Abstract
Background
Pelvic and acetabular fractures are associated with significant morbidity and mortality. The efficacy of 3D printing technology in improving surgical outcomes for these fractures has not been conclusively demonstrated.
Purposes
This study aimed to evaluate the efficacy of 3D simulation and printing in managing AO/OTA 61C and 62 B/62C pelvic and acetabular fractures.
Methods
A retrospective cohort study was conducted on patients aged >18 years with AO/OTA 61C and 62 B/62C fractures treated at a tertiary hospital between 2017 and 2022. Outcomes included postoperative complications, the need for total hip arthroplasty (THA), and quality of life measures, including the EuroQol Visual Analogue Scale (EQ-VAS), the EuroQol 5-Dimension 5-Level utility score (EQ-5D-5 L), and the Majeed pelvic score, assessed at 6 months, 1 year, and 2 years post-surgery.
Results
92 patients were analyzed, with 28 in the 3D printing group and 64 in the traditional treatment group. The mean age was 44.9 years. After adjusting for confounders, no significant differences were observed between the two groups in EQ-VAS, Majeed Pelvic Scores, or EQ-5D-5 L utility scores at 2 years (all p > .05). No significant differences were observed in the risk of THA or postoperative complications between the two groups.
Conclusions
The use of 3D printing for complex pelvic and acetabular fractures did not demonstrate an improvement in the primary outcomes of this study, including complications and the need for THA. This indicates that benefits of 3D printing observed in preoperative planning did not translate into improved clinical outcomes. Further research with larger, randomized trials is still warranted.
Introduction
The incidence of pelvic and acetabular fractures in adults has increased in the past decade.1,2 These fractures are often associated with higher morbidity and mortality owing to the anatomical position of the acetabulum, complex anatomical relationships of the pelvis with multiple vital organs and structures, and the traumatic nature of these injuries. 3 After stabilizing the vital signs, these fractures are typically surgically managed, primarily for achieving bone union, reducing the fracture, avoiding complications, and ultimately improving the patient’s quality of life.4,5 A comprehensive understanding of complex fracture patterns is essential to develop personalized surgical plans.
Surgeons can identify fracture fragments, conduct fracture mapping, and perform virtual simulations of complex fractures using a 3D simulation software in orthopedic preoperative planning. 6 The models built using this software can be used for preoperative planning, including pre-contouring plates and screw lengths.7–9 Compared with traditional surgical approaches, 3D printing technology in surgery improves intraoperative parameters, such as reducing surgical duration and blood transfusion and enhances patient outcomes.10,11 However, some studies reported conflicting results, showing no significant differences in intraoperative and functional recovery outcomes between the two approaches.12,13
Most current studies utilize the mirror-imaging technique which utilizes a 3D printed, healthy hemipelvis, for simulation.14–16 Although this approach is simple and effective, it has several limitations. Anatomical variations can occur between the hemipelvis and the fractured pelvis. Additionally, pelvic and acetabular fractures are often caused by trauma, and patients may have fractures on both sides, rendering the mirror imaging technique unsuitable for 3D printing and simulation. Furthermore, most 3D printing and fracture mapping studies have focused on simple pelvic and acetabular fractures.17,18 Data are limited on the long-term follow-up of patients undergoing 3D printing and fracture mapping, with most studies concentrating on intraoperative parameters and postoperative reduction quality.19,20
We compared the intraoperative parameters and long-term outcomes between traditional surgical methods and 3D printing techniques to evaluate the efficacy and efficiency of 3D printing for complicated fractures. We employed novel simulation algorithms developed in-house, such as mesh grid optimization and bone density evaluation, to determine the optimal plate and screw placement in complex pelvic or acetabular fractures. These techniques allow accurate fracture fragment placement and stable plate and screw positioning. This study aimed to assess the impact of virtual reduction simulation, bone density evaluation, and 3D printed models on surgical duration, intraoperative blood loss, functional recovery, and reduction quality in patients with complex comminuted pelvic and acetabular fractures.
Methods
Study design
This retrospective analysis of prospectively collected patient data included patients aged >18 years with complex pelvic or acetabular fractures admitted to our hospital, Taiwan, between January 2017 and May 2022. Patients had to choose between 3D printing and the traditional method, as the national insurance did not cover 3D printing, making it a self-funded option.
Ethics statement
The study was conducted in accordance with the principles of the Declaration of Helsinki. All study participants provided written informed consent, and the study protocol was approved by the local institutional review board of our University Hospital. (IRB number: CMUH108-REC3-144).
Patient selection
The inclusion criteria were (1) patients aged 18 years and older, (2) traumatic pelvic or acetabular fractures classified as 61C or 62B and above according to the 2018 AO Foundation/Orthopaedic Trauma Association (AO/OTA) classification, 21 and (3) a minimum of 48 months of postoperative follow-up.
The exclusion criteria were patients (1) with pathological or non-traumatic pelvic or acetabular fractures, (2) with incomplete follow-up details, (3) who declined surgical management, (4) whose treatment strategy included external fixation, and (5) with documented preoperative infections—such as urinary tract infections, wound infections, or systemic infections.
Surgical treatment and protocols
For the traditional group, the plates and screws were determined intraoperatively during surgery. In the 3D printing and fracture mapping group, the bone model was segmented and processed from computed tomography (CT) images using imaging software (Avizo 6.0, FEI, USA; Geomagic Design X, 3D Systems, USA). This process involved identifying fracture fragments, classifying fractures, and simulating initial fracture reduction. The software facilitated easier identification of bony fragments by color tagging.
Two novel simulation algorithms, the mesh grid optimization and bone density evaluation techniques, were applied. The mesh grid optimization technique allowed the segmentation, differentiation, and identification of each fracture fragment from the CT images. A mesh grid of the fracture fragments, pelvis, and acetabulum was overlaid onto the surfaces, and the algorithms enabled automatic grid comparison to identify the best-matched grids and fragments (Figure 1(a)). Grid surface information, such as edge alignment, curvature alignment, and grid fit, was analyzed; the selected best fit model aided in optimal reduction identification. Shows (a) the optimization of mesh grid to identify comminuted fragments and simulate optimal reduction; (b) pelvis model with the best-reduced fit; (c) the evaluation of bone density converted using Hounsfield unit of CT for evaluating the placement of screws and plates.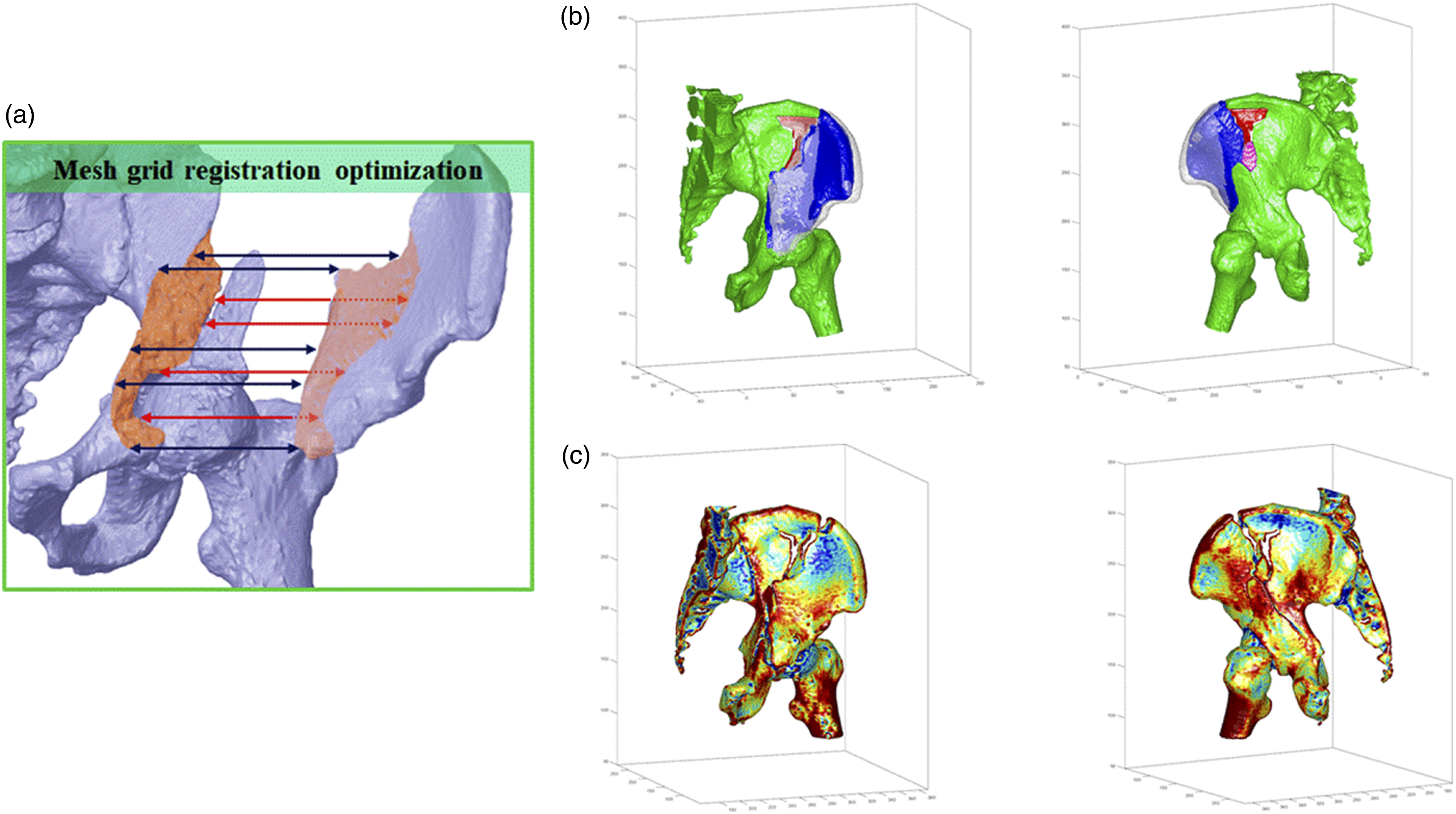
An in-house algorithm was developed to evaluate bone density using the Hounsfield units of individual pixels from CT images (Figure 1(b) and (c)) to determine the proper placement of screws and plates and reduce the risk of screw loosening. 22 During the simulation, screws and plates were placed in high bone density areas. Our algorithm analyzed the screw-bone contact surface area mass and compared it with our established screw-bone physiological database to determine the optimal placement. Additionally, we analyzed the pull-out stiffness of the screws at specific positions using a finite element mechanical analysis.
The reduced model was shown as a color-mapping module before the 3D printing of a 1:1 pelvic model (Figure 2(a)). The model was printed using a 3D printer (Object 500 Connex 3; Stratasys, USA) with a heat-resistant material for subsequent sterilization. These models were used for preoperative planning, allowing for the pre-contouring of plates and determining simulation-based appropriate screw sizes (Figure 2(b)). The model was also used for team briefings and communicating with patients and their family members. Subsequently, the models and surgical equipment were sterilized. Shows (a) the pre- and post-reduction of pelvis fractures; (b) the preoperative determination and pre-contouring of plates and screws using a 3D printing model.
Figure 3 illustrates the comprehensive management of a 27-year-old male with a complex pelvic fracture, using these techniques in conjunction with minimally invasive surgery. Shows (a) (b) a 27-year-old male with a left pelvic and acetabular fracture with multi-fragmentary anterior column involvement; (c) Post-reduction simulation was performed using our grid optimization algorithm; (d) hemi-pelvis was reduced using 3D printing; the fracture fault lines and placement of the plates and screws were engrooved onto the model; (e) Our bone density evaluation allowed us to place multiple screws at areas with the thickest cortex; (f) Minimal invasive surgery with pararectus approach anteriorly and direct posterior approach posteriorly was used on this patient. The length of the incision was approximately about 8.5 cm (the length of a credit card) on both side.
Drainage tubes were placed for 48 h postoperatively. Sensation, motor function, and lower limb circulation were regularly monitored during hospitalization. A postoperative radiograph was obtained 24 h after the surgery, and the patient was discharged with oral medications upon stabilization. Stable conditions were identified by stable vital signs, clean wounds, adequate analgesia, and suitability for home care.
The patient was scheduled for regular outpatient department follow-up with routine radiography and CT examinations and at 1, 3, 6, 12, 24, 36, and 48 months after surgery. Simultaneously, patients were asked to complete questionnaires to assess their recovery status and quality of life. Routine rehabilitation exercises were prescribed to promote the recovery of pelvic function. The results section presents the recorded complications and their management.
Data collection
We collected data on demographic and clinical variables, including age, sex, and comorbidities such as preoperative transarterial embolization (TAE), diabetes mellitus (DM), chronic kidney disease (CKD), and hypertensive cardiovascular disease (HCVD). We also recorded characteristics of the injury, including the Injury Severity Score (ISS) and AO/OTA classification, as well as intraoperative parameters, including the amount of blood loss and surgical duration. Trauma severity was evaluated using the ISS, an international scale modified from the Abbreviated Injury Scale, 23 which divides the body into six parts: the head, neck, chest, abdomen, limbs, and body surfaces. The total ISS was obtained by squaring and summing the highest scores for each part. An ISS >15 points is considered major trauma; therefore, this study used an ISS of 16 as the critical value. The amount of blood loss and surgical duration were recorded as intraoperative parameters. Subsequent analysis utilized a cutoff of 500 mL of blood loss and 4 h of surgical duration. The 500-cc threshold was determined based on the American College of Surgeons Advanced Trauma Life Support classification of hemorrhage severity, which indicates that a 10-15% blood volume loss (500-750 cc) in a healthy 70 kg adult leads to physiological changes. The 4-h surgical duration cutoff was based on a review by Cheng et al., which indicated increased postoperative complications with surgeries lasting 2-4 hours. 9
Outcomes
Postoperative complication rates, the need for total hip arthroplasty (THA), and quality of life measures—including the EQ-VAS and EQ-5D-5 L utility scores—as well as functional outcomes assessed using the Majeed pelvic score, were evaluated as primary outcomes. Patients completed the Mandarin version of the questionnaire to assess these scores. The EQ-VAS and EQ-5D-5 L evaluated perceived health status, while an orthopedic surgeon assessed the Majeed pelvic score to evaluate functional outcomes. A cutoff age of 60 years was used in the subsequent analysis due to the well-documented significant increase in mortality and morbidity risks beyond this age.
Statistical analysis
We presented the numbers and percentages of categorical variables, including sex, comorbidities, 2018 AO/OTA classification, complications, ISS, blood loss, and surgical duration, grouped using 3D printing and traditional methods. Continuous variables, including age, ISS score, blood loss, and surgical duration, were reported as means and standard deviations (SD) or medians with interquartile ranges (IQR) based on data distribution (normal or skewed). Differences in categorical variables between the 3D printing and non-3D printing groups were compared using the chi-square test and Fisher’s exact test, whereas independent t-tests or Mann–Whitney U tests were used to compare continuous variables between the two groups.
Outcomes at 6-month and 1- and 2-year follow-up, including EQ-VAS, Majeed pelvic score, and EQ-5D-5 L, were presented as mean ± SD. The mean differences in outcomes between the two groups estimated using linear regression analyses stratified by the AO/OTA classification, age, ISS, blood loss, and surgical time, were used to determine their impact on pelvic and acetabular fractures separately.
Complications, including the need for THA, were also evaluated using logistic regression analysis, and the odds ratio (OR) and 95% confidence interval (CI) for the 3D printing group compared with the traditional group were reported. All analyses were performed using IBM SPSS software (version 22.0; IBM Corp., Armonk, NY, USA), and p-values <0.05 for two-tailed tests were considered statistically significant.
Results
Patient characteristics
Demographic characteristic of patients compared between receiving 3D printing model and traditional approaches.
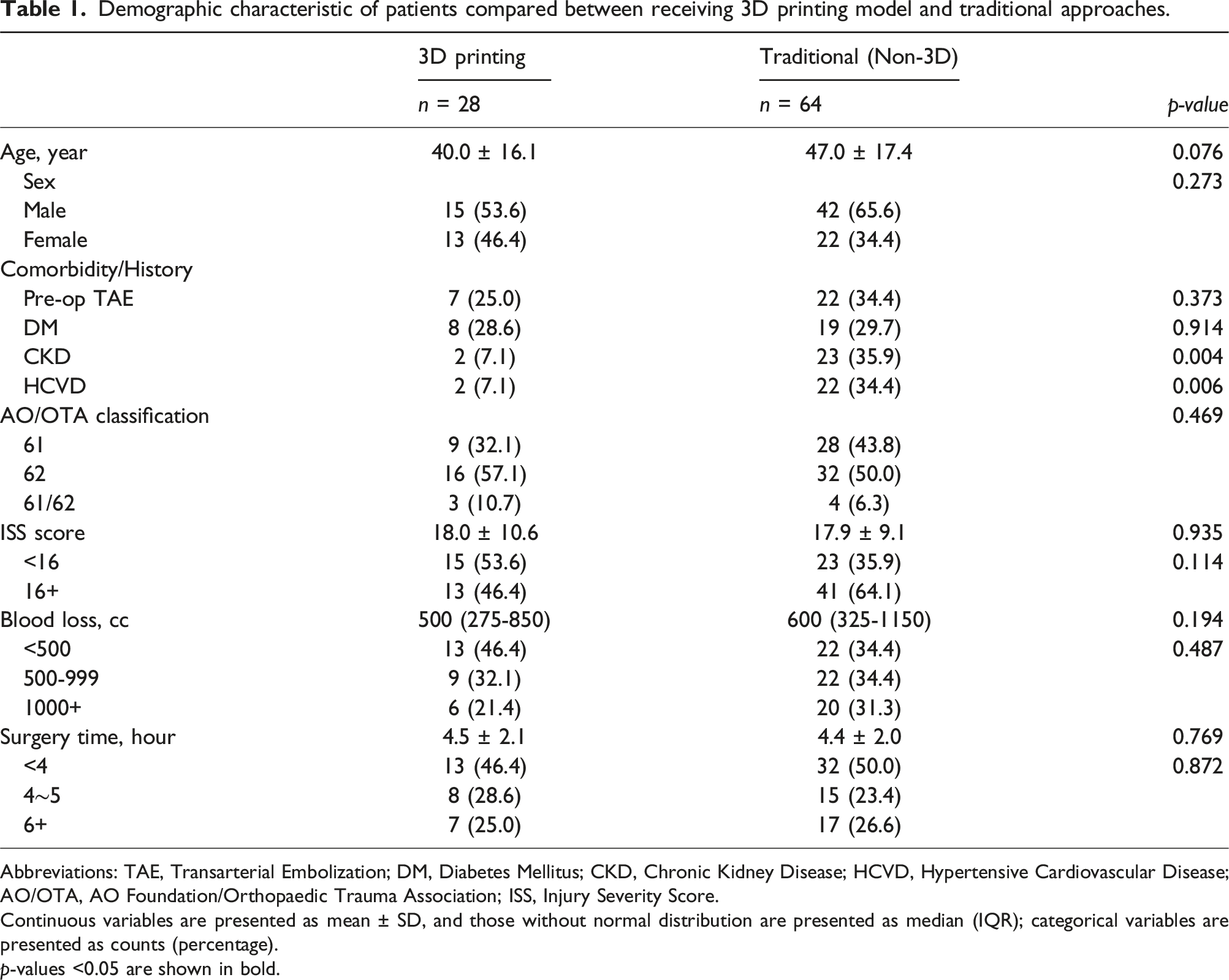
Abbreviations: TAE, Transarterial Embolization; DM, Diabetes Mellitus; CKD, Chronic Kidney Disease; HCVD, Hypertensive Cardiovascular Disease; AO/OTA, AO Foundation/Orthopaedic Trauma Association; ISS, Injury Severity Score.
Continuous variables are presented as mean ± SD, and those without normal distribution are presented as median (IQR); categorical variables are presented as counts (percentage).
p-values <0.05 are shown in bold.
Clinical outcomes
The clinical outcomes of patients comparing between receiving 3D printing-model and traditional approaches.

Abbreviation: EQ-VAS, EuroQol Visual Analogue Scale; EQ-5D-5 L, EuroQol 5-Dimension 5-Level; THA, Total Hip Arthroplasty.
Continuous variables are presented as mean ± SD [valid n]; categorical variables are presented as counts (percentage).
p-values <0.05 are shown in bold.
Although the traditional group showed a higher numerical rate of postoperative THA compared to the 3D printing group (17.2% vs 3.6%), the difference was not statistically significant (p = .098). In the traditional group, postoperative complications included five cases of neurological injury, one vascular injury, one instance of screw loosening, and one bladder injury. (Table 2)
Estimated difference of postoperative outcomes compared between patients receiving 3D printing model and traditional approaches by linear regression.

Abbreviation: EQ-VAS, EuroQol Visual Analogue Scale; EQ-5D-5 L, EuroQol 5-Dimension 5-Level; CI, Confidence Interval; AO/OTA, AO Foundation/Orthopaedic Trauma Association; ISS, Injury Severity Score.
Estimates are the mean differences between 3D printing and traditional approach, that is mean 3D -mean traditional .
aAdjusting for CKD and HCVD, the significant covariates identified in the univariate analysis.
bOnly AO/OTA classifications 61 and 62 were analyzed due to the limit number of patients in the 61/62 classification.
Risk estimate for THA and any complication compared by treated approaches and AO/OTA classification.

Abbreviation: OR, Odds Ratio; aOR, adjusted Odds Ratio; CI, Confidence Interval; THA, Total Hip Arthroplasty; AO/OTA, AO Foundation/Orthopaedic Trauma Association.
aIncluding THA, neurological injury, vascular injury, screw loosening, and bladder injury.
bAdjusting for CKD and HCVD, the significant covariates identified in the univariate analysis.
cOnly AO/OTA classifications 61 and 62 were analyzed due to the limit number of patients in the 61/62 classification.
Discussion
The incidence of pelvic and acetabular fractures has been increasing over the past decade because of increasing cases of low- and high-energy injuries, such as falls and road traffic accidents.2,24 Current publications on 3D printing for these fractures have shown conflicting results regarding improvements in intra-and postoperative outcomes. 25 With these studies as foundation, the current study included patients with complex pelvic or acetabular fractures and had a minimum follow-up period of 48 months. Additionally, our study is the first to apply novel simulation algorithms such as mesh grid optimization and bone density evaluation in 3D simulations and printing for complex pelvic and acetabular fractures.
Specifically, our intraoperative results showed no statistically significant differences in the ISS, blood loss, and surgical duration between the groups. However, the additional bone density evaluation conducted in this study allowed for strategic hardware placement, enhancing screw fixation by targeting higher-density areas, which could contribute to the reduction of screw loosening, especially in cases with poor bone quality. Further stratification revealed that intraoperative parameters did not affect the postoperative outcomes, except in the group exceeding surgical durations of 6 hours.
Recent advancements in 3D printing have demonstrated its potential to refine surgical planning and minimize intraoperative variability. 26 Dust et al. reported that 3D-printed models significantly improve inter- and intra-rater reliability in classifying fracture patterns, which is critical for accurate preoperative planning. 27 Similarly, Betancourt et al. highlighted that 3D-printed anatomical models enhance surgical planning by optimizing hardware placement and reducing errors. Integrating these quantitative metrics into 3D printing-assisted workflows could standardize fracture reduction protocols and improve procedural consistency, ultimately enhancing patient outcomes. 28
Our analysis revealed no statistically or clinically significant differences in the need for THA and postoperative complications between the 3D printing group and the traditional group, despite previous studies suggesting that surgeries employing 3D printing technology, including revision total hip and knee arthroplasty, are associated with lower complication rates compared to traditional methods.29,30 This may be attributed to the limited sample size. Future studies with larger cohorts are warranted to confirm these findings
Downey et al. found that 3D models did not alter the surgeon’s initial approach. However, usage of 3D models significantly improved the classification of fractures compared with usage of CT images alone.19,31 The fracture classification accuracy was found to be 11% with X-ray images alone and 30% with CT images. 32 Our preoperative simulations also enhanced the visualization of fracture locations, enabling minimally invasive approaches to reduce complex comminuted fractures. This resulted in a postoperative wound size approximately equal to the size of the credit card with optimal patient satisfaction and minimal postoperative analgesia usage. Traditional approaches for posterior column fractures, such as Kocher–Langenbeck and modified Stoppa, often increased the complication rates and duration of hospital stays. By utilizing our mesh grid algorithm, we achieved improved fragment matching, which is critical for comminuted fractures. This algorithm helps identify key fragments and determine their optimal positioning, thereby enhancing surgical planning accuracy.
We applied a modified version of the fracture technique proposed by Yang et al. to our 3D model.33–35 After reduction and alignment with landmarks, such as the sciatic notch and ischial tuberosity, fracture fault lines were identified and traced onto pelvic models. In future simulations, including additional details such as muscle origins, insertions, and nearby vascular and neural structures could further enhance surgical planning by aiding the identification of critical soft tissues. Although multiple publications use mirroring as a reduction technique, our study did not directly compare mirroring with our non-mirroring simulation. However, we hypothesized that non-mirroring provides better reduction quality because of the asymmetry of some pelvis.
Although our study did not demonstrate clear advantages in clinical outcomes from the use of 3D printing for pelvic and acetabular fractures, the most recent literature has reported potential benefits. As highlighted by Moorthy et al., 3D printing-assisted surgeries maintain safety while achieving perioperative outcomes comparable to conventional methods, even in early adoption phases. 36 Furthermore, Si et al. point out significant reductions in operative time, intraoperative blood loss, and fluoroscopy usage, enhancing procedural efficiency and patient safety. 37 The integration of 3D printing with robotic-assisted surgery, as explored by Jing et al., may further improve precision and safety, potentially leading to notable advancements in minimizing intraoperative complications and optimizing screw placement. 38 In summary, the advancements in 3D printing enhance surgical precision, yet the long-term benefits remain unclear and require further rigorous investigation. This technology’s ongoing evolution signals its potential to transform orthopedic surgery, emphasizing the need for targeted research to fully exploit its capabilities in complex fracture management.
Strengths and limitations
This study has several notable strengths. It is the first to integrate novel and existing simulation techniques, such as mesh grid optimization and bone density evaluation, in the treatment of complex comminuted fractures. Theoretically, the use of 3D printing offers trauma surgeons an additional tool to enhance surgical precision by improving their understanding of bony anatomy, fracture fragments, and the quality of reducible or available bone—factors that are critical for optimizing treatment outcomes. However, to date, few real-world studies have explored this approach. Our study highlights the role of advanced simulation algorithms in enhancing fracture visualization and optimizing surgical planning. Furthermore, the inclusion of patients with at least 48 months of postoperative follow-up provided robust, long-term data with a high follow-up rate, adding to the reliability of the findings.
However, this study has several limitations. First, it is not a randomized controlled trial (RCT). Case-control studies were chosen due to ethical concerns regarding randomization, as highlighted by Cao et al.39,40 Although parameters such as operation duration, blood loss, and quality of life outcomes were assessed, the evaluation of reduction quality on CT scans—a critical outcome in this context—was not included. Additionally, pelvic and acetabular fractures were not studied separately, and the use of different approaches for these fracture types might have confounded the analysis. Importantly, the two study groups were developed based on patient choice influenced by expense, introducing a significant potential bias in patient-related outcomes. All patients in the 3D-printed group were aware of their cohort assignment, which may have introduced selection bias. Patients willing to pay for 3D printing might represent a subset with more complex fractures or higher socioeconomic status, potentially impacting the outcomes. Furthermore, confounding variables, such as the degree of comminution of the articular surface and initial displacement on CT scans, were not controlled for, which could have influenced the results. Lastly, this is also a single-center study, which may limit the external validity of the findings. Consequently, the results may not be generalizable to other settings or patient populations. Future research should focus on multicenter trials with rigorous designs, larger and more diverse populations, and comprehensive data collection. Such studies should also evaluate the effectiveness of 3D printing technology among surgeons with varying levels of experience to better understand its broader applicability.
Conclusions
Our study evaluates the integration of 3D printing technology in the management of complex pelvic and acetabular fractures. While this technology enhances preoperative visualization and could potentially improve the efficiency and precision of surgical procedures, our findings did not demonstrate consistent or clinically meaningful improvements in outcomes compared to traditional methods. This suggests that while 3D printing is promising, especially in complex cases where detailed anatomical understanding is crucial, it is still in the early phases of development and validation. The benefits observed in preoperative planning did not translate into improved clinical outcomes in our study. Consequently, further research, including randomized controlled trials that carefully consider potential covariates, is critical to fully understand and validate the impact of 3D printing technology in trauma care.
Footnotes
Acknowledgments
We would like to thank China Medical University Hospital, Taichung, Taiwan for their support in this work.
Authors’ contributions
Conceptualization; T.L.L., C.T.S., Y.C.F., C.H.T., Data curation; C.H.T., Formal Analysis; K.X.V.L., T.L.L., C.H.T., Funding acquisition; C.T.H., Y.W.C., Y.C.F., H.T.C, Investigation; T.L.L., Y.C.F., C.H.T., Methodology; C.T.S., C.H.T., Project administration; H.T.C, Resources; Y.W.C., Y.C.F., H.T.C., Software; C.T.S., Supervision; Y.C.F., H.T.C., C.H., Validation; T.L.L., C.T.S., Y.W.C., H.T.C., C.H.T., Visualization; C.T.S., C.H.T., Writing – original draft, Writing – review & editing; K.X.V.L, C.H.T.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
