Abstract
Introduction
Ankle fracture with syndesmotic injury and deltoid ligament (DL) rupture results in severe instability. Traditionally, the syndesmosis is stabilized with distal tibiofibular screw fixation.1,2 Historical evidence suggests that after fibular reduction and tibiofibular fixation, DL repair is unnecessary, as the ligament heals indirectly with good clinical results.3–7 However, distal tibiofibular screw fixation has several deficiencies. Firstly, the tibiofibular screws can fail and break after long-term loading; hence, some surgeons still prefer to remove syndesmotic screws in clinical practice,8,9 although a second operation consumes medical resources and increases the costs and surgical risks. In addition, early weight-bearing after distal tibiofibular screw fixation often causes screw breakage, increasing the difficulty of removing the internal fixation. Secondly, the patient should not weight-bear for at least 6 weeks after tibiofibular fixation,8,9 which is not conducive to functional exercise and rehabilitation, and thus may lead to joint stiffness, muscle atrophy, and osteoporosis. Thirdly, if open reduction of the syndesmosis is not performed, malreduction and screw breakage can occur during placement of the tibiofibular screws.10–13
In clinical practice, we found that after fibular fixation in Danis-Weber type C ankle fracture with both syndesmotic and DL injury, the outcome of simple DL augmentation was similar to that of tibiofibular fixation. Furthermore, studies have reported that DL repair without tibiofibular fixation achieves satisfactory clinical results.14–17 Since 2014, we have treated more than 50 such ankle fractures with DL augmentation instead of tibiofibular fixation, and achieved satisfactory clinical outcomes at an average follow-up of 24 ± 12 months (range 6–40 months). 18 Our research team has pioneered the use of deep DL augmentation as a replacement for tibiofibular screw fixation in Weber C injuries. 18
Previously, DL repair often merely involved suturing the superficial layer, or using suture anchors to link the broken ends of the medial malleolus and the superficial layer of the ligament,19,20 resulting in limited effectiveness. The deep layer of the DL cannot easily be directly repaired due to its horizontal course within the narrow space between the medial malleolus and talus. The DL augmentation technique places the suture anchor on the talus side, with a hole drilled through the medial malleolus; this achieves a direct bone-to-bone connection between the talus and medial malleolus, which significantly increases the strength of the repair.18,21
Whether DL augmentation provides adequate strength to replicate the sufficient ankle stability provided by tibiofibular fixation depends on the technical execution, and there are many issues that require further investigation. The principle behind this technique is to mimic the deep anatomical course of the DL, yet there are notable differences between the suture anchor and the actual ligament tissue.21,22 The insertion point of the ligament on the talus side is a broad and flat area, whereas the suture anchor insertion point resembles a narrowed point. Although the choice of insertion point is likely to influence the repair strength, this issue has not yet been investigated. 23 Previously, our research team often selected the center of the insertion footprint on the talus side for suture anchor placement during clinical procedures. However, our preliminary biomechanical experiments revealed that more anterior placement of the anchor may offer better resistance to talus external rotation and anterior dislocation.
In the present study, we aimed to compare the external rotation stability between different insertion sites of the suture anchor in DL augmentation in a cadaveric ankle fracture model.
Materials and methods
This study evaluated formalin-fixed unpaired cadaveric lower limbs disarticulated at the knee joint (from the tibial plateau to the foot). The donors had no history of musculoskeletal injury or operation to the lower extremity. The specimens were prepared by removing the soft tissues surrounding the ankle and lower leg while preserving the ligaments and interosseous membrane. The forefoot was then removed, while the proximal tibiofibular joint was unconstrained to facilitate normal motion of the fibula relative to the tibia. The subtalar joint was solidly stabilized on each specimen using Kirschner wires (K-wires) to prevent additional subtalar joint movement. An ankle injury model with both syndesmotic disruption and DL injury was constructed by sectioning the inferior tibiofibular ligament (including the anterior inferior tibiofibular ligament, posterior inferior tibiofibular ligament, and interosseous ligament), the tibiofibular interosseous membrane up to 6 cm proximal to the tibial plafond, and the superficial and deep DL. The fibular was left intact to simulate fibular fracture fixation (Figure 1). Fixation of the fibula and augmentation of the DL.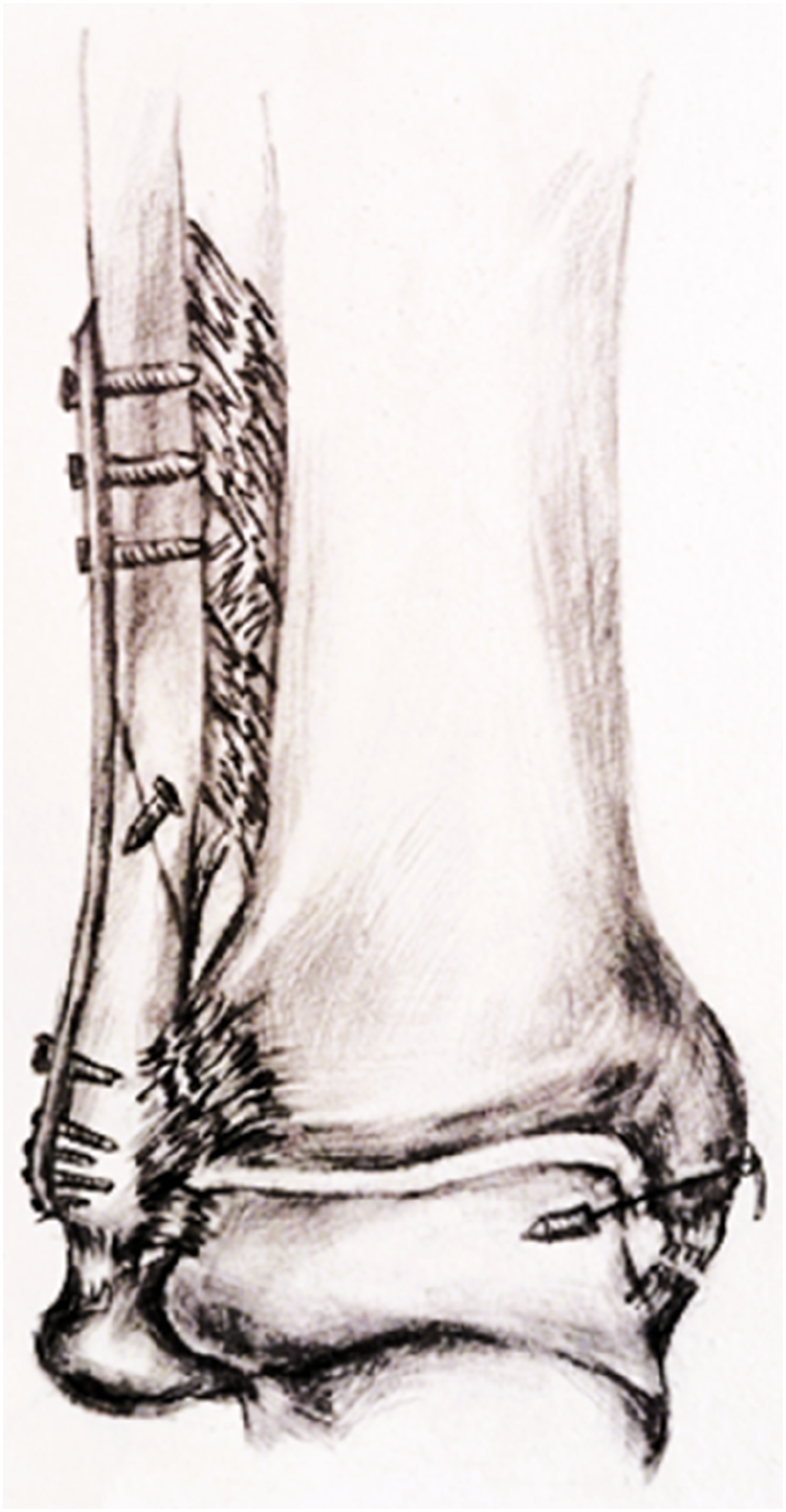
A 2.5-mm K-wire was used to drill a hole at the two test points on the talus, parallel to the talus dome, followed by the placement of a 5.0-mm suture anchor (Tianxing, Beijing, China). A 2.0-mm K-wire was used to drill two oblique holes through the medial malleolus (one at the inter-colliculus groove and one at the posterior colliculus) in the direction of DL insertion. The four wires connected to the anchor were then drawn out through the two holes, with two wires in each hole. Finally, the suture was secured on the medial tibial cortex at the ankle in an inversion position, while the superficial DL was sutured directly if possible (Figure 1).
The following two suture anchor positions were tested. In the experimental group, the anchor was positioned anterior to the perpendicular line at the position of the holes drilled through the medial malleolus, and in the anterior one-third of the deep layer of the DL insertion footprint. The suture anchor was oriented from the anterior-caudad direction to the posterior-cranial direction. In the control group, the anchor was placed in the center of the insertion footprint of the DL. The specimens in the two groups were tested in random order.
In the external rotation stress test, the tibial plateau was fixed solidly by K-wires on the custom-built clamps fastened to a hydraulic loading frame (ElectroPuls E10000; Instron, Norwood, MA, USA). The hindfoot was secured to custom-built clamps in a neutral position using K-wires (Figure 2). A compressive load of 150 N was applied along the long axis of the tibia in 2 min and maintained throughout each trial. The tibia was internally rotated at a rate of 1°/second until a resistive moment of 4.0 N·m was reached, in accordance with a previously reported non-destructive protocol.24–27 External rotation test.
Vernier calipers were used to measure the following parameters at the beginning and end state of each trial: medial clear space (MCS, the distance between the talus and the medial malleolus at 5 mm caudal to the superior margin of the talus), tibiofibular clear space (TCS, the distance between the tibia and the internal margin of the fibula at 1 cm cranial to the tibial articular surface). All measurements were carried out twice by an experienced researcher and the average values were used in the analyses.
The parameters were compared between the two groups using the Wilcoxon rank-sum test for paired samples. p < .05 was taken to indicate statistical significance in all tests. All analyses were performed using IBM SPSS Statistics (IBM Corp., Armonk, NY, USA).
Results
Comparison of measurements before and after the external rotation tests in the two groups.

Values expressed as median (quartile). Significant p values are typed in bold font.
Comparison of measurements in the two groups in external rotation tests.
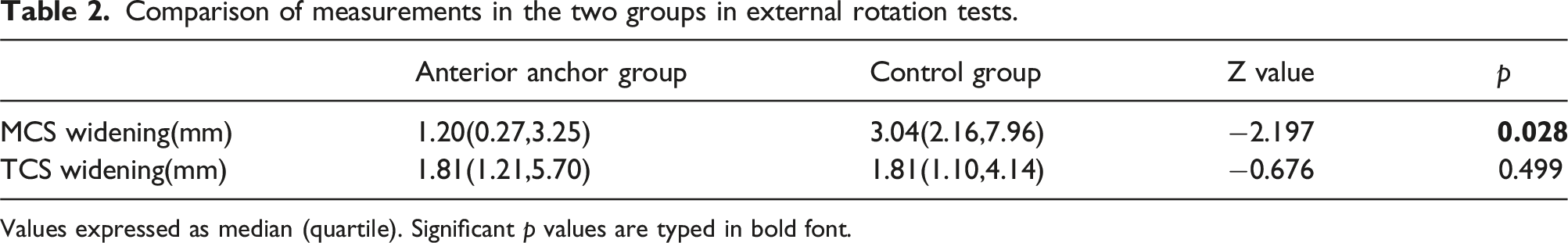
Values expressed as median (quartile). Significant p values are typed in bold font.
Discussion
Currently, many surgeons prefer to use screw fixation to repair an acute ruptured syndesmosis, although a suture-button is also available.28–32 However, distal tibiofibular screw fixation has many defects, such as the need for a second operation, avoidance of postoperative weight-bearing,8,9 risk of malreduction, and risk of screw breakage.10–13 We have introduced a approach for mortis stability using deep DL augmentation and superficial DL repair that aims to provide medial column stability, indirectly reduce the syndesmosis, and achieve primary stability of the ankle. 18
In the preliminary experiment, we observed that an anterior anchor position and a knot tied in the varus position led to less tibiofibular separation (Figure 3) and less anterior dislocation (Figure 4). Similarly, in the present study, the widening of the MCS was significantly smaller in the experimental group than in the control group. This suggests that anterior placement of the suture anchor may provide better resistance to external rotation forces and anterior dislocation tendencies of the talus than the traditional method used in the control group. Additionally, during the external rotation experiment, all MCSs and TCSs increased, except for the MCS of the experimental group, which remained statistically unchanged. This further highlights the superiority of anterior placement of the suture anchor in resisting external rotation forces. (a) Posterior anchor position with the knot tied in the neutral position. (b) Posterior anchor position with the knot tied in the varus position to make it tighter. (c) Anterior anchor position with the knot tied in the varus position, leading to less tibiofibular separation.
The potential mechanism by which anterior placement of the suture anchor enhances the resistance to external rotation may involve the following reasons. Firstly, Weber type C fractures and some Weber type B fractures with concomitant tibiofibular injuries primarily endure forces of eversion and external rotation, which means that the anterior part of the DL that bears tension during external rotation may become more damaged. Therefore, the anterior part of the DL should be the primary focus of repair. Lee et al. 33 found a significant difference between unstable lateral malleolus fractures and stable fractures solely in anterior DL injuries on preoperative MRI. They believed that for simple lateral malleolus fractures accompanied by DL injuries, repairing only the “anterior DL” achieves adequate valgus stability. 33 Secondly, ankle fractures occur with posterolateral dislocations, where the distal fibula and the talus move posteriorly. After fibular fixation, movement in the posterolateral dislocation is restricted. Therefore, the talus, which is wider anteriorly and narrower posteriorly, has to move anteriorly. Consequently, a force is required to limit this anterior movement, which justifies the overcorrected anterior positioning of the suture anchor.
External rotation force is a common and significant force in Weber type C ankle fractures with both tibiofibular and DL injuries, and the commonly used eversion and external rotation stress test also applies external rotation force to the foot; therefore, it is crucial to restore the ability of the ankle to resist external rotation. The deep layer of the DL itself plays a crucial role in resisting external rotation and lateral shift of the talus. Therefore, it is of great clinical significance to increase the resistance to external rotation by placing the suture anchor anteriorly during the repair of the DL.
In the preliminary experiment, we found that the widening of the TCS was smaller in the experimental group than in the control group (Figure 3). However, in the present study, there was no significant difference between the two groups in the widening of the TCS during the rotation tests, suggesting that the two methods were equally effective in resisting tibiofibular separation. The reasons that the experimental results did not meet our expectations may be due to the small number of experimental cases and deviations in the experimental process and measurements, which did not reflect statistical differences. It is possible for further testing of a large sample size to reveal a marked difference between the two groups in the widening of the TCS. (a) Posterior anchor position with the knot tied in the neutral position. (b) Posterior anchor position with the knot tied in the varus position to make it tighter. (c) Anterior anchor position with the knot tied in the varus position, leading to less anterior dislocation of the talus.
This study provides biomechanical evidence of the advantage of anterior placement of suture anchors. Presently, there is a lack of research to explain the phenomena discovered in this study. Although further clinical investigation is essential, the present study provides valuable information that may be used to select the optimal suture anchor position. However, our study also has some limitations. Due to the limited number of available specimens, we used formalin-fixed rather than fresh-frozen specimens. Nevertheless, a previous study showed no significant difference between the experimental data obtained from fresh and preserved specimens. 34 The present study also evaluated older specimens that had osteoporosis, which meant that the suture anchor was prone to cutting through the bone, affecting the experimental accuracy and potentially causing measurement errors. Finally, the small sample size may have affected the accuracy of our results. The present findings require validation in future biomechanical and clinical studies with larger sample sizes.
In conclusion, the present study suggests that anterior placement of the suture anchor in DL augmentation may offer improved stability against external rotation forces in ankle fractures with syndesmotic and DL injuries. The present findings provide biomechanical evidence supporting the potential advantages of this technique.
Footnotes
Acknowledgements
Author contributions
All authors contributed equal to this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This study was supported by the Capital’s Funds for Health Improvement and Research (Code 2022-2-1121) and Beijing Hospitals Authority Clinical medicine Development of special funding support (Code: ZLRK202311).
Ethical statement
Data Availability Statement
All datasets are deposited in additional supporting files.
