Abstract
Introduction
Proximal femoral tumor resection poses a major challenge in orthopaedic oncology, particularly in very young pediatric patients (<9 years), where the need to preserve function and limb length collides with the limited availability of reconstructive options (3D-printed implants, endoprostheses, biological constructs such as vascularized fibular grafts, and allograft-prosthetic composites) or leads to amputation.
Objective
To describe in detail the surgical technique for proximal femoral tumor resection and reconstruction using an allograft-prosthetic composite (APC) with a proximal humerus allograft plus a hip prosthesis, documenting its applicability and outcomes regarding function, allograft survival, and oncologic disease status in two pediatric cases treated at a national cancer referral center in Bogotá, Colombia.
Methods
We report two pediatric cases and detail the surgical technique used for proximal femoral reconstruction after tumor resection, based on an APC (proximal humerus allograft + hip prosthesis). Data were collected from medical records with radiological and functional follow-up.
Results
In both patients, proximal femoral reconstruction using our technique yielded a stable construct with preservation of limb function (MSTS >72), documented graft incorporation at 3 months, local disease control, and only one recorded complication (contact dermatitis).
Conclusion
Proximal femoral reconstruction using an APC with a proximal humerus allograft is a viable and safe surgical option in pediatric patients with Ewing sarcoma. In our experience, the technique was successfully applied in two patients, allowing limb preservation with a favorable course. Thus, it offers a functional and reliable option for limb salvage in selected cases.
Keywords
Introduction
Osteosarcoma and Ewing sarcoma are the two most common primary bone sarcomas in the pediatric and young-adult population, and because of their location they pose a therapeutic challenge due to the segment’s biomechanical demands and the need for reconstructions that preserve length, support, and mobility.1,2
Comprehensive oncologic management combines chemotherapy, wide resection, and functional reconstruction, and the choice of technique depends on patient age, tumor location, and implant availability.3,4
In very young pediatric patients, conventional modular prostheses have limitations (available sizes, risk of physeal injury, loss of bone stock, or long-term loosening), making their use problematic at extreme ages.5–7
Biologic reconstructions and allograft–prosthetic composites (APCs) are valuable alternatives because they allow the reconstruction of the soft tissues, preserve bone stock, and improve function; nevertheless, they carry risks (resorption, nonunion, fracture, infection). 8 In the absence of viable reconstructive options, radical procedures such as amputation or rotationplasty remain considerations, albeit with greater functional and psychosocial implications.9,10
Against this backdrop, developing techniques that broaden reconstructive options in pediatric patients is crucial. The technique we describe—an allograft–prosthetic composite based on a proximal humerus allograft—aims to provide a reproducible and biologically favorable option within the few alternatives available for this challenging segment.
Preoperative planning
A multidisciplinary plan was carried out to define resection margins, allograft dimensions, prosthesis size, and the overall reconstructive strategy, a prior request was made to the bone bank for the available allografts along with their respective dimensions, and in these cases, we selected the grafts that best matched the measurements of the host bone, using length, outer diameter, and medullary canal diameter as reference parameters. We elected not to reconstruct the acetabular component and instead to use a bipolar hemiarthroplasty. Other authors have reported that, in young patients treated with bipolar hemiarthroplasty, adding an acetabular component entails a significant risk of acetabular erosion, migration or loosening, and higher revision rates. Studies such as Dudani et al. show a low incidence of erosion when the acetabulum is omitted or preserved, whereas cases with an acetabular component tend to experience greater mid-term complications. Therefore, in our two cases, avoiding acetabular reconstruction and using a bipolar hemiarthroplasty is consistent with the prior evidence (Figure 1). Pelvis and femur MRI. (a) Coronal; (b and c). Axial.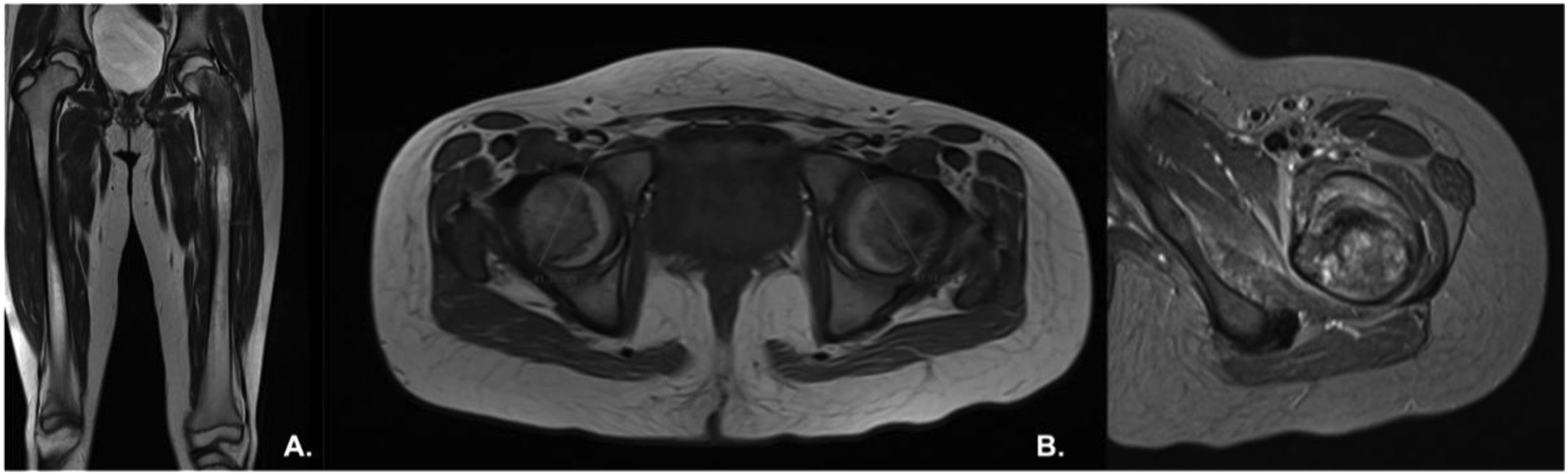
Approach
The procedure was performed under general anesthesia with the patient in the supine position and a bolster placed under the left buttock (Figure 2). An anterolateral (Watson–Jones) approach was used, starting the incision at the anterior superior iliac spine (ASIS) and extending distally to include the biopsy tract. During layered dissection, the lateral femoral cutaneous nerve was identified and protected. Separation of the tensor fasciae latae and gluteus mediusallowed exposure of the joint capsule. The rectus femoris was carefully retracted medially, avoiding tension on the femoral nerve and preserving its insertion. The iliopsoas was identified along the anterior aspect of the joint and kept intact throughout the exposure. The external rotators were selectively released from their femoral insertions while protecting the posterior course of the sciatic nerve, permitting safe access to the proximal femur. Patient in supine position, with a lump under the left buttock.
First stage: Tumor resection
Dissection of the rectus femoris is performed, followed by identification, protection, and detachment of the gluteus medius, adductor magnus, and psoas. Tumor resection is carried out using the vastus lateralis, vastus intermedius, and vastus medialis as surgical margins. A femoral osteotomy is performed 15 cm distal to the tip of the greater trochanter, securing a 2-cm bony margin.
Removal of the tumor specimen is achieved via a hip dislocation maneuver and controlled traction using a bone clamp from distal to proximal, after careful identification and protection of the femoral neurovascular bundle and the sciatic nerve. A capsulotomy is performed, preserving a remnant for subsequent reconstruction, and the ligamentum teres is resected, enabling en bloc resection of the specimen, which is sent to pathology (Figure 3). (a) Resected specimen; (b) Iliopsoas (arrow), adductor magnus (double arrow), gluteuus medios (asterisk).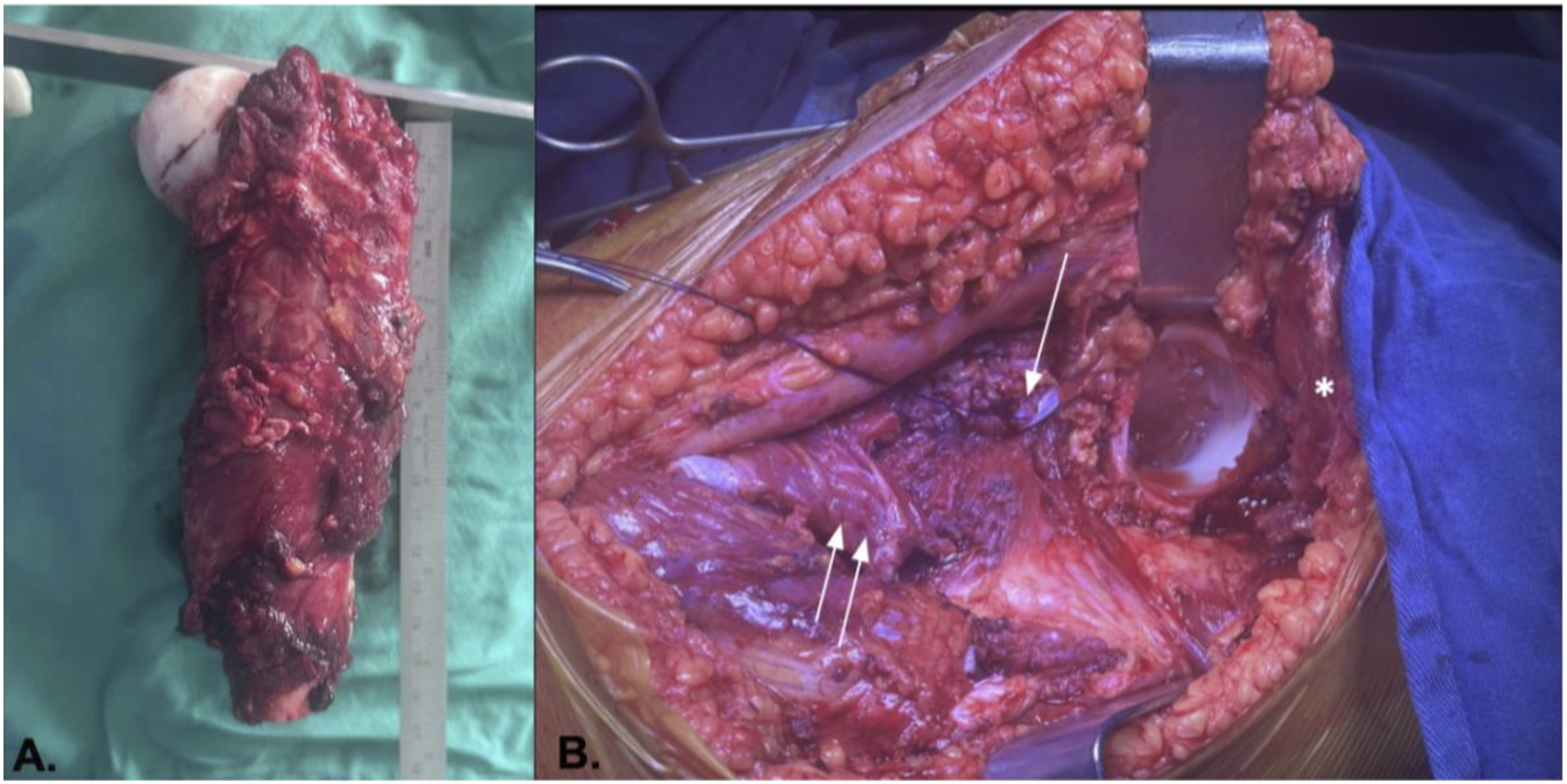
Second stage: Reconstruction
The proximal humerus allograft is prepared and soaked in normal saline containing gentamicin 240 mg and cefazolin 2 g as antibiotic prophylaxis (Figure 4(a)). A bayonet osteotomy is fashioned at 15 cm of allograft length, leaving a 2-cm distal segment intended for contact with native bone (Figure 4(b)). (a) Proximal humerus allograft; (b) Bayonet osteotomy; (c) Alloprosthetic composite; (d) Capsular reconstruction, E. Augmentation with osteosynthesis in the alloprosthesis-native bone transition zone.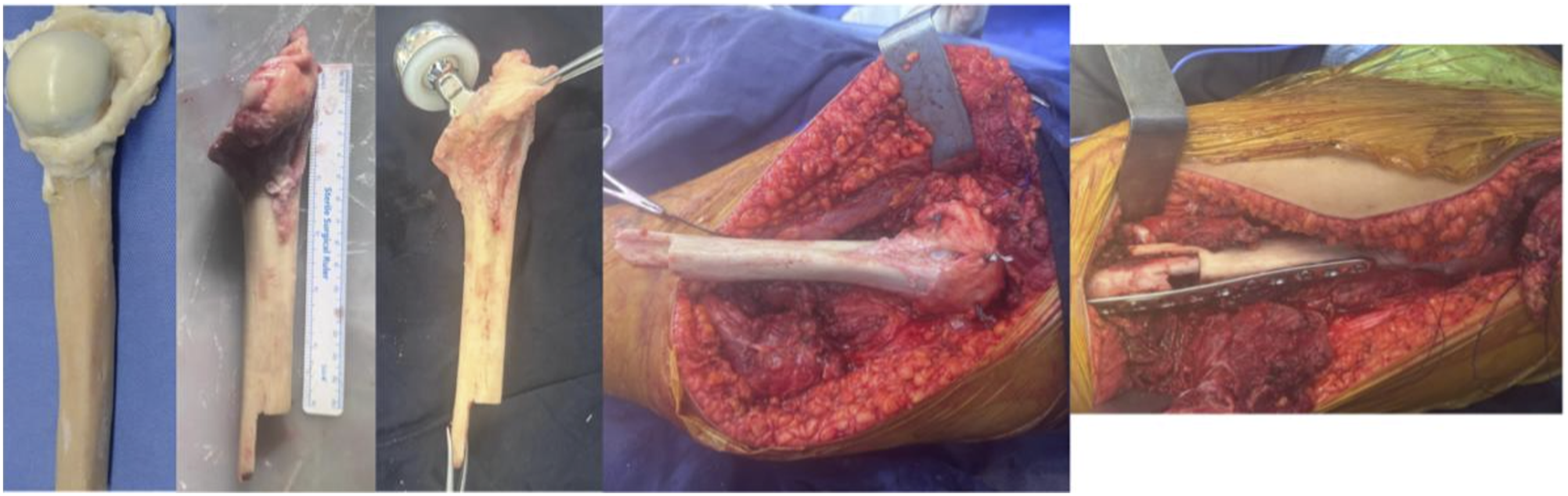
Ex vivo, on the back table, the allograft–prosthetic composite (APC) is assembled using a box osteotome/entry awl, followed by progressive broaching and rasping. Trialing with stem #1 was adequate, and we selected a cemented, neck-preserving femoral stem. Reconstruction was completed with a negative-offset modular head and a 41-mm bipolar cup, with reduction and stability tests yielding satisfactory results (Figure 4(c)).
The femoral canal is cemented using antibiotic-loaded bone cement, and the final stem, cup, and head are implanted. Alloprosthetic reduction into the acetabulum is complemented with capsular reconstruction, using the patient’s capsular remnant and multiple suture loops at 12, 3, 6, and 9 o’clock, anchored to the rotator cuff footprint and the allograft capsule to achieve stable containment (Figure 4(d)).
The distal junction of the APC is secured with two interfragmentary compression screws across the bayonet osteotomy. The allograft–host interface is protected with an LCP reconstruction plate, fixed with three proximal and three distal screws, to promote incorporation and protect the APC from early mechanical failure. Limb stability, length, and alignment are verified (Figure 4(e)).
Myotendinous reconstruction is performed with transfer of the gluteus medius, adductor magnus, and psoas, as well as reinsertion of the vastus lateralis, gracilis, and gluteus maximus. The hip capsular remnant is also sutured to the capsular remnant of the proximal humerus allograft (Figure 5). Postoperative X-rays.
Tips and tricks
Contour the distal allograft in a bayonet configuration to maximize contact area and stability. Augment with osteosynthesis (plate fixation) to protect the transition zone between the allograft and native bone. Cement the allograft canal to provide immediate prosthetic stability. Reinforce stability with capsular reconstruction by suturing the hip capsular remnant to the capsular remnant of the proximal humerus allograft. Include in the myotendinous reconstruction the gluteus medius and maximus, adductor magnus, hamstrings, and psoas, ensuring adequate muscular coverage. Maintain close clinical and radiographic surveillance in the early postoperative period, focusing on fixation stability, consolidation of the graft–host interface, and early detection of infectious complications.
Case reports
Case 1
Nine-year-old pediatric patient diagnosed with Ewing sarcoma of the proximal femur, treated with neoadjuvant chemotherapy. At week 13 of treatment, a wide resection with negative margins and reconstruction with an allograft–prosthetic composite were performed as described, the rehabilitation protocol consisted of nighttime use of an abduction pillow, restricted weight-bearing, and physical therapy focused on optimizing hip range of motion for 45 days, followed by weight-bearing as tolerated and progressive ambulation using a walker at the end of this period. At the 6-months follow-up, the patient demonstrated a favorable course (MSTS >72), graft incorporation at 3 months, no infection, no local recurrence, and a single complication—contact dermatitis, which resolved (Figure 6(a)). (a) Patient follow-up radiograph Case 1; (b) Patient follow-up radiograph Case 2.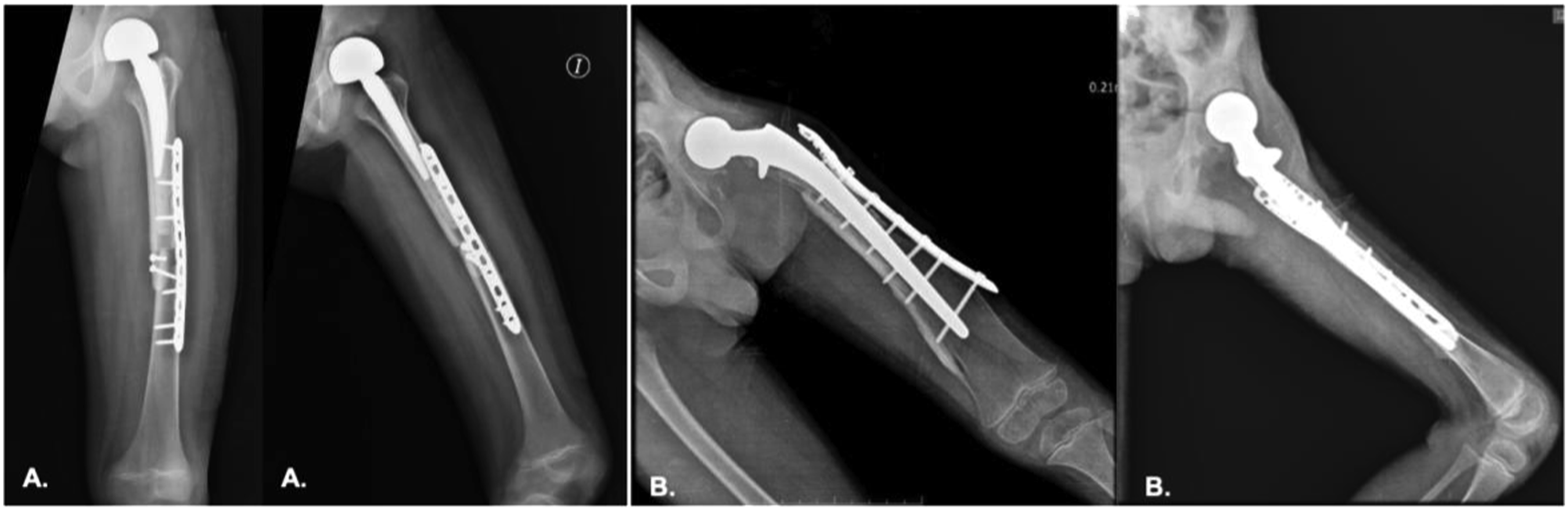
Case 2
A two-year-old pediatric patient diagnosed with Ewing sarcoma of the proximal femur, after initial oncologic therapy, wide tumor resection and reconstruction with an allograft–prosthetic composite were carried out, the rehabilitation protocol consisted of nighttime use of an abduction pillow, restricted weight-bearing, and physical therapy focused on optimizing hip range of motion for 45 days, followed by weight-bearing as tolerated and progressive ambulation using a walker at the end of this period. At the 6-months follow-up, the patient showed a favorable course (MSTS 83), graft incorporation at 4 months, no infection, no local recurrence, and no complications (Figure 6(b)).
Discussion
Proximal femoral tumor resection in pediatric patients at extreme ages is a major challenge, as reconstructive options must restore function, preserve limb length, and avoid mutilating procedures. The use of massive allografts and their combination with prosthetic components has been reported with acceptable functional outcomes in pediatric and young-adult populations.7,11–13
The aim of this technique is to offer a biological and mechanically stable alternative when modular prostheses are not viable, either because of a mismatch between implant size and the immature skeleton or due to limitations in availability. In our cases, we opted to use proximal humerus allografts combined with a hip prosthesis, because the available proximal femur allografts are excessively large for pediatric patients at extreme ages, such as those presented here (<9 years). This decision enabled better anatomic adaptation and cortical congruence without compromising mechanical stability or limb length. Both patients were at the +1 percentile for height and weight according to Colombian growth and development guidelines (Case 1: Weight 35 kg, Height 140 cm; Case 2: Weight 12 kg, Height 85 cm), which facilitated dimensional compatibility between the proximal humeral allograft and the resected femur (Case 1: Total femur length 34 cm, Resected segment 15 cm; Case 2: Total femur length 28 cm, Resected segment 12 cm). To date, there are no prior reports in the literature describing the use of a proximal humerus allograft combined with a hip prosthesis in an extreme pediatric population. This approach therefore represents a safe and reproduciblealternative in skeletally immature patients when conventional options—such as femoral allografts or pediatric modular prostheses—are not feasible or are disproportionate anatomically.7,11–13
In a previous case report from our surgical group, we demonstrated the feasibility of this reconstructive strategy and underscored the importance of precise preoperative planning and myotendinous reconstruction as a pillar of dynamic stability. 10 The present series reinforces those findings and adds evidence regarding the applicability of allograft–prosthetic composite reconstruction in pediatric patients, with satisfactory short-term functional outcomes.
From a technical standpoint, employing a bayonet osteotomy not only facilitates congruence between the graft and native bone, but also increases the contact surface and therefore the mechanical stability of the union. This surgical detail, together with supplementary fixation using a reconstruction plate and interfragmentary compression screws, is a decisive factor for construct integration.12,13
Capsular and myotendinous reconstruction—including the gluteus medius, gluteus maximus, adductor magnus, psoas, and hamstrings—plays a key role in restoring function by providing active stability and helping to reduce the risk of dislocation. This strategy becomes even more relevant in pediatric patients at extreme ages, given the reduced soft-tissue volume and the difficulty in achieving adequate coverage.11,12
Although our results are encouraging, inherent risks must be considered, such as infection, graft fracture, prosthetic component loosening, and limb-length discrepancy in long-term follow-up.13,14 Accordingly, periodic clinical and radiological surveillance is essential to detect early complications and ensure the durability of the reconstruction.
Our findings are consistent with the international literature and reinforce the utility of allograft–prosthetic compositereconstruction as a valid technique in selected pediatric scenarios.
Conclusions
Proximal femoral reconstruction using an allograft–prosthetic composite with a proximal humerus allograft and a hip prosthesis is a reproducible and functional alternative in pediatric patients with Ewing sarcoma. In our experience, it enabled local disease control, limb preservation, and satisfactory functional outcomes, with early graft incorporation and a low complication rate.
This technique offers biological advantages—restoration of tendon insertion sites and preservation of bone stock—together with immediate mechanical stability provided by the prosthetic component. Additionally, the decision not to reconstruct the acetabular component and to use a bipolar cup aligns with prior reports indicating higher risks of chondrolysis and acetabular loosening in young patients when conventional acetabular components are used.15,16 Although larger series and longer follow-up are needed to establish durability, the present experience provides evidence that this strategy can be considered a safe and reliable option for limb salvage in selected pediatric scenarios.14,17
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
