Abstract
Objective
To investigate the effects of denosumab on pain relief, bone mineral density (BMD), and refracture risk during long-term follow-up in patients with osteoporotic vertebral compression fractures (OVCF) after percutaneous vertebroplasty, and to identify factors associated with refracture.
Methods
This retrospective study included 396 OVCF patients who underwent percutaneous vertebroplasty and received denosumab between January 2021 and June 2023. Patients were classified into a completed-treatment group (n = 184) and a discontinued-treatment group (n = 212). After 1:1 propensity score matching, 101 patients were included in each group. Changes in visual analog scale (VAS) scores and lumbar spine and femoral neck T-score were compared over 24 months. Refracture risk was assessed using Kaplan–Meier analysis and multivariable Cox regression in the matched cohort.
Results
After matching, baseline characteristics were well balanced between groups. Over 24 months of follow-up, the completed-treatment group showed significantly lower VAS scores and greater improvements in lumbar spine and femoral neck T-score compared with the discontinued-treatment group. The incidence of refracture was significantly lower in the completed-treatment group. In the PSM cohort, multivariable Cox regression analysis demonstrated that completed denosumab treatment was independently associated with a lower risk of refracture (HR = 0.314, 95% CI 0.125–0.792, P = 0.014).
Conclusion
In patients with OVCF treated with percutaneous vertebroplasty, persistent denosumab therapy for at least 24 months is associated with sustained pain relief, improved BMD, and a lower risk of refracture, underscoring the importance of long-term treatment adherence.
Introduction
Osteoporosis is a common systemic metabolic disease among middle-aged and elderly populations, characterized by reduced bone mass, microarchitectural deterioration, and decreased bone strength, ultimately leading to a markedly increased risk of fractures. Vertebral compression fracture (VCF) is one of the most common complications of osteoporosis, with features of high incidence, severe disability, and frequent recurrence. 1 Studies have shown that after the first vertebral fracture, the risk of subsequent fractures increases significantly, forming a vicious cycle that severely compromises quality of life and even increases mortality. 2 Therefore, effective pain relief, improvement of bone mineral density (BMD), and reduction of refracture incidence are key goals of current clinical treatment and research.
Current therapeutic strategies for osteoporosis and related fractures mainly include basic supplementation (such as calcium and vitamin D) and antiresorptive drugs. Denosumab, a humanized IgG2 monoclonal antibody, exerts its effects by specifically binding to receptor activator of nuclear factor-κB ligand (RANKL), thereby inhibiting osteoclast differentiation, maturation, and activation. This process effectively reduces bone resorption, increases BMD, and lowers fracture risk. 3 Compared with traditional bisphosphonates, denosumab offers advantages such as subcutaneous administration, a convenient dosing interval of once every 6 months, and minimal renal toxicity, making it increasingly utilized in clinical practice. 4
Multiple clinical trials have confirmed that denosumab significantly increases BMD in the lumbar spine and femoral neck and reduces the risk of both vertebral and non-vertebral fractures in postmenopausal women with osteoporosis.5,6 Other studies have also demonstrated its efficacy in improving bone metabolism markers, making it particularly suitable for patients at high risk of fractures. 7 However, most existing studies have focused on general osteoporotic populations, and systematic evaluations of its long-term efficacy in patients who have already sustained vertebral compression fractures, whether after surgical or conservative treatment, remain limited. In addition, the occurrence of osteoporotic fractures is closely associated with various clinical factors, including age, sex, BMD level, prior fracture history, lifestyle habits, and comorbid chronic diseases. 8 Identifying these potential risk factors is essential for early intervention and individualized treatment strategies, which can further reduce fracture incidence and improve prognosis.
Therefore, this study retrospectively analyzed patients with osteoporotic vertebral compression fractures (OVCF) who received denosumab treatment. Based on mid-to long-term follow-up data, we comprehensively evaluated its effects on pain relief, BMD changes, and refracture prevention, and further explored the factors influencing refracture. The findings are expected to provide valuable clinical evidence for the long-term standardized management of patients with osteoporotic fractures.
We hypothesized that patients who completed long-term denosumab therapy (≥24 months) would achieve greater improvements in bone mineral density and experience a lower risk of refracture compared with patients who discontinued treatment prematurely.
Materials and methods
Study population
This was a single-center retrospective cohort study that included 396 patients with osteoporotic vertebral compression fractures (OVCF) who were diagnosed and treated at our hospital between January 2021 and June 2023. All patients received at least one injection of denosumab and completed ≥24 months of clinical follow-up.
Sample size determination: According to previous reports, the incidence of refracture within 2 years of denosumab treatment in patients with osteoporosis is approximately 3%–6% [1–3]. Based on prior institutional data and the expected effect size, the required sample size was calculated using the Cox proportional hazards model (α = 0.05, power = 80%), yielding a minimum sample size of 350 patients. Considering potential loss to follow-up and incomplete data, a total of 396 patients were ultimately included to ensure study power.
Study period. Patients were enrolled from January 2021 to June 2023. Follow-up and outcome ascertainment continued until June 30, 2025 (administrative censoring), allowing up to 24 months of follow-up for the last enrolled patient.
Inclusion criteria: (1) Meeting diagnostic criteria for OVC 9 and confirmed by X-ray, computed tomography (CT), or magnetic resonance imaging (MRI); (2) underwent percutaneous vertebroplasty at our hospital and initiated denosumab treatment postoperatively with complete treatment records; (3) follow-up time ≥24 months; (4) complete clinical and imaging data available.
Exclusion criteria: (1) secondary osteoporosis (e.g., multiple myeloma, bone metastases, parathyroid disease); (2) severe hepatic or renal dysfunction (Child–Pugh class C liver function or estimated glomerular filtration rate [eGFR] <30 mL/min); (3) systemic rheumatoid arthritis, connective tissue disease, or other disorders affecting bone metabolism; (4) prior treatment with other specific anti-osteoporosis drugs (e.g., bisphosphonates, teriparatide) for >6 months; (5) severe complications or death during follow-up resulting in missing data; (6) incomplete data or insufficient follow-up.
Data collection
Baseline clinical and laboratory data were obtained from the hospital electronic medical record and imaging systems, including sex, age, body mass index (BMI), smoking history, alcohol consumption, comorbidities (hypertension, diabetes mellitus, rheumatoid arthritis, etc.), prior vertebral fracture history, baseline BMD (lumbar spine T-score [L1–L4] and femoral neck T-score), initial fracture location, and fracture segment (L3 and above, L2, T11, T12–L1). Laboratory indices included serum calcium and serum creatinine. Treatment compliance data included the total number of denosumab injections and the injection interval. Follow-up data included refracture occurrence (defined as a new vertebral compression fracture during follow-up confirmed by imaging and treated surgically) and the time of refracture.
Treatment protocol
All patients received denosumab therapy following vertebroplasty (60 mg subcutaneous injection every 6 months, commercial name Prolia®). From the initiation of denosumab, all patients were additionally prescribed oral calcium carbonate (1,000 mg/day of elemental calcium) and cholecalciferol (vitamin D3, 800 IU/day) to maintain stable serum calcium levels and reduce the risk of hypocalcemia. This supplementation regimen was adhered to throughout follow-up, with regular BMD monitoring and outpatient evaluations. No other specific anti-osteoporosis drugs were used during the study period.
Patient grouping and definition of treatment persistence
Patients were grouped according to their persistence with denosumab treatment during follow-up. 10 A total of 406 patients who underwent percutaneous vertebroplasty and received denosumab were initially assessed. Patients with intermediate treatment persistence, defined as receiving three denosumab injections and/or a treatment duration of 12–24 months, were excluded from the primary analysis (n = 10) to allow a clear distinction between high- and low-persistence treatment patterns.
After exclusion of this intermediate group, the final analytic cohort consisted of 396 patients. These patients were categorized into two groups: the completed-treatment group, comprising patients who received ≥4 injections and maintained treatment for ≥24 months (n = 184), and the discontinued-treatment group, comprising patients who received ≤2 injections and/or had a treatment duration of <12 months (n = 212).
11
The patient selection and grouping process is shown in Figure 1. Flowchart of patient selection and grouping according to denosumab treatment persistence.
Refracture was defined as the occurrence of a new vertebral compression fracture during treatment, confirmed by imaging (CT, MRI, or X-ray), requiring surgical intervention (e.g., repeat vertebroplasty), and verified by comparison with previous imaging. 12
Ethics and quality control
The study protocol was approved by the Ethics Committee of our hospital. The study was conducted in strict accordance with the ethical principles of the Declaration of Helsinki. Given its retrospective design, all data were extracted from prior clinical records and anonymized to protect patient privacy. Written informed consent was waived for the following reasons: the study involved no direct patient contact or intervention, posed no risk to patient rights or welfare, and requiring informed consent from all participants would have rendered the study infeasible. To ensure data quality, two researchers independently extracted and entered the data, followed by cross-checking. Any discrepancies were resolved through discussion or consultation with a senior investigator, ensuring accuracy and reliability.
Statistical analysis
All statistical analyses were performed using R software (version 4.3.1). Continuous variables were expressed as mean ± standard deviation (±SD) or median (interquartile range) [M (P25, P75)], and categorical variables were expressed as frequency and percentage [n (%)]. Between-group comparisons were performed using the independent-sample t test, Mann–Whitney U test, or χ2 test, as appropriate. To control for potential confounding factors in between-group comparisons, propensity score matching (PSM) was performed using a logistic regression model. A 1:1 nearest-neighbor matching algorithm with a caliper width of 0.2 of the standard deviation of the propensity score was applied. Covariate balance after matching was assessed using standardized mean differences (SMDs), with an SMD <0.1 considered indicative of adequate balance. After matching, longitudinal changes in pain scores and bone mineral density (BMD) were analyzed using linear mixed-effects models, with time, treatment group, and the time × group interaction included as fixed effects. Refracture incidence between groups was evaluated using Kaplan–Meier survival analysis, and differences were compared using the log-rank test. To identify independent risk factors for postoperative refracture, Cox proportional hazards regression analyses were performed in the entire unmatched cohort. This approach was used to evaluate baseline clinical and biological predictors of refracture at the individual level while preserving statistical power. Hazard ratios (HRs) and 95% confidence intervals (CIs) were calculated. A two-sided P value <0.05 was considered statistically significant.
Results
Comparison of baseline characteristics
Comparison of baseline characteristics.
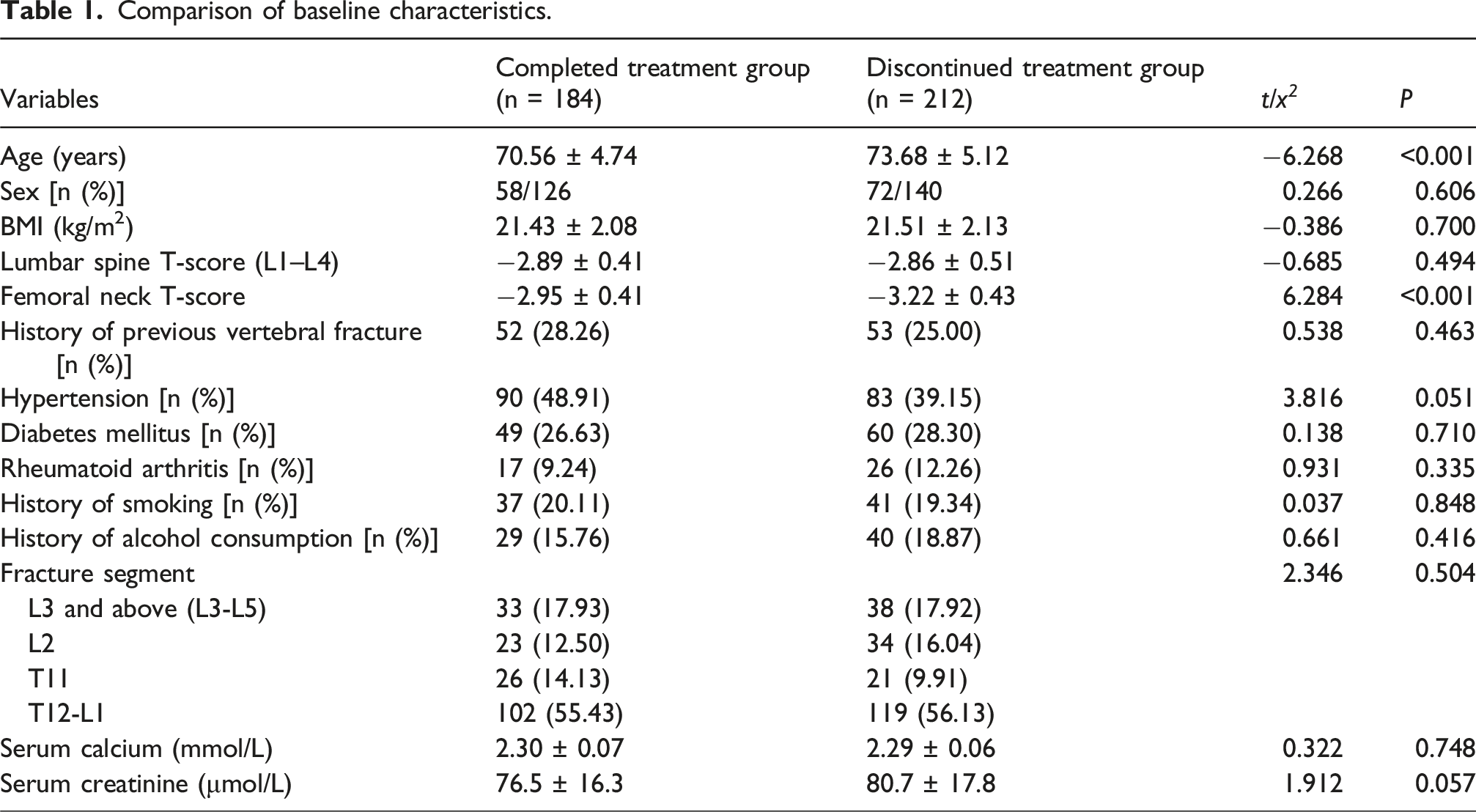
Baseline characteristics after PSM
To control for baseline differences, 1:1 nearest-neighbor PSM was performed. Matching variables included age, sex, BMI, lumbar spine T-score (L1–L4), femoral neck T-score, history of previous vertebral fracture, hypertension, diabetes, rheumatoid arthritis, smoking, alcohol consumption, fracture segment, serum calcium, and serum creatinine. The caliper was set at 0.2 of the standard deviation of the propensity score to reduce matching error.
Baseline characteristics after propensity score matching (n = 101 per group).


Balance and Distribution Analysis of PSM. Note: (a) SMD Balance Plot; (b) Propensity Score Q–Q Plot; (c) Propensity Score Density Plot. P: Completed treatment group; C: Discontinued treatment group.
Pain score changes
Linear mixed-effects model results for VAS scores.
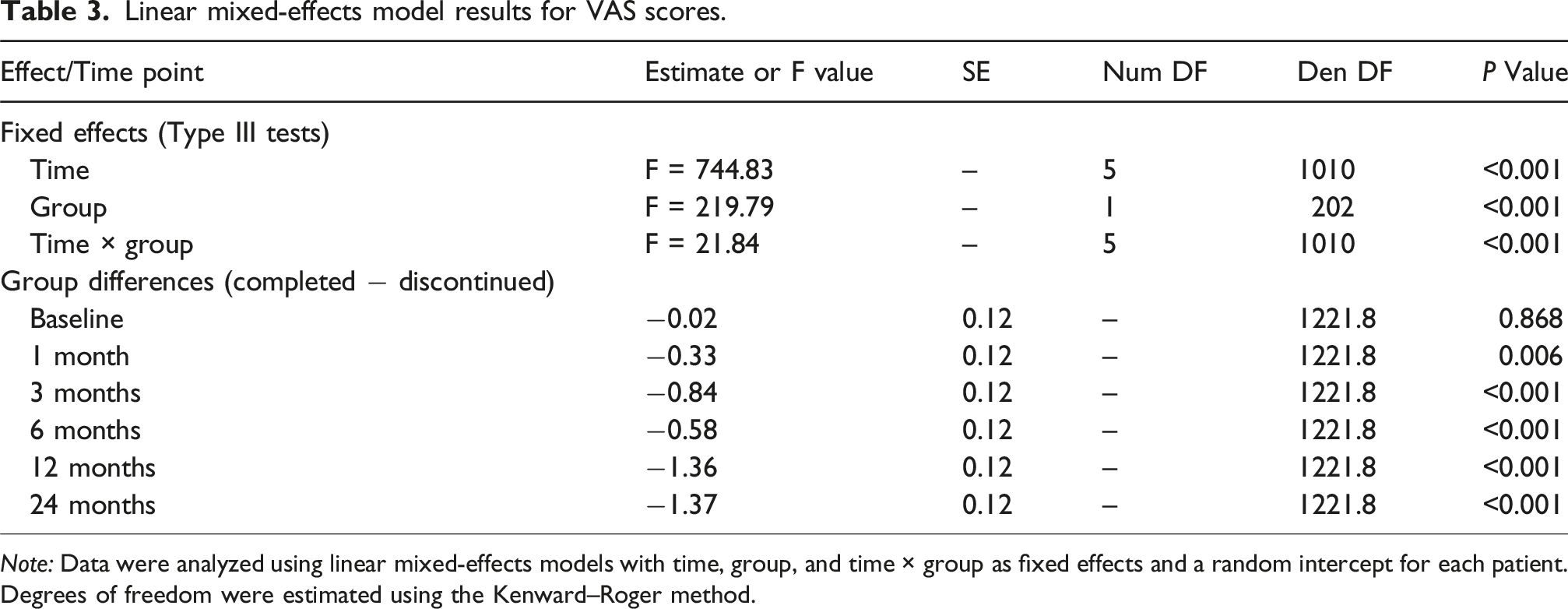
Note: Data were analyzed using linear mixed-effects models with time, group, and time × group as fixed effects and a random intercept for each patient. Degrees of freedom were estimated using the Kenward–Roger method.
Post hoc comparisons showed no significant difference in VAS scores between the two groups at baseline. From 1 month onward, the completed-treatment group consistently exhibited significantly lower VAS scores than the discontinued-treatment group, with the between-group difference progressively increasing over time, reflecting greater and more sustained pain relief associated with long-term denosumab treatment (Table 3).
Changes in BMD
Linear mixed-effects model results for lumbar spine and femoral neck BMD.
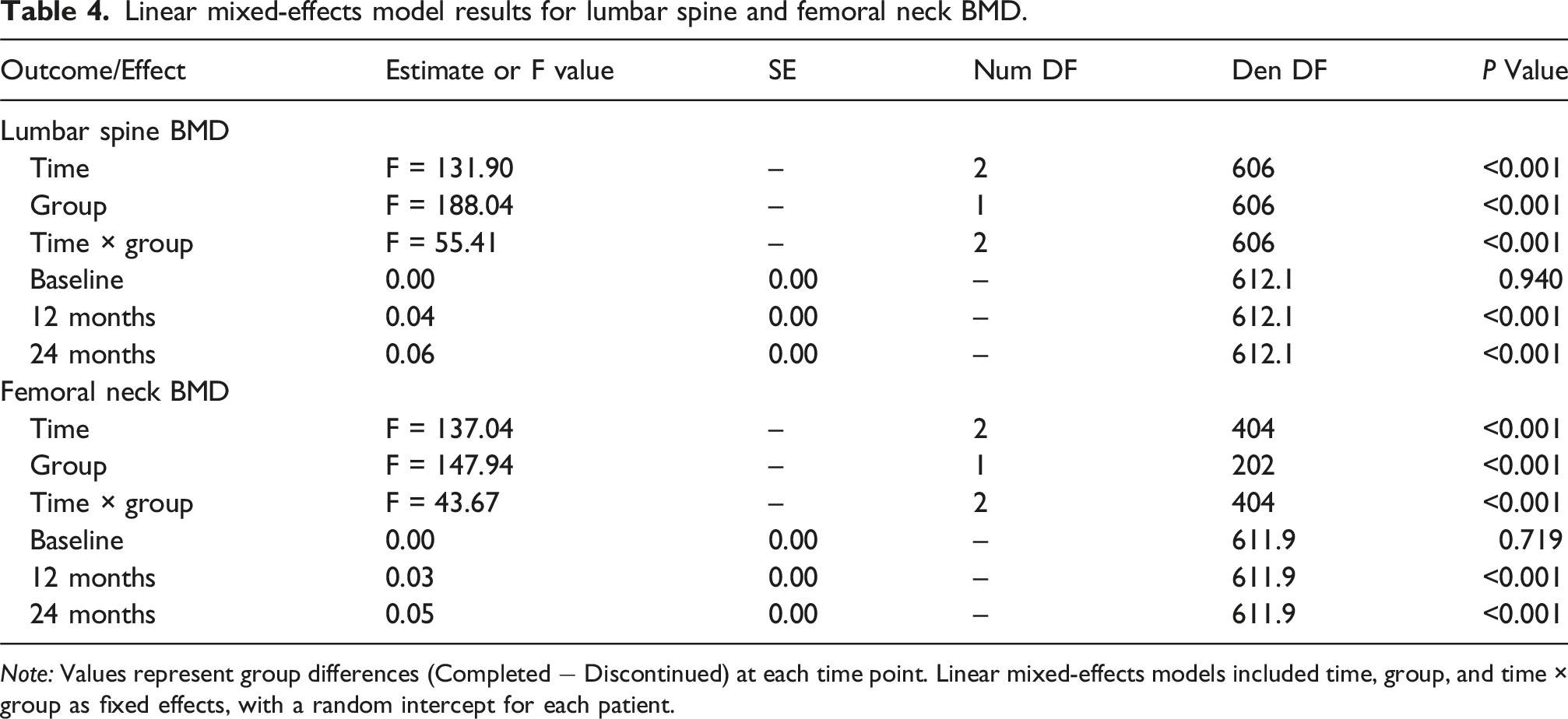
Note: Values represent group differences (Completed − Discontinued) at each time point. Linear mixed-effects models included time, group, and time × group as fixed effects, with a random intercept for each patient.
Post hoc analyses revealed no significant differences in lumbar spine or femoral neck T-score between the two groups at baseline. In contrast, the completed-treatment group showed significantly greater increases in both lumbar spine and femoral neck T-score at 12 and 24 months compared with the discontinued-treatment group, reflecting superior skeletal benefits associated with sustained denosumab therapy (Table 4).
Postoperative refracture of OVCF
Comparison of postoperative refracture in OVCF patients between groups.

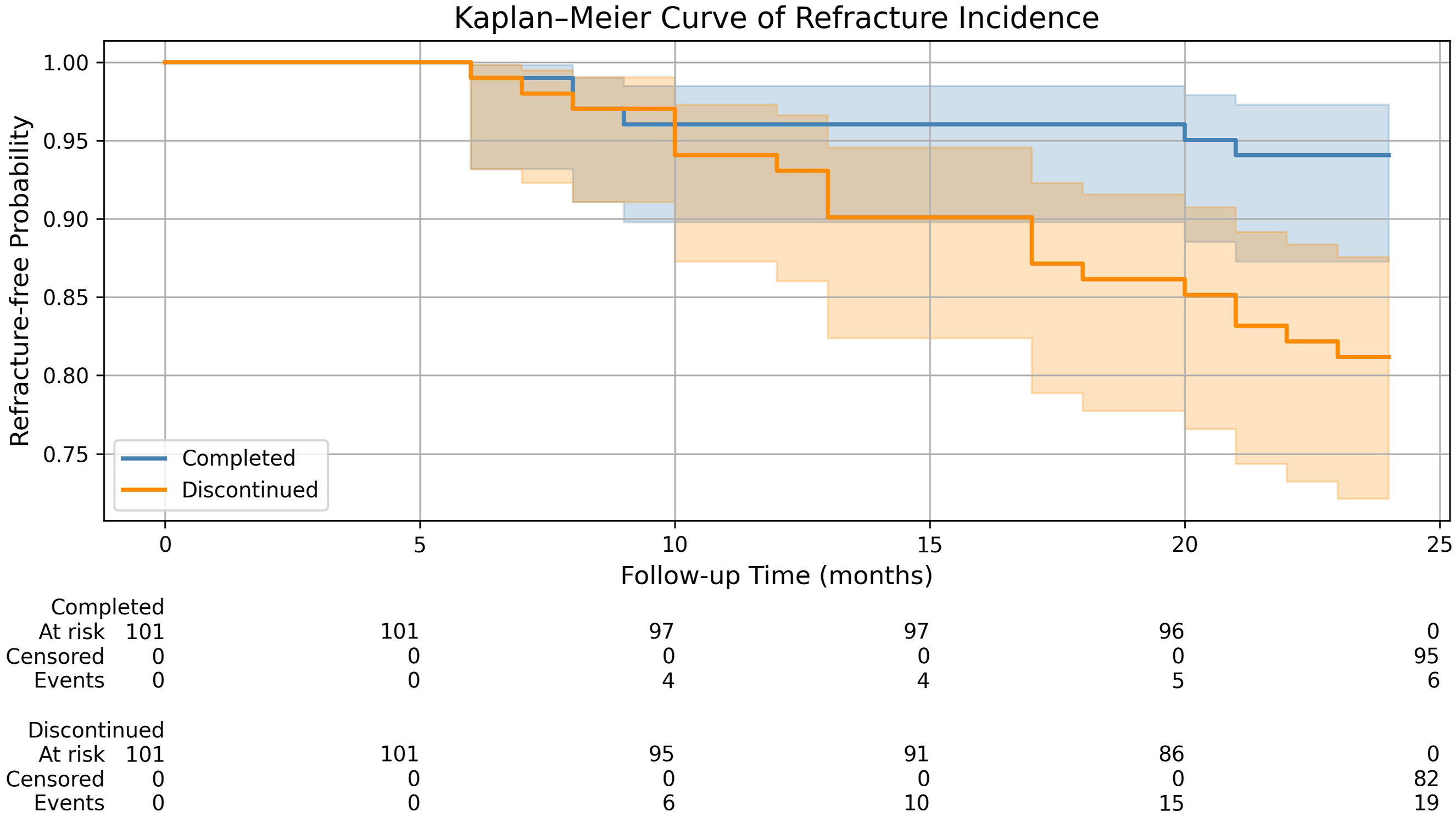
Kaplan–Meier curve of cumulative postoperative refracture incidence over 24 Months after matching.
Risk factors for refracture in the propensity score–matched cohort
In the propensity score–matched cohort, multivariable Cox proportional hazards regression analysis was performed to identify factors associated with refracture risk. After adjustment for age, sex, baseline femoral neck T-score, fracture extent, and prior fracture history, completed denosumab treatment was independently associated with a significantly lower risk of refracture (HR = 0.314, 95% CI 0.125–0.792, P = 0.014).
Multivariable Cox regression analysis for refracture risk in the propensity score–matched cohort.

Discussion
In this propensity score–matched retrospective cohort study, we found that completion of at least 24 months of denosumab therapy after vertebral augmentation was independently associated with a significantly lower risk of subsequent vertebral fractures, together with sustained improvements in bone mineral density. These findings highlight the clinical importance of treatment persistence in patients with OVCF.
Patients with vertebral compression fractures remain at high risk of subsequent fractures even after surgical intervention. Therefore, long-term anti-osteoporotic therapy postoperatively is crucial for improving BMD and reducing fracture recurrence. Denosumab, due to its potent and reversible inhibition of bone resorption, is considered an important therapeutic option for patients at high fracture risk. However, in clinical practice, patient compliance varies considerably, and the impact of compliance on long-term BMD improvement and subsequent fracture risk lacks sufficient evidence. This is particularly relevant in populations undergoing vertebral augmentation, where insufficient treatment compliance may offset the drug’s inherent bone-protective effects. Based on this rationale, the present study used propensity score matching to evaluate BMD changes and subsequent fracture occurrence over a 24-month follow-up in patients with different levels of denosumab treatment persistence, and to analyze independent risk factors for refracture.
This study followed 396 patients with OVCF to explore the relationship between denosumab compliance and subsequent fracture risk. In the unmatched cohort, patients who completed denosumab treatment exhibited a significantly lower 24-month incidence of subsequent fractures compared with those who discontinued treatment. After propensity score matching and multivariable Cox regression analysis, completed denosumab treatment remained independently associated with a lower risk of refracture. These findings underscore the importance of sustained antiresorptive therapy in reducing fracture recurrence in this high-risk population.
Although baseline femoral neck T-score was included as a matching variable in the propensity score matching procedure, it did not show a statistically significant association with refracture risk in the multivariable Cox model performed in the matched cohort. Propensity score matching was applied to balance baseline characteristics between treatment groups, whereas Cox regression analysis in the matched cohort was used to identify factors associated with refracture risk at the individual level. These analytical approaches are therefore complementary and methodologically consistent.
These findings are highly consistent with previous randomized controlled trials and real-world studies. Some studies have shown that denosumab significantly reduces the risk of new vertebral fractures during continuous therapy, accompanied by progressive increases in lumbar spine and femoral neck T-score. 13 Conversely, after discontinuation, bone turnover markers rebound to above baseline within 3–6 months, and BMD declines to near or even below pre-treatment levels within 12 months, markedly increasing fracture risk in the short term.14,15 In this study, the increased fracture risk in the discontinued-treatment group is likely related to such “rebound bone loss,” in which osteoclast activity overshoots after discontinuation, leading to rapid bone loss and increased fragility.
Denosumab functions by reversibly inhibiting the binding of RANKL to RANK, blocking osteoclast formation and activity, thereby significantly reducing bone resorption. 16 Under continuous therapy, trabecular number and connectivity increase, cortical microarchitecture improves, and overall bone strength is enhanced. 17 However, once treatment is stopped, this inhibitory effect is quickly reversed, osteoclast formation becomes overactive, bone resorption rises sharply, and rapid bone loss ensues. 18 Therefore, denosumab’s bone-protective effect depends on sustained treatment, which is corroborated by the results of this study.
Previous studies have proposed baseline femoral neck T-score ≤ −2.80 as a critical threshold for predicting post-PKP subsequent fractures in OVCF patients, 19 suggesting that patients with low BMD should initiate and adhere to long-term antiresorptive therapy as early as possible. Although baseline femoral neck T-score was not an independent predictor of refracture risk in the present multivariable analysis, patients with low bone mass remain clinically vulnerable to fracture recurrence. In conditions of low bone mass, trabecular disruption and cortical thinning are more severe, and even with anti-osteoporotic therapy, bone formation and repair lag behind bone resorption. 20 Treatment interruption further accelerates bone loss and increases fracture risk. Clinically, it is therefore essential to emphasize compliance management in patients with low bone mass and high fracture risk, with enhanced monitoring and interventions when necessary to ensure continuous and effective bone protection and minimize fracture recurrence.
Compared with similar domestic and international studies, the strength of this study lies in the use of propensity score matching, which better balanced baseline characteristics between groups and reduced confounding bias, allowing a more accurate assessment of the true impact of compliance on clinical outcomes. Previous domestic retrospective studies also indicated that standardized denosumab use leads to better BMD improvement and lower fracture incidence, but most did not use matching methods and were subject to potential confounding. 21 The refined analytical strategy in the present study further enhances the robustness of the findings.
Moreover, this study suggests that clinicians should pay close attention to factors affecting treatment compliance, including economic burden, drug side effects, insufficient patient disease awareness, and logistical barriers such as transportation. Measures such as patient education, follow-up reminders, and financial support can effectively improve compliance. For patients in whom treatment interruption is unavoidable, planning sequential anti-osteoporotic therapy in advance is recommended to mitigate post-discontinuation bone loss and fracture risk, thereby optimizing long-term bone health management. 22
Several limitations of this study should be acknowledged. First, this was a single-center retrospective study. Although propensity score matching was used to reduce confounding, the influence of unmeasured factors such as actual calcium and vitamin D intake, physical activity level, and fall risk cannot be entirely excluded. Second, the follow-up period was limited to 24 months, which may not reflect longer-term outcomes. Third, bone turnover markers and bone microstructural imaging were not available, limiting further exploration of the biological mechanisms underlying the rebound phenomenon after denosumab discontinuation. Finally, the specific reasons for treatment interruption were not analyzed in detail, which warrants further investigation in future studies. In addition, refracture in the present study was defined as a new vertebral compression fracture requiring surgical intervention. Vertebral refractures managed conservatively were not included, which may have led to an underestimation of the true refracture incidence.
Conclusion
This study indicates that OVCF patients who maintain regular denosumab therapy for at least 24 months after vertebral augmentation experience significant BMD improvement and a lower risk of subsequent fractures. Treatment discontinuation is associated with an increased fracture risk, highlighting the clinical importance of adherence management. Emphasis should be placed on sustained antiresorptive therapy and appropriate sequential treatment strategies prior to discontinuation to minimize rebound bone loss and fracture risk. Future multicenter, prospective studies are warranted to further validate these findings and to optimize long-term management strategies following denosumab therapy.
Footnotes
Ethical considerations
The study protocol was approved by the Ethics Committee of Sichuan Provincial Orthopedic Hospital (No. KY2025-079-01). The study was conducted in strict accordance with the ethical principles of the Declaration of Helsinki. Given its retrospective design, all data were extracted from prior clinical records and anonymized to protect patient privacy.
Consent to participate
Written informed consent was waived for the following reasons: the study involved no direct patient contact or intervention, posed no risk to patient rights or welfare, and requiring informed consent from all participants would have rendered the study infeasible.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Chengdu Municipal Health Commission - Chengdu Medical Research Project (No. 2023018).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data supporting our findings are available from the corresponding author upon request
