Abstract
Background
The study aims to evaluate the influence of tranexamic acid (TXA) on clinical outcomes in patients with elbow stiffness undergoing elbow arthrolysis.
Methods
A systematic review of records up to December 2024 was conducted to screen research examining the role of TXA in patients with elbow stiffness undergoing elbow arthrolysis. The primary outcomes of interest included blood transfusion, hematoma formation, operative time, postoperative pain measured by the Mayo Elbow Performance Score (MEPS), Visual Analog Scale (VAS), total blood loss, changes in hemoglobin, drain output, and complications.
Results
Following an extensive search of relevant databases, a total of seven studies involving 995 patients (491 in the TXA group and 504 in the non-TXA group) undergoing elbow arthrolysis were included. The pooled analysis showed that TXA administration was associated with a significant reduction in total drain output (MD = −55.34, 95% CI: −80.67 to −30.02, p = .0001) and blood loss (MD = −39.07, 95% CI: −69.71 to −8.43, p = .01) compared to non-TXA group. Furthermore, patients treated with TXA had higher postoperative hemoglobin levels (MD = 11.73, 95% CI: 8.74 to 14.73, p = .00001). No significant differences were observed in operative time, tourniquet time, or functional outcomes as measured by MEPS, VAS, or range of motion (ROM). The pooled results indicated that TXA was associated with a significant reduction in hematoma formation (RR = 0.43, 95% CI: 0.19 to 0.97, p = .04) compared to the non-TXA group. However, no significant differences were found in other complications.
Conclusion
The perioperative use of tranexamic acid is an effective strategy to reduce postoperative drainage in elbow arthrolysis while preserving joint function.
Introduction
Elbow stiffness is a prevalent and debilitating orthopedic condition that often primarily results from distal humeral fractures or elbow dislocations. 1 These traumatic injuries disrupt the normal anatomy of the elbow joint, leading to restricted motion, pain, and functional impairments. As a consequence, patients experience significant limitations in performing daily activities such as lifting, eating, and dressing, which can profoundly impact their quality of life.1,2 The onset of heterotopic ossification (HO) or fibrous adhesions 3 following surgery may further exacerbate the loss of range of motion (ROM), thereby contributing to long-term disability. Thus, elbow stiffness not only impairs patients' physical health but also imposes a psychological burden, with many individuals experiencing distress due to the prolonged nature of the disability.
Recovery from elbow stiffness is often protracted, requiring intensive rehabilitation aimed at restoring joint mobility and function. Patients typically undergo extended rehabilitation periods that involve painful and time-consuming physical therapy. This prolonged recovery process can delay a patient’s return to work or recreational activities, thus affecting overall well-being and placing substantial strain on healthcare resources. Elbow arthrolysis, a primary surgical treatment for elbow stiffness, is performed to release contracted soft tissues and enhance joint mobility. However, this procedure presents certain challenges. A primary concern following elbow arthrolysis is excessive postoperative bleeding, which can complicate recovery and impede rehabilitation. Additional common postoperative complications, including drainage, swelling, and inflammation, may further hinder the healing process. These complications often lead to extended hospital stays, increased healthcare costs, and prolonged rehabilitation, highlighting the need for effective strategies to mitigate these issues and facilitate a more rapid recovery.
Tranexamic acid (TXA), a synthetic derivative of the amino acid lysine, has emerged as a promising agent for managing bleeding and enhancing postoperative recovery. 4 The efficacy of TXA in reducing blood loss and improving surgical outcomes has been well-established across various specialties, particularly in high-risk procedures such as hip and knee arthroplasty. 5 Its use has been endorsed by international guidelines,6–8 including those from the American College of Surgeons and the European Society of Anaesthesiology, both of which recommend TXA in surgeries with high bleeding risks. Many bone-related diseases9–11 have been shown to be associated with immune inflammation, and anti-inflammatory treatments12–15 have demonstrated positive effects in managing these conditions. Interestingly, emerging studies suggest that TXA may possess additional anti-inflammatory effects 3 that extend beyond its role in hemostasis, including the reduction of postoperative swelling and pain.
In recent years, the use of TXA in elbow arthrolysis has garnered increasing attention. A previous randomized controlled trial (RCT) 16 demonstrated that intravenous TXA significantly reduced postoperative drainage and pain in patients undergoing open elbow arthrolysis, while also improving early joint function. A subsequent study 17 examined the effects of intravenous TXA on drainage and functional outcomes following traumatic elbow stiffness release. In addition to improving short-term recovery, TXA may play a role in addressing the long-term complications of elbow arthrolysis, such as the recurrence of HO. A recent study 18 introduced a protocol for a RCT investigating the potential of TXA to reduce the recurrence of HO following open elbow arthrolysis. Furthermore, a recent RCT by 19 evaluated the effects of TXA on elbow swelling and early functional recovery after arthroscopic arthrolysis.
This meta-analysis aims to further clarify the role of TXA in reducing postoperative drainage, preventing complications, and preserving joint function in patients undergoing elbow arthrolysis for elbow stiffness. By synthesizing the results of multiple studies, this analysis will provide evidence-based insights to guide clinical practice, thereby helping to optimize recovery and improve patient outcomes following this common orthopedic procedure.
Materials and methods
Search strategy and study selection
A systematic review of the literature was performed following the guidelines outlined in the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement 20 and the AMSTAR (Assessing the Methodological Quality of Systematic Reviews) checklist. 21 Any disagreements encountered during the process were resolved by consensus or through the judgment of an arbitrator. 22 Informed consent and ethical approval were not required, as all data used in current study were extracted from published articles.
A comprehensive search was performed across three medical databases—PubMed, Embase, and Cochrane Library—from inception through December 31, 2024. The search strategy was developed a priori and executed using the following search string: (“tranexamic acid” OR “TXA” OR “elbow”) AND (“elbow” OR “elbow arthrolysis” OR “open arthrolysis” OR “elbow stiffness”). No restrictions were applied with regard to language or publication date. To ensure the comprehensiveness of the search strategy, a senior librarian was consulted. In addition, keyword-based searches were supplemented by hand screening of the bibliographies of the retrieved articles.
Inclusion and exclusion criteria
Studies were included if they met the following inclusion criteria: (1) Participants were patients with elbow stiffness; (2) The study evaluated the efficacy of TXA in the treatment of elbow stiffness; (3) The study included a non-TXA control group for comparison; (4) Sufficient information was provided to obtain the outcome estimates of interest; (5) The study design was randomized or controlled.
Studies were excluded if they met any of the following criteria: (1) The study was a systematic review/meta-analysis, letter, editorial, elemental analysis, or case report; (2) The study was presented as a poster or at a conference.
Data extraction
The extracted data were obtained from the included study, which were organized into a dedicated spreadsheet for the pooled analysis. The detailed data included basic information, such as the name of first author, study design, inclusion period, sample size, demographic characteristics (age and sex) of the included participants, dominant hand, body mass index (BMI) (kg/m2), causes of elbow stiffness, surgery approach, TXA administration and dosage, follow-up data, hemoglobin levels, Mayo elbow performance score (MEPS), visual analogue scale (VAS), and range of motion (ROM). Each study was then analyzed to assess endpoint outcomes of interest, including operative time, tourniquet time, total drain output, blood loss, hemoglobin levels, hematocrit, ROM, MEPS, VAS, and complications, such as ulnar nerve symptom, recurrent HO, hematoma, and infection.
Risk of bias assessment
First, the revised Cochrane “Risk of bias” tool for randomized trials (RoB 2.0) 23 was employed to assess the risk of biases within individual studies. This tool evaluates five specific domains: bias arising from the randomization process, interventions, missing data outcome, outcome measurement, and selection of the reported result, which is classified as having low risk, some concern, or high risk.
Second, the Cochrane Risk of Bias in Non-randomized Studies of Interventions (ROBINS-I) tool 24 was used to evaluate the risk of bias, which addresses the risk of bias in relation to confounding, participant selection, classification of interventions, deviations from intended interventions, missing data, outcome measurements, and selective reporting. A judgment of risk is categorized in each domain as low risk, moderate risk, serious risk, or critical risk.
Statistical analysis
Statistical analyses and forest plots were conducted using Revman Manager (Version 5.3.5 (Cochrane Collaboration, London, UK). Indicators of interests were depicted using forest plots, with the mean difference (MD) for continuous variables and risk ratio (RR) for dichotomous variables. 25 Between-study heterogeneity was assessed using I2. An I2 value of 0%–40% indicated no substantial heterogeneity, whereas values greater than 40% indicated significant heterogeneity. 26 A two-tailed p-value threshold of <0.05 was applied for statistical significance.
Results
Study selection
The search of three medical databases identified 2053 potentially relevant citations, with an additional record generated by manually screening reference lists. After reviewing title and abstract, 2032 citations were excluded due to duplication or irrelevance. According to the eligibility criteria, the remaining 22 citations were retrieved in full text for further evaluation. Of these, 12 were considered irrelevant, one
18
was a protocol, and two27,28 were reviews, leading to their exclusion. After the full-text review, a total of 7 studies,16,17,19,29–32 encompassing 995 patients, were deemed eligible for inclusion in the quantitative analysis. A PRISMA flow chart is provided in Figure 1. Flow diagram of the literature search and selection process.
Study characteristics and patient demographics
The basic characteristics of the included studies.
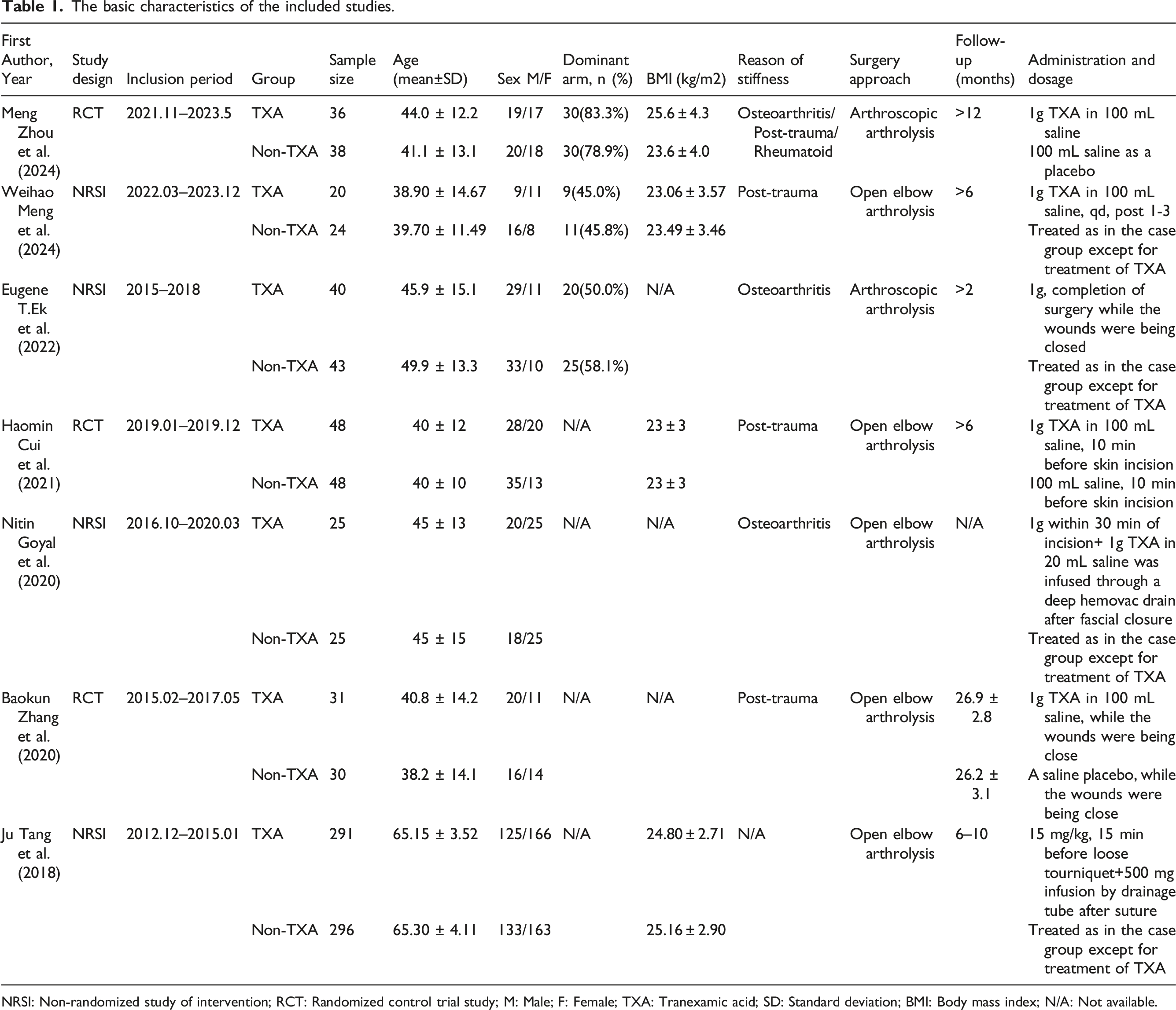
NRSI: Non-randomized study of intervention; RCT: Randomized control trial study; M: Male; F: Female; TXA: Tranexamic acid; SD: Standard deviation; BMI: Body mass index; N/A: Not available.
The basic information of participants from the included studies.

N/A: Not available; TXA: Tranexamic acid; VAS: Visual analog scale; MEPS: Mayo Elbow Performance Score.
Risk of bias
The revised Cochrane RoB 2 tool revealed that three RCTs16,19,30 were assessed to be of low risk of bias. According to the ROBINS-I tool for non-randomized studies of interventions (NRSIs), the majority of the included studies were deemed to have a moderate risk of bias.17,29,31,32 Overall, study assessment with the ROBINS-I tool indicated a moderate risk of bias among the majority of the included studies (Figure 2). Traffic-light plots for risk of bias assessment. (a) RoB 2 assessment of the the RCTs, (b) ROBINS-I assessment of NRCTs.
Operative time and tourniquet time
A total of four studies,16,17,30,31 involving 251 patients, assessed operative time, and no significant difference was observed between the two groups (MD [95% CI], 0.23 [−5.03, 5.50]; I2 = 0%; p = .93; Figure 3). Tourniquet time was analyzed in two studies,16,30 and no significant difference was found between the two groups (MD [95% CI], −0.80 [−10.01, 8.41]; I2 = 86%; p = .86; Figure 3). , involving 163 patients, assessed the ROM. Forest plots of the pooled results when assessing the indicators of interest, including operative time and tourniquet time.
Drain output (mL)
Total drain output was reported in seven studies.16,17,19,29–32 TXA was associated with a reduction in total drain output compared to the non-TXA group (MD [95% CI], −55.34 [−80.67, −30.02]; I2 = 84%; p = .0001; Figure 4). The pooled analysis of drain output at 24 hours after surgery, based on three studies,16,17,30 indicated that TXA could significantly reduce drain output at this time point compared to the non-TXA group (MD [95% CI], −70.41 [−121.75, −19.07]; I2 = 93%; p = .007; Figure 4). A similar result was observed with respect to drain output at 48 hours after surgery. Forest plot of the pooled results when assessing the indicators of interest, including total drain output.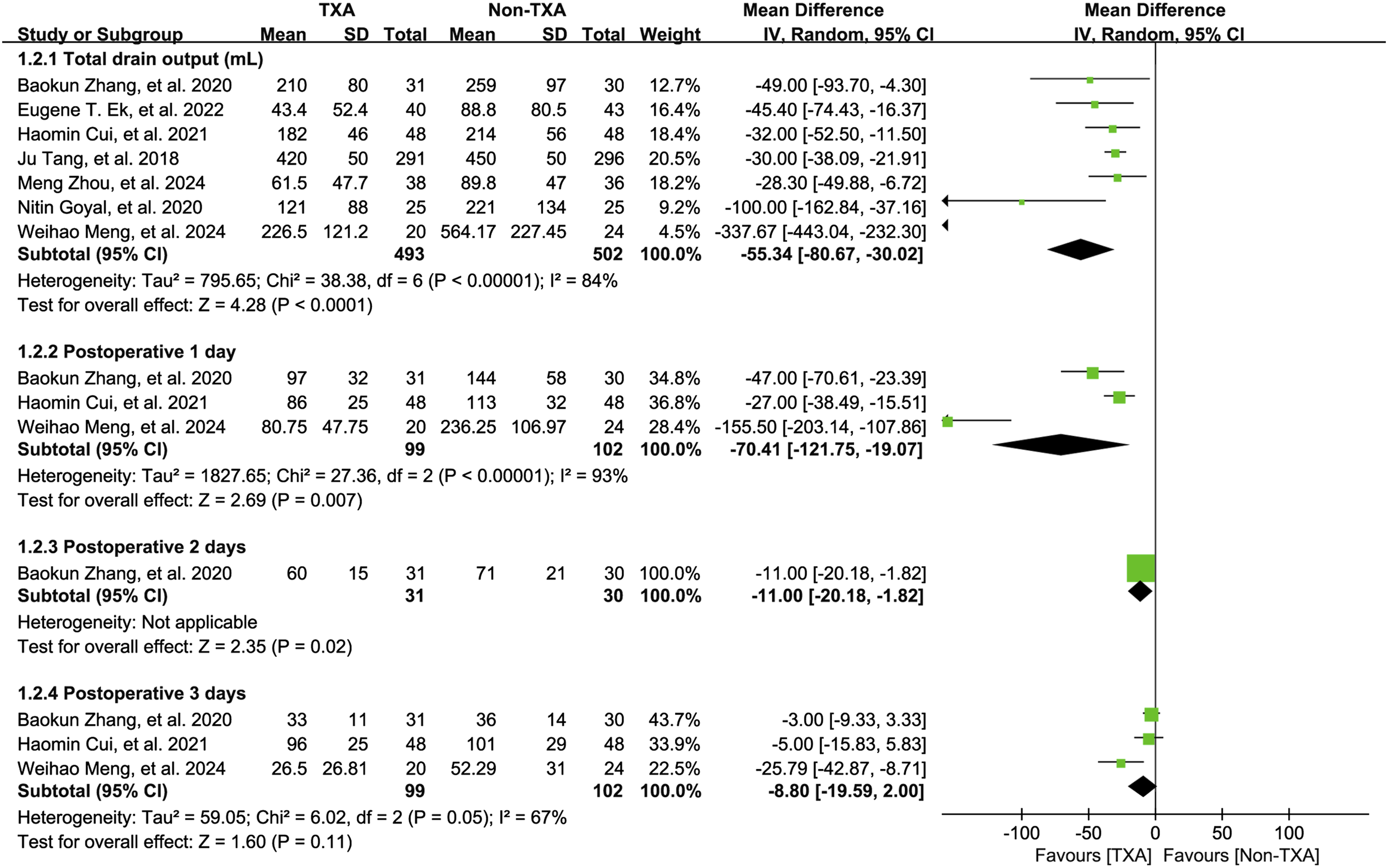
Subgroup analysis based on surgical approach (Open elbow arthrolysis vs Arthroscopic arthrolysis, Figure S1) for the evaluation of drain output demonstrated that the heterogeneity deceased from 84% to 0%, which indicated the source of heterogeneity and a consistent trend of comparison. Similarly, subgroup analysis based on TXA dosing (1g vs others, Figure S2) for the evaluation of drain output revealed that the heterogeneity deceased from 84% to 25%, which indicated the source of heterogeneity and a consistent trend of outcome.
Total blood loss, hemoglobin and hematocrit
A total of four studies,16,30–32 involving 995 patients, assessed total blood loss, and a significant reduction was observed in the TXA group compared to the non-TXA group (MD [95% CI], −39.07 [-69.71, −8.43]; I2 = 90%; p = .01; Figure 5). Forest plot of the pooled results when assessing blood loss, hemoglobin, and hematocrit.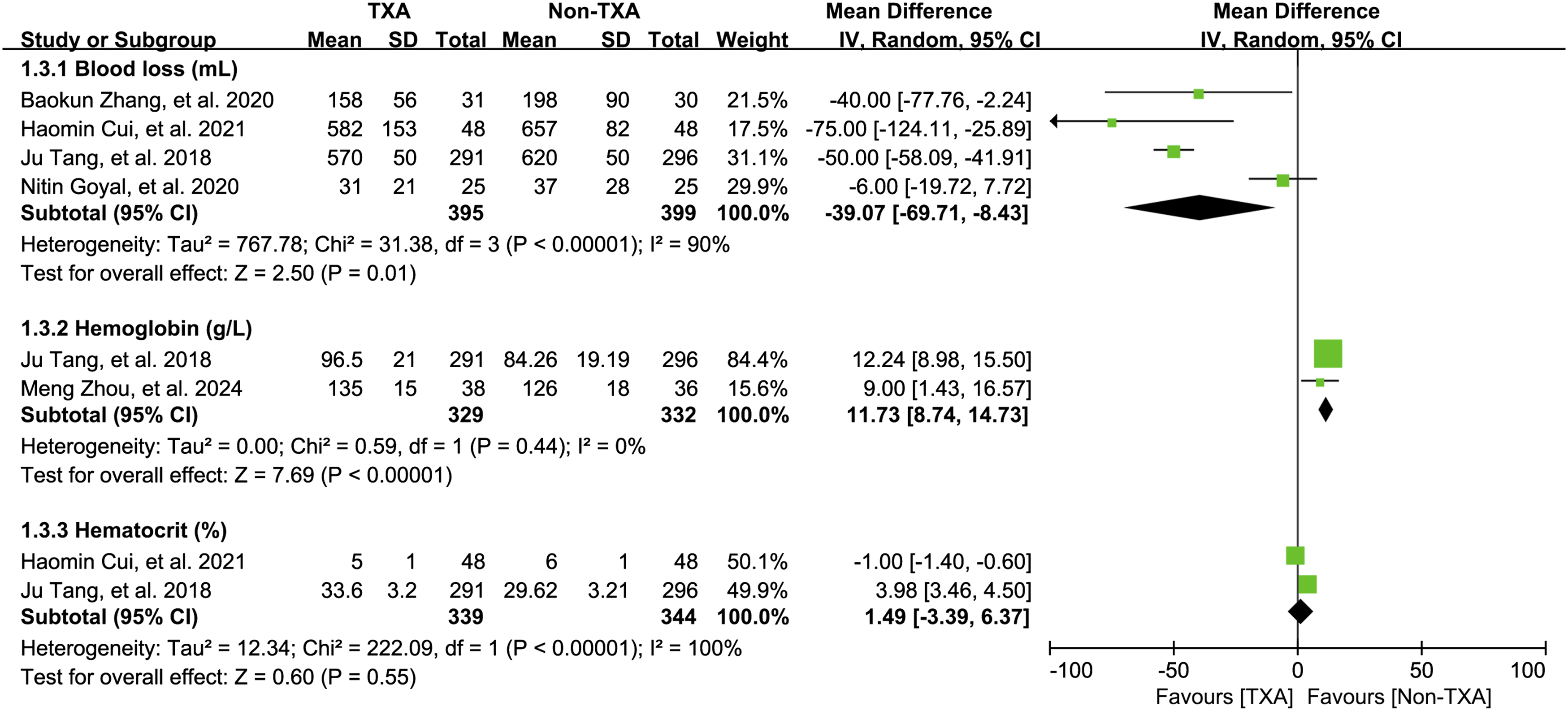
Two studies reported the postoperative levels of hemoglobin19,32 and hematocrit.16,32 The pooled data demonstrated that hemoglobin levels were significantly higher in the TXA group compared to non-TXA group (MD [95% CI], 11.73 [8.74, 14.73]; I2 = 0%; p < .00001; Figure 5). However, no significant difference was found in hematocrit levels.
Range of motion
A total of four studies,16,17,19,30 involving 163 patients, assessed the ROM. The pooled results indicated no significant difference in flexion-extension and pronation-supination between the two groups (Figure 6). Forest plots of the pooled results when assessing range of motion.
MEPS and VAS
Three studies16,19,30 involving 231 patients were included to evaluate MEPS at 6 months postoperatively. No significant difference was observed between the TXA and non-TXA groups (MD [95% CI], 0.58 [−1.63, 2.79]; I2 = 0%; p = .61; Figure 7). Forest plots of the pooled results when assessing MEPS.
Two studies19,29 involving 170 patients assessed VAS scores at 6 months postoperatively. No significant difference was found between the two groups (MD [95% CI], −0.35 [−0.96, 0.25]; I2 = 0%; p = .25; Figure 8). Forest plots of the pooled results when assessing VAS scores.
Complications
The complications from the included studies.

TXA: Tranexamic acid.

Forest plots of the pooled results when assessing complications.
Publication bias
Egger’s test results indicted no publication bias in this pooled analysis when evaluating indicators of interest including operative time, tourniquet time, drain output, hemoglobin, hematocrit, functional outcomes measured by MEPS and VAS and ROM, and complications. The results of these tests are presented in Figures S3–S9.
Discussion
This meta-analysis aimed to evaluate the impact of TXA on postoperative drainage, total blood loss, range of motion, and complications following elbow arthrolysis, a common procedure for addressing elbow stiffness. While elbow arthrolysis is highly effective in improving joint mobility, it is often associated with complications such as excessive bleeding, swelling, and prolonged recovery.1,2 These challenges highlight the importance of exploring strategies to minimize complications and promote faster recovery. The findings of this meta-analysis confirm that TXA plays a significant role in reducing postoperative drainage, minimizing total blood loss, and lowering the incidence of certain complications, while preserving functional outcomes such as range of motion.
The results of the meta-analysis revealed no significant difference in operative time between the TXA and non-TXA groups. These findings suggest that TXA administration does not prolong the surgical procedure, which is particularly relevant in orthopedic surgeries, where prolonged operation times can increase the risk of postoperative complications such as infections and extended anesthesia exposure. Similarly, no significant difference in tourniquet time was observed between the two groups, further supporting the notion that TXA does not impact the procedural aspects of elbow arthrolysis. This conclusion aligns with previous studies19,29 that have demonstrated the neutral effect of TXA on surgery duration in other surgical specialties.
The significant reduction in drain output observed in the TXA group is one of the most notable findings in this meta-analysis. Postoperative drainage is a common issue following elbow arthrolysis, 33 often leading to complications such as wound infections, 34 delayed healing, and longer hospital stays. The pooled analysis demonstrated that TXA significantly reduced the total drain output, with the most substantial reduction occurring within the first 24 hours post-surgery. This outcome aligns with previous studies16,30 that have highlighted TXA’s efficacy in reducing postoperative drainage by inhibiting fibrinolysis, thereby minimizing excessive blood loss. Reduced drainage is associated with decreased postoperative swelling, lower pain levels, and shorter hospital stays—factors that are crucial for optimizing patient recovery.
Total blood loss is a critical outcome in major orthopedic surgeries, particularly in procedures such as elbow arthrolysis, which involve manipulation of joint tissues and the surrounding soft tissues. 34 The analysis revealed that the TXA group experienced significantly less blood loss compared to the non-TXA group. This finding is consistent with multiple studies across various orthopedic procedures, including hip and knee arthroplasty, where TXA has been shown to significantly reduce blood loss.19,29 TXA exerts its effect by inhibiting the fibrinolytic pathway, preventing premature clot breakdown and thereby stabilizing the formation of clots at the surgical site. 35 This reduction in blood loss subsequently decreases the need for blood transfusions and minimizes the risk of postoperative anemia, a condition that can delay rehabilitation and impede functional recovery. 17
The analysis of postoperative hemoglobin levels revealed a significant advantage for the TXA group, with higher hemoglobin levels compared to the non-TXA group. This finding suggests that TXA effectively mitigates the impact of blood loss during surgery, preventing substantial drops in hemoglobin that could lead to anemia and delayed recovery. Interestingly, no significant difference was observed in hematocrit levels between the two groups. Although hematocrit is a more indirect measure of blood volume, its response to TXA may be less sensitive than that of hemoglobin, which more directly reflects blood loss and recovery from anemia. Similar findings have been reported in other studies examining TXA’s role in reducing blood loss and enhancing post-surgical recovery.16,17
A primary objective of elbow arthrolysis is to restore functional ROM. The meta-analysis revealed no significant difference in ROM between the TXA and non-TXA groups, suggesting that while TXA may reduce perioperative complications such as bleeding and drainage, it does not have a direct effect on joint mobility restoration. This result aligns with studies indicating that TXA primarily impacts perioperative factors rather than directly enhancing the mechanical aspects of joint function.16,19 The restoration of ROM is primarily influenced by surgical technique and postoperative rehabilitation, which includes physical therapy and joint mobilization exercises.
The MEPS and VAS scores, which evaluate joint function and pain levels, respectively, were assessed in the included studies. The pooled data from both the MEPS and VAS scores revealed no significant difference between the TXA and non-TXA groups at 6 months postoperatively. This suggests that while TXA may reduce blood loss and drainage, it does not significantly impact long-term functional recovery or pain levels in patients undergoing elbow arthrolysis. The lack of difference in MEPS and VAS scores may be attributed to the multifactorial nature of pain and functional recovery, which are more heavily influenced by factors such as the extent of soft tissue release, rehabilitation, and the presence of complications like heterotopic ossification. 19 Although this meta-analysis found that TXA significantly reduced postoperative drainage and blood loss, it did not lead to improvements in long-term ROM or pain scores (MEPS and VAS). This may be because TXA’s primary benefit lies in controlling perioperative bleeding, rather than directly influencing joint mechanics or pain pathways. Recovery of ROM is mainly determined by the extent of surgical release and adherence to postoperative rehabilitation, while pain is influenced by multiple factors including inflammation, nerve irritation, and individual pain thresholds. Therefore, while TXA helps create a more favorable early recovery environment, it does not significantly alter long-term functional outcomes.
The analysis of complications showed a significant reduction in hematomas in the TXA group compared to the non-TXA group. Hematomas are a common postoperative complication that can lead to pain, swelling, and delayed recovery. By reducing blood loss and promoting clot stability, TXA effectively minimizes hematoma formation, which could otherwise impair recovery and increase the risk of wound infection. However, no significant differences were observed in other complications such as ulnar nerve symptoms, recurrent HO, or infection. This finding is consistent with other studies that have demonstrated that TXA can reduce bleeding-related complications without affecting long-term outcomes, such as the recurrence of HO. 16 The absence of a significant impact on these complications suggests that while TXA can reduce immediate surgical risks, it does not prevent long-term complications associated with elbow stiffness and arthrolysis.
While most included studies reported functional outcomes at 6 months postoperatively, early postoperative functional data (1–3 months) were limited. These early data could offer more insight into the short-term benefits of TXA on recovery timelines, such as reduced swelling and pain that might facilitate earlier mobilization. Future research should prioritize early functional assessments to better understand TXA’s role in accelerating recovery following elbow arthrolysis.
In the included studies, TXA was administered either intravenously, topically, or in combination, with dosages ranging from 10 to 20 mg/kg IV or 1–3 g for topical use. However, due to variations in protocols, there is no consensus on the optimal dosing strategy for elbow arthrolysis. Evidence from larger orthopedic procedures suggests that a single preoperative intravenous dose of 15 mg/kg or 1 g may be both safe and effective. Further well-designed randomized trials are needed to determine the most appropriate dosing and route of administration to standardize TXA application in elbow surgery.
The underlying etiology of elbow stiffness (i.e., osteoarthritis vs post-traumatic) may influence surgical bleeding and response to TXA. We reviewed the original studies to determine whether subgroup data were available based on the cause of elbow stiffness. However, unfortunately, the available data were insufficient for a robust quantitative subgroup analysis, as most studies did not stratify outcomes by underlying diagnosis. Nevertheless, numerous studies indicates that post-traumatic stiffness often involves greater soft tissue scarring and heterotopic ossification, which may be associated with increased intraoperative bleeding. Theoretically, such patients might derive greater benefit from TXA, but conclusive subgroup comparisons cannot yet be made due to the lack of stratified data. This point may be an important direction for future research, emphasizing the need for studies that explicitly stratify patients by etiology when evaluating the efficacy of TXA in elbow arthrolysis.
Despite the promising results, this meta-analysis has several limitations that warrant attention. First, the quality of the included studies was not uniformly high. While several RCTs were included, some non-randomized studies were also part of the analysis, which may introduce potential biases. Additionally, significant heterogeneity was observed in some outcomes, such as drain output and blood loss, indicating that the results should be interpreted with caution. The variability in surgical techniques, TXA dosages, and follow-up durations across studies may have contributed to this heterogeneity. Future studies with more standardized protocols are necessary to validate the current findings. Furthermore, the included studies had differing follow-up periods, and the long-term effects of TXA on functional outcomes, such as ROM and HO recurrence, remain unclear.
Conclusion
Tranexamic acid (TXA) appears to be an effective adjunct in reducing postoperative drainage, blood loss, and hematomas following elbow arthrolysis for elbow stiffness. While it does not significantly influence long-term functional outcomes, such as ROM or pain (as measured by MEPS and VAS), its role in minimizing bleeding-related complications can contribute to a faster recovery and a reduced hospital stay. The findings from this meta-analysis provide robust evidence supporting the use of TXA in elbow arthrolysis and may guide clinical practices aimed at optimizing patient outcomes. However, further high-quality RCTs with standardized protocols and extended follow-up periods are needed to fully assess the long-term benefits and limitations of TXA.
Supplemental Material
Supplemental Material - Tranexamic acid: A strategy to decrease postoperative drainage in elbow arthrolysis while preserving joint function
Supplemental Material for Tranexamic acid: A strategy to decrease postoperative drainage in elbow arthrolysis while preserving joint function by Bao Zhao, Jinlei Dong, Guoming Zhang, Lianxin Li, Dongsheng Zhou, Shun Lu and Fanxiao Liu in Journal of Orthopaedic Surgery
Footnotes
Author contributions
LFX, LS and LLX contributed to the idea of this study. LFX and LS searched literatures and screened them independently. LFX, LS, and ZB screened data and made tables. LFX, LS, and ZB played an important role in analyzing the outcomes. LFX, LS, and LLX conducted the data analyses and made graphs. All authors wrote the first draft and polished and approved the final version. The authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The design, collection, analysis, and interpretation of the data in the study were financially supported by the Natural Science Foundation of Shandong Province (No.: ZR2021QH307), the Shandong Province Major Scientific and Technical Innovation Project (No.:2021SFGC0502), the Jinan Clinical Medical Science and Technology Innovation Plan (No.: 202328065), Shandong Traditional Chinese Medicine Science and Technology Development Plan Project (No.: 2019-0301). The authors, their immediate families, and any research foundations with which they are affiliated have not received any financial payments or other benefits from any commercial entity related to the subject of this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
