Abstract
Background
The autologous peroneus longus tendon (PLT), which is a common graft in sports medicine. However, superior fulcrum reconstruction (SFR) using autologous PLT for irreparable massive rotator cuff tears (IMRCT) have not been reported. The objective of this study is to histologically evaluate the effect of the SFR using autologous PLT on tendon-to-bone healing.
Methods
30 mature New Zealand white rabbits were used in this study, and were used to build the model of irreparable massive rotator cuff tears, which were randomly divided into two groups of 15 rabbits in each group, with superior fulcrum reconstruction (SFR group) and superior capsule reconstruction (SCR group). Five rabbits in each group were sacrificed at 4, 8 and 12 weeks postoperatively for histological assessment.
Results
In the histological analysis, there was no significant difference in the modified tendon maturation scoring system and semiquantitative evaluation of the cartilage staining area at 4 weeks. For 8 weeks and 12 weeks, SFR group confirmed a higher scoring level than SCR group. In the Immunohistochemical evaluation, the SFR group showed better angiogenesis and higher expression of collagen I compared to the SCR group at 8 weeks and 12 weeks.
Conclusion
The superior fulcrum reconstruction using autologous PLT can promote a better biological healing process. It could be used as a clinical option for irreparable massive rotator cuff tears.
Keywords
Introduction
The prevalence of rotator cuff tears (RCT) is well-documented to escalate with advancing age. 1 Recent statistics 2 reveal that 8% of the United States population experiences shoulder pain and dysfunction, with rotator cuff tears accounting for 50% of these cases. Currently, there exists a lack of consensus regarding irreparable massive rotator cuff tears (IMRCT), characterized by involvement of two or more tendons or a tear measuring ≥5 cm.3,4 Treatment modalities for IMRCT primarily encompass partial repair, tendon transfer, patch augmentation, superior capsule reconstruction (SCR), subacromial balloon implantation, and reverse shoulder arthroplasty. 5 However, these procedures are associated with elevated postoperative retear rates, graft absorption, infection, non-union between graft and bone, immune rejection, and fat infiltration, among other challenges.6–9 Hence, there is an urgent clinical demand for a surgical approach that significantly alleviates shoulder pain, enhances shoulder mobility, and restores overall shoulder function.
In 1988, Gerber 10 proposed the utilization of latissimus dorsi transfer as a therapeutic approach for massive tears of the rotator cuff. This marked the gradual rise in attention to the concept of the “rotator cuff force couple balance.” The balance of the rotator cuff musculature, particularly the force couple balance in the coronal plane, is critical for maintaining glenohumeral joint stability and functional integrity.11,12 The reconstruction of the rotator cuff force couple balance encompasses both static and dynamic approaches. The former includes several techniques such as SCR, patch augmentation, and subacromial balloon implantation, developed to center the humeral head and thereby restore the force couple. However, there is a need for further exploration of the biomechanical mechanisms and postoperative efficacy of these methods.13,14 Dynamic reconstruction primarily focuses on tendon transfer, intending to restore the functionality of the torn rotator cuff by modifying the natural starting and ending points of the original tendon surrounding the rotator cuff. This intervention aims to revive the normal function of the shoulder. It is essential to highlight that tendon transfer induces changes in the original anatomical structure, and the strength of the transplanted tendon is intrinsically lower than that of the original tendon. 15
Base on this, Ning 16 et al. developed a superior fulcrum reconstruction (SFR) technique. This procedure utilizes the autologous peroneus longus tendon (PLT) to establish a suture-bridge configuration at the proximal humerus, wherein redesigned bone tunnels optimize stress distribution at the tendon-bone interface. Subsequently, Li 17 et al. conducted a 1-year clinical follow-up study documenting favorable short-term postoperative outcomes following SFR. Biomechanically, Wang 18 et al. implemented a cadaveric study utilizing autografts PLT for modified superior capsule reconstruction (mSCR). In consistent with the study of Cline 19 et al., their study indicates that the mSCR technique holds promising clinical prospects in IMRCT. From a biological healing standpoint, the bone tunnel fixation technique substantially augments the tendon-bone contact area, thereby mitigating limitations inherent to conventional anchor fixation—including localized stress concentration and insufficient interfacial contact. These benefits have been demonstrated through biomechanical analyses and in vivo studies.20,21 Moreover, the autologous PLT offers superior biomechanical properties (high tensile strength and elastic modulus) and clinical utility (low donor-site morbidity, excellent biocompatibility), with well-documented efficacy in knee and ankle ligament reconstruction.22–24
Against this backdrop, our study delves into the application of autologous PLT in the SFR of a rabbit model with irreparable massive rotator cuff tears. Our objective is to conduct a histological evaluation of the impact of PLT in SFR on tendon-to-bone healing. Our hypothesis posits that the use of PLT in SFR would positively influence tendon-to-bone healing, positioning PLT as a viable graft option for irreparable massive rotator cuff tears. Our findings could potentially contribute to the promotion of SFR in clinical practice.
Materials and methods
Animal model setup
According to established protocols in the field of prior research,
25
this study selected 30 skeletally mature New Zealand white rabbits, with an average weight of 3.2 kg, procured from our Animal Experiment Center. All 30 rabbits were employed to create an irreparable massive rotator cuff tears model, randomly assigned to two groups. Each group comprised 15 rabbits, designated as the SFR group and the SCR group (Figure 1), and there were no significant differences in the background characteristics of the subject animals between the two groups. Furthermore, ethical approval for this study was obtained from the Animal Experiment Ethics Committee at our institution, the number was LLSC-YY2019-54. Thirty experimental rabbits underwent anesthesia through intravenous injection of 25% ethyl carbamate (5 ml/kg) and subsequent intramuscular injection of 400,000 U of penicillin G for antibiosis. All experimental groups had the animals positioned in left-lateral decubitus, with the right shoulder designated as the surgical side. Following a meticulous process of shaving and skin preparation in the surgical area, the rabbits were securely fixed to the operating table. The surgical area underwent iodine volt disinfection twice. A 3-cm anterolateral incision was skillfully executed on the shoulder, revealing and subsequently splitting the deltoid. Identification and detachment of the supraspinatus and infraspinatus from their insertion on the proximal humerus followed. Through this incision, distal lateral segments measuring 10 mm in length were precisely excised, along with the underlying capsule, utilizing a sharp scalpel. This procedure was carried out to induce supraspinatus and infraspinatus tendon defects.
26
Experimental procedure. IMRCT, irreparable massive rotator cuff tears; SFR, superior fulcrum reconstruction using peroneus longus tendon; SCR, superior capsule reconstruction using autologous fascia lata.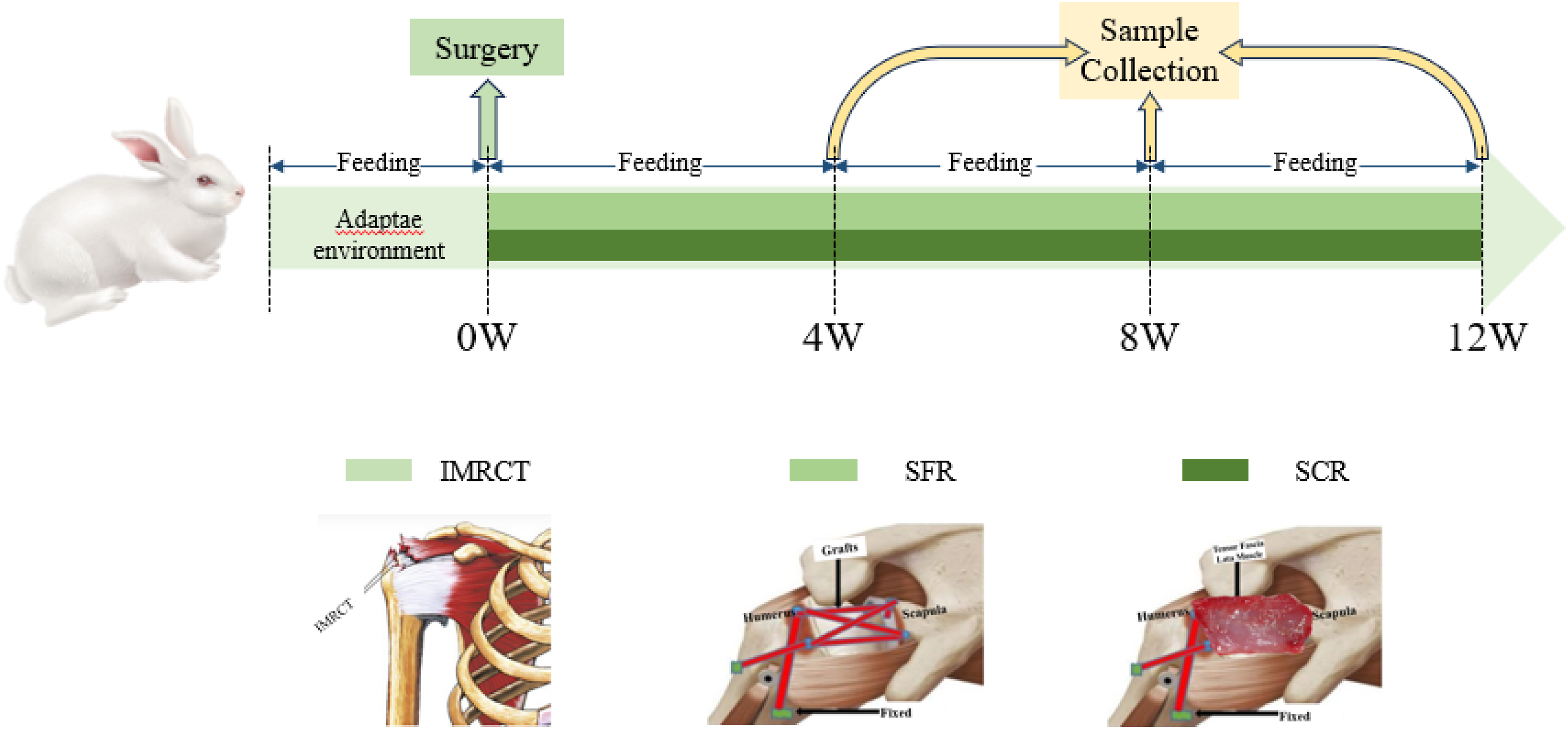
Surgical procedure
Surgical procedure. (a): Identification and isolation of the peroneus longus tendon; (b): The bone tunnels were in the humeral head; (c): The braided autologous peroneus longus tendon passed the bone tunnels and was tied on the proximal lateral humerus; (d): The harvested tissue of autologous fascia lata; (e): The bone tunnel was in the superior glenoid and (f); The fixed fascia lata.
Specimen collection
At 4, 8, and 12 weeks postoperatively, an overdose of carbon dioxide was administered to sacrifice five rabbits in each group. The entire humerus, tendon-to-bone junction, and graft were meticulously separated for gross observation. Subsequently, all specimens were fixed in 4% paraformaldehyde to facilitate histological observation.
Histological evaluations
Histological evaluation was conducted on five specimens from each group at 4, 8, and 12 weeks postoperatively. These specimens underwent fixation in 4% paraformaldehyde for 3 days, followed by decalcification for 30–35 days at room temperature in 10% EDTA. Upon completion of the decalcification process, the specimens underwent sequential dehydration in varying concentrations of ethanol, ultimately being paraffin-embedded. Sections (5 μm) were precisely cut along the long axial coronal plane of the humerus. Staining procedures included Hematoxylin and Eosin (H&E), Masson trichrome, Safranin O/Fast Green, and Picrosirius Red. Additionally, immunohistochemistry involved the assessment of CD31 (a vascular endothelial cell marker) to evaluate vascularity at the tendon-to-bone healing site. Collagen I and collagen III were employed to assess the maturity of tendon-bone healing and changes in collagen. Two pathologists, blinded to the treatment conditions, conducted examinations of all sections. Histological sections were captured using a light microscope (NI-U; Nikon).
Hematoxylin and Eosin (H&E) staining evaluation
The modified tendon maturation score system.

C: continuity; I: ingrowth; F: fibrocartilage; T: tidemark; +: positive; −: negative.
Safranin O/Fast green staining evaluation
Safranin O/Fast Green staining was employed to visualize fibrocartilage regeneration at the tendon-to-bone healing site, where the regenerated tissue exhibited a red color against a green background. 30 A semiquantitative analysis was conducted using metachromasia as an indicator of fibrocartilage formation. A representative slice illustrating tendon-to-bone healing was selected, and the metachromasia area was carefully outlined at ×40 magnification. Subsequently, digital images were imported into ImageJ software (National Institutes of Health) to directly obtain measurement results.
Masson trichrome staining evaluation
Masson trichrome, a classical three-color staining method widely utilized in connective tissue studies, stands as one of the primary techniques for visualizing fibers within tissues. In this staining method, collagen fibers exhibit shades of blue or green, while muscle fibers present a red hue. The application of Masson trichrome allowed for the examination of cellularity, continuity, direction, and regularity of collagen fibers at the tendon-to-bone healing area. Subsequent observation and analysis of these results were conducted under a light microscope.
Picrosirius red staining evaluation
To scrutinize the composition of collagen fibers and measure collagen birefringence, Picrosirius Red staining was applied. The examination was conducted using polarized light microscopy (Eclipse E800; Nikon). Consistent with prior findings, collagen I exhibited robust yellow-red birefringence, while collagen III fibers manifested a subdued greenish color. 28 For a quantitative assessment of collagen fiber composition, ten areas (50 × 50 μm) within the tendon-to-bone healing site were randomly chosen. These images were then processed in the Image J software system (National Institutes of Health), and grayscale values were calculated. Elevated grayscale values indicated a higher presence of collagen I components.
Immunohistochemical evaluation
This study encompassed two distinct immunohistochemical examinations. The first focused on CD31, a vascular endothelial marker, to analyze vascularity at the tendon-to-bone healing site. The second examination involved collagen I and collagen III, assessing the maturity of the tendon-to-bone healing process. For the CD31 immunohistochemical analysis, paraffin-embedded sections underwent paraffin removal and rehydration, followed by antigen repair. A citric acid solution was configured, placed in a pressure cooker, and boiled under high pressure for a two-minute hot repair. Subsequently, these sections were blocked with 3% peroxide for 20 min at room temperature, followed by a wash with phosphate-buffered saline (PBS). The next steps involved incubating the sections with the anti-rabbit CD31 primary antibody for 60 min at 37°C, followed by the addition of the anti-rabbit secondary antibody for 20 min at 37°C. A DAB color developer was introduced for an additional 10 min at room temperature, followed by a thorough rinse with running water for 5 min. The final step comprised counterstaining with hematoxylin.
To evaluate the maturity at the tendon-to-bone healing site, immunohistochemical analysis of collagen I and collagen III was conducted using their respective polyclonal antibodies as primary antibodies. The procedures for paraffin removal, rehydration, and blocking endogenous peroxidase activity mirrored those employed for CD31. Following this, sections underwent an additional 2-h blocking step with goat serum at 20°C to 25°C. Subsequently, they were incubated overnight with the primary antibody at 4°C, followed by an additional 1-h incubation with the anti-rabbit secondary antibody at 20°C to 25°C. Chromogen was applied to the tissue sections for 10 min at room temperature, and a thorough rinse in running water for 5 min ensued. The concluding step involved counterstaining all sections with hematoxylin. Additionally, in the preliminary experiments, we performed immunohistochemical staining on untreated, isotype-matched rotator cuff tissues as negative controls and explored the working concentrations for CD31, COL1, and COL3 antibodies. This established the foundational staining conditions for subsequent experiments, as detailed in Figure S1.
Statistical analysis
Statistical analyses were conducted using GraphPad Prism 8.0 software, presenting the data as the mean ± standard deviation. To compare histological outcomes between two groups, the Student t-test was employed, and a significance threshold of p < .05 was established to identify statistically significant differences.
Results
Macroscopic observations
Postoperatively, no retears were observed in any of the rabbits. However, two rabbits—one from the SFR group and one from the SCR group—were identified with infections in the right shoulder during the harvest and were consequently excluded from the experiment. Fortunately, no signs of infection were detected in the remaining rabbits. Importantly, all grafts demonstrated continuous integration with the humerus in every shoulder at the time of sacrifice, as illustrated in Figure 3. Macroscopic view of the harvested specimens after surgical operation. A-C: SFR group at 4, 8 and 12 weeks. D-F: SCR group at 4, 8 and 12 weeks. SFR group: superior fulcrum reconstruction; SCR group: superior capsule reconstruction.
Histological analysis
H&E staining and modified tendon maturity scores
The examination of HE-stained sections revealed notable distinctions in tendon-to-bone healing between the two experimental groups, as depicted in Figure 4. At the 4-week mark, the tendon-to-bone healing interface exhibited the presence of inflammatory cells and granulation tissue, with a limited number of chondrocytes and osteoblasts in both groups. By the 8-week stage, inflammatory cells diminished at the healing interface, while osteoblasts and chondrocytes emerged in both groups. However, in the SFR group, the healing interface resembled normal tendon healing, characterized by irregularly organized collagen fibers and cells. In contrast, the SCR group continued to display a healing interface filled with fibrovascular granulation tissue. By the 12-week milestone, inflammatory cells were scarcely observed in both groups. In the SFR group, the healing interface matured, exhibiting continuous and neatly aligned collagen fibers, with well-organized osteoblasts and chondrocytes. Meanwhile, the SCR group still presented irregular collagen fibers and chondrocytes at the healing interface. Moreover, the SFR group demonstrated higher scores than the SCR group at 8 weeks (21.84 ± 0.45 points vs18.36 ± 0.64 points, p = .041) and 12 weeks (26.69 ± 0.48 points vs20.16 ± 0.57 points, p = .018). However, there were no significant score differences between two groups at 4 weeks (11.72 ± 0.64 points vs10.94 ± 0.66 points, p = .564) (Table 2) (Figure 5). Hematoxylin and eosin staining at the tendon-to-bone healing site (×20). A-C: SFR group at 4, 8 and 12 weeks. D-F: SCR group at 4, 8 and 12 weeks b, bone; i, interface; t, tendon; SFR, superior fulcrum reconstruction; SCR, superior capsule reconstruction. Scale bar: 100 μm. Comparison of the Modified Tendon Maturation Scoring System between the SFR group and SCR group (N = 5). SFR group: repair with superior fulcrum reconstruction; SCR group: repair with superior capsule reconstruction. Safranin O/fast green staining at the tendon-to-bone healing site (×20). A-C: SFR group at 4, 8 and 12 weeks. D-F: SCR group at 4, 8 and 12 weeks b, bone; i, interface; t, tendon; SFR, superior fulcrum reconstruction; SCR, superior capsule reconstruction. Scale bar: 100 μm.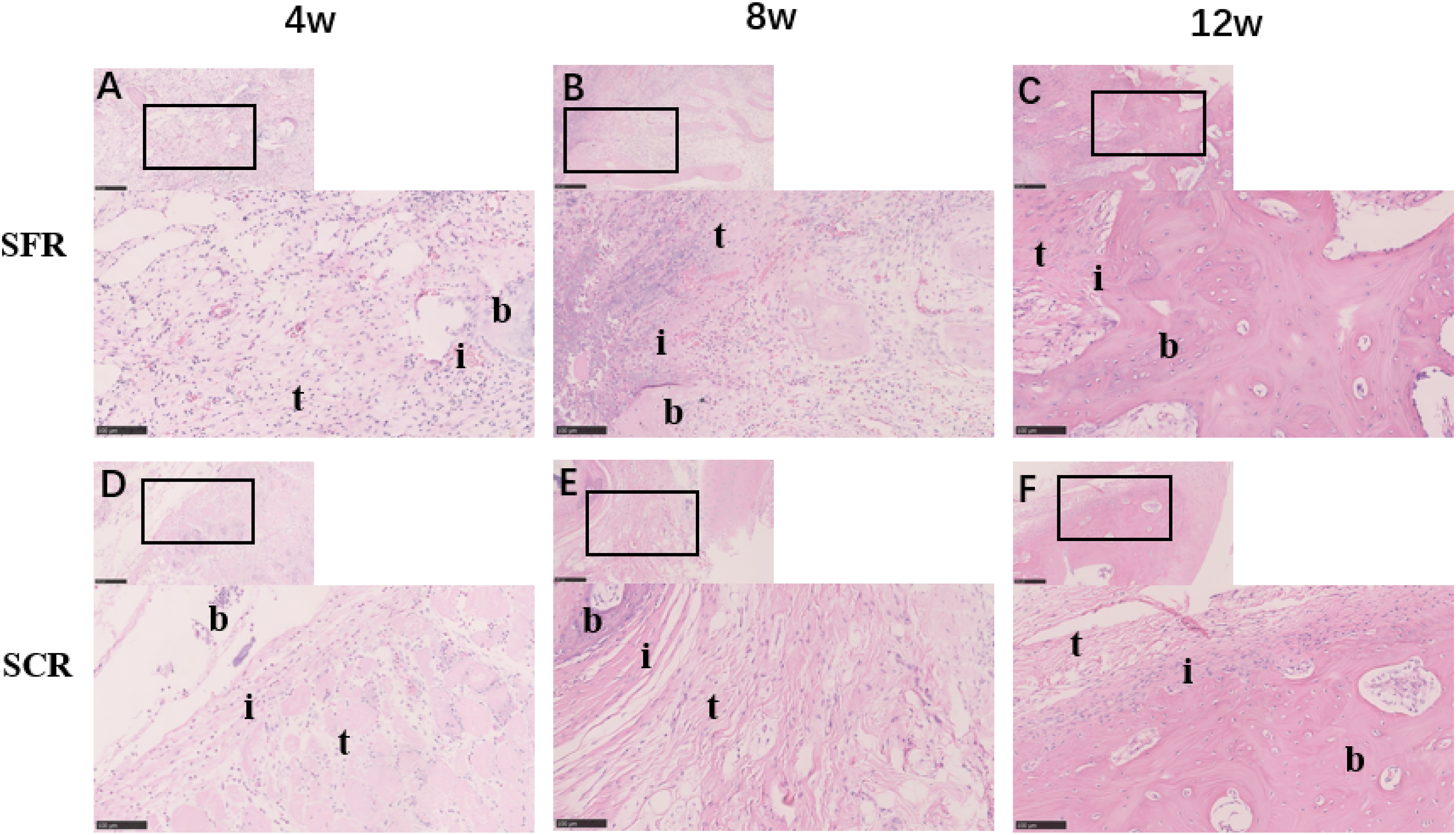


Metachromasia
Semiquantitative Evaluation of the Cartilage Staining Area between the SFR group and SCR group (N = 5).
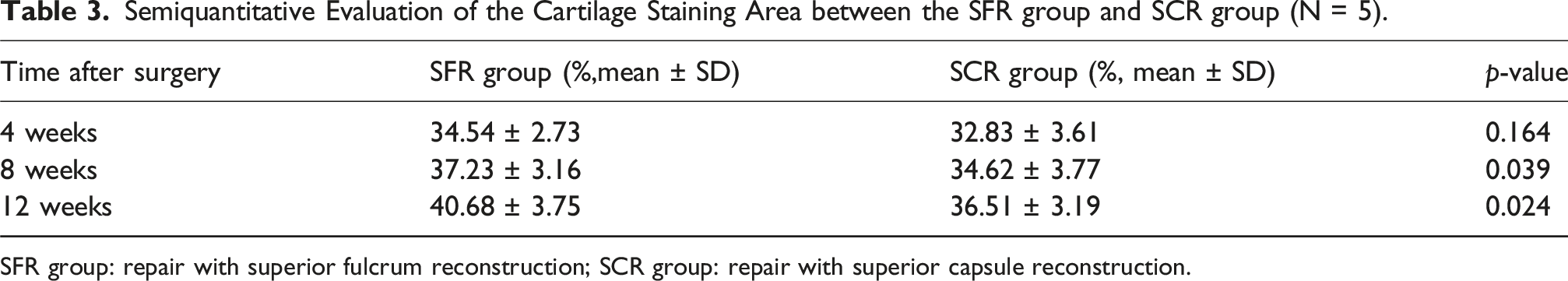
SFR group: repair with superior fulcrum reconstruction; SCR group: repair with superior capsule reconstruction.
Collagen composition and fiber organization
Over the course of this study, Masson trichrome staining was employed to assess the fiber organization at the tendon-bone healing interface in both experimental groups (Figure 6). Notably, no discernible distinctions in fiber organization were observed between two groups at the 4-week time point. However, a significant enhancement in regularity and organization was noted in the SFR group compared to SCR at both 8 and 12 weeks. At 12 weeks, the total area of Masson trichrome staining at the tendon-bone interface exhibited an increase in both SFR and SCR, with SFR demonstrating a significantly larger overall area. Examining the representation of collagen type I through areas of yellow-red strong birefringence, no significant differences were identified between the groups at the 4-week mark. Additionally, the alignment of collagen fiber bundles at the tendon-bone healing interface appeared irregular. Moving to the 8-week interval, SFR exhibited the disappearance of the tendon-bone interface, predominantly featuring collagen type I, interspersed with types II and III. Conversely, in SCR, the tendon-bone interface remained visible, displaying a superior arrangement of collagen, primarily composed of type III. At the 12-week juncture, the tendon-bone interface became imperceptible in both groups, showcasing a direct insertion of well-organized type I collagen in large bundles (Figure 7). Masson trichrome staining at the tendon-to-bone healing site (×20). A-C: SFR group at 4, 8 and 12 weeks. D-F: SCR group at 4, 8 and 12 weeks b, bone; i, interface; t, tendon; SFR, superior fulcrum reconstruction; SCR, superior capsule reconstruction. Scale bar: 100 μm. Picrosirius red staining at the tendon-to-bone healing site (×20). A-C: SFR group at 4, 8 and 12 weeks. D-F: SCR group at 4, 8 and 12 weeks b, bone; i, interface; t, tendon; SFR, superior fulcrum reconstruction; SCR, superior capsule reconstruction. Scale bar: 100 μm.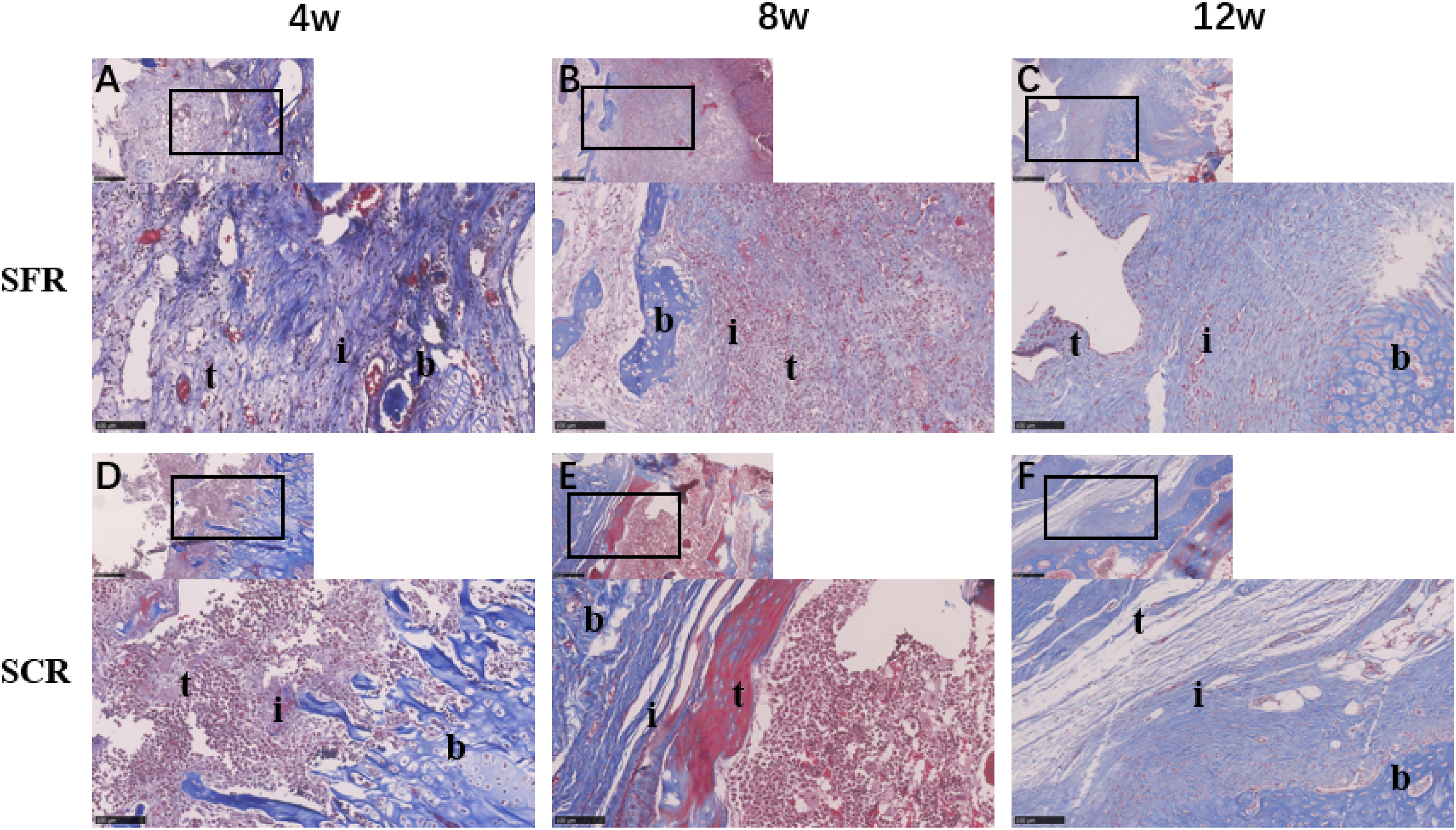

Immunohistochemical analysis
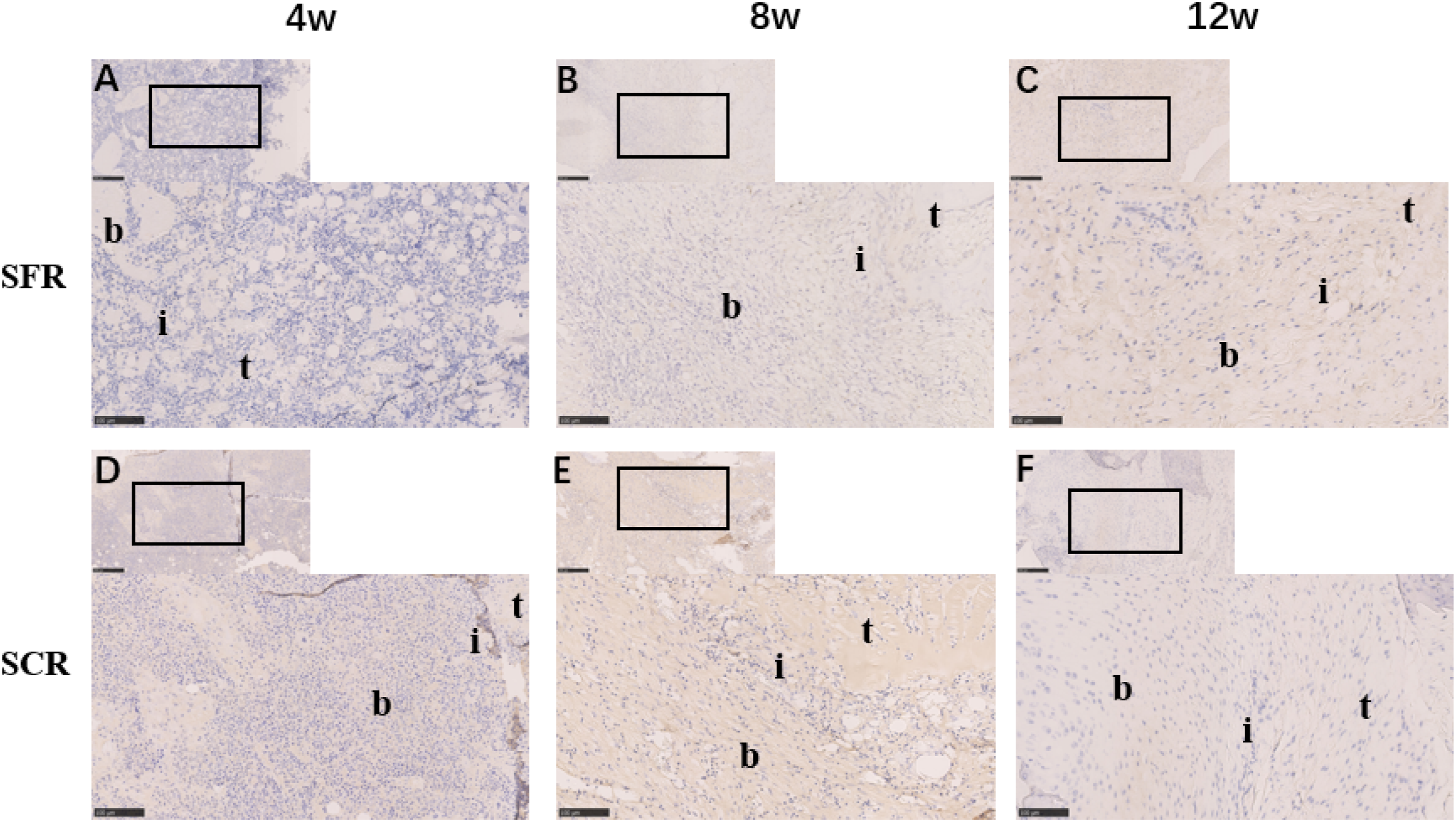
Exploration of angiogenesis, assessed through immunohistochemistry using an endothelium-specific anti-CD31 antibody, revealed a robust influx of blood vessels into the healing area. Notably, SFR exhibited a higher degree of vascularity compared to SCR at 4 weeks. In SFR, the peak of angiogenesis occurred around 4 weeks, followed by a rapid decline, resulting in significantly lower vascularity than SCR at 8 weeks. By the 12-week mark, minimal angiogenesis was evident in both groups (Figure 8). Over time, turning to collagen type composition, an increase in collagen I density was observed (Figure 9), while collagen III density exhibited a decrease (Figure 10). Subsequently, newly formed fibrocartilaginous columns displayed positive staining for collagen III, there no significant differences between the two groups were noted at 4 weeks (32.48% ± 3.76% vs 32.86% ± 3.24%, p = .072). However, at 8 weeks (25.13% ± 2.89% vs 30.08% ± 3.71%, p = .024) and 12 weeks (20.94% ± 1.93% vs 23.67% ± 2.69%, p = .041), the SCR groups demonstrated significantly higher collagen III density compared to the SFR groups. Conversely, the collagen I density of the SFR group surpassed that of the SCR group at 4 weeks (25.73% ± 2.61% vs 21.35% ± 3.67%, p = .045), 8 weeks (27.93% ± 3.45% vs 24.28% ± 3.79%, p = .035), and 12 weeks (33.86% ± 2.47% vs 29.53% ± 1.94%, p = .029). Notably, only at 8 weeks, the collagen III density of the SCR group was significantly higher than that of the SFR group. Refer to Table 4 for a comprehensive presentation of these results. Immunohistochemistry staining of endothelium-specific anti-CD31 antibody at the tendon-to-bone healing site (×20). A-C: SFR group at 4, 8 and 12 weeks. D-F: SCR group at 4, 8 and 12 weeks b, bone; i, interface; t, tendon; SFR, superior fulcrum reconstruction; SCR, superior capsule reconstruction. Scale bar: 100 μm. Immunohistochemistry staining of collagen I at the tendon-to-bone healing site (×20). A-C: SFR group at 4, 8 and 12 weeks. D-F: SCR group at 4, 8 and 12 weeks b, bone; i, interface; t, tendon; SFR, superior fulcrum reconstruction; SCR, superior capsule reconstruction. Scale bar: 100 μm. Immunohistochemistry staining of collagen III at the tendon-to-bone healing site (×20). A-C: SFR group at 4, 8 and 12 weeks. D-F: SCR group at 4, 8 and 12 weeks b, bone; i, interface; t, tendon; SFR, superior fulcrum reconstruction; SCR, superior capsule reconstruction. Scale bar: 100 μm. Semiquantitative Evaluation of the Collagen Staining Area between the SFR group and SCR group (N = 5). SFR group: repair with superior fulcrum reconstruction; SCR group: repair with superior capsule reconstruction.


Discussion
Upon conducting histological analysis in this investigation, a pronounced enhancement in tendon-to-bone integration was evident within the SFR group at both 8 and 12 weeks postoperatively. The pivotal outcomes of our study affirmatively validate the superior efficacy of SFR in fostering tendon-to-bone healing, encompassing advancements in the maturation of the tendon-to-bone interface, fibrocartilage formation, vascularity, and the orientation and composition of collagen fibers. Specifically, histological evaluations unveiled the assimilation of SFR into a structure mirroring a normal tendon healing interface by the 8-week mark postoperatively. These notable findings provide robust support for our initial hypothesis. This hypothesis posited that the implementation of the PLT in SFR would exert a beneficial influence on tendon-to-bone healing, thereby establishing the viability of PLT as a graft in irreparable massive rotator cuff tears (IMRCT). Notably, at the 4-week timepoint, the SFR group demonstrated no statistically significant differences in mature tendon scores or fibrocartilage-stained area compared to the SCR group. Although these quantitative outcomes did not reach conventional thresholds of statistical significance, absolute expression values revealed a consistent upward trend in the SFR cohort relative to SCR controls. This observed discrepancy may be attributable to the limited sample size in the current preclinical investigation. To address this limitation and validate these preliminary findings, we plan to expand the cohort size in subsequent animal studies and initiate a multicenter randomized clinical trial to comprehensively evaluate the long-term therapeutic efficacy and biological mechanisms of the SFR technique.
Effective interface healing significantly influences the prolonged success of IMRCT repair,31–34 with various clinical treatments available, such as partial repair, tendon transfer, patch augmentation, SCR, subacromial balloon implantation, and reverse shoulder arthroplasty.5,35 Among these alternatives, SFR presents distinct advantages for clinical utilization, supported by four key considerations. Firstly, the accessibility of the PLT from the external malleolus facilitates ease of use. Secondly, the autologous nature of the PLT eliminates the risk of immune responses. Thirdly, the establishment of bone tunnels on the glenoid and humeral head sides enhances the transplant process, maximizing the contact surface between the tendon and bone. Li X conducted an animal study, demonstrating that an into-tunnel rotator cuff repair induces a unique tendon-bone healing pattern, characterized by prominent fibrocartilage regeneration and superior healing strength compared to onto-surface repair. Fourthly, the natural release of growth factors from the bone marrow during SFR contributes to promoting healing at the tendon-to-bone contact surface. 36 Wang J’s research emphasized the role of insulin-like growth factor I (IGF-1) in maintaining and regenerating the adult skeleton, illustrating the reciprocal regulation between the hematopoietic and skeletal systems. 37 In summary, the promising outcomes of SFR, particularly in IMRCT scenarios, warrant continued investigation into the valuable aspect of PLT-to-bone healing over the long term.
SCR is a popular technique that has promising short-term results in the treatment of IMRCT. The superior capsule of the shoulder is an anatomically and histologically distinct layer and is different from the tendon layer that attaches to a substantial area of the greater tuberosity. The superior capsule and supraspinatus are important stabilizers and depressors of the humeral head that provide a stable fulcrum for the deltoid muscle. 38 Superior glenohumeral translation at 5° and 30° of abduction has been shown biomechanically in cases of superior capsular defects that cause superior humeral escape. 39 Another biomechanical cadaveric study showed that SCR with a collagen graft restored the superior stability in IMRCT. 40 But SCR establish “surface cover” through the “surface contact” in biomechanics and tendon-bone interface histology has serious defects, which leads to graft tear, absorption and creep of the graft at the healing site of the tendon-bone connection site after repair. 41 Given to the disadvantages of the biomechanics and histology in the tendon-bone interface after SCR, we firstly design SFR for the treatment of irreparable massive rotator cuff tears in rabbits. Autologous PLT is used to establish tendon “bridge covering” which can play the role of lever “fulcrum”, so as to obtain stronger initial mechanical strength, better biomechanical function, instead of relying on SCR “surface covering” above shoulder joint with poor tendon-bone healing and insufficient initial strength.
This study focuses on exploring the efficacy of utilizing autologous PLT as a graft for tendon-to-bone healing. Traditionally employed in ACL reconstruction for the knee, 42 PLT presents several compelling reasons for its selection in this context. Firstly, the ready accessibility of PLT is a notable advantage. Additionally, scholarly investigations reveal its regenerative capacity, with PLT exhibiting growth even 52 months post-surgery for ACL reconstruction, as observed through long-term MRI follow-ups. 43 Secondly, PLT offers ample length and mechanical strength, essential for restoring the force couple balance in the rotator cuff. Cadaver studies affirm that PLT meets the length and strength requirements for ACL reconstruction.44,45 Thirdly, resecting PLT does not compromise ankle stability or induce gait alterations.46–48 Potential compensatory mechanisms, such as the contributions of peroneus brevis and abductor digiti minimi, may mitigate the impact on eversion strength and plantar flexion.47,49 However, the long-term biomechanical impact of PLT harvest on highly active patients (e.g., athletes, laborers) requires careful prospective evaluation. Therefore, thorough patient counseling and stringent selection criteria would be essential in initial clinical trials. Finally, the composition of PLT, characterized by regular dense connective tissue rich in collagen fibers (mainly collagen I), aligns in parallel bundles along stress directions. The investigation highlights a healing process where collagen III is initially predominant in the early phase of tendon-to-bone healing, gradually replaced by collagen I. This pattern mirrors findings in previous studies, reinforcing the specificity of the healing process. 15 In summary, SFR utilizing PLT demonstrates robust tendon-to-bone healing capabilities, emphasizing its potential as a promising approach in rotator cuff repair.
This investigation presents several noteworthy limitations. Firstly, the rabbit model utilized in this study focused on acute irreparable massive rotator cuff tears, but the use of an acute rabbit tear model does not fully replicate the chronic degenerative pathology typically seen in human IMRCTs, where tendon quality, retraction, and fatty infiltration complicate repair. Nonetheless, our primary objective was to explore tendon-to-bone healing employing the PLT and emulate the clinical context of resurfacing and decorticated footprint. The selection of an acute rotator cuff model was deemed appropriate for addressing these research objectives. Secondly, the study did not incorporate biomechanical analyses to assess the mechanical properties associated with tendon-to-bone healing. This omission limits a comprehensive understanding of the graft’s strength and stability in the context of rotator cuff tears. Thirdly, the sample size of this study is relatively small. While significant differences were found, the modest sample size may have limited our ability to detect more subtle differences, particularly at earlier time points. Lastly, although the rabbit rotator cuff tear model shares considerable similarities with human shoulders, inherent anatomical and histological differences exist. It is crucial to recognize these distinctions, as they introduce variations that may impact the direct extrapolation of findings to human clinical scenarios. Based on the aforementioned findings, future studies will focus on establishing an animal model of chronic massive rotator cuff tears and conducting comprehensive biomechanical testing to validate the efficacy of the SFR technique under realistic pathological conditions and to elucidate its mechanisms in restoring glenohumeral stability and resistance to failure. Furthermore, expanding the sample size and implementing multi-center collaborations will enhance the generalizability of our conclusions. Most importantly, we will conduct prospective comparative studies to evaluate SFR against existing mainstream SCR approaches, thereby more accurately defining its clinical advantages and appropriate indications.
Conclusions
In the context of a rabbit model simulating acute irreparable massive rotator cuff tears, the utilization of autologous PLT for SFR exhibited favorable tendon-to-bone healing outcomes, as evidenced by histological evaluations. Notably, this study marks the pioneering introduction of SFR as a novel approach to enhance rotator cuff repair. The promising histological results suggest that SFR holds potential as a viable option for addressing irreparable massive rotator cuff tears. This innovative technique may offer a valuable contribution to the repertoire of treatments available for such challenging clinical scenarios.
Supplemental Material
Supplemental material - Superior fulcrum reconstruction using autologous peroneus longus tendon for irreparable massive rotator cuff tears in rabbits: A comparative study
Supplemental material for Superior fulcrum reconstruction using autologous peroneus longus tendon for irreparable massive rotator cuff tears in rabbits: A comparative study by Cheng Peng, Kehao Wang, Zhengming Zhu, Hongyan Li, Guang Chen, Lingchao Kong, Rende Ning in Journal of Orthopaedic Surgery
Footnotes
Acknowledgements
The authors would like to thank all staff members in the participating departments.
Ethical considerations
The study was approved by the Ethics Committee of Anhui Medical University (approval no. LLSC-YY2019-54). All methods were conducted in accordance with the relevant guidelines and regulations.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Anhui Provincial Key Research and Development Plan (NRF: 202104j07020057) and the Applied Medical Research Project of Hefei Municipal Health and Wellness Commission (NRF: Hwk2021yb003).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
IACUC information
The Third Affiliated Hospital of Anhui Medical University Animal Care and Use Committee IACUC NO: KNU 2021-0702.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
