Abstract
Objectives
To report on the process and results of clinical application of 3D printed patient-specific metal plates in orthopaedic trauma.
Materials and Methods
A retrospective review was conducted on 11 pilot patients managed with 3D printed Patient-Specific Plates (PSPs) together with 3D printed guides, 3D printed bone models and 3D virtual planning for malunions or acute fractures. There were six clavicles, three forearm shafts, one ankle, and one tibial plateau. The surgeon and the engineering team collaborated on the designing of customized treatment solutions for each case.
Results
From obtaining the CT data to manufacture of all 3D printed parts, the required ranged from 3 to 43 days (mean 16 days) and the cost ranged from 1590 to 4100 USD (mean 1360 USD). All 11 patients were followed up from 6 to 24 months. All patients had bony union, good to excellent clinical outcomes documented from clinical scores and with no complications. Challenges with the workflow were collected qualitatively and listed out.
Conclusion
Implementation of 3D printing technology with customized patient specific implants appears to be beneficial with good outcomes in patients with complex anatomy and malunions. We have described the logistical and regulatory considerations. Further studies should be conducted to demonstrate its cost-effectiveness compared to conventional solutions.
Keywords
Introduction
Three-dimensional printing (3DP) or additive manufacturing (AM) is a recognized technology which enhances patient care in complex maxillofacial, vascular, plastic, and orthopedic surgery.1–3 The success of this technique hinges on accurate anatomical visualization, surgical execution, and enhanced positioning of implants. Numerous studies have demonstrated the efficacy of 3D virtual planning and 3D printed bone model and 3D printed jig for the management of complex fractures and malunions in various regions, including the pelvis and acetabulum,4–7 pilon, 8 tibial plateau, 9 clavicle 10 forearm and other regions.11,12 Recent reviews conclude that 3D and AM techniques significantly reduce operative duration, blood loss, and radiation exposure.13,14
The challenges of corrective osteotomy include several critical considerations. Cuts should be accurately aligned with the center of rotation of angulation (CORA) which is the site of maximal angular deformity. Proper bone contact at the osteotomy site must be ensured while preserving blood supply to minimize complications. Additionally, minimizing the defect size is crucial for optimizing healing. Wedge osteotomies are the most common yet present specific challenges; closing wedges shorten bone length and require two highly accurate cuts for adequate contact, while opening wedges can create defects and soft tissue tension. Single-cut rotational osteotomies are advantageous as they enhance bone contact while minimizing bone loss. However, they require meticulous planning guided by geometrical principles, often involving specialized software. AM surgical jigs have been shown to enable precise osteotomy and placement of screw holes, facilitating reduction and precise implant positioning.
Mechanically stable fixation facilitates early rehabilitation and bone union. Manually contoured reconstruction plates are made from annealed alloys with high ductility to facilitate easy contouring. However, the low yield strength makes them prone to deform under physiological loads and less suitable for long bone mid-shaft fixation. 15 Anatomically pre-shaped locking plates based on average body sizes have been the standard for many years. These are made from stiff cold-worked alloys which can withstand high yield strength but can be difficult to shape during surgery. 16 Shaping of implants using patient specific plastic bone models digitally reconstructed from the contralateral side is a recognized technique to resolving the above difficulties.17–19
Alternatively, AM Patient Specific Plates (PSPs) can be fabricated to perfectly match patient anatomy. 3D printed metal parts using powder bed laser sintering technology typically have high yield strength, 20 making them ideal for malunion correction and fracture fixation in long bones with complex anatomy. While the role of AM patient specific implants is well established in craniomaxillofacial bone reconstruction procedures,3,21,22 its application remains at infancy in orthopedics. The perceived challenges include a long production time, questionable longevity, lack of financial reimbursement, and medical device regulation hurdles. Here, we describe a series of illustrative cases where we have overcome these obstacles, highlighting the practicality and benefits of AM PSPs.
Materials and methods
This retrospective study investigates the application of AM PSPs in orthopedic trauma surgery, 11 consecutive patients treated between October 2018 and June 2023 across two centers were included. The study had ethical approval obtained from two ethics committees: the Institutional Review Board of the Medical Research and Invention Center, Bhumibol Adulyadej Hospital, Directorate of Air Medical Service, Royal Thai Air Force, Thailand (IRB No.12/63), and the University of Hong Kong, Hospital Authority Hong Kong West Cluster, Hong Kong, China (UW 20-496). All patients provided written informed consent.
AM PSP is indicated in patients with acute fractures or malunions where high accuracy for fragment positioning is preferred while implant contouring is perceived to be challenging. The additional benefits are minimized surgical dissection, and straightforward anatomical restoration guided by perfectly pre-contoured implant. Specific uses cases include acute clavicle fractures and malunions, forearm fractures and malunions and periarticular malunions. In these locations, fluoroscopic clues for anatomical axis restoration are vague and direct visual assessment is limited. This study emphasizes qualitative reporting of workflows together with quantitative analysis of outcomes. The limited number of cases highlights the exploratory nature of this technique and the challenges of compiling a larger sample size
Workflow
The workflow for PSPs in orthopedic trauma surgery relies on the close collaboration between surgeons and engineers. This partnership ensures the accurate execution of the treatment plan and adherence to manufacturing and regulatory standards. Engineers bring expertise in CAD/CAM, material science and metallurgy, while surgeons provide insights into anatomical structures, clinical needs, and surgical techniques.
The process begins with acquiring computed tomography (CT) imaging data of both the fractured and contralateral normal bone. CT scans are conducted using standard imaging protocols with a maximum slice thickness of 1 mm to achieve high spatial resolution. The data (DICOM format) is processed using Avizo software (Thermo Fisher Scientific, MA, USA) or Mimics Software (Materialise NV, Leuven, Belgium) to create a detailed 3D model in a process called bone segmentation. Bone segmentation is performed based on Hounsfield unit thresholds judged case by case depending on the bone density. The CT scans include the contralateral bone which is segmented, virtually mirrored and superimposed to serve as a normal reference.
Engineers from a local patient specific implant manufacturer (Meticuly, Thailand or Koln 3D Medical, Hong Kong) worked together with surgeons to define the osteotomy plane, simulate fragment reduction, and implant positioning. In malunions, the location of the osteotomy is at the center of rotation of angulation (CORA), defined by the site of maximum angular deformity. The deformity is often multiplanar, simultaneously affecting the sagittal, coronal and axial planes. After completing the virtual reduction, the PSP is designed to be placed on it using ANSYS software (Ansys Inc., PA, USA) or 3-Matics software (Materialise NV, Leuven, Belgium). Surgeon’s preferences are considered in each case, including the approach, size and placement of K-wires, saw blade thickness, and the location and number of screws.
Titanium alloy (Ti-6Al-4V) PSPs must withstand physiological loads, while maintaining interfragmentary compression, and minimizing prominence. Using Finite Element Analysis (FEA) simulation, the implant’s mechanical integrity is validated under physiological loading conditions with a safety factor of at least 2x. FEA simulation allows the designer to improve the mechanical properties by altering its shape and eliminating stress risers, maximizing fatigue strength. 23 The FEA certified implant is then fabricated via powder base selective laser sintering (Concept Laser, Germany, or 3D Systems, USA) using medical-grade titanium alloy powder, with subsequent post-processing steps including heat treatment, machining of locking screw threads, polishing, and autoclave sterilization.
3D-printed polymer bone models and patient-specific cutting and drilling guides, known as Surgical Assisting Kits (SAKs), are designed as the final step after planning the osteotomy and fixation. These guides are surface matched to fit the patient’s anatomy and provide precise positioning for osteotomy cuts and screw holes. Intraoperative use of SAKs facilitates precise drilling, bone cuts, accurate fracture reduction, and optimal implant placement. SAKs and bone models are manufactured using fused filament fabrication 3D printers (Zortrax, Poland) or (Stratasys FDM, USA) with biocompatible Polyamide-12 (Nylon) or Acrylonitrile Butadiene Styrene polymer (ABS-M30i), these jigs undergo validation through mock surgeries with plastic bone models before they are sterilized for on-table use Figure 1. Flowchart illustrating the key stages of 3DP-PSP workflow: design, manufacturing, quality assurance, and implementation. Steps requiring surgeon involvement are highlighted in bold, while others can be supported by technical and engineering staff.
Regulatory and quality control considerations
The implementation of customized AM implants follows five steps for regulatory approval and quality control. Firstly, the use of such implants must be complied with local regulation defined by healthcare authorities, such as the Thai Food and Drug Administration or the Hong Kong Department of Health. Extensive pre-market biomechanical and chemical testing for each specific design is time consuming. In our localities, extensive pre-market testing can be exempted following provisions like the “Custom Device Exemption” guidance by the US FDA (2013-D-1601, Section 520b) and the European Union’s I.117/163 Annex XIII “Procedure for Custom-Made Devices” requirements if these implants are used in unique conditions in limited number of patients and are directly prescribed by surgeons.
Secondly, authorization is required from the hospital’s internal regulatory board. In our case, we have also obtained blanket approval form the ethics committee for PSPs in procedures involving complex fracture fixation and malunion surgeries. These procedures are conducted as clinical studies, mandating post-implantation follow-up and surveillance to identify and report potential issues.
Third, the manufacturing processes adhere to established industrial standards. Fabrication workflow is certified under EN ISO 13485, which governs quality management systems for medical devices, including design, manufacturing, and packaging by experienced manufacturers and ISO 19227 for cleanliness of orthopedic implants. All 3D printing machines must undergo routine maintenance and calibration, with validation tests ensuring geometric precision between printed samples and CAD data. Implant materials comply with ISO-10993 for chemical biocompatibility. These implants are fabricated using raw materials conforming to ASTM standards for orthopedic applications, specifically ASTM F136/UNS R56401 for Wrought Titanium-6 Aluminum-4 Vanadium ELI Alloy for Surgical Implant Applications.
Fourth, the manufacturing quality is pre-validated using standard shaped coupons of typical dimensions which undergo destructive mechanical testing, including single-load failure and cyclic fatigue performance to verify that yield strength and fatigue strength requirements are met. The testing protocols ASTM F382 (Specification and Test Method for Metallic Bone Plates) and ASTM F3001 (Standard Specification for Additive Manufacturing Titanium-6 Aluminum-4 Vanadium ELI) are used. Key mechanical properties include four-point bending yield strength (>1025 MPa), elastic modulus (>110 GPa), and fatigue strength (>500 MPa at 1,000,000 cycles). Since individual implant designs are not subjected to time consuming fatigue testing due to obvious impracticality. The gross shape and dimensions of implants must stay similar with small discretional variations allowed per clinical scenario.
Fifth and finally, in silico simulated testing is performed for each case. This non-destructive and resource-efficient approach applies Finite Element Analysis (FEA) to each clinical scenario, referencing literature-established physiological loads across long bones with a safety factor of twice the expected loads. Although clinical regulatory standards for such testing remain undefined, this process is undertaken as a final due-diligence measure.
Results
Demographics data, and details including clinical outcome scores and challenges of the patients receiving 3DP-PSP.

Clavicle fractures
Six clavicle fractures were treated, comprising of three acute fractures, one delayed union and two symptomatic malunions. Virtually mirrored 3D models of the contralateral side were used for all patients to guide reduction since symmetrical length and curvature are desired. Separate SAKs were used for screw holes at the proximal and distal ends and osteotomies at the CORA. Large single incisions were avoided and Minimally Invasive Plate Osteosynthesis (MIPO) was performed with incisions parallel to the skin crease. Reduction is performed by placement of screws compressing the implant into pre-drilled holes, followed by neutralization locking screws. Postoperative radiographs confirmed satisfactory reduction, and all patients achieved good to excellent shoulder function without complications Figure 2. Corrective osteotomy of clavicular malunion, (a) Model of reduced clavicle with PSP (b) Malunion model with intra-operative positioning drill guides (black arrows) and corrective osteotomy guides (white arrows), (c) Model used for incision planning, (d) Corrective osteotomy with clips for securing the guides to bone, (e) Minimally invasive submuscular osteosynthesis with PSP, (f) Radiograph showing symmetrical restoration of the anatomy.
Malunited forearm fractures
Three ulna and two radial shaft malunions were corrected in three patients. All had an epiphysial deviation of at least 12 mm at one end when the other end is superimposed with the mirrored contralateral model. All patients had minor angular deformities of less than 5° which are subtle and difficult to assess on plain radiographs. All patients presented with restricted pronation supination and discomfort at the proximal or distal radio-ulna joints. In all cases, the surgical plan aimed for symmetrical restoration of length, axis and rotation as this is believed to improve function. One patient with a chronic proximal radioulnar joint dislocation required additional, open reduction and annular ligament reconstruction. One patient with malunited previous plate fixation of both bones had osteotomy and exchange to a PSP, previous screw holes were reused to preserve bone stock and prevent future per-implant fractures. In all five long bones, implant guided deformity correction was successful. All patients had uneventful union and satisfactory functional recovery with no complications over a follow-up period of 9 to 24 months Figure 3 and 4. Corrective osteotomy of forearm malunion, (a) deformed bone superimposed on the mirror image of contralateral side to evaluate the deformity, (b) preoperative planning and design of malunion correction, SAKs and PSPs for surgical treatment using the ulnar for illustration. (c) preoperative and (d) post-operative radiographs showing restoration of the forearm bowing. Forearm shaft malunion with pre-existing metal implant. (a) axis deviation of 12 mm at distal ulna when superimposed to 3D contralateral mirror, cortical irregularities are caused by metal signal artefacts. (b) axis deviation 16 mm at proximal radius. (c) intraoperative photo of 3DP at hand, comparing to the existing implant with screw holes to be reused. (d) X-ray before correction showing subtle insufficient radial and reversed ulna bowing. (e) X-ray showing correction of above deformities and clinical restoration of functional pronation and supination range after osteotomy healing.

Intraarticular revision of internal fixation
Two patients required intra-articular correction and revision of internal fixation. The first involved a 17-year-old female with a malreduced supra-syndesmotic pronation-external rotation bimalleolar fracture. The deformities to be corrected included fibula shortening, syndesmosis malposition, and ankle joint subluxation. Again, the preoperative plan aimed for symmetrical restoration of joint anatomy. The syndesmotic screw had to avoid previous screw holes for a better purchase. The syndesmosis is openly released, distractors were used to grossly restore fibula length, followed by a more precise SAK and implant directed reduction. The syndesmotic screw was removed at 12 weeks. At 6 months postoperatively, the fracture had healed completely with the patient enjoying excellent ankle function Figure 5. PSP in mal-reduced ankle fracture. (a) Drilling guide for fibula plate and syndesmotic screw, avoiding existing screw holes, (b) pre-operative planning for anatomical reduction of the fibula length and syndesmosis, (c) PSP and drill guides used intra-operatively, (d) radiograph before surgery showing mal-reduction of syndesmosis and fibula shortening, (e) Radiograph after surgery showing restored alignment.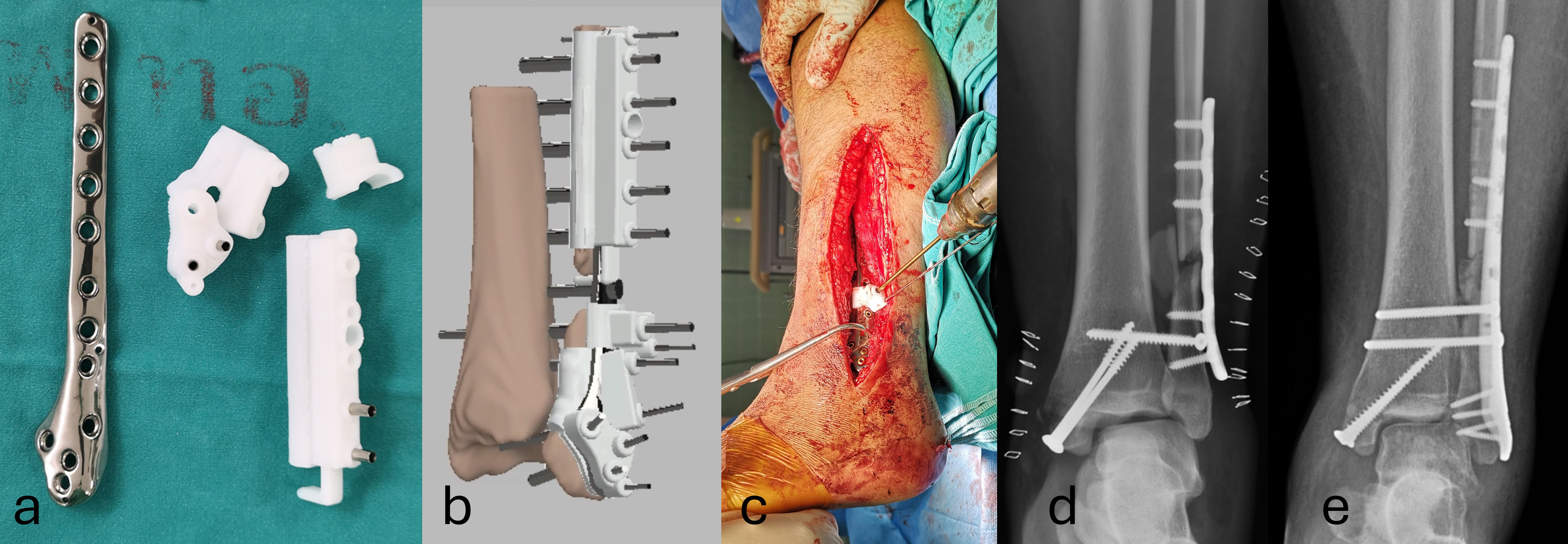
The second case involved a 52-year-old male with a malunited bicondylar tibial plateau fracture, presenting with knee joint subluxation, instability and varus malalignment. Preoperative simulations guided the design of a PSP for posteromedial buttress. Fluoroscopy monitoring was necessary during osteotomy to avoid damage to the femoral condyles. Osteotomy and fixation were executed using SAKs and the PSP, achieving proper limb axis alignment and a smooth joint surface. At the 2-year follow-up, the patient demonstrated full knee range of motion, good alignment, and excellent function Figure 6. Mal-reduced tibial plateau fracture, (a) Pre-operative design of customized surgical solution with customize plate, guides and the preoperative planning of intraarticular osteotomy, (b) Finite element analysis of PSP under a load of 120 kg, (c) Intraoperative fluoroscope of cutting guide, (d) fluoroscopic monitoring of intraoperative placement of the guides and bone cut, (d) pre-operative X-ray showing the a varus mal-reduced deformity of the medial plateau and, (e) post-operative 2 years with full union, noting the use of medial PSP and a lateral conventional locking plate.
Discussion
3D technology is adopted in orthopaedic trauma with substantial known benefits through virtual planning, AM bone models and surgical guides.4–12,17 Mirrored contralateral normal models are used to assist surgeons in visualizing complex fracture configurations in regions such as the pelvis and acetabulum24,25 and for pre-contouring commercially available plates in long bones such as the clavicle and forearm, enhancing reduction and implant fit.10,17–19,26 These tools aids the less experienced surgeon, 27 and have shown to drive efficiency and good outcomes.28,29 The use of free software and rapid production using low-cost desktop printers further improves the accessibility of the technology. 29 However, the adoption of customized AM implants for fracture treatment is not widespread due to a series of logistical hurdles, and thus in this article, we have described the step-by-step workflow to aid readers adopt this technique.
AM implants are widespread in craniomaxillofacial surgery21,22 with its challenges and benefits well described more than a decade ago. 22 Yet, for orthopedics, the use of PSP is seldom reported. We have found a few case series reporting its uses in the forearm,26,30 clavicle 31 and acetabulum. 32 These regions have complex or subtle curvatures which make perfect contouring of implants challenging. These studies report favorable outcomes with PSPs, highlighting their high anatomical accuracy and intraoperative ease of use due to pre-contoured fit, which reduces the need for additional adjustments during surgery. Good outcomes include reliable restoration of forearm pronation supination26,30 with a corrective error of less than 1.8° measured in post operative CTs when compared to the digital plan 30 with excellent functional outcomes. 26 Improvement in material science, the availability of 3D printers and software, and understanding in quality control and regulations means PSPs can be more widespread. In the above studies, PSPs, AM surgical guides (SAKs), bone models and virtual preoperative planning are four integral components of the treatment workflow. We have demonstrated their effectiveness for addressing complex fractures and malunions in various regions following the same workflow. All steps from planning, reduction, bone cutting, and implant placement are stereotactic guided, ensuring that length, axis, rotation and articular congruity are reliably addressed. Sterilized bone models are useful for trial reductions, clamp positioning, and reference for implant placement. Soft tissue incision can be minimized using separate smaller SAKs. Implant guided reduction of bone fragments appears to be intuitive when the screw holes are accurately pre-positioned. The workflow minimizes repetitive attempts at reduction, fluoroscopy, implant contouring and positioning, resulting in good overall efficiency. Safety is demonstrated with all patients achieving union and good to excellent functional outcomes with no complications.
The preparation time for PSPs remains variable, the minimum is 3 days for acute fractures when the team devotes extra effort. For malunions, a more conventional work schedule is adopted and this averaged 16 days. Hence, PSP solutions are more pragmatic when performed as elective procedures. PSPs were deliberately designed to be compatible with standard commercially available screws and surgical instruments.
The challenges for using PSPs include the steep learning curves for CAD/CAM software. Additional time and resources are required for image acquisition, processing, and PSP manufacturing. Collaboration between surgeons and engineers cannot be substituted, as they each contribute their unique expertise in anatomy, surgical techniques, biomechanics, materials science, manufacturing and quality control. Surgeons’ preferences for the surgical approach and available tools must be considered when designing the SAKs. Both surgeons and engineers must have a certain degree of fluency regarding digital tools, biomechanics and surgical anatomy to communicate effectively. PSPs, and SAKs may not fully accommodate the bone surfaces due to image segmentation errors in osteoporotic bone or from metal artefacts and minor adjustments, such as by manual removal of interfering callus may be necessary. Fortunately, we have not encountered a need to contour the PSPs in any of our patients.
Funding for this work was provided through research grants and public healthcare reimbursement. PSPs and may not be similarly reimbursable by all healthcare systems. The cost for PSPs was comparable to the commercially available locking plates in our localities but the price varied greatly between studies conducted in different regions. Debates persist regarding their cost-effectiveness compared to traditional methods. Logistical and regulatory barriers in some regions may limit their use. Hospitals equipped with a centralized point-of-care 3D printing office may enhance communications, logistics, and quality control, as subspeciality departments may lack the manpower and infrastructure needed to routinely implement these processes. 33
Our case series is limited by its retrospective design, absence of a control group, and small sample size, precluding meaningful statistical analysis. Future research should focus on a larger cohort, registries and comparative analyses to evaluate the long-term outcome, complications and cost-effectiveness of patient specific bone fixation implants. Advances in artificial intelligence, software, virtual mechanical simulations, manufacturing techniques and biomaterials should further streamline the process and lower costs.
Conclusion
We have illustrated the promising potential of patient specific plates in fracture surgery and our findings are in consensus with literature. The four integral components are 3D virtual preoperative planning, AM bone models, AM surgical guides and AM PSPs. The importance of collaborative workflows and adherence to regulatory standards is underscored and described. The technique is applicable to various fracture types and malunions, demonstrating high precision, reasonable efficiency, safety and good outcomes.
Footnotes
Acknowledgments
We wish to thank Dr G. Lamar Robert. PhD for reviewing and English proof of our paper. Generative AI (Microsoft Copilot) is used for English optimization, spelling and grammatical checking only. Generative AI is not used for content creation or online search for information.
Authors contribution
The authors confirm contribution to the paper as follows: study conception and design: R. Jarayabhand, C. Puncreobutr, B. Lohwongwatana; data collection: R. Jarayabhand, C. Jiamton Y, C Fang; analysis and interpretation of results: R. Jarayabhand, C. Jiamton Y, T Apivatthakakul; draft manuscript preparation: R. Jarayabhand, T Apivatthakakul. C Fang, All authors reviewed the results and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Christian Fang received research support from Johnson & Johnson Depuy Synthes, Koln 3D Medical, Brainlab, Agnovos, the AO Foundation and is speaker for Zimmer Biomet, Johnson and Johnson and Shareholder of Lifespans
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
The dataset for the current study is available from the corresponding author on reasonable request.
