Abstract
Introduction
Loose bodies (LBs) are fragments of cartilage or bone that have become detached within the joint space; they can free float or lodge at certain positions or locations within the joint. Milgram et al. 1 categorized LBs in the synovial joints into three types based on their formation mechanism: osteochondral or chondral fracture, synovial chondromatosis, and degenerated joint surfaces due to osteoarthritis (OA). Furthermore, they explained that each type may have a different tissue origin. Extra-articular LBs of the ankle joint can result from various pathological processes, including synovial inflammation and (osteo)chondromatosis, and are considered both the result and agent of ankle OA. 2
In our clinical experience, we have frequently encountered cases in which LBs were discovered in the posterior ankles of patients who underwent surgical treatment for varus ankle OA. Although the main focus in these cases remains on the surgical treatment of ankle OA, we have wondered about the nature of concomitant LBs. However, information regarding the association between ankle OA and LBs is limited through existing research. This has prompted questions regarding the potential association between the characteristics of these LBs and ankle OA.
Consequently, this study aimed to investigate the characteristics of extra-articular posterior ankle LBs in patients who underwent surgical treatment for varus ankle OA. Furthermore, we sought to determine whether there are variations in the characteristics of LBs based on the degree of ankle OA. We hypothesized that the number of LBs may increase with the progression of ankle OA and that their location may exhibit specific patterns of change. Additionally, through this study, we expected to provide guidance on the optimal approach for LB removal when performing surgical treatment on varus ankle osteoarthritis with extra-articular posterior ankle LBs.
Materials and methods
Ethical considerations
This study was conducted in accordance with the principles of the Declaration of Helsinki and was approved by the Institutional Ethics Review Committee. Written informed consent was obtained from all enrolled patients.
Patient selection
We retrospectively analyzed 265 patients who underwent surgical treatment for varus ankle OA, including total ankle replacement arthroplasty, ankle arthrodesis, and supramalleolar osteotomy, between March 2011 and February 2023. Among them, we specifically focused on patients with confirmed posterior LBs on preoperative ankle imaging including radiographs, computed tomography (CT) or magnetic resonance imaging (MRI). The exclusion criteria were as follows: (1) individuals with systemic inflammatory diseases, such as rheumatoid arthritis or gout, and (2) individuals with a previous history of bony or soft tissue trauma that could have affected foot and ankle alignment. We categorized the entire patient cohort into four groups according to the degree of ankle OA to investigate the characteristics of LBs based on the severity of ankle OA (Takakura stage II, IIIA, IIIB, and IV).
Assessments
First, we collected demographic data, including age at operation, sex, body mass index (BMI), and affected side (right or left) from the patients’ medical records. Functional outcomes were assessed using the Foot and Ankle Ability Measure (FAAM) subscale scores for Activities of Daily Living (ADL) and Sports.3,4 The FAAM scores ranged from 0 to 84 and 0 to 32 for the ADL and Sports subscales, respectively. All evaluations were based on self-administered questionnaires completed by the patients.
The severity of ankle OA was assessed using the Takakura grading system,
5
which was based on standing ankle anteroposterior and lateral radiographs. Before surgery, all patients underwent CT and MRI of the affected ankle joint. CT revealed the number and size of LBs, focusing specifically on those located in the posterior ankle; the os trigonum was not included in the quantification of the number of LBs. The measurement of LB size involved determining the longitudinal distance on the sagittal image of the CT scan and selecting the maximum value for each LB (Figure 1). MRI was used to precisely identify the location of each LB, which was broadly classified into three groups: posterior recess (Figure 2), within the flexor hallucis longus (FHL) tendon sheath (Figure 3), and within the flexor digitorum longus (FDL) tendon sheath (Figure 4). Measurement of loose body size. The maximum value of the longitudinal distance of each loose body was selected to determine the longitudinal distance in the sagittal computed tomography image. Loose bodies in the posterior recess. Lateral radiography (a), computed tomography (b), sagittal T1-weighted (c), sagittal T2-weighted fat-suppressed (d), axial T1-weighted (e), and coronal T1-weighted (f) magnetic resonance images show multiple ossified loose bodies with marrow fat content (arrows) in the posterior recess. Loose bodies within the flexor hallucis longus tendon sheath. Computed tomography (a), axial T1-weighted (b), axial T2-weighted fat-suppressed (c), and sagittal T1-weighted (d) magnetic resonance images show multiple conglomeratic ossified loose bodies (arrows) with marrow fat content within the flexor hallucis longus tendon sheath. Loose bodies within flexor digitorum longus tendon sheath. Computed tomography (a) and axial T1-weighted (b) magnetic resonance images show two calcified and ossified loose bodies (arrows) in the flexor digitorum longus tendon sheath (arrowhead).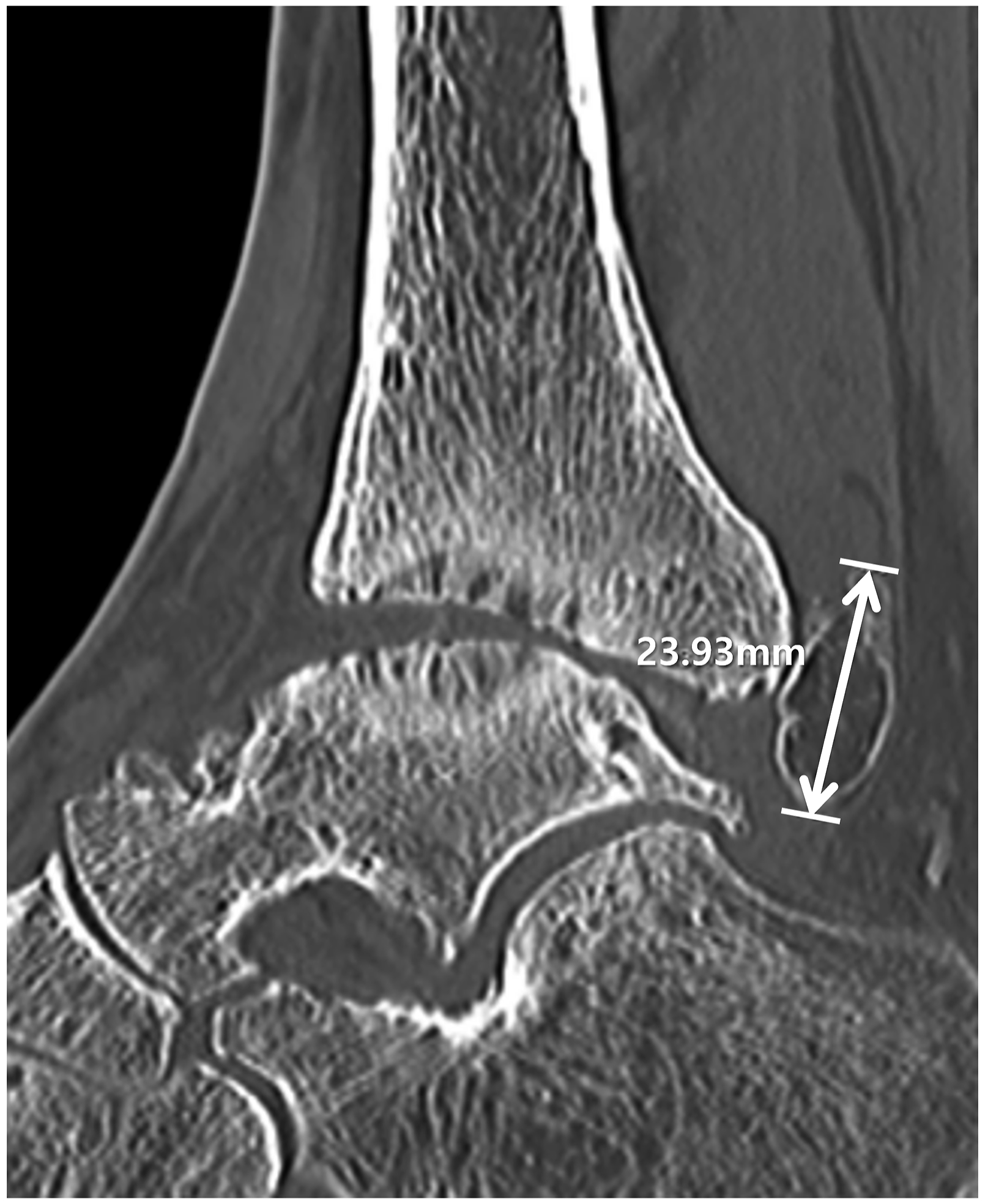



Statistical analyses
Descriptive analysis was performed using SPSS version 21 (IBM Corp., Armonk, NY, USA) for all variables, including the mean and standard deviation, or frequency. Data normality was tested using the Kolmogorov–Smirnov test. The Kruskal–Wallis test was used to compare continuous variables of LBs (size, total number, and number per person) among the four subgroups according to the Takakura stage. Post-hoc analysis was performed using the Mann–Whitney test.
Results
Characteristics of study patients
The patient selection algorithm is illustrated in Figure 5. Initially, 63 of the 265 patients (23.8%) were selected; however, after applying the exclusion criteria, a final cohort of 50 patients was included. The patient selection algorithm. Initially, 63 of the 265 patients (23.8%) were selected; however, after applying the exclusion criteria, a final cohort of 50 patients was included.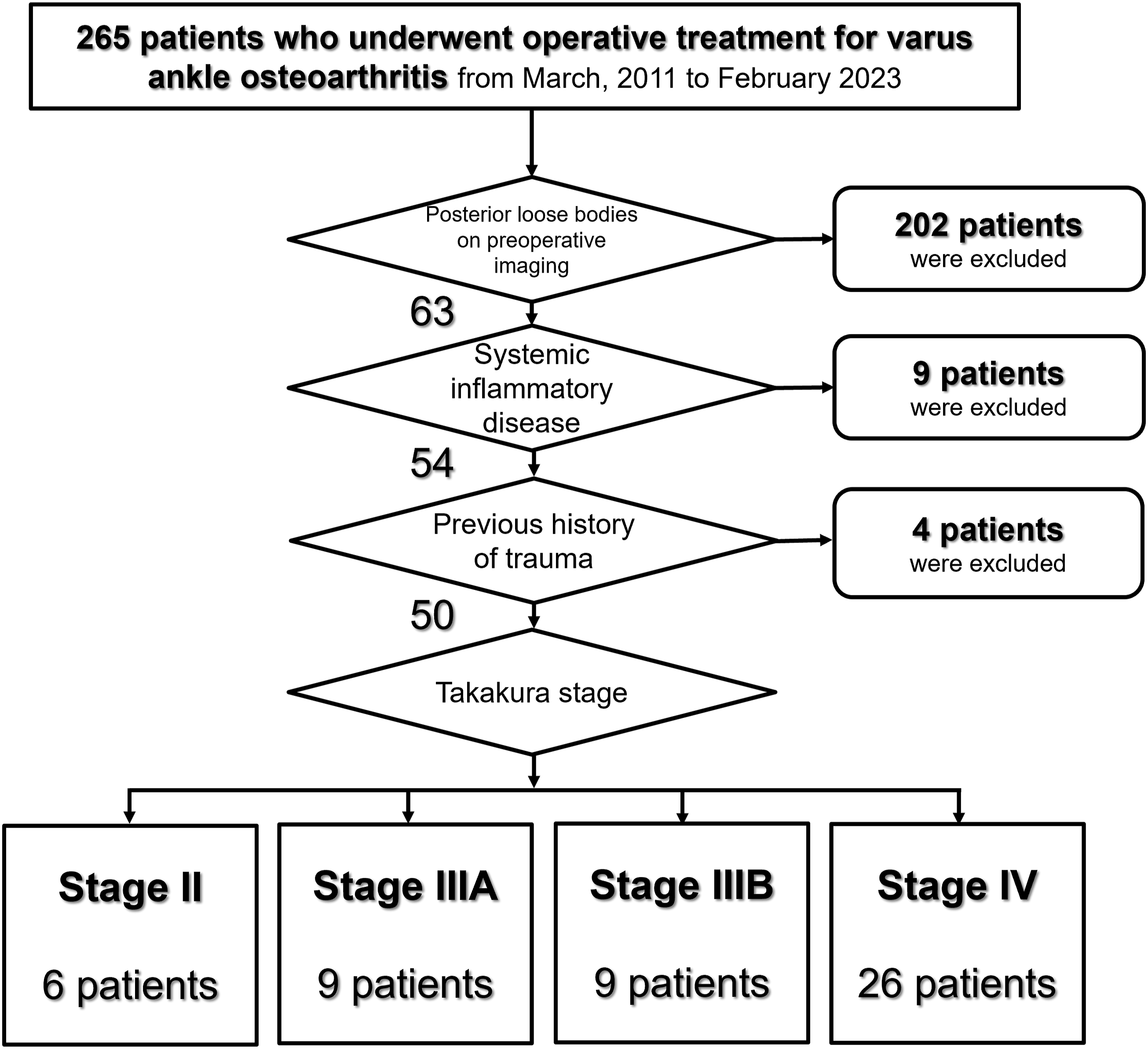
Characteristics of the 50 study patients.

BMI: body mass index; FAAM: foot and ankle ability measure; ADL: activities of daily living; FHL: flexor hallucis longus; FDL: flexor digitorum longus.
Values are presented as means ± standard deviations or number of cases (%), as appropriate.
Characteristics of loose bodies according to takakura stage
Characteristics of loose bodies according to takakura stage.

N: number of ankles; FHL: flexor hallucis longus; FDL: flexor digitorum longus.
Values are presented as means ± standard deviations or number of cases (%), as appropriate.
Although a limitation exists in the small number of stage II, IIIA, and IIIB patients, the location of LBs showed that ‘within the posterior recess’ was the most common in stage II, whereas ‘within the FHL tendon sheath’ was the most common in stage IIIA, IIIB, and IV. In cases where LBs were found within the FDL sheath, one patient each in stages IIIA and IIIB and two patients in stage IV were observed, while no patients in stage II exhibited such findings.
Discussion
Our study revealed that 23.8% of the patients undergoing surgical treatment for varus ankle OA exhibited posterior ankle LBs. A significant number of patients presented with multiple LBs, of which 76.8% were located in the FHL sheath. The LBs occasionally reached a substantial size, exceeding 2 cm, and there was a trend of increasing size in the advanced Takakura stages. Notably, LBs were most commonly found within the FHL sheath in the advanced Takakura stages (IIIA to IV) while a significant proportion of LBs in stage II were situated in the posterior recess, suggesting the possibility that these LBs may initially originate near the joint and gradually aggregate in a dependent position connected to the FHL tendon sheath.
LBs are soft tissue fragments that move freely within the body cavity, typically within the synovium. LBs occurring in the ankle joint are often derived from transchondral fractures or osteochondral lesion, or may arise from osseous metaplasia in synovial tissue, such as in synovial chondromatosis. 6 These LBs, generated through such processes, can traverse both within and outside the joint, resulting in symptoms such as pain, swelling, and episodes of locking and catching. In addition to the symptoms directly caused by LBs, manifestations may also result from the associated conditions that lead to the formation of these LBs.
Synovial chondromatosis is an uncommon, benign neoplastic process characterized by the development of hyaline cartilage nodules in the subsynovial tissue. It is classified into primary and secondary synovial chondromatosis based on the underlying causes, including disease, trauma, and joint inflammation. 7 Bursal synovial chondromatosis occurs within the intraarticular synovial tissue of a joint. 8 Conversely, if it originates from the extra-articular synovial tissue present in a tendon sheath, it is termed tenosynovial chondromatosis.9–14 Lui 15 categorized tenosynovial chondromatosis within the FHL sheath into zones 1–3 based on its location. Zone 1 corresponds to the area behind the ankle joint distal to the sustentaculum tali, zone 2 extends from the sustentaculum tali to the master knot of Henry, and zone 3 encompasses the region distal to the knot of Henry. To date, some studies have introduced methods for removing chondromatosis using tendoscopy based on the specific location15–18 and the high postoperative local recurrence rate. 19 Our research exclusively investigated cases falling within zone 1 according to Lui’s classification, specifically targeting LBs within this zone for a detailed examination. This distinct characteristic sets our study apart from previous research endeavors, emphasizing a unique approach to the analysis of zone 1 LBs. The findings of our investigation provide novel insights into the characteristics of these specific LBs and make a valuable contribution to the existing body of knowledge in this field.
Although the number of cases corresponding to Takakura stage II in our study was limited, making it challenging to draw concrete conclusions, an intriguing observation emerged regarding the predominant location of LBs within the posterior recess. Specifically, our findings suggest that a significant proportion of LBs in stage II are situated in the posterior recess, raising the possibility that these LBs may initially originate near the joint and gradually aggregate in a dependent position connected to the FHL tendon sheath. Furthermore, as there is a connection between the ankle joint and the FHL tendon sheath in approximately 17% of the general population, 20 it is challenging to conclusively determine from the results of our study whether a LB occurring within the FHL tendon sheath originates as tenosynovial chondromatosis within the FHL tendon sheath itself, or it was originated from the synovium, cartilage, or osteophytes of the ankle joint and subsequently migrated into the FHL tendon sheath. However, the observation that the majority of extraarticular posterior LBs in varus ankle OA are present within the FHL tendon sheath is considered a novel finding of this study. This highlights the importance of opening the FHL tendon sheath when surgeons plan the surgical removal of LBs while performing concomitant surgery to treat OA.
In this study, the size of the LB tended to increase with the progression of arthritis severity (as indicated by the Takakura stage). The growth of ossified LBs is a dynamic process influenced by mechanical, biological, and pathological factors. Once an ossified LB develops, the ongoing deposition of calcium and other minerals can continue, leading to an increase in its size. Another reason is the continued mechanical stress and microtrauma which can stimulate further ossification and calcification, leading to growth over time. Additionally, LBs can receive nutrients from the synovial fluid, which may contribute to their growth. The synovial fluid provides a conducive environment for the LB to accumulate more minerals, leading to further ossification. In some cases, the LB can become encased in synovial tissue, which may undergo metaplasia and lead to further ossification (synovial metaplasia).1,21
The current study has several limitations, including a small sample size, and high number of excluded patients. Additionally, it would have been beneficial to classify LBs into chondral and osseous fragments based on their radiographic characteristics. In the early stages of our research, we attempted to perform this categorization; however, because of the difficulty in distinguishing such characteristics in very small fragments, we ultimately excluded this result from our analysis. Nevertheless, despite these challenges, this study represents an initial attempt to investigate the characteristics of LBs in patients with varus ankle OA based on the degree of ankle OA, which adds significance to our findings.
In conclusion, our study revealed that a considerable proportion of patients with varus ankle OA had extra-articular posterior LBs (23.8%). The LBs were predominantly located within the FHL tendon sheath (76.8%). From Takakura stages IIIA and above, patients often presented with an average of two or more LBs, and there was a significant increase in the size of LBs observed in Takakura stages IIIB and IV. We hope that this information will serve as valuable reference material for clinicians treating patients with varus ankle OA who have extra-articular posterior LBs.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
