain education and drug therapy forms the basic treatment for KOA. The 2019 Osteoarthritis Research Society International non-surgical guidelines conditionally recommend duloxetine for patients with multi-joint OA and/or comorbidities such as depression. 26 In several randomised controlled trials, regular administration of duloxetine to patients with KOA has shown efficacy in reducing pain, regardless of a diagnosis of NP or CS.12,27–32 However, in this randomized controlled trial, the ITT analysis showed no significant difference in longitudinal VAS trajectories between the groups. Although pain improved over time in both groups, the absence of a group-by-time interaction indicates that the intervention did not confer additional benefit in subjective pain reduction. This discrepancy may stem from differences between the current study population that underwent HTO and the general KOA population on whom duloxetine has typically been tested. Another reason for this discrepancy may be attributed to the proportion of patients with NP or CS among the study population. While approximately 20% of patients with KOA are reported to have features of NP and CS,33,34 the proportion in the current study was lower (PainDETECT ≥14 points: 5.4% in the control group and 13.2% in the duloxetine group; Central Sensitisation Inventory ≥40 points: 8.1% in the control group and 7.9% in the duloxetine group). Thus, in our hospital, patients with NP or CS may have been more likely to avoid surgery through pharmacological management, including agents targeted at NP and CS. Therefore, while duloxetine may be effective in patients with NP or CS,35,36 the small proportion of such patients in this cohort may have limited the detection of between-groups differences. Interestingly, Otsuki et al. 27 reported that perioperative duloxetine administration—initiated 2 weeks before surgery and continued for 2 weeks postoperatively at a fixed dose of 40 mg/day—significantly reduced early postoperative pain and NSAID consumption. This differs fundamentally from the present study, in which duloxetine was initiated only after surgery and titrated to 60 mg over 12 weeks. The preoperative initiation in Otsuki’s protocol may have allowed sufficient time for activation of descending inhibitory pathways before surgical insult, potentially attenuating CS during the acute postoperative phase. In contrast, postoperative-only administration in the current trial may have limited its capacity to modulate acute nociceptive input immediately after HTO. Therefore, differences in treatment timing and perioperative exposure may explain the discrepancy between the two studies.
Another possible explanation for the limited analgesic effect of duloxetine is the type of postoperative pain. The most common pain types in KOA are nociceptive pain and NP, 2 and different mechanisms may respond to different medications. 37 In the current study, duloxetine may have been less effective because nociceptive pain may have been more primary than nociceptive pain due to an acute response to surgical insult, including osteotomies, for the first 2 weeks after surgery. However, during the third postoperative week, when patients typically begin full weight bearing in rehabilitation, the duloxetine group required fewer rescue doses of acetaminophen, suggesting that NP may be associated with pain during this phase. Additionally, the significantly lower rVAS at 6 months in the duloxetine group suggests that the administration of duloxetine for 3 months may have exerted a desensitising effect on neural circuits, producing sustained relief from NP till 6 months postoperatively. These findings suggest that duloxetine may be more beneficial in the subacute phase, when NP replaces nociceptive pain as the dominant pain type. However, Taken together, it is important to determine whether the pain is primarily NP or nociceptive for selecting appropriate pharmacological management.
However, diagnosing NP in patients with KOA remains challenging, as the underlying mechanisms are not yet completely understood. OA is characterised by cartilage degeneration, synovitis, bone marrow lesion, osteophyte, and joint stiffness. However, there are poor associations between the radiological assessment of KOA and the clinical symptoms. 17 In contrast to articular cartilage, the synovium membrane, Hoffa’s fat pad, meniscus outer region, and subchondral bone are highly innervated by nociceptors, and inflammation in these structures (e.g., synovitis) is associated with clinical pain 38. It has also been reported that NP does not correlate with the MRI-based OA Knee Score. 17 Thus, to diagnose NP, subjective rather than objective assessments are necessary. The PainDETECT, 39 Douleur Neuropathique 4 questionnaire, 40 Neuropathic Pain Questionnaire, 41 and Neuropathic Pain Scale 42 are widely used scoring systems to differentiate NP in patients with KOA. Similarly, CS, which is also associated with KOA pain, should be assessed using instruments such as the Central Sensitisation Inventory. 43
This study has some limitations that must be acknowledged. First, First, the prevalence of possible NP and CS in our cohort was relatively low. Because duloxetine primarily targets NP mechanisms, 35 the limited proportion of patients with NP or CS features may have attenuated the observable treatment effect. In particular, only a small number of patients met commonly used thresholds for neuropathic pain or central sensitization (e.g., PainDETECT ≥14 or CSI ≥40), rendering any post hoc subgroup analysis statistically underpowered and potentially unreliable. Therefore, we did not perform additional subgroup analyses. Future studies employing stratified randomization or enrichment designs targeting patients with established NP or CS characteristics may better clarify the therapeutic role of duloxetine following HTO. Second, because of the lack of blinding in the duloxetine group, there is a possibility of placebo or nocebo effects, especially during and shortly after the intervention period. However, these effects are unlikely to have influenced the primary endpoint at 6 months following HTO, when the difference in rVAS was statistically significant. Third, the effects of concomitant treatments, including the use of acetaminophen as a rescue medication and physical therapy received for pain management after discharge, were not analyzed and may have influenced pain assessments. Notably, a significant difference in rescue medication use between the two groups was observed during the third postoperative week prior to discharge. Therefore, continued monitoring of rescue medication use after discharge would have been important to better understand the sustainability of this difference. However, such post-discharge tracking was not performed, which represents a limitation of the present study. Fourth, this study may have been underpowered for subgroup analysis because patients with OA and osteonecrosis or subchondral insufficiency fracture of the knee were included, which may have influenced the results. Fifth, to examine the efficacy of duloxetine, a subgroup analysis of patients with NP may be useful; however, the proportions of such patients was too small to provide sufficient statistical power. Further studies are warranted to investigate the efficacy of duloxetine for pain management in patients undergoing HTO. Lastly, due to the open-label design, performance and detection bias cannot be excluded. Future double-blind, placebo-controlled trials are warranted.
Conclusion
This study demonstrated that routine postoperative administration of duloxetine did not significantly modify the overall longitudinal trajectory of postoperative pain following HTO. Although a modest reduction in pain at rest was observed at 6 months, this was not accompanied by a higher proportion of patients achieving clinically meaningful improvement. Duloxetine was associated with reduced rescue medication use during the early postoperative period; however, its overall analgesic benefit appeared limited.
These findings suggest that routine administration of duloxetine after HTO may not be warranted in unselected patients. Given the relatively low prevalence of preoperative NP in this cohort, duloxetine may be more appropriately considered in patients with established NP features. Further studies are needed to clarify the optimal patient selection and timing of duloxetine therapy following HTO.
Footnotes
Author notes
Regular postoperative administration of duloxetine has a limited analgesic effect on postoperative pain after high tibial osteotomy: An open-label, randomised controlled trial.
Ethical considerations
This randomized controlled trial was approved by the institutional review board of Ogaki Tokushukai Hospital (TGE01451-066).
Consent to participate
Patients were accepted into the study after obtaining informed consent from them or their relatives based on voluntariness. The study protocol followed the tenets of the Declaration of Helsinki. Our study adheres to CONSORT guidelines.
Author contributions
Hiroyasu Ogawa was responsible for the organisation and coordination of this study. All authors contributed to the management of this study and the acquisition, analysis and interpretation of the data. All the authors have approved the manuscript for publication.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
