Abstract
Background
Traumatic arthritis leads to progressive cartilage degeneration, ultimately causing joint dysfunction. Due to cartilage’s limited ability to self-repair, damage often extends to bone and joint tissues. This study investigates the correlation between prostaglandin-endoperoxide synthase 2 (PTGS2) expression and cartilage injury in traumatic arthritis, aiming to assess PTGS2 as a potential biomarker for early diagnosis.
Methods
We analyzed data from 181 patients diagnosed with traumatic arthritis between January 2022 and August 2024. Serum levels of PTGS2 and cartilage biomarkers were measured and compared between patients with and without cartilage injury. Logistic regression and ROC curve analysis were used to evaluate the diagnostic efficacy of PTGS2.
Results
PTGS2 levels were significantly higher in patients with cartilage injury compared to those without (p < 0.05). Correlation analysis showed that PTGS2 was positively correlated with cartilage biomarkers (COMP, CTX-II) and identified as an independent risk factor for cartilage injury. ROC analysis revealed that PTGS2 had good diagnostic performance with an AUC of 0.820, sensitivity of 94%, and specificity of 68.42%.
Conclusion
PTGS2 is a promising biomarker for diagnosing cartilage injury in post-traumatic osteoarthritis and may serve as a target for early diagnosis and treatment.
Introduction
Post-traumatic osteoarthritis (PTOA) is primarily caused by traumatic events such as traffic accidents, high-impact sports, and military training, with a particularly high incidence among young individuals and athletes. PTOA can affect any joint in the body; however, weight-bearing joints are most frequently involved.1,2 It is estimated that approximately 12% of symptomatic osteoarthritis cases in weight-bearing joints, including the hip, knee, and ankle, are attributed to PTOA. 3 In the early stages, most patients exhibit no apparent clinical symptoms, while pain progressively intensifies in the middle and late stages of the disease. Additionally, some patients experience involvement of subchondral bone and surrounding ligaments, ultimately leading to progressive cartilage degeneration.4,5 Due to the lack of blood supply to cartilage, its repair ability is limited, so it is difficult for damaged cartilage to recover itself. If the lesion is not intervened in time, it may further involve adjacent bones and joint tissues, affecting limb function and quality of life.6,7 In recent years, with the in-depth study of PTOA cartilage injury, how to diagnose PTOA cartilage injury early and explore its potential pathogenesis has become an important focus. Currently, the diagnosis of PTOA primarily relies on imaging modalities such as X-ray, computed tomography (CT), and magnetic resonance imaging (MRI). While these techniques provide valuable diagnostic insights, they are limited in their ability to accurately assess early-stage cartilage damage. 8 Emerging evidence suggests that ferroptosis, a regulated form of iron-dependent cell death, plays a crucial role in osteoarthritis progression. Articular cartilage consists of chondrocytes embedded within an extracellular matrix, the homeostasis of which is maintained through a dynamic balance between matrix synthesis and degradation. 9 Ferroptosis impairs the antioxidant defense mechanisms of chondrocytes, leading to oxidative stress and cellular dysfunction. Consequently, patients with systemic ferroptosis may experience accelerated articular cartilage degeneration, exacerbating joint damage and promoting the onset and progression of osteoarthritis following injury or trauma. 10
Ferroptosis is a newly discovered form of programmed cell death that is different from traditional apoptosis, necrosis or autophagy, which is characterized by the generation of intracellular iron-dependent reactive oxygen species and lipid peroxidation. 11 In PTOA cartilage injury, the specific mechanism of ferroptosis has not been fully elucidated, and the exploration of its biomarkers has gradually become a hot topic. Among them, prostaglandin-endoperoxide synthase 2 (PTGS2) is a key indicator. PTGS2 is an important enzyme involved in inflammatory response and lipid metabolism, and plays an important role in ferroptosis with a dual role. 12 On one hand, as a key catalytic enzyme involved in oxidative metabolism, the upregulation of PTGS2 expression is closely associated with the accumulation of lipid peroxidation products, suggesting that PTGS2 may serve as a central mediator in the ferroptosis-inducing signaling pathway. On the other hand, its overexpression is often accompanied by iron-dependent cellular damage and accelerated programmed cell death, further establishing PTGS2 as a critical biomarker of ferroptosis.13,14 Recent studies have identified PTGS2 as a key regulator of ferroptosis, with its role being particularly pronounced in arthritis pathogenesis. 15 Therefore, exploring the expression of PTGS2 in traumatic arthritis and its association with cartilage degeneration may not only enhance our understanding of PTOA pathogenesis but also provide novel targets for early diagnosis and therapeutic intervention. This study focuses on PTGS2 as a ferroptosis-related biomarker, analyzing its serum expression patterns in patients with traumatic arthritis, assessing its correlation with cartilage damage, and evaluating its diagnostic potential for PTOA-related cartilage injury. Through a systematic analysis of these factors, we aim to contribute valuable insights for future research on the pathophysiology and clinical management of PTOA.
Materials and methods
General information
Patients with traumatic arthritis admitted to our hospital from January 2022 to August 2024 were selected as the research subjects. The sample size was calculated using the estimated rate n = Z2·P·(1-P)/d2, where n is the sample size; Z is the quantile of the standard normal distribution with a 95% confidence level, Z = 1.96; P is the prevalence, and the prevalence of PTOA is 37.9% 16 ; the allowable error range of d is set to 6.95%, and considering the dropout rate of 15%, the required sample size is 187. The study was conducted in accordance with the Declaration of Helsinki and approved by the institutional ethics committee (Approval No. JPH202412233). Written informed consents from all patients were obtained in any experimental work with humans.
PTOA diagnostic criteria (independent of arthroscopy): PTOA was diagnosed based on (i) a documented history of joint trauma; (ii) persistent osteoarthritis-compatible symptoms (pain, stiffness, functional limitation) lasting ≥3 months after injury; and (iii) imaging evidence of osteoarthritis, defined as Kellgren–Lawrence (KL) grade ≥2 on weight-bearing radiographs (MRI was not part of the protocol). Arthroscopy was not used to establish the diagnosis of PTOA; rather, it was performed for clinical indications and used in this study to characterize cartilage status. Accordingly, participants who met clinical/imaging criteria for PTOA but had no macroscopic or microscopic cartilage damage at arthroscopy were classified as “PTOA without arthroscopic cartilage injury.”
Inclusion criteria: (1) Meeting the PTOA diagnostic criteria above and confirmed by imaging techniques such as computed tomography (CT) or X-ray (weight-bearing radiographs preferred for KL grading); (2) History of trauma affecting at least one weight-bearing joint (knee, ankle, or hip) due to external mechanical forces, unbalanced weight distribution, or gravitational insufficiency; (3) Intact cruciate and collateral ligaments without evidence of insufficiency.
Exclusion criteria: (1) Presence of acute inflammatory diseases or joint infections; (2) History of neurological diseases or muscle weakness in the hip, knee, or ankle; (3) Immature skeletal development or congenital malformations; (4) Significant impairment of vital organ function, including renal failure or liver dysfunction; (5) History of prior lower limb surgery; (6) Diagnosis of systemic or inflammatory arthropathies, including rheumatoid arthritis, gout, diabetes-related arthropathy, suppurative arthritis, and non-traumatic arthritis; (7) Presence of bone-related conditions such as tuberculosis or bone malignant tumors; (8) Osteoporosis-induced arthritis.
Imaging
All participants underwent weight-bearing radiographs (anteroposterior and lateral; patellofemoral skyline or ankle mortise views when applicable). Computed tomography (CT) was obtained only when clinically indicated (e.g., fracture planning, assessment of intra-articular comminution or malalignment). Radiographic findings were abstracted with a standardized template (Kellgren–Lawrence grade, osteophytes, joint-space narrowing, subchondral sclerosis/cysts, and malalignment). Radiographs were reviewed preoperatively to inform care; arthroscopy served as the reference standard for cartilage surface status. A qualitative correlation between radiographic severity (KL grade) and arthroscopic cartilage grade (Outerbridge) was recorded at the joint level.
Patient identification and selection
Patient identification and selection. Consecutive patients evaluated for PTOA and screened from outpatient clinics and inpatient admissions logs. Eligibility was assessed sequentially against the prespecified inclusion/exclusion criteria (see above). Screening and enrollment proceeded in four stages: (1) records screening; (2) clinical/imaging verification of PTOA; (3) exclusion of systemic/inflammatory arthritis and prior lower-limb surgery; and (4) confirmation of data completeness (clinical labs and serum biomarkers). A CONSORT-style flow diagram details the number assessed for eligibility, excluded with reasons at each stage, enrolled (n = 181), and analyzed in the cartilage-injury versus no-injury subgroups.
Definition of “traumatic arthritis.” In this study, “traumatic arthritis” refers to post-traumatic osteoarthritis attributable to joint trauma. All included cases were PTOA following an acute index injury (e.g., fracture, dislocation, or ligamentous injury).
Rationale for PTOA without arthroscopic cartilage injury. PTOA was diagnosed clinically and radiographically; arthroscopy was used only to characterize cartilage status. Consequently, some PTOA patients had no macroscopic/microscopic chondral defects at arthroscopy and were categorized as “PTOA without arthroscopic cartilage injury.” Synovial-fluid aspiration or cartilage biopsy solely for research was not mandated by protocol; intra-articular tissue sampling occurred only when clinically indicated.
Grouping and clinical data collection
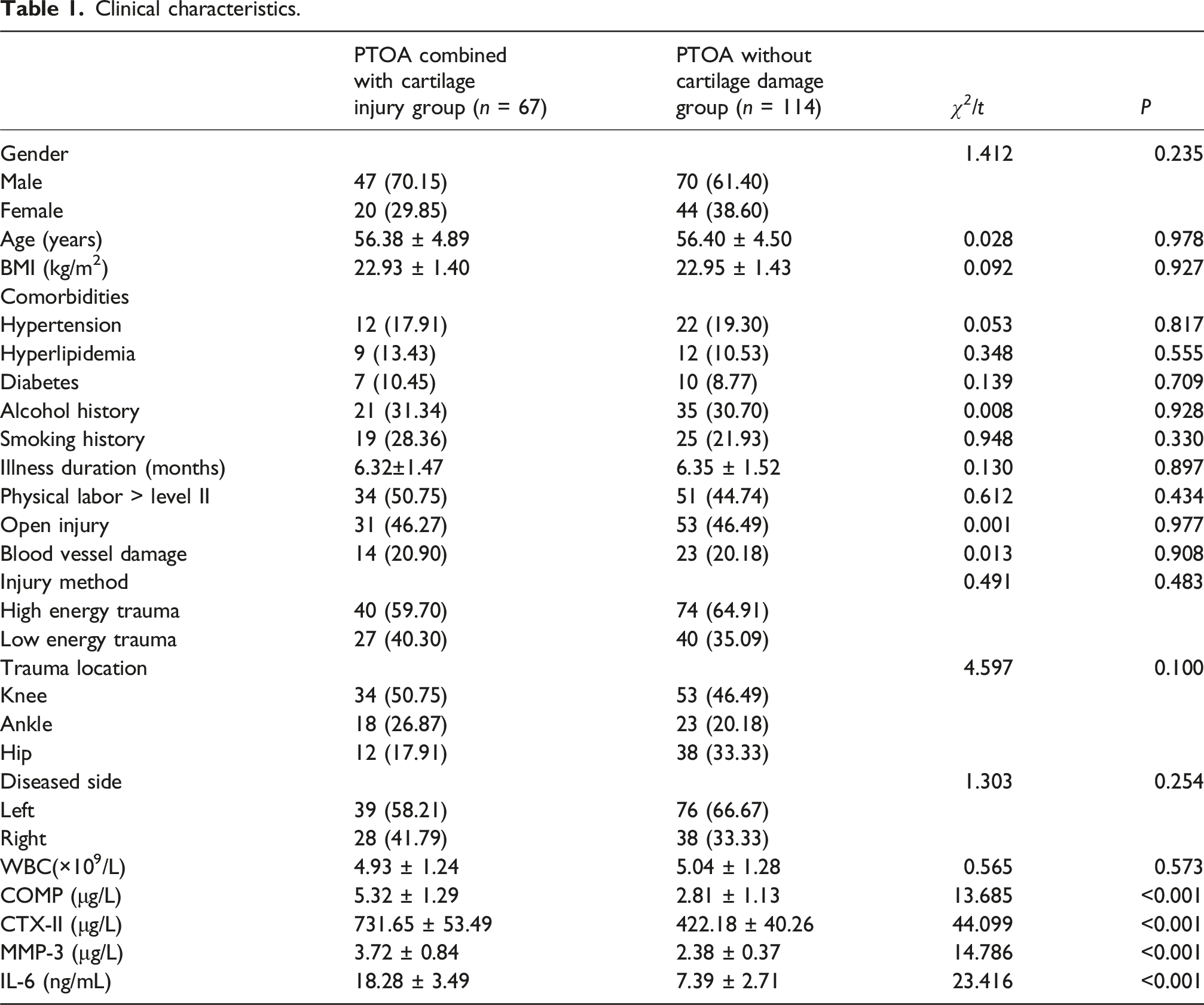
Totally, 181 patients with traumatic arthritis were included according to the inclusion and exclusion criteria. Arthroscopic examination was performed after admission. The diagnostic criteria for cartilage injury were as follows: rough cartilage surface, cracks, cartilage detachment or subchondral bone exposure under the microscope were considered cartilage injury. During arthroscopy, cartilage damage was graded using the Outerbridge classification (Grade 0–IV) as the primary system; when sufficient detail was available, the International Cartilage Repair Society (ICRS) grading (Grade 0–4) was also recorded in a sensitivity analysis. For patients without visible lesions, the grade was recorded as Outerbridge 0 (ICRS 0). Two experienced surgeons, blinded to laboratory data, performed grading; disagreements were resolved by consensus. The patients were divided into groups according to whether they were combined with cartilage injury, and were divided into PTOA combined with cartilage injury group and PTOA without cartilage injury group. Data such as age, gender, BMI, comorbidities (hypertension, hyperlipidemia, diabetes), drinking history, smoking history, illness time, physical labor > II level, open injury, vascular injury, injury method, trauma location, disease side, white blood cell (WBC), and interleukin-6 (IL-6) levels were collected.
Serum PTGS2 and cartilage damage markers detection
All subjects need to fast for 10 h. In the morning of the next day, 6 mL of fasting venous blood was collected and centrifuged at 4°C and 3000r/min for 10 min to remove impurities. The upper serum was taken and stored at −80°C. Serum PTGS2, COMP, CTX-II, and MMP-3 were measured using commercial sandwich ELISA kits according to the manufacturers’ instructions (PTGS2: CUSABIO, Wuhan, China; Cat. CSB-E10103h; COMP: R&D Systems/Bio-Techne, Minneapolis, MN, USA; Quantikine Cat. DCMP0; CTX-II: Thermo Fisher Scientific/Invitrogen, Waltham, MA, USA; Cat. EEL037; MMP-3: R&D Systems/Bio-Techne, Minneapolis, MN, USA; Quantikine Cat. DMP300), with personnel blinded to clinical and arthroscopic data. Serum was allowed to clot prior to centrifugation; aliquots were limited to a single freeze–thaw cycle, and grossly hemolyzed/lipemic samples were excluded; time from collection to freezing and storage duration were recorded. Each analyte was assayed in duplicate on 96-well plates with 7–8 point calibrators and two in-house pooled serum controls (low/high); standard curves used four-parameter logistic (4PL) regression, with plate acceptance criteria of R2 ≥ 0.99, control recoveries 85%–115%, and duplicate CV ≤10%; samples with duplicate CV >15% or recovery outside 80%–120% were reassayed and excluded for that analyte if still out of range. Lot-verified analytical performance was: PTGS2—LOD 2.0 pg/mL, reportable range 5–200 pg/mL, intra-assay CV ≤6.5%, inter-assay CV ≤9.0%; COMP—LOD 0.20 µg/L, range 1–50 µg/L, intra-assay CV ≤5.8%, inter-assay CV ≤8.7%; CTX-II—LOD 0.02 µg/L, range 0.05–3.0 µg/L, intra-assay CV ≤6.0%, inter-assay CV ≤9.5%; MMP-3—LOD 0.10 µg/L, range 0.30–20 µg/L, intra-assay CV ≤5.2%, inter-assay CV ≤8.3% (units: PTGS2 pg/mL; COMP, CTX-II, MMP-3 µg/L). To mitigate batch effects, samples were randomized across plates/batches by group (injury vs no-injury); assays were completed across [N] plates and [K] reagent lots with pooled-serum bridging controls on every plate and Levey–Jennings monitoring, yielding inter-plate CVs ≤ [7–10] % for all analytes; plate/batch IDs were recorded for sensitivity analyses. Values below LOD were imputed as LOD/√2; values above range were diluted and re-assayed per kit IFUs; pre-specified outlier rules (±3 SD after QC) triggered repeat testing, and confirmed outliers were retained with flagging. Only serum was assayed in this study; synovial fluid and cartilage tissue were not routinely collected or analyzed for PTGS2 or the other markers. This design choice limits direct inference about local (intra-articular) ferroptosis–systemic linkage and is acknowledged in the Discussion/Limitations.
Flow chart
Figure 1 shows the flow chart of this research. It reports counts screened, excluded with reasons, enrolled (n = 181), and analyzed by arthroscopic cartilage-injury status. Study flow diagram: Screening, exclusions (with reasons), and final numbers analyzed for patients with and without cartilage injury (arthroscopy-defined). Abbreviations: PTOA, post-traumatic osteoarthritis.
Statistical analysis
SPSS 23.0 was used to process the data, and GraphPad Prism was used to generate the figures. In this manuscript, s denotes the sample standard deviation. The data that met the normal distribution were expressed as (
Results
Clinical characteristics
Among the 181 patients diagnosed with post-traumatic osteoarthritis (PTOA), the mean age was 56.39 ± 9.39 years. The cohort included 124 males and 57 females, of whom 67 patients exhibited cartilage damage. Based on the presence or absence of cartilage injury, patients were stratified into two groups, and their clinical characteristics were compared. Trauma type. All enrolled cases were acute trauma progressing to PTOA.
Clinical characteristics.
Arthroscopic grading/staging of cartilage damage
Cartilage status was classified intraoperatively using the Outerbridge system (Grade 0–IV) as the primary grading method; when feasible, the International Cartilage Repair Society (ICRS) grading (Grade 0–4) was also recorded in a sensitivity analysis. Participants who met clinical/imaging PTOA criteria but had no macroscopic or microscopic cartilage damage at arthroscopy were recorded as Grade 0 (“PTOA without arthroscopic cartilage injury”). The grade distribution is summarized in Table S1. Radiographs and arthroscopy demonstrated overall qualitative concordance for identifying moderate-to-severe lesions, with most discordances occurring in low-grade disease.
Expression of serum ferroptosis-related index PTGS2 in patients with PTOA cartilage injury
The PTGS2 level in the PTOA combined cartilage injury group was (34.20 ± 5.39) pg/mL, and in the PTOA not combined cartilage injury group was (12.38 ± 2.91) pg/mL. The PTGS2 level in the PTOA combined cartilage injury group was significantly higher than that in the PTOA not combined cartilage injury group (p < 0.05, Figure 2). Serum PTGS2 levels by cartilage-injury status. Bar plot of PTGS2 (pg/mL) comparing patients with versus without cartilage injury. Bars show group mean; error bars show ± SD; dots represent individual participants. Groups compared with a two-sided test (see Methods). Significance is indicated by a single asterisk: P < 0.05 (*). Cartilage injury was graded per Outerbridge (0–IV) as the primary system and ICRS (0–4) where available. Preoperative imaging consisted of weight-bearing radiographs for all participants and CT in selected cases; arthroscopy served as the reference for cartilage surface status.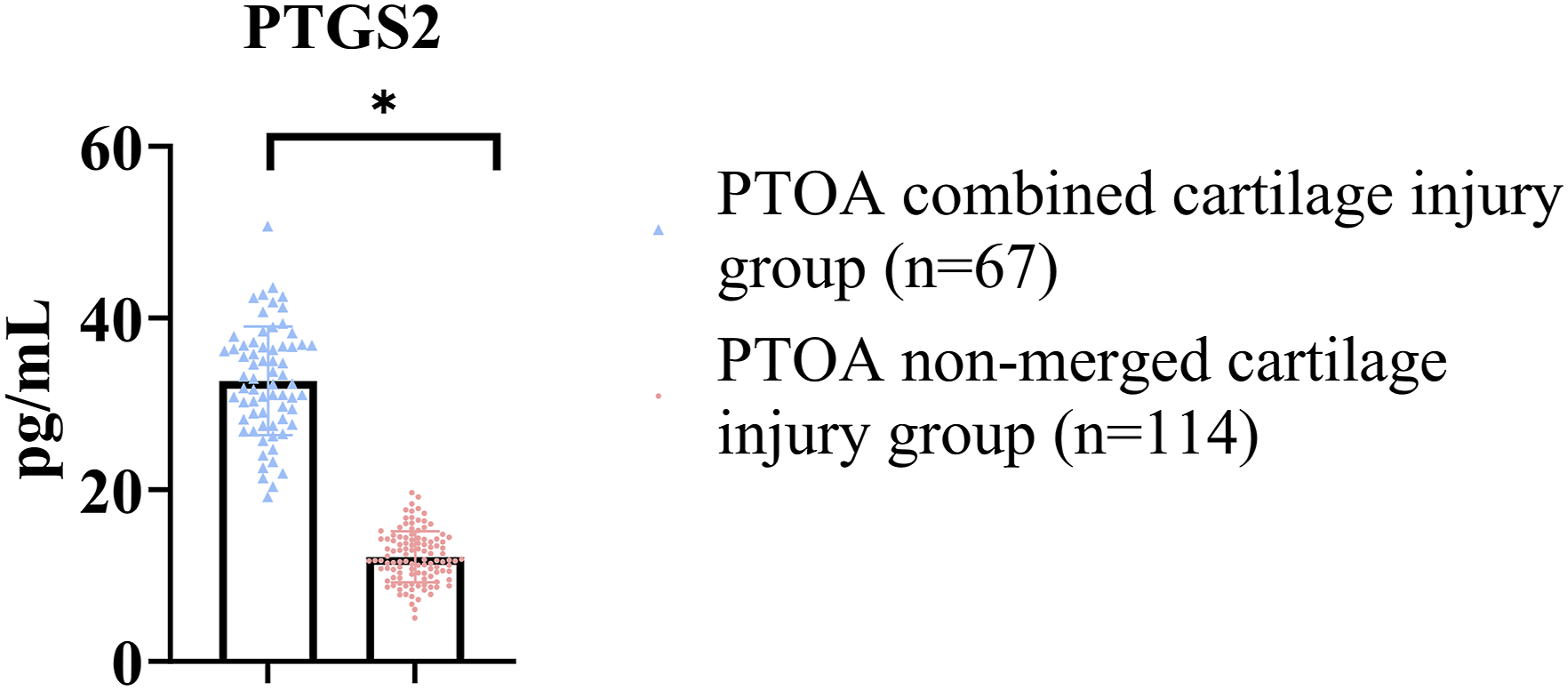
Severity analysis of PTGS2 across cartilage grades
Beyond presence/absence, PTGS2 levels differed across Outerbridge grades (overall comparison by Kruskal–Wallis, p < 0.05). Post-hoc Dunn tests (Bonferroni-adjusted) showed higher PTGS2 in moderate-to-severe damage (Outerbridge ≥2) versus Grades 0–1 (adjusted p < 0.05). A monotonic increase in PTGS2 with increasing grade was observed (Jonckheere–Terpstra trend test, p < 0.05).
Correlation analysis of serum iron death related index PTGS2 and cartilage markers
To further explore the relationship between PTGS2 and cartilage injury, Pearson correlation analysis showed that PTGS2 was positively correlated with COMP and CTX-II (p < 0.05). There was no correlation between PTGS2 and MMP-3 (p > 0.05), as shown in Figure 3. In addition, PTGS2 correlated positively with arthroscopic cartilage grade (Spearman’s test, p < 0.05). Correlations between PTGS2 and cartilage-degradation markers. Scatter plots showing (A) PTGS2 (pg/mL) versus COMP (µg/L), (B) PTGS2 (pg/mL) versus CTX-II (µg/L), and (C) PTGS2 (pg/mL) versus MMP-3 (µg/L). Dots denote individual participants; line indicates least-squares fit with 95% CI. Each panel reports the correlation coefficient (r) and two-sided P.
Analysis of influencing factors of cartilage injury caused by PTOA
Analysis of influencing factors of cartilage injury caused by PTOA.

Efficacy analysis of PTGS2, a serum iron death-related index, in diagnosing cartilage injury caused by PTOA
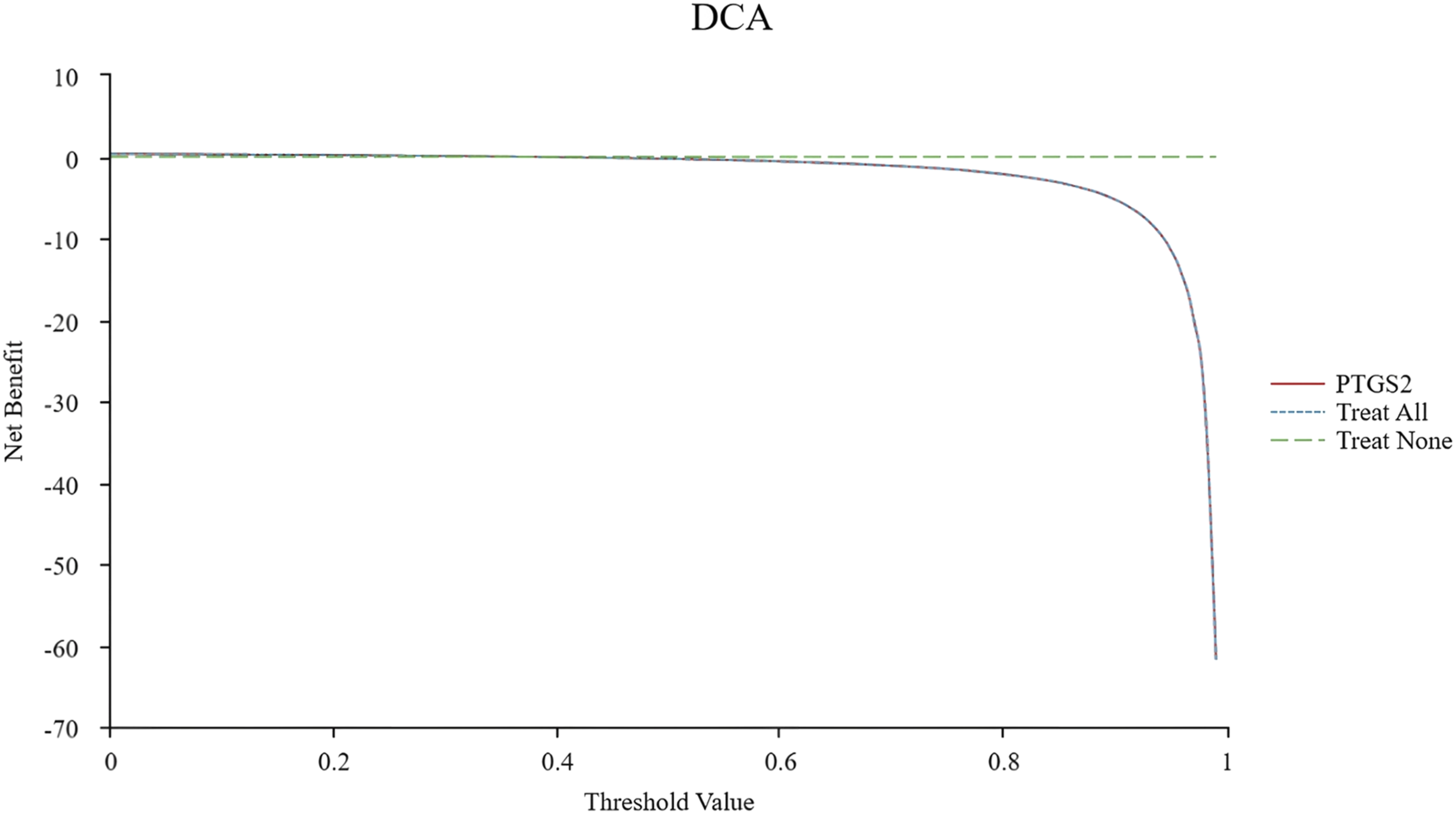
Receiver operating characteristic (ROC) curve analysis demonstrated that PTGS2 exhibited strong diagnostic performance for cartilage injury in PTOA, with an area under the curve (AUC) of 0.820 (95% CI: 0.758–0.991). The sensitivity and specificity were 94.00% and 68.42%, respectively, with a Youden index of 0.625. The optimal cutoff value for PTGS2 was determined to be 15.73 pg/mL, as shown in Figure 4. For clinical interpretability, PTGS2 also discriminated moderate-to-severe damage (Outerbridge ≥2), as shown in an exploratory ROC analysis (Fig. S1). Additionally, decision curve analysis (DCA) indicated that PTGS2 provided a favorable net benefit for diagnosing cartilage damage in PTOA when the threshold probability ranged from 0.6 to 1.0, further supporting its potential clinical utility (Figure 5). Diagnostic performance of PTGS2 for cartilage injury. Receiver operating characteristic curve plotting Sensitivity (—) versus 1 − Specificity (—) for PTGS2 (pg/mL). AUC with 95% CI is reported; the point marker indicates the Youden-index cut-off with corresponding sensitivity/specificity. Decision-curve analysis (DCA) of PTGS2-based classification. Net benefit across Threshold Probability (%) for a PTGS2-based strategy versus treat-all and treat-none. If the software reports benefit per 100 patients, the y-axis is labeled Net Benefit (per 100 patients).
Discussion
Cartilage is the only cell type in articular cartilage, and the survival of chondrocytes plays an important role in ensuring the integrity of articular cartilage and the balance in joint. Some studies found that a large number of ferroptosis (iron death) death markers were accumulated in the chondrocytes with iron death in osteoarthritis, especially in the damaged position. Further single cell RNA sequencing research was carried out on the chondrocytes with iron death, and it was found that a large number of iron death genes were preferentially expressed in the chondrocytes with iron death.17,18 Scholars such as X. Yao 19 have implicated ferroptosis in chondrocytes as a potential contributor to the progression of osteoarthritis. Their findings suggest that iron-handling pathways may exert a protective, anti-osteoarthritic effect within the body. Under conditions of inflammation and iron overload, chondrocytes can undergo ferroptosis, which could accelerate osteoarthritis progression by disrupting fundamental cellular mechanisms. This process also may inhibit the expression of type II collagen in chondrocytes cultured in vitro, further impairing cartilage integrity and function. Blocking the sensitivity of chondrocytes to ferroptosis has been reported in preclinical work to mitigate osteoarthritic changes, and have certain protection for chondrocytes, so our data should be interpreted as associative and hypothesis-generating rather than causal, so it is speculated that iron death is involved in the occurrence of cartilage injury. 20
Among many iron death markers, PTGS2 is widely concerned and used as one of the key markers of iron death. PTGS2 is a downstream effector molecule of iron death, which directly reflects the occurrence of iron death. Compared with other ferroptosis markers, such as lipid peroxidation and intracellular iron ion levels, PTGS2 exhibits higher specificity. Its upregulation is strongly associated with ferroptosis, while it remains largely unaltered in other forms of regulated cell death, making it a more reliable indicator of ferroptosis-related cellular processes. 21 In the process of iron death, the increased activity of PTGS2 may lead to a large number of lipid peroxides and accelerate the formation of iron-dependent non-apoptotic cell death, which is mainly caused by the excessive accumulation of lipid peroxides and reactive oxygen species in iron metabolism, so the change of PTGS2 activity may affect the process and result of iron death.22,23 In our results, we found that the serum PTGS2 level of patients with PTOA combined with cartilage injury was significantly higher than that of patients without cartilage injury (p < 0.05). We distinguished diagnostic criteria for PTOA based on clinical history and imaging from arthroscopic characterization of cartilage. This explains why some PTOA patients exhibited no macroscopic/microscopic chondral defects at arthroscopy (early PTOA may present with symptoms, synovitis, meniscal/subchondral changes, or MRI abnormalities before overt surface lesions). By incorporating standardized grading (Outerbridge/ICRS) and analyzing severity rather than presence/absence alone, we observed a dose-response relationship between serum PTGS2 and cartilage damage. In our cohort, higher serum PTGS2 was associated with cartilage injury and with higher arthroscopic grades; these observational findings are correlative and do not establish a causal role for ferroptosis in PTOA. PTGS2 is a key molecule related to iron death, and its increased expression may be closely related to the occurrence of cartilage injury. 24 Ferroptosis is a form of cell death characterized by iron-dependent lipid peroxidation, and the significant increase of PTGS2 suggests that it may participate in the pathological process of cartilage injury in patients with PTOA by promoting lipid peroxidation and inflammatory reaction. 25 Local–systemic linkage. While serum PTGS2 correlated with arthroscopic severity, serum may incompletely capture intra-articular ferroptotic activity. Future studies should include paired sampling of serum and synovial fluid with PTGS2 quantification, and PTGS2 assessment in cartilage tissue (e.g., immunohistochemistry, Western blot/qPCR) obtained during clinically indicated arthroscopy to test concordance across serum–synovial–cartilage compartments.
In order to further verify the relationship between PTGS2 and PTOA, the markers of cartilage injury, COMP, CTX-II and MMP-3, were screened, all of which were important factors of cartilage injury, and their abnormal increase reflected cartilage matrix degradation and chondrocyte damage. COMP is an extracellular matrix protein secreted by chondrocytes, which exists in articular cartilage in large quantities. When cartilage undergoes damage or degeneration, COMP is released into the synovial fluid and bloodstream. Its upregulation serves as an indicator of altered chondrocyte metabolism and structural degradation of the cartilage matrix, reflecting progressive joint deterioration. 26 CTX-II is a small molecular fragment secreted in the degradation of type II collagen, which can reflect the degradation degree of cartilage matrix. It is found that type II collagen is the key structural protein of cartilage and CTX-II is an important marker in cartilage degeneration, and the level of CTX-II is positively correlated with the severity of cartilage degeneration. 27 MMP-3 is an important matrix metalloproteinase, which can degrade many components in cartilage matrix, including type II collagen and aggrecan. In arthritis and other diseases, the expression of MMP-3 increased significantly, which accelerated the degradation of cartilage matrix. 28 Therefore, MMP-3 is not only one of the main drivers of cartilage injury, but also considered as a potential marker of disease diagnosis and treatment targets. 29 Our Pearson correlation analysis further indicated that PTGS2 was positively correlated with cartilage markers COMP and CTX-II (p < 0.05), but not significantly correlated with MMP-3 (p > 0.05). This result suggests that PTGS2 may be involved in cartilage injury by influencing some cartilage metabolism processes. Due to the high expression of iron death related index PTGS2 in the process of arthritis and cartilage injury, and its pro-inflammatory effect will accelerate the degradation of cartilage matrix and promote the secretion of cytokines. 30 COMP and CTX-II are important markers in cartilage metabolism, which reflect the synthesis and metabolic degradation of cartilage matrix respectively. 31 There is a positive correlation between PTGS2 and these two markers, suggesting that PTGS2 may directly or indirectly affect the degradation and reconstruction of cartilage matrix by regulating the metabolic activity of chondrocytes in the process of joint degeneration. However, the correlation between PTGS2 and MMP-3 was not statistically significant. While MMP-3, a key matrix-degrading enzyme, contributes to cartilage degeneration, its regulatory mechanisms may operate independently of the PTGS2 signaling pathway. Nevertheless, further research is required to determine whether a potential interaction exists between PTGS2 and MMP-3 in the context of cartilage degradation.
To clarify the risk factors of cartilage injury in PTOA, logistic regression was used to analyze the effects of various influencing factors. Among them, COMP, CTX-II, MMP-3, IL-6 and PTGS2 were all confirmed as important risk factors for cartilage injury (p < 0.05), and the OR value of PTGS2 was 2.672 (95% CI: 1.397 - 5.113), which showed that they had significant independent effects on the occurrence of PTOA cartilage injury. This result re-emphasizes the key role of PTGS2 in the pathological mechanism of cartilage injury. However, due to the limited sample size, constructing a multi-index joint prediction model may introduce overfitting or instability, potentially compromising its predictive reliability. Additionally, previous studies have shown that the expression levels of certain cartilage markers can be influenced by multiple factors, leading to poor stability and limiting their clinical applicability. In contrast, PTGS2 has a well-defined role in cartilage injury and demonstrates high stability and specificity across different studies. Therefore, this study focused on evaluating the predictive value of PTGS2 as a single biomarker rather than developing a multi-index joint prediction model. Furthermore, given the complexity of multi-index models, their clinical utility may be restricted. PTGS2, as a single biomarker, offers a simpler and more stable detection method, making it more feasible for routine clinical application.
Interpretation and scope
Our results position serum PTGS2 as a candidate biomarker that correlates with arthroscopic cartilage damage and with established markers of matrix turnover. However, the present study is observational and cannot determine whether ferroptosis is causally upstream of cartilage degeneration in PTOA. The associations we report should therefore be viewed as hypothesis-generating, pending mechanistic validation in cell and animal models and in longitudinal human studies.
Therapeutic implications (hypothesis-generating)
If future studies substantiate a causal contribution of ferroptosis to chondral pathology, pharmacologic approaches that modulate ferroptotic pathways e.g., lipid peroxidation inhibitors, iron chelation, or GPX4 pathway augmentation could plausibly attenuate cartilage damage. At present, these strategies remain speculative and require rigorous preclinical testing and clinical trials to determine efficacy and safety in PTOA.
This study has some limitations that should be acknowledged. First, this was a single-center study, which may limit generalizability. Second, the precise molecular mechanisms underlying PTGS2-mediated ferroptosis were not extensively explored. Third, due to study constraints, validation in animal or cellular models was not performed. Fourth, a formal radiograph–arthroscopy correlation was not prespecified and is acknowledged as a limitation. We did not prespecify a formal, compartment-level statistical correlation between radiographic severity and arthroscopic grades; this should be quantified in future work. Given the sample size and the EPV constraint, the primary model was intentionally parsimonious; however, we confirmed robustness in adjusted sensitivity models. Residual confounding is still possible. Additionally, PTGS2 (and other markers) were measured only in serum; synovial fluid and cartilage tissue were not assayed, limiting inference about whether systemic levels mirror intra-articular ferroptosis. This motivates future paired serum–synovial–cartilage validation with correlation to Outerbridge/ICRS grades. Future work should include multi-omics approaches (e.g., transcriptomics, metabolomics) and longitudinal follow-up to clarify PTGS2’s role in PTOA-related cartilage injury and its clinical utility.
Conclusion
This study revealed the important role of PTGS2 in the cartilage injury of PTOA and found that PTGS2 increased in the cartilage injury of PTOA, and was positively correlated with cartilage markers, which was one of the risk factors of cartilage injury and had a certain diagnostic efficiency. Taken together, our findings support PTGS2 as an associative serum signal of cartilage injury severity in PTOA. While they align with a mechanistic model involving ferroptosis, causality cannot be inferred from this cross-sectional analysis. Future mechanistic experiments and longitudinal studies are needed to test whether targeting ferroptosis can modify disease trajectories.
Supplemental Material
Supplemental Material - Expression of serum ferroptosis related index PTGS2 in patients with traumatic arthritis and its correlation with secondary cartilage injury and diagnostic efficiency analysis
Supplemental Material for Expression of serum ferroptosis related index PTGS2 in patients with traumatic arthritis and its correlation with secondary cartilage injury and diagnostic efficiency analysis by Zhiyong Zhu, Yong Tang, Kaiyuan Wu, Fan Zhu, Zhiliang Luo, and Xiaoming Fu in Journal of Orthopaedic Surgery
Footnotes
Ethical considerations
The current study was approved by the Ethics Committee of the 922th Hospital of Joint Logistics Support Force, PLA (approval number JPH202412233).
Consent to participate
Written informed consents from all patients were obtained in any experimental work with humans.
Author contributions
Conceptualization: Zhiyong Zhu, Yong Tang, Xiaoming Fu; Data Curation: Zhiyong Zhu, Yong Tang, Kaiyuan Wu, Fan Zhu, Xiaoming Fu; Formal Analysis: Zhiyong Zhu, Yong Tang, Zhiliang Luo, Xiaoming Fu; Funding Acquisition: Xiaoming Fu; Investigation: Zhiyong Zhu, Yong Tang, Kaiyuan Wu, Xiaoming Fu; Methodology: Zhiyong Zhu, Yong Tang, Fan Zhu, Xiaoming Fu; Project Administration: Xiaoming Fu; Resources: Zhiyong Zhu, Yong Tang, Zhiliang Luo, Xiaoming Fu; Software: Zhiyong Zhu, Yong Tang, Fan Zhu, Xiaoming Fu; Supervision: Zhiyong Zhu, Yong Tang, Kaiyuan Wu, Xiaoming Fu; Validation: Zhiyong Zhu, Yong Tang, Zhiliang Luo, Xiaoming Fu; Visualization: Zhiyong Zhu, Yong Tang, Kaiyuan Wu, Fan Zhu, Xiaoming Fu; Writing - Original Draft: Zhiyong Zhu, Yong Tang, Kaiyuan Wu; Writing - Review & Editing: Fan Zhu, Zhiliang Luo, Xiaoming Fu.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
