Abstract
Objectives
Platelet-rich plasma treatment delays the need for total knee replacement in patients with knee osteoarthritis. However, its use and preparation remain controversial. The aim of this study was to investigate the relationship between anticoagulant use in the preparation of platelet-rich plasma and post-treatment pain in patients with knee osteoarthritis. Additionally, we explored the efficacy of platelet-rich plasma over medium- and long-term follow-up periods and identified other factors that may affect treatment outcomes.
Methods
In this retrospective study, 225 patients with knee osteoarthritis, who underwent knee platelet-rich plasma treatment from June 2021 to January 2022, were examined at three study centres. Patients were categorised, based on the type and amount of anticoagulant used during platelet-rich plasma preparation, into 4% sodium citrate (SC) 0.6 mL, 4% SC 1 mL, 4% SC 2 mL, heparin 0.1 mL, and heparin 0.2 mL groups. We analysed the patients’ basic information, pain after treatment, and inflammatory markers (i.e., interleukin 6, tumour necrosis factor-α, and hypersensitive C-reactive protein) in the joint fluid via enzyme-linked immunosorbent assay and joint fluid crystallisation. Additionally, we assessed the patients’ Western Ontario and McMaster University scores and minimal clinically significant differences after treatment.
Results
Patients in the 4% SC 0.6 mL and heparin 0.1 mL groups experienced less pain after platelet-rich plasma treatment than did patients in the high-dose anticoagulant group. The joint fluid of patients with pain in these groups had lower levels of inflammatory markers. Patients treated with SC had slightly better medium- and long-term therapeutic outcomes than did patients treated with heparin. Patients with poorly controlled hyperuricemia also experienced pain after platelet-rich plasma treatment.
Conclusions
The results suggest that platelet-rich plasma prepared using high-dose anticoagulants or administered to patients with poorly controlled hyperuricaemia may lead to moderate-to-severe knee pain and joint effusion after joint puncture therapy. Platelet-rich plasma had a therapeutic effect on knee osteoarthritis; however, its efficacy gradually decreased over time. SC anticoagulant is more suitable for platelet-rich plasma preparation than is heparin. Further studies are needed to understand the safety and the various factors influencing platelet-rich plasma therapy.
Introduction
Knee osteoarthritis (KOA), the most common joint disease in older adults, 1 is caused by systemic, local, and mechanical-/trauma-promoting factors. 2 Early diagnosis and intervention can prevent its progression. 3 However, because of poor blood supply, the articular cartilage cannot regenerate effectively after injury, thereby leading to irreversible damage and progressive loss of joint function that ultimately requires total knee replacement (TKA).4,5 Platelet-rich plasma (PRP) treatment delays the need for TKA in patients with KOA. 6 PRP is a biological agent that contains growth factors, regulates joint homeostasis, promotes healing of the articular cartilage and other tissues, and controls intra-articular inflammation.7–10
The use of PRP remains controversial; however, it has recently been widely applied in clinics. Riewruja et al. 11 analysed the spectrum of cytokines, chemokines, and growth factors in PRP and platelet-poor plasma derived from patients with KOA; they found that PRP contained significantly higher concentrations of several factors. Intra-articular PRP injection reduces pain and improves joint function. Yurtbay et al. 12 conducted a randomised controlled trial (RCT) comparing PRP with placebo for the treatment of patients with KOA; they found that PRP was more effective than a placebo and that multiple doses increased the therapeutic effect and duration. However, Tomicic et al. 13 conducted an RCT comparing PRP and saline control treatment for patients with KOA and found that PRP was not better than placebo in terms of pain and functional improvement within 24 weeks after treatment; furthermore, the PRP group had a higher frequency of mild transient pain.
The preparation of PRP remains controversial, with most research focusing on the centrifugation method and the content of white blood cells.14,15 However, the anticoagulants used for blood product preparation may not be safe for humans.16,17 This issue has largely been ignored in studies related to PRP preparation and KOA therapy. We have observed that different doses of anticoagulants for PRP preparation are strongly correlated with adverse reactions after joint puncture, such as joint swelling and pain. Ethylenediaminetetraacetic acid (EDTA)-dependent pseudothrombocytopenia often occurs when EDTA is used as an anticoagulant, leading to difficulties in the clinical control of PRP quality.18,19 Therefore, in this study, we selected two common anticoagulants for PRP preparation: sodium citrate (SC) and heparin. We collected data from patients at three PRP treatment centres to observe the effects of different anticoagulant doses in patients with mild-to-moderate KOA. We aimed to investigate the impact of anticoagulant use in PRP preparations on the therapeutic efficacy and potential complications associated with the treatment. Additionally, we examined whether metabolic or systemic diseases in patients with KOA pose a risk to PRP treatment.
Methods
Patients
In this retrospective study, we examined patients with KOA who underwent knee PRP treatment at the PRP Treatment Clinic of the Department of Joint Surgery at three study centres from June 2021 to January 2022. These patients had a Kellgren–Lawrence (K/L) rating of grades II–III (i.e., one knee) and no thrombocytopenia. Patients with KOA who had complete treatment data [including name, sex, age, body mass index (BMI), initial Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) score, K/L grade on knee X-ray, platelet count, PRP preparation, and treatment records] and who completed telephone follow-up were screened. Finally, the treatment records of 225 patients with KOA were obtained. All PRP preparations were performed by skilled practitioners, and joint punctures were performed by senior orthopaedic surgeons in each centre. All patients were routinely followed up after treatment, with an average follow-up period of 12 months. The PRP treatment clinics of the three research centres used 4% SC at a concentration of 136 mmol/L or used a heparin anticoagulant with a relative molecular weight of 12,500 U to prepare PRP. Based on the PRP preparation records, the dosage of anticoagulant in each group was recorded as follows: (1) 4% SC 0.6 mL group; (2) 4% SC 1 mL group; (3) 4% SC 2 mL group; (4) heparin 0.1 mL group; and (5) heparin 0.2 mL group (i.e., a total of five groups). The study complied with international standard regulations for clinical research, as outlined in the latest revisions of the Declaration of Helsinki and Good Clinical Practice Regulations, and was approved by the Ethical Committee. Written informed consent was obtained from all patients before treatment initiation.
Preparation of platelet-rich plasma
The preparation of PRP was based on the secondary centrifugation method recommended by Landersberg.20,21 Under the guidance of experts from the blood transfusion department, a 3.8% SC 1:9 anticoagulant tube, 1:4 anticoagulant tube, or 1:16 (i.e., 0.56:9, 0.6:9 for easy operation) ratio of in vitro anticoagulant, or 12,500 U heparin anticoagulant made up to a volume of 0.1–0.2 mL, based on the manufacturer’s instructions, can be used for 9 mL blood anticoagulant in vitro. A total of 36 mL of venous blood was collected from the patient and centrifuged at 280 × g for 5 min. The upper platelets containing the plasma were extracted and centrifuged at 300 × g for 8 min. The upper platelet-deficient plasma was discarded, and the lower PRP was collected. Taking approximately 5 mL of the PRP preparation, the current recommended platelet concentration is three to five times the normal venous blood platelet concentration. 22 All PRP preparations were attained using strict aseptic procedures. PRP was prepared within 30 min of venous blood collection and immediately administered via intra-articular injection therapy to complete treatment under optimal PRP conditions.
Treatment method
The superior patellar lateral approach was used to inject PRP into the knee cavity. A 2.5-mL syringe was used for treatment, and 1 mL of lidocaine was administered subcutaneously before injection to minimise pain. The needle could be directed down or up along the surface of the patella and into the joints. The advantage of the lateral approach is that no obvious pain occurs during treatment. When a large amount of effusion is in the suprapatellar bursa, it should be extracted first, and specimens should be collected for subsequent detection. Each patient received three injections of PRP into the knee cavity, with an interval of 2 weeks between each injection. Patients were observed for 30 min after the completion of treatment and were discharged without discomfort. If the patient experienced symptoms such as knee pain or swelling that could not be relieved after treatment, they were advised to visit the hospital as soon as possible for symptomatic treatment such as analgesia and extraction of knee effusion.
Evaluation of PRP platelet concentration in anticoagulant preparation
Routine blood analyses were conducted before the preparation of PRP in all patients to record platelet and other counts to prevent the treatment of patients with abnormal platelet counts. A 1-mL PRP sample was extracted under aseptic conditions for platelet count measurement after PRP preparation, and the concentration of the anticoagulant in each group was recorded.
Evaluation of therapeutic effect
The number and frequency of knee pain and swelling episodes within 3 days after PRP injection were observed in each group, and the visual analogue scale (VAS) scores and data on duration of pain in these patients were recorded. The number of patients in each group who abandoned follow-up treatment because of post-treatment pain was recorded. After the patients completed their treatment cycle, their knee joint conditions were evaluated at 6 and 12 months after treatment. We used WOMAC scores and minimum clinically important difference (MCID); based on previous research, the MCID of WOMAC score was 11.5. 23 We evaluated the medium- and long-term effects of PRP treatment on KOA and determined whether to proceed to the next cycle of treatment.
Test of knee effusion samples
For each group of patients with knee effusion after PRP treatment, samples were sent to the clinical laboratory after joint effusion extraction for the following tests: (1) routine examination of joint fluid (including fluid colour, clarity, coagulation, density, and microscopic cell count and classification); (2) joint fluid culture + drug sensitivity; (3) joint fluid biochemistry (including mucin clot formation test, protein quantification, and glucose quantification); (4) crystal detection of articular fluid; (5) inflammatory metabolic factors in the synovial fluid, including interleukin 6 (IL-6), tumour necrosis factor- α (TNF-α), and hypersensitive C-reactive protein (hs-CRP), were detected using enzyme-linked immunosorbent assay.
Statistical analysis
After data collection, SPSS (version 26.0; IBM, Armonk, NY, USA) was used for statistical analysis. The main dependent variables of clinical outcome were platelet count in PRP, WOMAC score, VAS pain score, number of short-term pain occurrences/cases, duration of pain, number of patients who abandoned treatment, amount of joint effusion, inflammatory markers in the joint fluid, and crystallisation in the joint fluid. All measurements are expressed as standard deviations. When the variance was homogeneous, analysis of variance (ANOVA) was used between groups, and the Bonferroni test was used for pairwise comparisons after the event. When the variances were inconsistent, Welch’s ANOVA was used between groups, and the Games–Howell test was used for pairwise comparisons. Paired-sample t-tests were used for paired comparisons. Statistical significance was set at p < .05.
Results
General situation of patients in each group
Overall, 184 of the 225 screened patients completed the post-treatment follow-up (Figure 1 and Table 1). No significant differences were noted in age, sex, BMI, K/L stage, pain duration, or basal platelet count between the groups (p > .05). The platelet count was significantly higher in the SC group than in the heparin group (p < .001), but no statistically significant difference existed between the three SC groups or the two heparin groups (p > .05). Flowchart of patient selection for the retrospective analysis. Overall, 225 patients were screened by three centres from 2021 to 2022 and their data were retrospectively analysed. Abbreviations: PRP platelet-rich plasma, SC sodium citrate, VAS visual analogue scale. Patients’ characteristics and groups. When the variance was homogeneous, ANOVA was used between groups, and the Bonferroni test was used for pairwise comparisons after the event. When the variances were inconsistent, Welch’s analysis of variance was used between groups, followed by the Games–Howell test for pairwise comparisons. Statistical significance was set at p < .05. Abbreviations: ANOVA analysis of variance, BMI body mass index, K/L Kellgren–Lawrence, PLT platelet, PRP platelet-rich plasma, SC sodium citrate, SD standard deviation, WB whole blood. aIndicates that the comparison between groups was statistically significant (based on Welch’s analysis of variance, p < .001). bThe results differed significantly between the heparin (0.1 mL) and SC groups (based on the Games–Howell test, p < .001). cThe heparin (0.2 mL) and SC groups differed significantly (based on the Games–Howell test, p < .001). Pairwise comparison of SC among the three groups revealed no statistically significant differences. No statistically significant difference existed between the heparin group and the other two groups.

Pain after PRP treatment
Short-term situation of each group after PRP treatment.

When the variance was homogeneous, ANOVA was used between groups, and the Bonferroni test was used for pairwise comparisons after the event. When the variances were inconsistent, Welch’s analysis of variance was used between groups, followed by the Games–Howell test for pairwise comparisons. The chi-square test was used to compare the classified data between groups, and the Bonferroni test was used for pairwise comparisons after the event. Statistical significance is set at p < .05.
Abbreviations: ANOVA analysis of variance, SC sodium citrate, VAS visual analogue scale, PRP platelet-rich plasma, SD standard deviation.
*Indicates a statistically significant difference between the two groups of low-dose anticoagulants and the two groups of sodium citrate high-dose anticoagulant.
Joint fluid test outcomes
Joint fluid test.

ANOVA was used to compare the variance between groups, and LSD or the Scheffe method was used to compare the two groups. If the variance was inconsistent, Welch’s method was used for the analysis, and the Games–Howell method was used for the post-test. The detection rate of uric acid crystals was analysed using Fisher’s exact test.
Abbreviations: ANOVA analysis of variance, hs-CRP hypersensitive C-reactive protein, IL-6 interleukin 6, LSD least significant difference, SC sodium citrate, TNF-α tumour necrosis factor- α
aIndicates a significant difference with the 4% SC (2 mL) group.
bIndicates a significant difference with the heparin (0.1 mL) group.
cIndicates a significant difference with the 4% SC (0.6 mL) group.
dIndicates a significant difference with the 4% SC (1 mL) group.
Medium and long-term follow-up outcomes
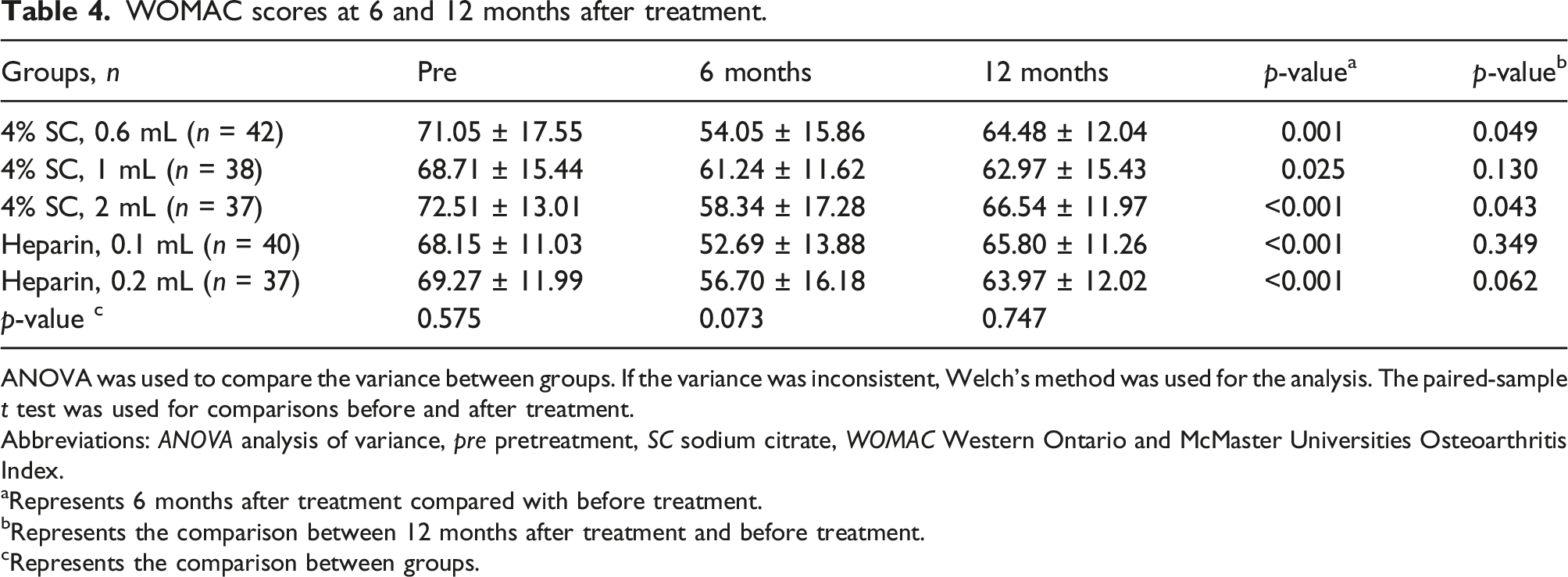
Figures 2 and 3 illustrate the changes in the WOMAC score and MCID, respectively, of patients in each group at 6 and 12 months of follow-up after PRP treatment. No significant differences were noted in the scores before treatment, at 6 months after treatment, and at 12 months after treatment between the groups. The symptoms improved at 6 months after treatment, with a statistically significant difference, compared with symptoms before treatment. However, the efficacy decreased at 12 months after treatment (Tables 4 and 5). WOMAC scores at each follow-up point. The change in WOMAC scores in each group. * Indicates a significant difference from baseline. No statistically significant differences were observed between the groups. Abbreviations: pre, pre-treatment; PRP, platelet-rich plasma; SC, sodium citrate; WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index. MCID at each follow-up point. The change in the proportion of MCID in each group. No statistically significant differences were observed between the groups. Abbreviations: MCID, minimal clinically important difference; PRP, platelet-rich plasma; SC, sodium citrate. WOMAC scores at 6 and 12 months after treatment. ANOVA was used to compare the variance between groups. If the variance was inconsistent, Welch’s method was used for the analysis. The paired-sample t test was used for comparisons before and after treatment. Abbreviations: ANOVA analysis of variance, pre pretreatment, SC sodium citrate, WOMAC Western Ontario and McMaster Universities Osteoarthritis Index. aRepresents 6 months after treatment compared with before treatment. bRepresents the comparison between 12 months after treatment and before treatment. cRepresents the comparison between groups. MCID at 6 and 12 months after treatment. The chi-square test was used to compare the classified data between groups, and the Bonferroni test was used for pairwise comparisons after the event. Statistical significance is set at p < .05. MCID minimum clinically important difference, PRP platelet-rich plasma, SC sodium citrate. aRepresents the comparison between 6 and 12 months after PRP treatment. bRepresents the comparison between groups.


Discussion
PRP has been extensively used for KOA treatment in joint surgery; however, numerous questions remain unanswered. The most important finding of this study is that the use of high doses of anticoagulants and poorly controlled hyperuricemia may be risk factors for PRP treatment of KOA. PRP is an invasive therapy that involves articular puncture to reach damaged joint tissues. Despite increased proficiency in the knee joint puncture technique, complications such as pain and swelling still exist. As a novel biological therapy, PRP has yielded many favourable results and has received encouraging evaluations for treating KOA. 24 However, a standardised preparation protocol for PRP remains elusive, and new complications need to be recognised and addressed. The preparation of PRP requires the use of anticoagulants; otherwise, the therapeutic platelets may be activated prematurely.
Anticoagulants differ in terms of their pharmacology, and no standard protocol exists for PRP preparation. These factors are unfortunately often overlooked in clinical practice. 25 A study by Görgü et al. on PRP injections into the face and scalp revealed that different anticoagulant components in PRP could affect patients’ post-treatment pain levels. 26 Invasive joint puncture therapy appears to be relatively safe for patients undergoing long-term anticoagulant therapy; however, anticoagulant drug adjustment should be based on specific circumstances. 27 The local risks of injecting the anticoagulant PRP directly into the joint via invasive methods have received little attention.
The aim of this study was to investigate the influence of anticoagulants on platelet content in PRP, their therapeutic effects, and short-term adverse reactions after treatment. We focused on patients with moderate-to-severe pain (i.e., VAS score ≥4; duration ≥3 days) after knee puncture because the pain caused by this procedure is generally mild. The results showed that SC and heparin anticoagulants can increase knee swelling and pain after treatment with higher PRP dosage; however, this situation was rare when using low dosages of anticoagulants. Regardless of the dosage of the anticoagulant used, the platelet content in the PRP met the treatment standard. Furthermore, no significant difference was noted in the medium- and long-term efficacy during follow-up.
Short-term knee swelling and pain after PRP treatment can be attributed to soft tissue injuries of varying degrees caused by the syringe needle in the joint cavity during joint puncture. The joint capsule, synovium, cartilage, and ligaments may be injured during this process. When excess anticoagulants enter the joint cavity along with PRP, local injury causes healing difficulties and persistent bleeding may occur, leading to swelling and pain. This study demonstrated that the high-dose anticoagulant group had joint fluid samples with higher amounts of joint effusion and inflammatory indicators, although the bacterial culture was negative. This finding indicated that joint synovitis in these patients arose from aseptic inflammation, which may be similar to the pathogenesis of synovitis caused by haemophiliac arthritis. 28 These conditions may gradually resolve as cytokines are released from PRP. 29 However, for these patients, this outcome represents a poor medical experience, and they may abandon follow-up treatment.
PRP prepared with low-dose anticoagulants may be less effective in producing a response in the joint cavity, resulting in fewer cases of knee swelling and pain. A clinical study by Duan et al. 30 demonstrated that PRP treatment improved neither pain levels nor function in patients with haemophiliac arthritis; however, their study did not detect changes in inflammatory indicators in the joint synovial fluid. In contrast, an RCT by Cole et al., 31 which included 111 patients with KOA, showed that PRP could improve the prognosis of patients with KOA and sustainably reduce inflammatory markers in the synovial fluid of the joint. These findings suggest that the anti-inflammatory properties of PRP may contribute to symptomatic improvement. This supports the findings of our study, compared with those of the study on haemophiliac arthritis. However, the injection of excessive anticoagulant may lead to intra-articular bleeding, producing negative effects on the joint. Du et al. 32 reported a method of preparing high-quality PRP by using a temperature change instead of an anticoagulant, but this method has not been widely adopted because of economic limitations. The mainstream method of PRP preparation still requires the addition of anticoagulants. However, further studies are needed to confirm whether anticoagulants directly stimulate the synovium in the joint cavity, lead to aseptic inflammation, or produce other biological effects.
Gout is a chronic disease caused by the precipitation and deposition of urate crystals in the joints or tissues resulting from long-term hyperuricaemia and triggering severe pain during acute attacks. Urate crystals are often found in the joint effusions of patients, providing strong evidence for the diagnosis of this disease. 33 In our study, urate crystals were detected in the synovial fluid of some patients with post-treatment joint pain and swelling. These patients received immediate intervention. PRP treatment was discontinued, symptomatic management was adopted and the fact that they had poorly controlled hyperuricemia was later confirmed. PRP is a high-concentration platelet plasma product; therefore, metabolites in plasma such as blood uric acid and free fatty acids are directly injected into joints along with the plasma, which may be one of the causes of acute joint pain in these patients. Anticoagulants may also induce aseptic inflammation and bleeding of the punctured synovial tissue in the joint, thereby contributing to pain.
Recognising the contraindications for PRP treatment in patients with KOA is critical. Few studies have comprehensively reported whether PRP treatment is effective or risky for patients with systemic inflammatory diseases or blood diseases such as rheumatoid arthritis. According to Spartalis et al., 34 after tumour resection, the presence of growth factors in PRP may promote the growth and recurrence of tumours, and using the blood products of tumour patients to treat their own diseases may also spread tumour cells. This factor is a safety issue that needs to be recognised in PRP treatment, especially because the popularity of PRP continues to increase in various therapeutic fields.
The medium- and long-term follow-up results of this study demonstrated that PRP exhibited a certain therapeutic effect, which persisted for more than 6 months, in patients with KOA. However, the curative effect of PRP had decreased after 12 months of follow-up and PRP prepared with SC appeared to have a more beneficial effect.
The current research partially supports the therapeutic effect of PRP in KOA 35 ; however, high-quality RCTs have reported conflicting results. For instance, Bennell et al. 36 only followed up participants for 12 months. The prospective and retrospective studies by Chopin et al. 37 and Saita et al., 38 respectively, demonstrated that variations in patients’ clinical characteristics resulted in differences in the therapeutic effects of PRP. Furthermore, Collins et al. 39 proposed that different types of PRP may be more appropriate for treating different diseases. These factors may explain why the efficacy of PRP treatment remains controversial.
This study has several limitations. First, the retrospective nature of the study precluded using other treatments as controls. Second, patient screening requirements limited the number of patients who successfully completed the study and retained complete data, and the follow-up period was also restricted. Third, further pathological studies are required to confirm the effects of injecting various anticoagulants and serum samples from patients with hyperuricaemia into the knee joint on the intra-articular structure. Fourth, although the preparation methods of PRP in all centres were consistent, differences in the doctors’ injection techniques may have affected the treatment outcomes.
In summary, high-dose anticoagulants in PRP or in patients with poorly controlled hyperuricaemia may result in moderate-to-severe knee pain and joint effusion after joint puncture therapy. Therefore, low-dose anticoagulants are preferred, and patients with uncontrolled hyperuricaemia should not undergo PRP therapy. PRP exhibits a therapeutic effect on KOA, but this effect diminishes over time. SC appears to be more suitable than heparin for PRP preparation.
Footnotes
Author’s note
All authors listed meet the authorship criteria according to the latest guidelines of the International Committee of Medical Journal Editors, and all authors are in agreement with the manuscript.
Authors’ contributions
All authors had full access to the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. Conceptualization, H.L; Methodology, Y.Z; Investigation, H.L., H.S., and S.W; Formal Analysis, Y.C., and H.Z; Resources, H.L., H.S., and S.W; Writing - Original Draft, Y,C; Writing - Review & Editing, Y.Z., and H.L; Visualization, Y.C., and H.L; Supervision, S.W., Y.Z., and H.L; Funding Acquisition, S.W., Y.Z., and H.L.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: that might pose a conflict of interest in connection with the submitted article related to the author or any immediate family members.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (82072501), the Hunan Provincial Natural Science Foundation project (Grant No. 2023JJ50247), the Research Program of the Health Commission of Hunan Province (Grant No. D202304076870), and the Science and Technology Program of Shaoyang City (Grant No. 2021071ZD).
