Abstract
Purpose
This study aimed to evaluate the efficacy and safety of an elastic constriction ring compared to traditional pneumatic tourniquets in patients undergoing surgery for closed distal radius fractures.
Methods
A total of 60 participants were enrolled, with 30 in the experimental group (elastic constriction ring) and 30 in the control group (traditional tourniquet). Key indicators assessed included preoperative exsanguination time, intraoperative blood loss, tourniquet-related complications, and so on.
Results
The experimental group demonstrated a significantly shorter average exsanguination time (8.36 ± 2.72 s vs 55.98 ± 3.61 s) and lower intraoperative blood loss (12.07 ± 6.54 mL vs 17.07 ± 8.29 mL). No significant differences were observed in tourniquet time or operation time between the groups. Besides, adverse events related to the device are minimal in two groups.
Conclusions
The elastic constriction ring is an effective alternative to traditional pneumatic tourniquets, offering significant advantages in reducing exsanguination time and intraoperative blood loss without increasing complication rates.
Brief introduction
The adoption of minimally invasive principles in clinical surgery has marked a progressive evolution in operative techniques, particularly in orthopedic limb surgeries. 1 Tourniquets have emerged as indispensable tools within this domain, primarily because of their ability to minimize bleeding and maintain a clear surgical field. 2 However, the selection of tourniquet type, application duration, inflation pressure, and the skill in application have frequently been associated with complications ranging from tourniquet slippage to suboptimal hemostasis.
In response to the need for more effective hemostatic solutions in surgery, a novel type of elastic constriction ring has been developed.3–5 It consists of an elastic constriction ring, an elastic sleeve, dual-colored tension straps, a handle, and plastic spacers. The tubular elastic sleeve encases the ring, forming a circular structure that wraps around the limb. The tension straps facilitate easy manipulation and work alongside the ring to compress the limb. The ring’s smaller diameter generates a stretching force that effectively expels blood from vessels as it rolls toward the proximal end, achieving integrated exsanguination and hemostasis. This system demonstrates high efficiency and ease of use, making it suitable for bloodless limb surgeries. 6 However, its efficacy and safety in various surgical contexts require thorough evaluation.
Closed distal radius fractures are among the most prevalent limb fractures, with an annual incidence rate of 0.04% to 1.10%.7,8 They are especially common in elderly patients, where osteoporosis increases the risk from minor falls, 9 and in younger athletes during sports activities. 10 These fractures significantly affect wrist function, impacting daily life and work. Consequently, they are crucial in orthopedic practice, requiring careful diagnosis and treatment. With the growing demand for early rehabilitation, minimally invasive approaches via a palmar surgical route have gained popularity, often necessitating the use of a tourniquet for a clear surgical field.11,12
This study employs a prospective randomized controlled design to evaluate the performance of a new elastic constriction ring compared to traditional pneumatic tourniquets. Conducted from May 2021 to December 2022, the study enrolled 60 patients with fresh closed distal radius fractures, who were randomly assigned to either the experimental group (elastic constriction ring) or the control group (pneumatic tourniquet). The focus was on exsanguination time, effectiveness, and complication rates, aiming to highlight the advantages of the innovative device in orthopedic surgery.
The hypothesis suggests that the elastic constriction mechanism will outperform traditional tourniquets by accelerating the exsanguination process, thus reducing intraoperative blood loss. It is anticipated that these benefits will not extend surgery duration or increase postoperative complications, providing a superior clinical alternative.
Materials and methods
Selection and recruitment of participants
CONSORT reporting guidelines were used to guide this study. 13 This prospective clinical trial was conducted exclusively at Cixi People Hospital Medical Health Group (Cixi People Hospital). The total study execution period spanned from January 1, 2021, to December 31, 2022. Subject recruitment commenced on January 12, 2021, and concluded on December 3, 2022. All interventions, data collection, and follow-up assessments were completed within the designated study period at this single-center site. No changes were made after trial commencement.
The sample size was determined a priori based on pilot data: traditional tourniquet group (n = 10) showed 19.2 ± 5.1 mL intraoperative blood loss (primary outcome) versus 14.3 ± 4.6 mL for the elastic ring group (n = 10). Using Zstats Storm Statistics Platform (https://zstats.medsta.cn/samplesize/) (research type = difference, two-tailed α = 0.05, 90% power), minimum requirements were 22.8/group for intraoperative blood loss. Considering the 15% cases of loss to follow-up and refusal to visit, at least 27 cases in the experimental group and 27 cases in the control group are ultimately needed. As a result, we enrolled 30 participants per group.
Participants should meet all of the following criteria for inclusion: (1) Unilateral fresh closed distal radius fractures caused by trauma; (2) Treatment involving the palmar radial approach and internal fixation using locking compression plates for distal radius fractures; (3) Complete closure of the epiphyses; (4) Time from injury to surgery ≤2 weeks; (5) Circumference of the upper forearm between 24 and 40 cm.
Exclusion criteria were applied if participants met any of the following conditions: (1) Open distal radius fractures or pathological fractures; (2) Concurrent ipsilateral ulnar fractures requiring surgical intervention; (3) Forearm circumference less than 24 cm or greater than 40 cm; (4) Presence of thromboangiitis obliterans, venous thrombosis, severe arteriosclerosis, or other vascular diseases in the affected limb; (5) Severe skin damage, limb infection, or malignant tumors in the affected limb; (6) Patients who have previously experienced cardiac risks, respiratory difficulties, or other severe discomfort due to the use of exsanguination devices; (7) Patients whose preoperative systolic blood pressure cannot be stably controlled below 180 mmHg; (8) Participation in other drug or device clinical trials within the last month of screening; (9) Other circumstances deemed unsuitable by the investigator.
Group allocation, randomization, blinding, standardization of interventions
Participants were allocated into two groups based on the intervention method: the experimental group utilized the elastic constriction ring provided by Cixi BLD Medical Instrument Co., Ltd, while the control group received the conventional exsanguination and hemostasis approach using a pneumatic tourniquet supplied by Zhejiang Guangci Medical Device Co., Ltd. The application of both the elastic constriction ring and the pneumatic tourniquet strictly adhered to the respective product manuals and established surgical standards.
The random allocation sequence was generated by an independent biostatistician using SPSS software (version 26.0), employing a block randomization design with variable block sizes of 4–6 participants. This approach ensured balanced group assignment (experimental vs control) throughout the recruitment period. Allocation concealment was strictly maintained using sequentially numbered, opaque, sealed envelopes. Each envelope contained the group assignment and was opened only after participant enrollment to prevent selection bias.
An independent biostatistician generated the randomization sequence without involvement in recruitment or intervention. Research staff enrolled eligible participants after confirming inclusion criteria and obtaining informed consent. A designated study coordinator—uninvolved in outcome assessment—assigned participants to interventions by opening the next sequentially numbered envelope, revealing group allocation (elastic constriction ring or pneumatic tourniquet). This separation of roles ensured impartial sequence implementation.
Due to the visible differences between devices, participants and surgeons could not be blinded to group assignments. However, outcome assessors (evaluating surgical field efficacy, complications) and data analysts remained fully blinded. Assessors received anonymized data without group identifiers, while analysts processed coded datasets. Blinding was maintained by restricting access to allocation records; only the study coordinator and surgical team knew group assignments during the trial.
To standardize procedural aspects, surgical teams received uniform training on device application and removal, and consistent surgical management practices were followed across both groups. This standardization aimed to minimize confounding factors, ensuring that any outcome differences were attributed to the exsanguination and hemostasis methods.
Surgical methodology
The procedure was performed under brachial plexus block, with the patient in a supine position. A volar approach utilizing the path of the flexor carpi radialis (FCR) was employed. The FCR tendon was retracted ulnarly, and the radial artery and vein were gently retracted radially to expose the pronator quadratus muscle. An incision was then made at the radial insertion of the pronator quadratus to reveal the fracture site. Following anatomical reduction, a volar plate was applied. The wound was thoroughly irrigated, the pronator quadratus was reconstructed, and the incision was closed in layers. The volar approach utilizing the FCR is a well-established and standardized surgical approach. Given the high familiarity of the operating surgeon with this technique, the risk of iatrogenic injury to major vessels is minimal. Consequently, to maintain a clear and clean surgical field, we elected to remove the tourniquet only after the incision was thoroughly irrigated and completely sutured. Besides, due to the specific surgical approach utilized, intraoperative bleeding is generally minimal. Consequently, surgical drains were not routinely inserted in either group.
In the experimental group, the new exsanguination device was used. As shown in Figure 1, after measuring the upper forearm circumference, the device was prepared and positioned. The surgeon pulled the handles to roll the elastic constriction ring proximally, while an assistant held the distal limb. The surgical site was moistened, and the sleeve was cut open. A plastic spacer was inserted post-procedure, and the ring was cut for removal. Operating procedure for the elastic constriction ring.
In the control group, a traditional pneumatic tourniquet was applied after padding the limb and performing exsanguination with an Esmarch bandage, followed by tourniquet inflation to achieve hemostasis. The tourniquet pressure was set to 250–300 mmHg, which is also consistent with the research of Pereira A et al. 14 After surgery, the tourniquet was deflated and removed.
Outcome measures
The assessment criteria encompassed both efficacy and safety indicators.
Efficacy indicators included preoperative exsanguination and hemostasis time, tourniquet time, operation time, intraoperative blood loss, and the effectiveness of exsanguination and hemostasis in the surgical field. Intraoperative blood loss was measured by the weight difference between soaked and dry gauze, plus the volume in the suction bottle. The effectiveness of exsanguination and hemostasis in surgical field was categorized as excellent (During surgery, no bleeding/bruising with clear anatomical layers; after decompression, slight skin pressure marks without redness/blisters, immediate hemodynamic restoration, and normal nerve function.), good (During surgery, slight bleeding without obstructing dissection; after decompression, noticeable pressure marks with mild redness/minimal blisters, prompt hemodynamic restoration, and intact nerve function.), or bad (During surgery, significant bleeding impeding visualization/procedure; after decompression, large/blistered skin lesions, limb congestion/swelling, and neurological deficits.).
Safety indicators focused on the rate of adverse events during the use of research medical devices. Adverse events, whether device-related or not, were categorized into mild (slight discomfort, no impact on daily activities), moderate (significant discomfort affecting regular activities), severe (hindering routine activities), and serious (resulting in death or major health deterioration). Device-related adverse events were identified as reactions that followed known patterns of the device, occurred in a reasonable timeframe post-treatment, could not be explained by other causes, and potentially improved after cessation of the treatment.
Statistical methods
Statistical analysis for results was performed using SPSS software (version 26.0), while graphical representations were generated with GraphPad Prism 9.
The chi-square test was employed to compare the gender distribution, AO fracture classification, and the incidence of complications related to the tourniquet between the experimental and control groups. While the exsanguination and hemostasis effect cannot be analyzed using the chi-square test because not all expected values exceed 1.0. Independent samples t-tests were used to compare the height, body mass index (BMI), basal blood pressure, circumference of tourniquet site, preoperative exsanguination and hemostasis time, tourniquet time, operation time, and intraoperative blood loss between the two groups. Welch’s t-test was utilized to compare the weight differences between the groups. Lastly, the Mann-Whitney test was used to compare the ages of the participants in the experimental and control groups, due to the non-normal distribution of age data. P-values <0.05 was considered to be statistically significant.
Results
Study population and baseline analysis
The entire study actually included a total of 60 enrolled participants (30 in the experimental group and 30 in the control group). All participants successfully completed the trial. Completed CONSORT flow chart was shown in Figure 2. CONSORT flow diagram of this study.
As shown in Figure 3(a), in the experimental group, there were 13 males (43.33%) and 17 females (56.67%), while the control group comprised 11 males (36.67%) and 19 females (63.33%). The gender distribution did not reveal any statistically significant differences between the two groups. Regarding age, the average for the experimental group was 53.87 ± 12.33 years, compared to 56.5 ± 9.24 years in the control group, with no significant difference observed (Figure 3(b)). When examining baseline blood pressure, the experimental group had an average of 17.41 ± 2.16 kPa, while the control group had a slightly higher average of 17.9 ± 2.22 kPa; again, these differences were not statistically significant (Figure 3(c)). For height, the experimental group averaged 161.9 ± 9.35 cm, whereas the control group’s average height was 160.6 ± 7.46 cm, with no significant disparity noted (Figure 3(d)). In terms of weight, the experimental group reported an average of 64.38 ± 11.40 kg, in contrast to 60.38 ± 7.66 kg for the control group, but this difference also lacked statistical significance (Figure 3(e)). Similarly, the average BMI in the experimental group was 24.4 ± 2.56 kg/m2, while the control group had a mean BMI of 23.4 ± 2.55 kg/m2, showing no significant difference between the groups (Figure 3(f)). Lastly, regarding the circumference of the tourniquet site, the experimental group had an average measurement of 24.8 ± 1.66 cm, compared to 24.26 ± 1.76 cm in the control group, with no significant difference found (Figure 3(g)). Demographic and baseline characteristics of participants. Distribution of gender (a), age (b), basal blood pressure (c), height (Td), weight (e), body mass index (BMI) (f), circumference of tourniquet site (g), and AO classification (h) across the two groups. Ns, no significance. P-values <0.05 were considered statistically significant (indicated with *), 0.01 (indicated with **), 0.001 (indicated with ***), and 0.0001 (indicated with ****).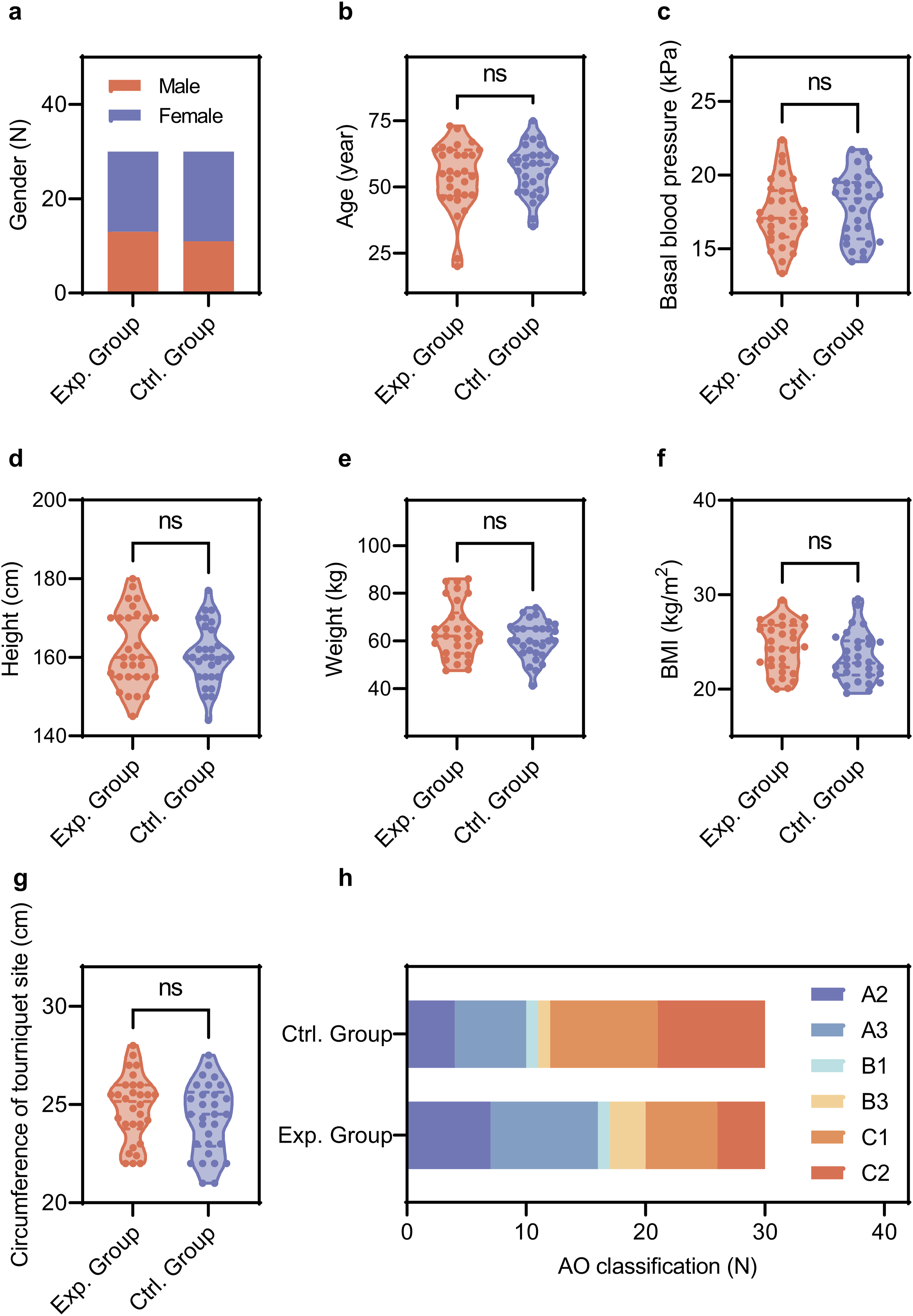
The AO classification system categorizes fractures by anatomical location and complexity, aiding in treatment decisions and outcome predictions. While variations in fracture types were observed—more cases of A2 and A3 in the experimental group and C1 and C2 in the control group—these differences were not statistically significant (Figure 3(h)). This suggests a similar distribution of fracture types across both groups, enabling a reliable comparison of treatment outcomes.
Efficacy and safety indicator analysis
For the effectiveness indicators, as illustrated in Figure 4(a), the experimental group required an average of 8.36 ± 2.72 s for preoperative exsanguination and hemostasis, significantly less than the 55.98 ± 3.61 s needed by the control group, with this difference proving statistically significant. However, the average tourniquet time in the experimental group was 65.03 ± 14.58 min, compared to 65.13 ± 12.71 min in the control group, indicating no significant statistical difference between the two groups (Figure 4(b)). Similarly, the average operation time for the experimental group was 63.00 ± 13.63 min, while the control group had a mean duration of 64.73 ± 14.36 min, and again, no significant difference was noted (Figure 4(c)). In terms of intraoperative blood loss (primary outcome), the experimental group exhibited an average blood loss of 12.07 ± 6.54 mL, which was significantly lower than the 17.07 ± 8.29 mL observed in the control group (Figure 4(d)). Regarding the effectiveness of exsanguination and hemostasis during the procedure, the experimental group recorded 29 excellent outcomes, 1 good, and 0 poor, while the control group had 24 excellent, 5 good, and 1 poor outcome. Although the number of excellent ratings in the experimental group exceeded that of the control group, there was no statistically significant difference between the two (Figure 4(e)). Comparison of efficacy and safety indicators between the experimental group and the control group. Comparison of preoperative exsanguination and hemostasis time (a), tourniquet time (b), operation time (c), intraoperative blood loss (d), and exsanguination and hemostasis effect (e) in the surgical field between the experimental group and the control group. (f) Occurrence of relative adverse events between the experimental group and the control group. Ns, no significance. P-values <0.05 were considered statistically significant (indicated with *), 0.01 (indicated with **), 0.001 (indicated with ***), and 0.0001 (indicated with ****).
In terms of safety indicators, the experimental group reported 5 cases of tourniquet-related complications, including slight numbness or pain occurring 1 to 2 days post-surgery. The control group experienced 2 cases of pain on the first day after the procedure. However, no statistically significant difference was found in the incidence of device-related adverse events between the two groups (Figure 4(f)).
Discussion
In this study, the efficacy and safety of the elastic constriction ring were systematically evaluated against traditional pneumatic tourniquets in patients with closed distal radius fractures.
The application of the elastic constriction ring in distal radius fracture surgeries presents several advantages. It significantly reduces exsanguination time compared to traditional pneumatic tourniquets, as demonstrated by statistically significant findings in this study. This efficiency stems from its integrated design, which streamlines the process by eliminating the need for protective padding, securing straps, limb elevation, and Esmarch bandages. Moreover, traditional pneumatic tourniquets may not always be sterile, potentially harboring bacteria, which necessitates the use of sterile dressings. 14 By positioning the tourniquet 5 cm proximal to the incision, the elastic constriction ring enhances the aseptic field and increases sterilization efficacy, providing more operational space for the surgeon. Finally, intraoperative blood loss was significantly lower in the experimental group due to the ring’s close adherence to the forearm, which delivers uniform pressure on underlying tissues, enhancing hemostatic effectiveness. The consistent pressure distribution of the elastic constriction ring minimizes variability associated with human factors, improving overall exsanguination efficiency compared to traditional methods.
While the elastic constriction ring offers several advantages, there are important considerations to keep in mind during its use. Firstly, the pressure value of the elastic constriction ring is relatively fixed, and its application must be adjusted based on the preoperative limb circumference. Although some literature suggests that the circumference of the limb at the tourniquet site can serve as a reference for inflation pressure, it is generally accepted that determining the pressure based on systolic blood pressure or limb occlusion pressure is more scientifically sound.15–17 This discrepancy could potentially lead to excessive or insufficient pressure, thereby impacting the effectiveness of hemostasis. Secondly, during the application of the elastic constriction ring, there is a possibility of exacerbating damage to the surrounding tissues, blood vessels, and nerves near the fracture site. Although we did not observe any nerve or vascular injury during our usage, we recommend that an assistant provide continuous traction to the fracture site while rolling the ring to minimize the risk of local tissue and vascular damage. Lastly, the elastic constriction ring does not display the duration of use, necessitating reliance on manual or external timing methods, and it cannot be reused, which somewhat limits its application in surgical settings.
While this study demonstrated significant advantages of the elastic constriction ring in exsanguination efficiency and hemostasis, several methodological considerations should be noted. The single-center design and moderate sample size (n = 60) may limit generalizability, though rigorous randomization and standardized protocols enhanced internal validity. Potential performance bias was mitigated through blinded outcome assessment despite the inherent inability to blind surgeons to device type. Strict adherence to predefined endpoints minimized selective reporting, and the consistent surgical approach across groups reduced confounding.
Measurement precision was optimized through protocolized techniques, though inherent variability in these clinical measurements exists. Statistical analyses revealed high precision in primary outcome, while secondary endpoints like hemostasis effectiveness showed greater variability. Although multiple comparisons were conducted, the prespecified primary endpoint maintained strong effect sizes without multiplicity adjustment, aligning with the trial’s exploratory aims. Future studies with larger cohorts could further refine precision estimates for secondary outcomes.
The results demonstrate applicability to adult patients undergoing volar plating for closed distal radius fractures within the forearm circumference range (24–40 cm), representing common clinical scenarios. While the single-center design may limit broad extrapolation, the inclusion of both osteoporotic elderly and younger trauma patients—coupled with consistent outcomes across AO fracture types (A2–C2)—supports relevance to diverse demographic and injury-severity subgroups. Standardized surgical protocols mirror real-world practice, though universality to open fractures, complex comorbidities, or non-specialized centers requires further validation.
Conclusion
In summary, the elastic constriction ring is a significant advancement in surgery, providing a time-efficient and user-friendly solution that effectively reduces intraoperative blood loss. Its design allows for consistent pressure application without the complex setup of traditional pneumatic tourniquets, thus enhancing surgical efficiency and maintaining a low incidence of tourniquet-related complications. However, limitations such as a small sample size and insufficient follow-up duration must be acknowledged. Further research with larger cohorts and extended follow-up is needed to validate its safety and efficacy in diverse surgical contexts, ultimately improving patient care and outcomes.
Supplemental Material
Supplemental Material - A prospective randomized controlled study on the effect of a novel exsanguination tourniquet in fresh closed distal radius fracture surgery
Supplemental Material for A prospective randomized controlled study on the effect of a novel exsanguination tourniquet in fresh closed distal radius fracture surgery by Binbin Sun, Jianjie Xu, Jianhao Yu, Songlin Tong, Yi Zhang, Weifeng Zhou in Journal of Orthopaedic Surgery.
Supplemental Material
Supplemental Material - A prospective randomized controlled study on the effect of a novel exsanguination tourniquet in fresh closed distal radius fracture surgery
Supplemental Material for A prospective randomized controlled study on the effect of a novel exsanguination tourniquet in fresh closed distal radius fracture surgery by Binbin Sun, Jianjie Xu, Jianhao Yu, Songlin Tong, Yi Zhang, Weifeng Zhou in Journal of Orthopaedic Surgery.
Footnotes
Ethical considerations
This study was in accordance with the Declaration of Helsinki. Approval was granted by the Ethics Committee of Cixi People Hospital Medical Health Group (Cixi People Hospital) (Date. 12/25/2020/No. 2020-LP-KY008).
Consent to participate
Informed consent was obtained from all patients for being included in the study.
Author contributions
Binbin Sun: Funding Acquisition, Investigation, Writing Original Draft. Jianjie Xu: Statistics, Data Curation. Jianhao Yu: Investigation. Songlin Tong: Investigation, Software. Yi Zhang: Investigation. Weifeng Zhou: Funding Acquisition, Writing–Review & Editing. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Zhejiang Provincial Natural Science Foundation of China under Grant No. LGF20H060002, and Cixi City Agriculture and Social Development Science and Technology Program under Grant No. CN2020008.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated or analyzed during this study are included in this manuscript. The full trial protocol and the completed CONSORT checklist are available in the Supplemental Files.
Trial registration (Name,URL,Number,Date)
Supplemental Material
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
