Abstract
Background
Key instruments used in posterior cervical expansive open-door laminoplasty (CEOL), such as the high-speed drill (HSD) and the ultrasonic bone scalpel (UBS), are selected based on surgeon preference. However, skepticism remains among some surgeons regarding the superiority of the HSD over the widely used UBS, necessitating a scientific evaluation of its practical utility.
Objective
To evaluate the efficacy and safety of the ultrasonic bone scalpel compared to the high-speed drill in cervical expansive open-door laminoplasty using a posterior cervical approach in a prospective randomized controlled trial.
Methods
This prospective study, conducted after obtaining prior informed consent, involved 40 patients diagnosed with cervical degenerative diseases who underwent cervical expansive open-door laminoplasty between July 2018 and June 2021. The patients were randomly divided into two groups: the ultrasonic bone scalpel group (n = 20) and the high-speed drill group (n = 20). Surgeons could not be blinded due to the nature of the intervention, but outcome assessors were blinded to group allocation where feasible. Surgical time, open-door time, intraoperative blood loss, postoperative drainage loss, post-operative complications, and functional outcomes were compared between the two groups.
Results
No statistically significant difference was observed between the two groups in terms of intraoperative blood loss, post-operative complications, screw migration, plate fracture, or spinal cord expansion. However, the ultrasonic bone scalpel group demonstrated significantly shorter surgical time, open-door time, and reduced postoperative drainage loss compared to the high-speed drill group (p < .05). The reduced drainage volume may be attributed to thermal sealing of small vessels by the UBS rather than solely reduced tissue trauma. No significant differences were observed in the Visual Analog Scale (VAS) scores, bony union at the hinge, or Japanese Orthopedic Association (JOA) score improvement rates between the two groups at 1 week, 3 months, and 1 year postoperatively. While the UBS group showed numerical reductions in complication rates (e.g., dural tear: 0% vs 5%, p = .311), these differences did not reach statistical significance, likely due to the limited sample size.
Conclusions
The study concluded that USB significantly reduces operation time, open-door time, and postoperative drainage loss. However, the study’s limited sample size may have been insufficient to detect differences in complication rates between groups.
Introduction
With the aging population and widespread use of electronic devices, the prevalence of cervical degenerative diseases (CDD), such as cervical spondylotic myelopathy (CSM), 1 ossification of the posterior longitudinal ligament (OPLL), 2 and cervical canal stenosis (CCS), 3 is steadily rising. The CDD is a leading cause of nerve and spinal cord injury, particularly in middle-aged and elderly individuals, with symptoms often manifesting as neck and shoulder pain, as well as nerve root and spinal cord compression syndromes.4,5
For patients with severe CDD and significant nerve compression, conventional treatments are often ineffective, making surgical decompression the primary option. Among the surgical techniques, posterior cervical approach is well-established for multilevel cervical spondylotic myelopathy, as it enlarges the spinal canal and alleviates nerve compression. This intervention is widely practiced and considered highly effective. 6
Two commonly employed posterior cervical decompression surgeries are cervical laminectomy and cervical laminoplasty. These procedures alleviate spinal cord compression by creating space for the spinal cord to shift backward, which reduces pressure from surrounding tissues. 7 Cervical laminectomy involves the removal of laminae to decompress the spinal cord. However, this approach is associated with significant complications, demanding high surgical expertise and specialized equipment. 8 As a result, ensuring a safe and effective laminectomy is crucial. 9
Cervical laminoplasty, on the other hand, is often preferred due to its lower incidence of complications, such as C5 nerve root palsy, when compared to laminectomy. 10 The technique relies on repositioning the lamina, with one side of the lamina acting as a hinge while the other is opened, expanding the spinal canal and indirectly decompressing the spinal cord. 11
Cervical expansive open-door laminoplasty (CEOL) has emerged as a safe and effective laminoplasty technique. Studies have indicated that CEOL is especially beneficial for patients with stable cervical alignment, good cervical lordosis, no significant neck pain, and ossified K-line negative OPLL. 12 Laminoplasty is a preferred intervention for patients with cervical myelopathy having multi-level stenosis presenting preserved sagittal alignment and minimal to no axial neck pain related to spondylosis.
It is important to note that complications such as epidural venous plexus rupture, cerebrospinal fluid leakage from dural tears, and spinal nerve root injury can occur when the medial cortical bone is cut during CEOL. 13 Traditionally, CEOL is performed using laminectomy rongeurs and high-speed drills (HSD). While laminectomy rongeurs require high operator effort and prolong decompression time, potentially exacerbating spinal cord compression. 14 The HSD improves surgical efficiency and reduces labor intensity. 15 However, use of HSD comes with high technical demands. Improper handling can result in rolling injuries to nearby soft tissues, including nerve tissue and spinal dura. 16 Additionally, heat generated by HSD may hinder bone healing and cause thermal damage to nerves.17,18 High labor intensity associated with HSD increases the risk of injury to critical structures like spinal cord, nerve roots, and dura, potentially impacting patient recovery and quality of life. 19
In recent years, ultrasonic bone scalpel (UBS) has gained much attention in spinal surgery, thanks to its innovative technology and precise cutting capabilities.20–22 The UBS uses ultrasonic frequencies to selectively cut bone tissue with minimal impact on surrounding soft tissues and generates less heat, reducing the risk of nerve and vascular injury. It also provides simultaneous hemostasis through reducing intraoperative bleeding and postoperative drainage. Furthermore, UBS improves surgical control, reduces difficulty, and lessens operator fatigue.23,24 However, it is important to acknowledge that UBS technology also has limitations, including a learning curve for surgeons transitioning from conventional techniques, higher initial equipment costs, and potential thermal risks if used improperly, as reported by Tarazi et al. (2018). 25
To objectively compare the clinical efficacy and safety of UBS versus HSD in CEOL for treating cervical degenerative diseases (CDD), a prospective randomized controlled study was conducted.
Materials and methods
Study design
To ensure smooth execution, all personnel underwent systematic professional training prior to the project’s commencement. The training and details of study staff is as under: Scientific Research Support Personnel: Training focused on tasks such as assigning patients to groups using numbered cards from a random number table, sealing the random number cards in envelopes, monitoring the project’s implementation, data collection, statistical analysis, and timely reminders for outpatient follow-up.
Outpatient physicians
Their responsibilities included confirming the eligibility of subjects based on diagnosis, inclusion, and exclusion criteria for CDD, explaining the study to participants, distributing the envelopes after informed consent, and collecting follow-up data.
Residents
They were responsible for re-confirming each subject’s condition and willingness to participate, arranging surgeries based on the group assignment indicated in the envelopes, and mastering both UBS and HSD techniques for CEOL surgery. It was necessary to ensure that all participants received uniform treatment, apart from the surgical methods.
Training was completed by June 2018, with all staff meeting the required standards to proceed with the study.
Blinding
Due to the nature of the surgical intervention, it was not possible to blind the operating surgeons to the treatment allocation. However, to minimize bias, outcome assessors who evaluated postoperative functional scores (VAS and JOA) and radiographic outcomes were blinded to the group allocation. Patients were also blinded to their group assignment throughout the study period.
Patient population
From July 2018 to June 2021, 40 patients with CDD who underwent CEOL were selected from the Department of Orthopedics.
Diagnostic criteria included
Typical clinical manifestations of cervical spinal cord damage, such as limb motor dysfunction, sensory disturbances, and abnormal reflexes. Imaging evidence of spinal cord compression consistent with clinical symptoms. Exclusion of other conditions such as amyotrophic lateral sclerosis, intraspinal mass, acute spinal cord injury, subacute combined degeneration of the spinal cord, syringomyelia, chronic polyneuropathy, and thoracic spinal stenosis.
General data collected for all patients included age, gender, body mass index (BMI), CDD etiology (CSM, OPLL, or CCS), preoperative Visual Analogue Scale (VAS) scores, preoperative Japanese Orthopaedic Association (JOA) scores for cervical spondylotic myelopathy, number of surgical segments, and spinal canal cross-sectional area.
The study was conducted in accordance with the Declaration of Helsinki and relevant ethical guidelines of West China Hospital (LY-GM-SDN-2016-01). Written informed consent was obtained from all participants.
Inclusion criteria
(1) Patients met the specified diagnostic criteria. (2) Presence of developmental cervical spinal stenosis. (3) Underwent CEOL with micro titanium plate internal fixation. (4) The surgical segments between C3 and C7. (5) Osteotomy performed using intraoperative ultrasound or high-speed grinding drill. (6) Patients provided consent for the surgery and had no contraindications.
Exclusion criteria
(1) History of trauma, including cervical spine fracture or dislocation. (2) Previous posterior cervical surgery. (3) Intraoperative lateral mass screw fixation, combined with anterior cervical fusion or facet.
Joint fusion surgery
(1) Diagnosed cervical cancer or tuberculosis. (2) Severe osteoporosis.
Randomization
Patients were randomly assigned to two groups using a computer-generated random number table, resulting in 20 patients in the UBS group and 20 patients in the HSD group, based on the intraoperative osteotomy tools used.
Sample size calculation
Based on previous studies comparing surgical techniques in cervical laminoplasty, we performed a power analysis to determine the appropriate sample size. Using operation time as the primary outcome measure, with an expected difference of 15 minutes between groups, standard deviation of 18 minutes, significance level (α) of 0.05, and power (1-β) of 0.8, we calculated that a minimum of 18 patients per group would be required. To account for potential dropouts, we enrolled 20 patients in each group. However, we acknowledge that this sample size may be insufficient to detect differences in secondary outcomes such as complication rates, which typically require larger sample sizes due to their lower incidence.
Surgical procedures
All surgical procedures were performed by a team of three senior spine surgeons, each with over 10 years of experience in cervical spine surgery. These surgeons had equivalent expertise in both UBS and HSD techniques, having performed at least 50 procedures with each instrument prior to the study. To minimize the impact of the learning curve and surgeon-dependent variables, each surgeon performed an approximately equal number of surgeries in each group, and the surgical technique was standardized across all operators. Additionally, the composition of the surgical team (including assistants and nursing staff) was kept consistent throughout the study period.
Under general anesthesia, the patient was positioned prone with a chest pillow, and the head was fixed using a Mayfield head frame, ensuring slight forward flexion of the neck. The procedure began with the dissection of the skin, subcutaneous tissue, and nuchal ligament. The paravertebral muscles were dissected on both sides of the spinous processes and the lamina, exposing the C3-C7 laminae. The interspinous ligaments at C2-C3 and C7-T1 were resected, and the ligamentum flavum was exposed. The ends of the spinous processes at C6-C7 were trimmed for evenness.
The right side was designated as the hinge side, while the left side served as the door side.
In the UBS group, an ultrasonic bone scalpel was employed for osteotomy, while in the HSD group, a high-speed grinding drill was utilized. A “V”-shaped bone groove was created at the medial margin of the cervical articular process on the hinge side, preserving the inner cortical bone. The lamina was then osteotomized at the medial edge of the cervical articular process on the open-door side and subsequently cut off. The C3-C7 lamina was opened approximately 2 cm from the hinge side, and care was taken to separate the dural sac and any interlaminar adhesions. In cases where five levels required decompression (C3-C7), all levels were addressed according to the same standardized technique.
A suitable micro titanium plate was selected, and its ends were fixed to the side block and the lamina on the door-opening side, maintaining the lamina in an expanded position. The incision was thoroughly rinsed, and a drainage tube was placed. The cervical muscles were carefully reconstructed at the C2 attachment point, followed by layered suturing of the incision.
Postoperatively, mannitol, dexamethasone, and neurotrophic medications were routinely administered for symptomatic treatment. On the first postoperative day, a neck support was provided, and the drainage tube was removed once the drainage volume was less than 50 mL.
Observation index
The study assessed various surgery-related indicators, including: • Surgery duration • Duration of the open-door procedure • Intraoperative blood loss • Postoperative drainage loss • Operation-related complications, such as spinal injury, dural tear, cerebrospinal fluid (CSF) leakage, axial symptoms, and C5 paralysis • Screw migration or plate fracture • Spinal canal expansion area
Functional outcomes were measured using the VAS score, the bony union at the hinge, and JOA score improvement rate, calculated as follows:
JOA Improvement Rate = [(Postoperative JOA Score−Preoperative JOA Score)/(17−Preoperative JOA Score)] × 100%
These outcomes were evaluated at 1 week, 3 months, and 1 year postoperatively.
Statistical analysis
Data were analyzed using SPSS 20.0 statistical software. Data were expressed as mean ± standard deviation for normal distribution and as median for non-normal distribution. Count data were represented as the number of cases (percentage). • The independent sample t test was employed to compare data with normal distribution between groups. • The Mann-Whitney U test was used for non-normal distribution comparison. • The chi-square test, continuity corrected chi-square test, and Fisher’s exact test were applied for count data comparison. • Paired sample t tests compared preoperative and postoperative data.
The significance level was set at α = 0.05 for all tests.
Results
Patient characteristics
Patient characteristics.

Operation-related characteristics
Operation-related characteristics.

ap < .05, statistically significant.
bFisher’s exact test.
Functional outcomes
There was no significant difference in VAS score between the two groups at 1 week, 3 months, and 1 year after surgery. There was no significant difference in lamina healing on the hinge side between the two groups at 1 week, 3 months, and 1 year after surgery. There was no significant difference in the rate of improvement of the JOA score between the groups at 1 week, 3 months, and 1 year after surgery.
Functional outcomes at different time points.
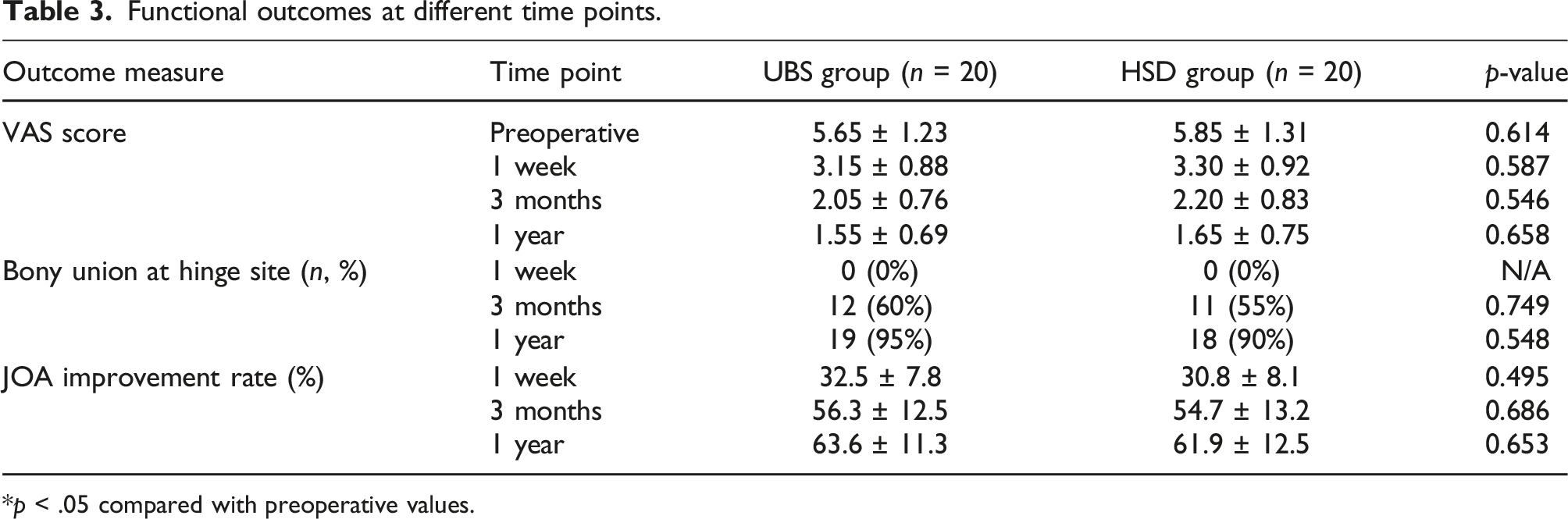
*p < .05 compared with preoperative values.
Typical case
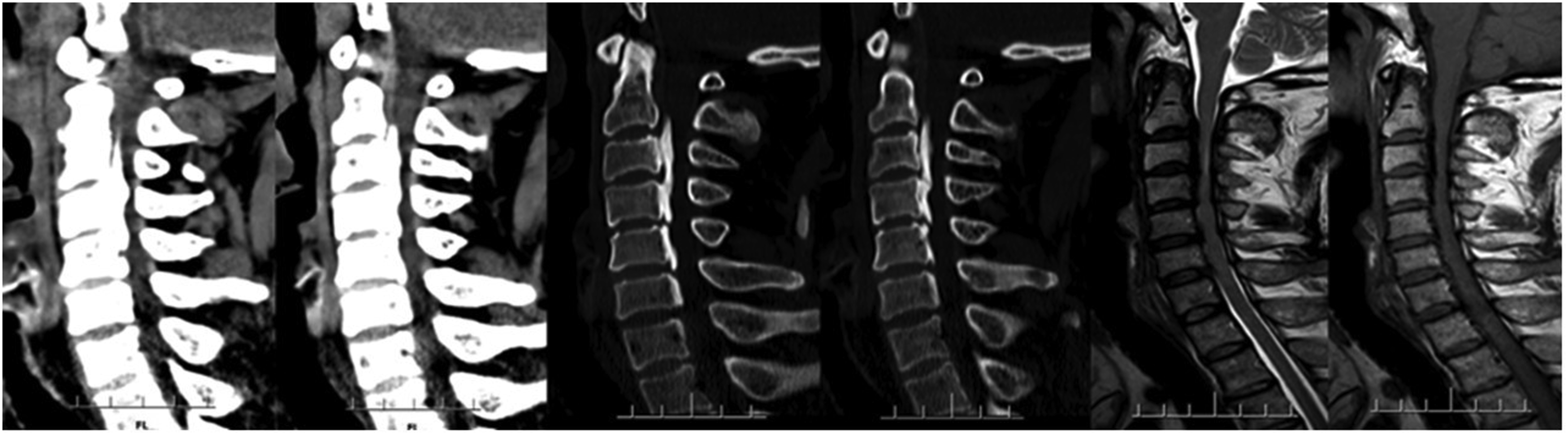
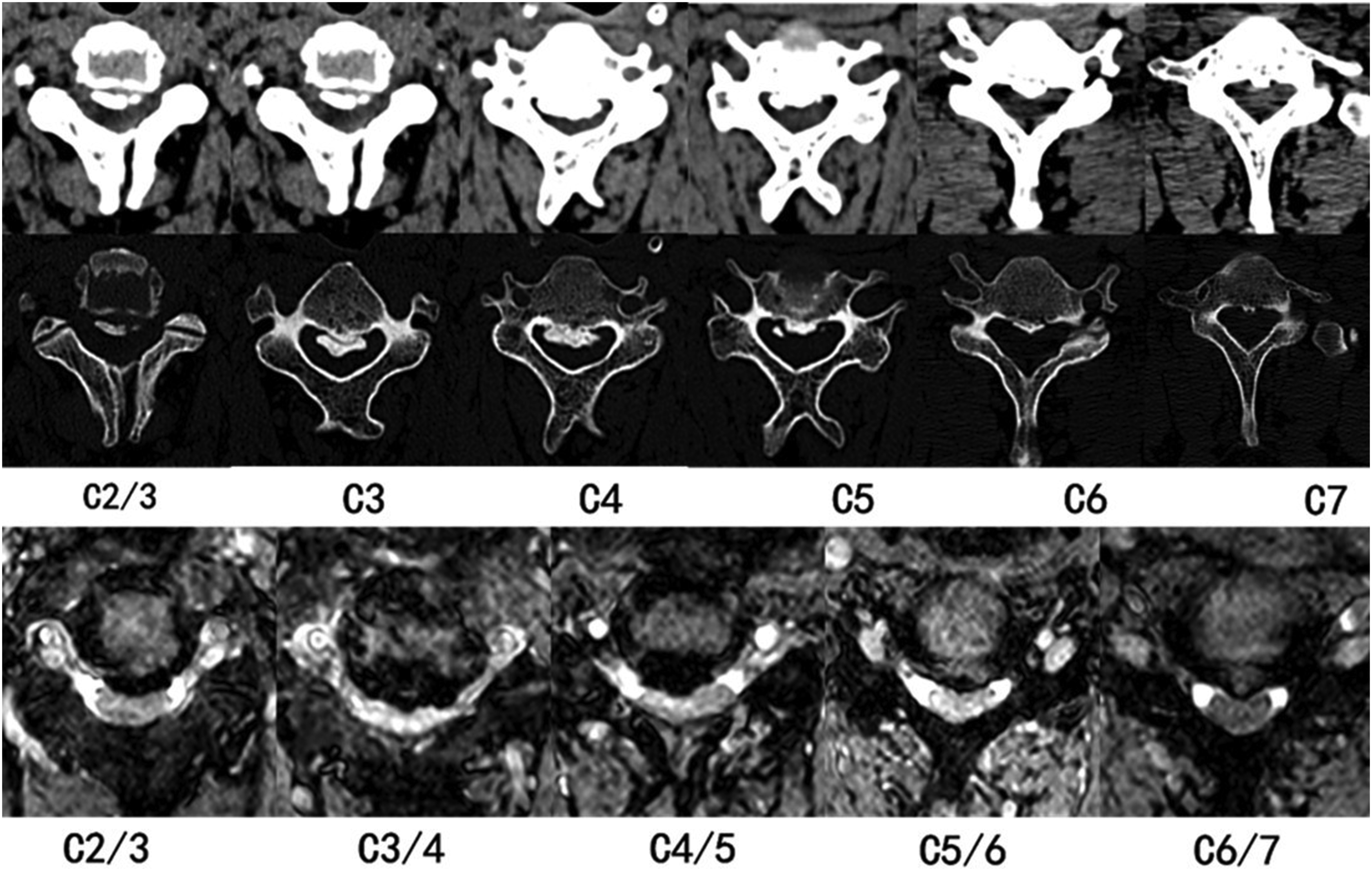
Typical case introduction: The patient Zhao, a 47 years old male, was admitted because of “numbness and weakness of extremities for 1 month”. Specialist physical examination: the sensory muscle strength of extremities was normal, bilateral biceps brachii, triceps brachii hyperreflexes, bilateral knee hop hyper-reflexes, Babinski sign positive and JOA score of 10. Figures 1–5 present the preoperative interpositive and dynamic X-ray, preoperative sagittal CT and Magnetic Resonance Imaging (MRI), preoperative axial CT and MRI, postoperative interpositive and dynamic X-ray, sagittal and axial CT, respectively. Preoperative interpositive and dynamic X-rays showing cervical spondylotic changes and preserved lordosis. Preoperative sagittal CT and MRI demonstrating multi-level spinal canal stenosis with cord compression. Preoperative axial CT and MRI at C4-5 level showing severe central canal stenosis with spinal cord compression. Postoperative interpositive and dynamic X-rays showing successful laminoplasty with mini-plate fixation and preserved cervical alignment. Postoperative sagittal and axial CT images demonstrating adequate spinal canal decompression and stable “open-door” position of the laminae at five levels (C3-C7).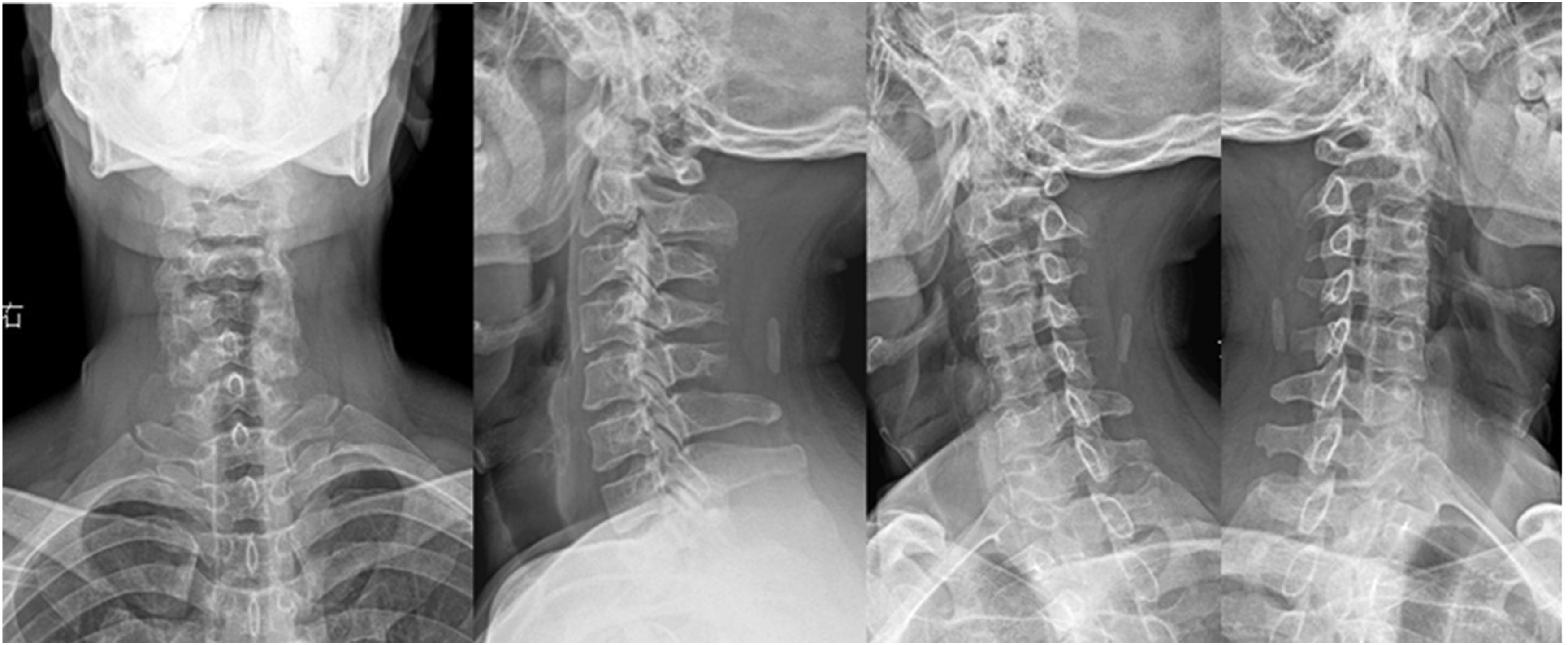


Discussion
Application of UBS in CEOL
The CEOL was first reported by the Japanese scientist Hirabayashi in 1978 and remains a classic surgical technique for posterior cervical decompression. This procedure effectively decompresses the spinal cord while preserving the structural integrity of the lamina, posterior muscles, and ligaments. Such preservation is beneficial in reducing the occurrence of complications, including cervical kyphosis and anterior oxidation, thereby maintaining biomechanical stability while reducing the incidence of postoperative complications. 26
In recent years, maturation of UBS technology and advances in surgical skills have led to its widespread application in CEOL for the treatment of CDD. Our findings are consistent with previous randomized controlled trials, such as Kim et al. (2020), who also reported significant reductions in operation time with UBS compared to HSD in cervical laminoplasty (146.5 ± 23.4 min vs 168.3 ± 28.6 min, p < .001). 27 However, our study extends these findings by demonstrating reduced postoperative drainage, which was not evaluated in the Kim et al. study. The UBS works by converting electrical energy into high-frequency vibrations through a piezoelectric transducer, achieving osteotomy through mechanical and cavitation effects. The mechanical effect involves the cutting force generated by the high-frequency vibration of the ultrasonic bone cutter head acting on the bone tissue. 28 In contrast, the cavitation effect occurs when tiny bubbles, called cavitation nuclei, in the fluid surrounding the tissue generate high temperature, pressure, strong tangential force, and shock waves under ultrasonic energy. This results in the emulsification and fragmentation of the tissue.
As orthopedic surgeons, it is imperative to critically evaluate the advantages and disadvantages of UBS in the context of CDD treatment. This discussion aims to provide valuable insights and improvement suggestions for optimizing surgical outcomes.
Safety evaluation of UBS in CEOL
This study found statistically significant differences in both the total operation time and the opening time between the ultrasonic bone scalpel (UBS) group and the high-speed drill (HSD) group (p < .05). While these differences may be partly attributed to the inherent characteristics of each device, it is important to acknowledge the potential influence of surgeon-dependent variables. We attempted to control for this factor by ensuring all participating surgeons had equivalent expertise with both instruments and by distributing cases evenly among them. Despite these controls, we recognize that individual variations in surgical technique and team coordination may still have minor influences on our results.
The UBS is an efficient bone-cutting tool that operates at a lower frequency than the natural frequency of soft tissue. When the ultrasonic bone knife collides with soft tissue, the tissue can absorb part of the energy through elastic vibration, which minimizes the risk of injury to the spinal cord and surrounding nerves. 29 Furthermore, small clearance of the ultrasonic bone knife allows for quick completion of the door shaft production using the “V” slotting method, thereby enhancing operation efficiency. 28
In contrast, the use of a high-speed drill requires an initial grooving step, followed by lamina cutting with a Kerrison rongeur, which may prolong the door-opening time. 30 Some studies have shown that the average time for single-segment laminar osteotomy using ultrasonic osteotomy is significantly shorter than that achieved with high-speed burr drilling, both in open-door extended spinal canal formation and in total laminectomy with internal fixation.31,32
Thus, advantages of the ultrasonic bone knife include its precision in operation and streamlined open-door technique, contribute not only to shorter overall operating time but also to a shorter hemostasis time and improved visibility of the surgical field, which fascilitates a smoother operation procedure.
The results of this study showed that intraoperative blood loss in the UBS group was comparable to HSD group. However, postoperative drainage volume in the UBS group was lower than in the HSD group, which contrasts with the results of some related studies.27,33,34 This discrepancy may be due to differences in measurement techniques and the specific mechanisms by which UBS reduces postoperative bleeding. The thermal effect of UBS likely provides better hemostasis of small vessels in the cancellous bone surface, which continues to benefit patients in the postoperative period. Additionally, the selective cutting property of UBS may result in less soft tissue disruption, contributing to reduced postoperative oozing.
The main factor influencing the intraoperative blood loss between the two groups is probably the measurement of the blood exposure from the cancellous bone on the laminectomy surface during the procedure. Throughout the entire CEOL procedure, whether in the UBS or HSD group, the treatment of cancellous bone oozing due to “cutting the lamina” inevitably results in the rupture of the venous plexus along the inner wall of the spinal canal during the subsequent “opening of the spinal canal.” This usually results in significant bleeding. Surgeons typically rely on automatic hemostasis at the site of the venous rupture by filling the spinal canal with a gelatin sponge or brain sponge.
While the fine cutting and thermal effects of the ultrasonic bone knife can provide effective local hemostasis to microvessels,35,36 the short cutting time minimizes its impact on the overall statistics of intraoperative blood loss. In contrast, the prolonged microvascular bleeding associated with the UBS likely contributes to the reduced postoperative drainage volume observed. With consistent standards for drainage tube removal, the use of the ultrasonic bone knife may reduce drainage tube indwelling time of the, thereby facilitating earlier patient rehabilitation.
Surgery related complications, such as spinal cord injury, dural tear, cerebrospinal fluid (CSF) leakage, axial symptoms, and C5 nerve root palsy, are critical indicators of assessing the safety of posterior cervical surgery. In this study, there were no spinal cord injuries, axial symptoms, or C5 nerve palsies both groups. However, there was one case of dural tear in the HSD group (p > .05). When the dural tear was identified during surgery, attempts were made to repair it, and dural pulsation was monitored. While there were no cases of CSF leakage in the UBS group, there was one case in the HSD group (p > .05). It is important to note that while our study showed numerical differences in complication rates favoring UBS (e.g., 0% vs 5% for dural tears), these differences did not reach statistical significance. This lack of significance is likely due to our limited sample size, which was powered primarily for detecting differences in operation time rather than rare complications. Larger multicenter studies would be needed to definitively establish whether UBS truly reduces complication rates compared to HSD.
Axial symptoms and C5 nerve root palsy significantly affect patients’ quality of life after posterior cervical surgery. The primary cause of both complications is believed to be nerve traction during the retraction of the cervical spinal cord after surgery.37–39 To mitigate these risks, careful attention was paid to preserving the hinge muscle-ligament complex and maintaining cervical lordosis during CEOL procedure in both UBS and HSD groups. 40 This approach effectively reduced the incidence of axial symptoms and C5 nerve palsy.
The occurrence of spinal cord injury, dural tears, and CSF leakage is often attributed to factors such as soft tissue adhesion within the spinal canal, but in many cases it is the result of improper surgical technique. While high-speed drilling is effective in quickly removing bone tissue, it can also cause collateral damage to surrounding soft tissues, including the dura mater, and can even directly injure the spinal cord. In addition, the heat generated during high-speed drilling can burn the spinal cord and nerve roots. 41
In contrast, the ultrasonic bone knife works by making longitudinal incisions in bone tissue through a reciprocating motion with a small amplitude, which minimizes the risk of damaging surrounding soft tissues, thereby reducing the likelihood of injury to the dura mater or spinal cord compared to traditional techniques using gun-type lamina rongeurs and high-speed drills.42,43 However, high-frequency vibration of the ultrasonic bone knife may generate heat during operation. Although it is equipped with an automatic spraying device to cool the surgical area and protect the surrounding soft tissues, 44 there have been reports of temperatures exceeding 70°C on the bone cutting surface when the ultrasonic bone knife is applied to the same area for a prolonged period of time, leading to possible dural injury. 45 As reported by Tarazi et al. (2018), improper use of UBS can generate significant thermal energy, emphasizing the importance of proper technique and continuous irrigation during use. 25
In one case involving a patient with severe callus hyperplasia at the lamina, the cutting surface of the ultrasonic bone knife showed a burnt yellow appearance due to excessive local heat. Fortunately, no CSF leakage was observed postoperatively after a timely stop-and-reassess approach was implemented. This incident underscores the high safety of the ultrasonic bone knife, although it still requires careful handling during surgery.
Effectiveness evaluation of UBS in CEOL
Regarding the efficacy of ultrasonic bone surgery after CEOL, the results of this study indicated that there was no occurrence of screw displacement or plate fracture in either UBS or HSD groups when cervical spine imaging was re-evaluated within 1 week of surgery. Both groups demonstrated significant improvements in spinal canal area, Japanese Orthopaedic Association (JOA) scores, and Visual Analog Scale (VAS) scores when compared to preoperative measurements, but no significant differences were observed between the two groups. Furthermore, the follow-up assessments of postoperative vertebral body healing at the portal axis showed no statistically significant differences, with a generally slow trend in overall healing.
It should be noted that the absence of significant differences in JOA and VAS score improvements between the two groups might be influenced by our relatively small sample size (n = 20 per group). While our sample size calculation was adequate for detecting differences in operation time based on previous studies, it may have been insufficient to detect smaller differences in functional outcomes. A larger multi-center study would be valuable to validate these findings and potentially identify subtle clinical benefits that our study may have missed.
These findings suggest that the choice of laminoplasty instruments does not affect clinical efficacy; both the ultrasonic bone knife and the high-speed drill can effectively achieve spinal canal decompression and facilitate clinical improvement. Although high-quality surgical instruments can provide ancillary benefits, the restoration of neurological function depends largely on the surgeon’s skills in spinal cord decompression techniques.
Healing of the lamina at the portal axis is critical for maintaining the open state of the lamina, which is essential for minimizing the risk of lamina reclosure and axis fracture complications. Therefore, it is important to understand how different surgical tools affect bone healing at the portal axis. The high temperatures generated by high-speed drilling may adversely affect healing in this area, while the cavitation effect of the ultrasonic bone knife could potentially lead to osteocyte necrosis, thereby affecting bone healing. 25
Animal studies have shown that while high-speed drilling promotes better healing of the cortical bone, ultrasonic bone knives are more effective in healing cancellous bone. 42 In addition, a randomized controlled trial involving 140 patients found that bone healing at the vertebral portal axis was adversely affected within 6 months in patients who went ultrasonic osteotomy. 29
Experience sharing
This study identified several limitations of UBS, leading to the following recommendations for enhancing its application in cervical expansive open-door laminoplasty (CEOL): (1) Progressive Cutting Technique: Implement a stepwise cutting approach, particularly as the osteotomy approaches the spinal canal. Surgeons should maintain control of the ultrasonic osteotome with one hand while stabilizing with the other, thereby preventing excessive force that could inadvertently compromise the spinal canal. (2) Cold Cutting Mode: Utilize the cold cutting mode to minimize friction between the ultrasonic bone knife and surrounding bone tissue, which can result in thermal expansion. This practice not only preserves the tool’s integrity but also reduces wear and costs associated with frequent replacements. (3) Continuous Motion of the Tool Head: Ensure that the tool head remains in motion with appropriate force to prevent it from staying in one position for too long, which can inhibit normal vibrations. This approach reduces the risk of thermal damage to adjacent tissues and enhances the efficiency of bone cutting. (4) Strategic Bone Incision Point: When cutting through the lamina, position the incision point inward and maintain a perpendicular angle to the lamina. This technique keeps the knife tip distanced from the venous plexus, thus minimizing the risk of short-term massive bleeding. However, care should be taken not to displace the incision point excessively inward to avoid dural injury. (5) Preoperative Imaging Enhancements: Improve preoperative imaging assessments to identify potential complications related to ossification of the dura mater, ligamentum flavum, or protruding cervical posterior longitudinal ligament. Awareness of these conditions is crucial, as they may lead to complications during ultrasound osteotomy. (6) Customized Knife Head Selection: Select knife heads of varying diameters and shapes based on the specific surgical needs. Spine surgeons often use flake knife heads to apply downward force during horizontal cuts. Caution is advised to prevent excessive downward pressure that may damage the spinal cord or nerves. Conversely, a hook cutter tip can be employed against the caudal edge of the lamina, requiring only transverse cutting forces, thus theoretically reducing the risk of nerve injury. (7) Use of Wet Cotton Pads: Place wet cotton pads near critical anatomical structures to protect them during surgery. In cases where a dural tear is detected, the operation should be halted immediately to prevent further spinal cord injury, and meticulous repair of the dura should be performed concurrently.
Limitations and future directions
This study has several limitations that should be acknowledged. First, despite our efforts to control for surgeon skill and learning curve effects, some degree of variability in surgical technique inevitably remains. The learning curve associated with UBS adoption was not specifically evaluated in this study, which may influence the generalizability of our results. 25 Second, our sample size, while statistically adequate for the primary outcome measures, may have limited our ability to detect more subtle differences in secondary outcomes such as functional improvement rates and rare complications. Based on our observed complication rates, a sample size of approximately 200 patients per group would be needed to detect statistically significant differences in dural tear rates with 80% power. Third, our follow-up period of 1 year, though sufficient for most clinical parameters, may be inadequate for fully assessing long-term outcomes such as adjacent segment degeneration or late-onset complications. Fourth, the cost-effectiveness of UBS versus HSD was not evaluated in this study. Given that UBS equipment requires a higher initial investment compared to traditional HSD, economic considerations may influence its adoption in various healthcare settings.
Future research should address these limitations through multi-center trials with larger sample sizes and longer follow-up periods. Additionally, more sophisticated outcome measures, including quality of life assessments and detailed radiological analysis of bone healing processes, would provide deeper insights into the comparative benefits of these surgical techniques. Cost-effectiveness analyses would also be valuable in determining the overall economic impact of choosing UBS over HSD, considering both direct costs and indirect benefits such as potentially shorter hospital stays and faster return to work. Studies specifically examining the learning curve for UBS adoption and strategies to optimize training would help facilitate wider implementation of this technology.
Conclusions
In conclusion, ultrasonic bone surgery (UBS) demonstrates significant efficacy in cervical spine surgery. Compared to high-speed drilling (HSD), UBS not only shortens laminectomy time but also reduces postoperative drainage and shows a trend toward fewer surgery-related complications, although the latter did not reach statistical significance in our study due to sample size limitations. These benefits contribute to faster patient recovery and reduced operator workload, highlighting the broader applicability of the UBS in spine surgery.
While our study controlled for surgeon-dependent variables, we acknowledge that individual surgical skill and team factors may still influence outcomes to some degree. Nevertheless, the consistent results across different surgeons in our study suggest that the advantages of UBS are likely device-related rather than operator-dependent. However, larger studies are needed to confirm whether UBS truly reduces complication rates and to better understand the cost-benefit ratio of this technology.
Moving forward, increasing the rate of hinge side healing and improving the quality of life after surgery are critical areas for further investigation. Continued exploration of UBS application techniques and innovative improvements to cervical expansive open-door laminoplasty (CEOL) will be central to in our future research efforts.
Footnotes
Acknowledgments
The authors would like to thank the editors and all reviewers for their constructive comments and valuable suggestions that helped improve the quality of this manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Xinjiang (2022D01F30) and The First Science and Technology Innovation Talent Selection Committee of Karamay City, Xinjiang.
Data Availability Statement
The data could be obtained by contacting the corresponding author.
