Abstract
Purpose
This study aimed to investigate changes in sarcopenia status and physical function after total knee arthroplasty (TKA) in patients with knee osteoarthritis (KOA).
Methods
This prospective observational study was conducted at a single general hospital. Seventy-one patients with KOA undergoing first TKA were classified into sarcopenia (n = 12) and non-sarcopenia (n = 59) groups based on the 2019 Asian Working Group for Sarcopenia criteria. Outcomes including walking speed, grip strength, skeletal muscle mass index (SMI), Knee Injury and Osteoarthritis Outcome Score (KOOS), knee joint muscle strength, and performance-based tests were evaluated preoperatively and at 6 and 12 months postoperatively. Analysis of covariance (ANCOVA) with age and sex as covariates was used to examine differences in improvement at 6 and 12 months. Clinical equivalence was evaluated using 90% confidence intervals and minimum clinically important differences.
Results
Of the 12 patients with preoperative sarcopenia, 10 (83.3%) improved at 12 months postoperatively. ANCOVA showed significant adjusted mean difference (aMD) of 0.31 for SMI at 12 months. No significant differences were found in grip strength (aMD: 0.60), KOOS pain (aMD: 0.01), or 40-m fast-paced walk test (aMD: −0.03). Clinical equivalence was observed for SMI, grip strength, KOOS pain, and 40-m fast-paced walk test, but not other outcomes.
Conclusions
In patients with KOA and sarcopenia, TKA combined with postoperative rehabilitation was associated with improvements in sarcopenia-related measures. Improvements in gait ability, grip strength, and appendicular skeletal muscle mass were comparable to those observed in non-sarcopenic patients.
Introduction
Knee osteoarthritis (KOA) is a degenerative disease characterized by the progressive wear of articular cartilage, causing pain, functional impairment, and difficulties to walk. 1 Total knee arthroplasty (TKA) is widely performed in patients with KOA to relieve pain and improve activities of daily living. 2 TKA has long-term functional durability, and many patients with KOA have reported pain relief and improvement in motor function. 3 The number of TKA procedures performed in Japan is projected to rise by approximately 30% between 2018 and 2030, with the greatest increase expected among individuals aged ≥75 years, indicating a progressive aging trend of the TKA population. 4
Sarcopenia is an age-related condition characterized by a loss of skeletal muscle mass, reduced muscle strength, and subsequent decline in physical function.5,6 The prevalence of sarcopenia among individuals aged ≥60 years in Asia has been reported to be approximately 16.5%, which is considered a significant public health issue. 7 In recent years, the association between sarcopenia and KOA has gained increasing clinical attention.8,9 In patients with KOA, reduced physical activity due to pain avoidance was observed to cause a decrease in lower limb muscle mass and strength.10,11 This decrease have been suggested to compromise knee joint stability and potentially contribute to the progression of KOA. 9 Conversely, pain and joint deformity caused by KOA are believed to accelerate the progression of sarcopenia, creating a vicious cycle between these two conditions. In older patients with similar conditions, achieving sufficient functional improvement with TKA alone may be difficult. Therefore, it is important to employ both interventions: TKA for joint deformity and pain, and rehabilitation for muscle weakness associated with sarcopenia and KOA.
Although the effectiveness of TKA for KOA has been demonstrated in many studies, few have examined whether sarcopenia improves after TKA. Furthermore, it has been suggested that even with sarcopenia, lower limb muscle strength and walking speed may improve after TKA. 12 However, some reports suggest that patients with sarcopenia show only minimal improvements in the Timed Up-and-Go Test, walking speed, and Timed Chair Rise. 13 Therefore, a consensus regarding the impact of preoperative sarcopenia on physical function after TKA is lacking. Furthermore, previous studies have not examined whether postoperative improvements exceed the minimum clinically important difference (MCID), nor have they clarified whether patients achieve equivalent outcomes from a clinical perspective, regardless of the presence of preoperative sarcopenia.
Therefore, we aimed to investigate whether the sarcopenic condition in patients with KOA undergoing TKA improves postoperatively if they have sarcopenia before surgery and examine the impact on postoperative physical function from the perspective of equivalence assessment.
Methods
This prospective observational study, reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines, was conducted at a single general hospital. The study was approved by the ethics committee of the authors’ affiliated institution, Nittazuka Medical Welfare Center Ethics Committee (approval number: Shinrin 2024-7). In addition, written informed consent was obtained from all participants after the purpose of the study had been explained.
The inclusion criteria are shown in Figure 1. This study enrolled 108 patients with KOA who underwent primary TKA performed by the same surgeon at a single institution between June 2023 and July 2024. The exclusion criteria were as follows: age ≤64 years (n = 5), rheumatoid arthritis (n = 13), history of neuromuscular disease (n = 4), and presence of pacemakers (n = 1), for whom body composition analyzers are contraindicated. Eighty-five individuals were included in this study, and baseline assessment of sarcopenia (physical performance, muscle strength, and skeletal muscle mass measurements) was conducted preoperatively. In accordance with the Asian Working Group for Sarcopenia 2019 Consensus Update (AWGS 2019),
5
the participants were divided into two groups: sarcopenia and non-sarcopenia. After excluding postoperative infection cases (n = 1) and those with missing data (n = 13), the final analysis included 12 patients in the sarcopenia group and 59 in the non-sarcopenia group. Inclusion Criteria. TKA: total knee arthroplasty, AWGS: Asian Working Group for Sarcopenia.
Sarcopenia assessment involved measuring physical performance by evaluating the gait speed. This method measures how long it takes to walk 6 m at a normal pace from a dynamic start without decelerating. The speed in m/s was calculated using the average value of the two trials. The cutoff value was set at less than 1.0 m/s. Muscle strength was measured using grip strength. Measurements were acquired twice, with the dominant and non-dominant hand, using a digital grip strength meter TKK5401D (Takei Scientific Instruments, Japan), and the average value was obtained. The measurement position was standing, with the elbow joint in extension. The cutoff values were set at less than 28 kg for men and less than 18 kg for women. Skeletal muscle mass was measured using the multifrequency bioelectrical impedance analysis (BIA) method. The amount of appendicular skeletal muscle mass measured by the BIA method is reported to correlate with the amount of appendicular skeletal muscle mass measured using dual-energy X-ray absorptiometry. 5 Limb skeletal muscle mass was measured using a multi-frequency body composition analyzer, MC-780A-N (TANITA, Japan). A body composition analyzer measures body composition using BIA. It is relatively inexpensive, easily portable, and considered an effective tool for diagnosing sarcopenia. 14 The skeletal muscle mass index (SMI) was calculated by dividing the measured appendicular skeletal muscle mass (kg) by the square of the height (m2). 15 The cutoff value for SMI was set at less than 7.0 kg/m2 in men and less than 5.7 kg/m2 in women.
Next, regarding the survey items, demographic characteristics, including age, sex, height, weight, body mass index (BMI), surgical procedure (unilateral TKA or bilateral TKA), OA grade of the operated and non-operated sides according to the Kellgren–Lawrence classification, total rehabilitation implementation time (number of sessions × 20 min), and preoperative biochemical data (C-reactive protein level, hemoglobin level, total protein level, and albumin level) were obtained from the medical records. Outcome measurements were conducted at baseline (preoperatively) and 6 and 12 months postoperatively. The measured items were walking speed, grip strength, SMI, and the patient-reported outcome measure (PROMs), the Knee Injury and Osteoarthritis Outcome Score (KOOS). In addition, physical function assessments were conducted, including knee joint muscle strength, the 30-s chair-stand test (30s CST), the 40-m fast-paced walk test (40m FPWT), and the stair-climb test (SCT).
KOOS is a PROMs used to assess knee joint disorders and treatment effects. It consists of five categories: 7 items on symptoms, 9 on pain, 17 on activities of daily living, 5 on sports and recreation, and 4 on quality of life. This assessment is considered highly reliable.16,17 The questionnaire is answered by the patient themselves on a five-point scale, from 0 (indicating no problems) to 4 (indicating severe problems). Responses were converted into a scale ranging from 0 to 100 points for each category. A score of 0 indicated severe knee problems, whereas 100 indicated no knee problems.
Knee extensor and flexor muscle strength were measured according to the method described by Ho et al. 12 The measurement position was a sitting posture with both feet off the floor, maintaining both the hip and knee joints at 90° flexion. The measurement was performed using an isometric muscle strength measurement device μTas F-1 (ANIMA Corporation, Japan). Isometric muscle strength for knee joint extension and flexion was measured three times on the operated and non-operated sides. The average value of the three measurements was multiplied by the lower leg length (m), and the resultant value was divided by the body weight (kg) to calculate the weight ratio (Nm/kg). In this study, the weaker side before surgery was examined in bilateral TKA cases, and the operated side was examined in unilateral TKA cases.
The Osteoarthritis Research Society International (OARSI) recommends three core performance-based tests—30s CST, 40m FPWT, and SCT—for the assessment of physical function in patients with KOA and those who have undergone TKA. 18 Measurements for each test were conducted according to the OARSI standards. The 30s CST used a chair with a backrest and a seat height of approximately 40 cm. The participants started in a seated position with their feet shoulder-width apart and arms crossed in front of the chest. After standing up fully and completely extending the hips and knees, the participant repeated the action of sitting back down until the buttocks made full contact with the seat surface for 30 s. The total number of repetitions performed in 30 s was measured by counting the number of cycles from standing up to sitting down. The 40mFPWT placed cones at both ends, spaced 10 m apart. The participants were instructed to walk between the cones as quickly as possible and make a U-turn around each end cone, and the time taken to complete two round trips was measured in seconds. Permission was granted to use a cane if necessary. Speed (m/s) was calculated by dividing the distance (40 m) by the measured time (s). The SCT measured the number of seconds required to ascend and descend a staircase with 11 steps, each 20 cm high. The use of handrails and canes was permitted when necessary. In all tests, the average of the two measurements was used.
The surgery was performed by an experienced orthopaedic surgeon. In all cases, the joint was exposed using the Medial Parapatellar Approach, and the implant used was the cruciate-retaining type (Zimmer Biomet, Persona CR). All the patients underwent the same postoperative rehabilitation program. They started standard postoperative rehabilitation protocols, including range of motion, muscle strengthening, weight bearing, and gait exercises, as tolerated on 1 day postoperatively.
Univariate analyses were performed between the two groups for baseline variables in the statistical analysis. Fisher’s exact test was performed on the categorical variables. Normality of continuous variables was assessed using the Shapiro–Wilk test. Variables that were normally distributed were analyzed using the two-sample t-test, while those that were not normally distributed were analyzed using the Mann–Whitney U test. The proportion of patients with improved sarcopenia after surgery was calculated to assess the changes in sarcopenia before and after the procedure.
For the outcome measures, analysis of covariance (ANCOVA) was performed using age and sex as covariates, and differences between the sarcopenia and non-sarcopenia groups at 6 and 12 months postoperatively were examined at a significance level of 5%. Furthermore, the adjusted mean difference (aMD) between the two groups was calculated. All aMDs were calculated as the difference between the mean values of the sarcopenia and non-sarcopenia groups. We also examined the clinical equivalence of each of these outcomes. The clinical equivalence margin was set to the MCID for each outcome based on previous studies. MCID was set at a walking speed of 0.1 m/s, 19 grip strength of 5.0 kg, 20 KOOS pain at 12 points, 21 KOOS symptoms at 9 points, 21 KOOS ADL at 10 points, 21 KOOS sports and recreation at 9 points, 21 and KOOS QOL at 14 points. 21 The MCID for the performance-based tests was set at 2.6 repetitions for the 30s CST, 22 0.2 s for the 40m FPWT, 22 and 2.6 s for the CST. 23 In this study, for SMI, knee extension muscle strength, and flexion muscle strength, for which MCID could not be obtained from the existing literature, a distribution-based indicator was used, and 0.5 times the standard deviation of all subjects at baseline was set as the MCID. This method has been reported to reflect clinically meaningful changes. 24 The MCID was set at 0.57 for SMI, 0.21 for knee extensor strength, and 0.09 for knee flexor strength. To verify the equivalence, the 90% confidence interval (CI) of the aMD was used, and if the 90% CI fell entirely within the predefined MCID, the two groups were considered clinically equivalent.
The statistical software used was EZR (ver. 1.68). 25 EZR is a statistical software that extends the functions of R and the R Commander.
Results
Baseline characteristics of the two groups.
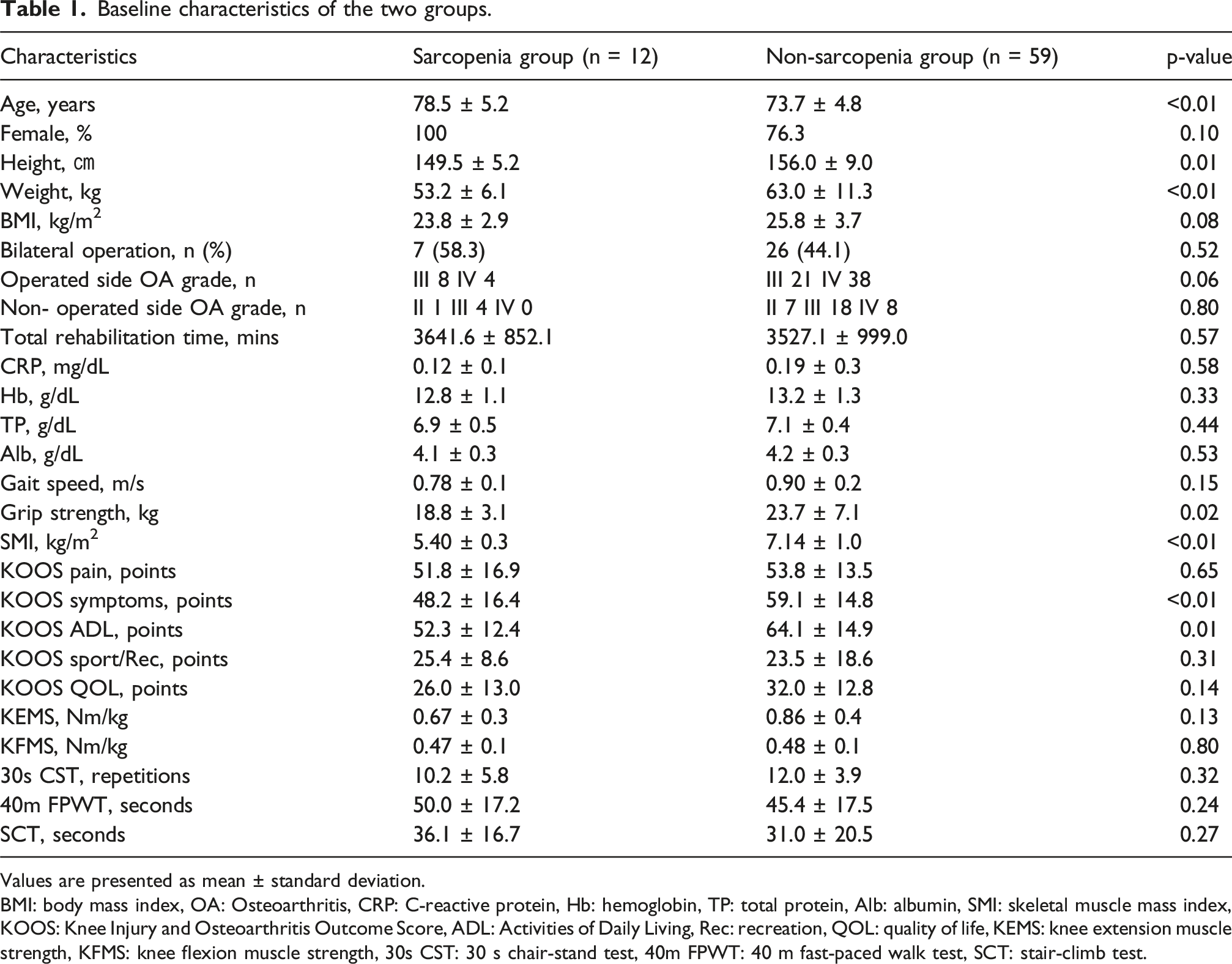
Values are presented as mean ± standard deviation.
BMI: body mass index, OA: Osteoarthritis, CRP: C-reactive protein, Hb: hemoglobin, TP: total protein, Alb: albumin, SMI: skeletal muscle mass index, KOOS: Knee Injury and Osteoarthritis Outcome Score, ADL: Activities of Daily Living, Rec: recreation, QOL: quality of life, KEMS: knee extension muscle strength, KFMS: knee flexion muscle strength, 30s CST: 30 s chair-stand test, 40m FPWT: 40 m fast-paced walk test, SCT: stair-climb test.
Changes in sarcopenia before and after surgery.

SMI: skeletal muscle mass index.
Clinical outcomes at 6 and 12 months after surgery.

aMD: adjusted mean difference, CI: confidence interval, MCID: minimal clinically important difference.
SMI: skeletal muscle mass index, KOOS: Knee Injury and Osteoarthritis Outcome Score, ADL: activities of daily living, Rec: recreation, QOL: quality of life, KEMS: knee extension muscle strength, KFMS: knee flexion muscle strength, 30s CST: 30 s chair-stand test, 40m FPWT: 40 m fast-paced walk test, SCT: stair-climb test.
For other outcomes, no significant differences were observed at 6 and 12 months postoperatively, and clinical equivalence was not demonstrated.
Discussion
Despite the limited number of patients with sarcopenia, this exploratory study provides clinically relevant insights into postoperative functional recovery after TKA. The focused cohort and detailed functional assessments enabled the identification of meaningful trends that may inform future prospective studies and individualized rehabilitation strategies. Among patients undergoing TKA for KOA, MCID-based clinical equivalence was observed for postoperative grip strength, skeletal muscle mass index, pain, and walking ability between patients with and those without preoperative sarcopenia. These findings suggest that, from a clinical perspective, patients with sarcopenia may achieve postoperative improvements comparable to those without sarcopenia through pain relief and increased activity following TKA. Furthermore, regarding changes in sarcopenia status before and after surgery, improvement was observed in 10 (83.3%) of 12 patients at 12 months postoperatively. While these results should be interpreted cautiously given the exploratory nature of the study, they suggest that TKA may contribute to favorable changes in sarcopenia status in selected patients. To the best of our knowledge, only one previous study has investigated changes in sarcopenia before and after TKA. Ho et al. reported that 5 of 19 patients with preoperative sarcopenia transitioned to non-sarcopenia at 12 months postoperatively. The improvement rate observed in the present study appears higher, although direct comparison is limited by differences in study design and patient characteristics.
As a factor in the improvement of sarcopenia, the pain relief effect of TKA is considered significant, as evidenced by the improvement in KOOS pain scores to a level comparable to that in the non-sarcopenia group. Furthermore, the improvement in walking ability and increased activity levels triggered by pain relief and postoperative rehabilitation further contributed to the increase in grip strength and skeletal muscle mass of the limbs. Ho et al. 12 reported that even in patients with KOA and concomitant sarcopenia, walking speed improved after TKA. In addition, Liao et al. 26 conducted a longitudinal study on the recovery of walking speed after TKA and reported that patients with sarcopenia or sarcopenic obesity tended to experience delayed recovery compared with those without sarcopenia. However, it has been reported that over time and with postoperative rehabilitation interventions, groups of patients with sarcopenia and sarcopenic obesity also showed a tendency to improve. 26 Therefore, even if a patient has preoperative sarcopenia, improvement in walking ability after TKA is supported by previous studies. In addition, grip strength is considered an important predictive biomarker for postoperative recovery in older adults, 27 and its correlation with mobility has been reported.5,28 Therefore, a positive relationship between improvements in walking ability and grip strength is believed to exist. Regarding the amount of appendicular skeletal muscle, Heo 29 and colleagues have investigated the impact of TKA for KOA on muscle mass. According to a previous report, the skeletal muscle mass of the limbs was gradually and continuously increased up to 24 months after TKA. As a factor in the increase in skeletal muscle mass in the limbs, pain reduction from TKA leads to increased activity levels, which is consistent with the findings of this study. However, in two cases, no improvement in sarcopenia was observed even 12 months after the surgery. In these two cases, the patients were older women (>80 years), and their preoperative SMI values were significantly below the cutoff value. These factors may interfere with the improvement of sarcopenia after surgery; therefore, future studies are necessary to address them to confirm this trend.
However, no equivalence was observed in the improvement of KOOS categories other than pain, knee joint muscle strength, 30s CST, and SCT. Regarding KOOS, Shon et al. 30 reported that up to 6 months after TKA, the sarcopenia group showed significantly poorer results in KOOS categories other than pain than the non-sarcopenia group. However, no significant difference was observed between the two groups at 12 months postoperatively. These findings show similar results regarding pain; however, they differ from the results of the present study in other categories. One possible reason for this is that no equivalence was observed in the improvement of the knee joint muscle strength, 30s CST, or SCT. Goodpaster et al. 31 confirmed that the rate of decline in muscle strength with aging is approximately three times faster than that in muscle mass. In addition, it has been stated that maintaining or increasing muscle mass does not necessarily prevent the decline in muscle strength with aging, and that a non-linear relationship exists between muscle mass and muscle strength. 31 This discrepancy suggests the involvement of factors that cannot be explained by muscle mass alone; in other words, it indicates reduced muscle quality. 31 Furthermore, factors such as neurological changes, inflammatory cytokines, and fat infiltration, which are unique to aging and KOA, have been identified as causes of decreased muscle quality. 31 Therefore, it is possible that not only muscle mass but also changes in muscle quality affecting muscle strength may have influenced improvements in higher KOOS categories, as well as in the 30s CST and SCT. The phase angle 32 is useful for assessing muscle quality. The phase angle is a clinically established indicator obtained using the BIA method and is considered an indicator of cellular health. In the future, by introducing phase angle into body composition assessment, it will be necessary to longitudinally investigate not only quantitative changes in muscle mass but also muscle quality to clarify its role in functional recovery after TKA.
In this study, we showed that even KOA patients with sarcopenia before surgery experienced similar improvements in pain relief, walking ability, grip strength, and appendicular skeletal muscle mass after TKA as those in the non-sarcopenia group. Furthermore, postoperative sarcopenia was improved. These results support the expectation that pain relief from TKA and improved activity levels from postoperative rehabilitation can be anticipated, regardless of the presence or absence of sarcopenia. However, in categories other than KOOS pain, namely knee joint muscle strength, 30s CST, and SCT, no equivalent improvement was observed, suggesting that the effects of sarcopenia may persist in these areas. It is possible that not only a reduced muscle mass but also a decline in muscle quality has a greater impact on more advanced physical conditions. In particular, decreased muscle quality can lead to reduced contraction speed and muscle efficiency and may continue to be a significant limiting factor for advanced physical function even after improvements in pain and instability. Therefore, in the recovery of physical function after TKA, it is important to implement rehabilitation interventions that focus on increasing muscle mass and improving muscle quality. In recent years, it has been reported that combining aerobic exercise with resistance training can decrease intramuscular fat and improve muscle fiber characteristics.33,34 In addition, neuromuscular activation using electrical muscle stimulation has been shown to reduce intramuscular fat and potentially improve muscle quality in older individuals and patients with low levels of physical activity. 35 These findings support the involvement of muscle quality, as suggested in this study. Therefore, to maximize functional recovery after TKA in patients with KOA and preoperative sarcopenia, implementing a comprehensive rehabilitation program that combines aerobic exercise and neuromuscular activation with conventional training aimed at increasing muscle mass would be effective. In the future, it will be necessary to introduce body composition assessment using the phase angle obtained by the BIA method and to longitudinally track quantitative changes in muscle quality to clarify the impact of muscle quality on the recovery process after TKA.
The limitations of this study include the small number of cases and the fact that the sarcopenia group had a higher proportion of women. This may limit the statistical power of the detection. In the future, large-scale, multicenter studies and advanced analyses that consider gender differences are necessary. In addition, reports demonstrating the relationship between TKA and sarcopenia are limited, and the establishment of unified evaluation criteria is required. After overcoming these challenges, identifying the factors that hinder recovery after TKA in patients with KOA and preoperative sarcopenia is an important topic for future research.
Conclusion
In patients with KOA who also had preoperative sarcopenia, pain relief from TKA and postoperative rehabilitation led to improvements in walking ability, grip strength, and limb skeletal muscle mass to a degree comparable to that in nonsarcopenic patients, thereby improving postoperative sarcopenia. However, no comparable improvements were observed in muscle strength or advanced physical function, which could be attributed to decreased muscle quality. These findings highlight the importance of comprehensive rehabilitation aimed not only at increasing muscle mass but also at improving muscle quality for the recovery of physical function after TKA.
Footnotes
Ethical considerations
This study was approved by the ethics committee at the authors’ affiliated institution, Nittazuka Medical Welfare Center Ethics Committee (approval number: Shinrin 2024-7).
Consent to participate
The purpose of the study was explained to the participants in writing, and their consent to participate was obtained.
Consent for publication
Written informed consent was obtained from all participants prior to study participation.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors disclose receipt of the following financial or material support for the research, authorship, and/or publication of this article: JSPS KAKENHI grant number JP23K10518 (to Y.S.).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors without undue reservation.
