Abstract
Objective
Activation of gap junction channels can induce neuropathic pain. Octanol can limit the conductance of gap junctions containing connexin 43 proteins. Thus, this study focused on the roles of octanol in chronic constriction injury (CCI)-induced peripheral neuropathy in mice and its mechanisms of action.
Methods
Male mice were assigned into control, sham, CCI, CCI + Octanol-20 mg/kg, CCI + Octanol-40 mg/kg and CCI + Octanol-80 mg/kg groups. CCI was performed by applying three loose ligations to mouse sciatic nerve, and the mice with CCI was administered with 20 mg/kg, 40 mg/kg, or 80 mg/kg octanol. The neuropathic pain development was examined by assessing thermal withdrawal latency, paw withdrawal mechanical threshold, and sciatic functional index. Histopathological changes were evaluated by hematoxylin and eosin staining. The phosphorylation of protein kinase B (Akt) and mammalian target of rapamycin (mTOR) was examined by western blotting. The expression of Akt and mTOR was also evaluated by immunofluorescence staining.
Results
Octanol alleviated the CCI-induced mechanical and thermal hyperalgesia and sciatic functional loss. Additionally, octanol relieved the CCI-induced abnormal histopathological changes. Mechanistically, octanol inactivated the Akt/mTOR pathway in the mice with CCI.
Conclusion
In conclusion, octanol can alleviate CCI-induced peripheral neuropathic by regulating the Akt/mTOR pathway and might be a novel pharmacological intervention for neuropathic pain.
Keywords
Introduction
Neuropathic pain arises due to damage of somatosensory function in central (CNS) and peripheral nervous systems (PNS) and is clinically manifested by allodynia, hyperalgesia and paresthesia. 1 Toxicity, surgery and trauma are three critical risk factors for neuropathic pain. 2 As estimated, neuropathic pain affects 7-10% of the global population, and this incidence is increasing as the population ages. 3 Currently, the clinical intervention of neuropathic pain remains elusive and it represents a huge unmet medical need . 4 Neurons in the CNS are free of regenerative capacity, 5 whereas strong regenerative ability occurs in neurons of PNS, which can lead to functional recovery following injury.6,7 Therefore, identification of novel therapeutic strategies for treating neuropathic pain caused by peripheral nerve injury is urgently needed.
Protein kinase B (Akt) represents a serine/threonine kinase that phosphorylates and regulates many substrates involved in cell growth, angiogenesis and metabolism. 8 The mammalian target of rapamycin (mTOR) pathway is found to be intimately linked with the Akt signaling. mTOR includes mammalian target of rapamycin complex 1 (mTORC1) and mammalian target of rapamycin complex 2 (mTORC2). mTORC1 is activated by phosphorylated Akt, whereas mTORC2 activates Akt by phosphorylating Ser473. 9 mTORC1 can negatively regulate catabolic processes such as autophagy. It can also directly regulate protein synthesis. 10 In addition to mTORC1, mTORC2 mainly regulates cell proliferation and migration and cytoskeletal remodeling. 11 Phosphatase and tensin homolog (PTEN) is a lipid phosphatase negatively regulated by the Akt/mTOR signaling. Previous studies have shown that PTEN depletion can promote peripheral nerve regeneration after sciatic nerve injury.12–14 Additionally, activation of mTOR/Akt signaling is suggested to augment axon growth potential and thus promote recovery from dorsal root crush injury. 15 Moreover, maresin 1 (MaR1) and Schwann-like cell conditional medium (SCLC-CM) can activate the Akt/mTOR pathway to promote angiogenesis and enhance neurological function recovery after peripheral nerve injury.16,17 Thus, the Akt/mTOR pathway is associated with the pathogenesis of peripheral neuropathy.
Gap junctions are macromolecular structures that regulate intercellular communication. As reported, gap junctions are constituted by connexin proteins which are assembled as hexamers to form connexons. Octanol is found to serve as a blocker of gap junctions containing connexin 43 proteins . 18 Under pathological conditions, gap junction channels are activated in the spinal cord and result in chronic pain states including neuropathic pain. 19 Carbenoxolone, a gap junction antagonist, can relieve neuropathic pain induced by chronic constriction injury (CCI) of the sciatic nerve. 20 The gap junction inhibitor octanol can prevent the oxaliplatin-induced neurotoxicity on peripheral nerve myelinates fibers. 21 Moreover, octanol can enhance the phosphorylation of Akt in experimental cerebral ischemia/reperfusion models. 22 However, whether octanol exerts protective effects against peripheral neuropathy through modulation of the Akt/mTOR pathway remains unknown. The experimental CCI model is a classical animal model of peripheral neuropathy. 23 Thus, this study was designed to detect the roles of octanol and its related mechanisms in neuropathic pain using CCI models. We hypothesized that the analgesic effect of octanol may be mediated by the Akt/mTOR pathway. This study might provide a theoretical basis for clinical application of octanol in neuropathic pain caused by peripheral nerve injury.
Methods
Animals
C57BL/6J mice (male, body weight 23-25 g; Charles River Laboratories, Beijing, China) were housed under standard conditions (25 ± 1°C, 50 ± 5% humidity, a 12-h light/dark cycle) with free access to food and water. The mice were acclimated for 7 days under aforementioned standard conditions prior to the experiments, and randomly divided into the Normal, the Sham, the CCI, the CCI + Octanol-L, the CCI + Octanol-M and the CCI + Octanol-H groups. Each group 8 mice. The experimental protocols were granted approval from the Ethics Committee of Hubei Provincial Center for Disease Control and Prevention (No. 202310089) and abided by the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
The chronic constriction injury (CCI) operation was performed in rats in accordance with a previous study with slight modification. 23 The mouse model has also been established involving the exposure of the common sciatic nerve at the level of the mid-thigh level and loose ligation of nerve with three ligatures.24,25 Briefly, the mice were anesthetized by inhalation of a mixture of isoflurane (4% for anesthesia induction and 1% for maintenance) and oxygen. The anesthetic was delivered by a facemask connected to a gas anesthesia machine in turn connected to the oxygen source and equipped with a precision vaporizer. The mice were constantly monitored to avoid excess cardiac or respiratory depression and insufficient anesthesia. After anesthesia, the left thighs were shaved and sterilized with 70% alcohol and iodine solution. Then, a blunt incision was made on left paw biceps femoral muscle to expose the sciatic nerve, followed by careful removal of muscle and facia that were attached to sciatic nerve. The nerve was loosely ligated with 4-0 catgut at three different points with about 1 mm distance from each successive point. The ligation intensity is appropriate when the nerve is slightly depressed, but the blood supply of the sciatic nerve is not blocked under a microscope. The ligatures were made proximal to trifurcation of sciatic nerve. This was carefully performed to prevent the disruption of epineural blood supply. Thereafter, the wound was sutured layer by layer, and iodine solution was applied superficially on skin. Finally, the mice were recovered from surgery on a homeothermic blanket for 2 days before behavioral testing. Sham group was operated to expose the sciatic nerve without ligation. The mice showing obvious hind limb paralysis in both hind limbs after CCI operation were included in this study, while the mice showing unilateral limb paralysis were excluded from the study. No animal was excluded in this study.
Drug treatment
The mice were randomly divided into 6 groups: normal, sham, CCI, CCI + Octanol (20 mg/kg), CCI + Octanol (40 mg/kg) and CCI + Octanol (80 mg/kg) group (8 mice per group). The mice in the normal group were not subjected to any surgical procedure. In the sham-operated mice, the sciatic nerve was only exposed but not ligated. The mice in the CCI group received exposure and three loose ligations of the sciatic nerve. Octanol (chemical structure, Figure 1) obtained from Sigma-Aldrich (Shanghai, China) was dissolved in 10% DMSO (Sigma-Aldrich) and then administered intraperitoneally into CCI-induced mice. The pharmacological treatment started the day after CCI induction and lasted 14 days. Chemical structure of octanol.
Behavioral examination
Thermal sensitivity and mechanical hyperalgesia were carried out on day 0 before CCI induction and on days 1, 7, 14, and 21 post-surgeries by an experimental who was blinded to the different treatments. The mice were given 2 days to acclimate before the testing. The thermal sensitivity was determined using a Hargreaves radiant heat apparatus as previously described. 26 Briefly, the animals were placed on a glass surface at 30°C while loosely restrained under a Plexiglass cage (12 × 20 × 15 cm3) and allowed to acclimate for 1.5 h on the day of testing. To test the thermal sensitivity in the animals, a radiant thermal beam was directed from below to the mid-plantar surface of the left and right hind paw to evoke a withdrawal response. The latency of paw withdrawal responses (the time between the stimulus onset and paw withdrawal responses) was recorded. A cutoff time of 20 s was used to avoid damage to the skin. Each hind paw was stimulated three times with an interval of at least 3 min and the three latencies obtained from each paw were averaged. Mechanical hyperalgesia of the left hind paw was assessed using Randall Selitto test apparatus. 27 Briefly, the left hind paw of was placed on the platform of Randall Selitto test apparatus and pedal switch operated to increase the pressure on the left hind paw. The withdrawal of left hind paw or vocalization response was recorded to assess the mechanical nociceptive threshold. To avoid tissue damage, a cut-off pressure of 450 g was maintained.
Assessment of sciatic functional index (SFI)
The physiological function of sciatic nerve was estimated by walking track analysis. 28 Briefly, the trials were done in an 8.2 × 42 cm corridor that was darkened at one end and covered with a sheet of white paper. The mouse hind paws were inserted in blue ink, and the animals were allowed to walk freely in the corridor. The following lengths were obtained: print length (PL), toe spread (TS), and intermediary toe spread (ITS). All the three measurements were taken from the experimental (E) and normal (N) sides. The factors were calculated as follows: print length factor (PLF) = (EPL-NPL)/NPL; toe spread factor (ITF) = (ETS-NTS)/NTS; intermediary toe spread factor (ITF) = (EIT-NIT)/NIT. These factors were then incorporated into the SFI formula: SFI = −38.3 × PLF+13.3 × ITF-8.8. An SFI of 0 is normal and of −100 indicates total impairment.
Histopathological evaluation
On day 21 after the behavioral procedures, the mice were anesthetized with 2.5% isoflurane and then transcranially perfused with 40 mL precooled precooled phosphate buffered saline (PBS) and 40 mL 4% cold paraformaldehyde. Subsequently, the L4-6 segments of the spinal cord were immediately dissected out. The spinal cords were then fixed in 4% paraformaldehyde at 4°C for 48 h, dehydrated in 30% sucrose at 4°C overnight, embedded in paraffin, and cut into 5-μm sections. Thereafter, the sections were sequentially immersed in xylene and gradient ethanol. After being washed with distilled water, the deparaffinized sections were stained with hematoxylin (Sigma-Aldrich) for 5 min, soaked in deionized water for 5 min, and stained with eosin (Sigma-Aldrich) for 1 min. The images were taken using a light microscope (Olympus, Tokyo, Japan).
Western blotting
The lumbar 4-6 superficial dorsal horn were homogenized in RIPA lysis buffer (Amylet Scientific, Wuhan, China) containing phenylmethylsulfonyl fluoride (MedChemExpress). An Enhanced BCA Protein assay kit (Yeasen) was used to determine the protein concentration. Protein (30 μg) were separated by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and then transferred onto polyvinylidene fluoride (PVDF) membranes. After being blocked using 5% skimmed milk., the membranes were incubated overnight with primary antibodies against mTOR (ab32028, 1:2500; Abcam), phosphorylated AKT (ab108266, 1:3000; Abcam), phosphorylated mTOR (ab109268, 1:5000; Abcam), GAPDH (ab9485, 1:2500; Abcam) and AKT (ab233755, 1:2000; Abcam) at 4°C, followed by incubation with secondary antibodies for 2 h at room temperature. The blots were developed using enhanced chemiluminescence (Yeasen) and imaged using the chemiluminescence detection system (Bio-Rad, Hercules, CA, USA).
Immunofluorescence staining
The superficial dorsal horn (L4-6) segments were fixed overnight with 4% paraformaldehyde, dehydrated in 30% sucrose in PBS at 4°C, embedded in paraffin and cut into 5-μm thick sections in a cryostat. The sections were then blocked with 5% bovine serum albumin for 1 h at 37°C. After being washed with PBS, the sections were incubated with anti-Akt (ab175354, 1:100; Abcam) and anti-mTOR (ab25880, 1:200; Abcam) primary antibodies at 4°C overnight. Afterward, the sections were washed with PBS and incubated with FITC-conjugated anti-rabbit secondary antibodies (ab150077, 1:500; Abcam) for 1 h at 37°C. The nuclei were counterstained with DAPI (Sigma-Aldrich) and washed thrice with PBS. Images were captured using a fluorescence microscope (Olympus). The fluorescence intensity was calculated using the Image Pro Plus software.
Statistical analysis
The data were analyzed using GraphPad Prism 8 (GraphPad Software, San Diego, CA, USA) and expressed as the mean ± standard deviation. All experiments were independently repeated in triplicate. The normality distribution was evaluated using the Kolmogorov-Smirnov Test of Normality. The p-value >0.05 indicated that the assumption of normality of data was consistent, and thus further testing could be performed. The data from the behavioral results were statistically analyzed by two-way analysis of variance followed by Bonferroni correction post hoc tests. Alterations of expression of the proteins detects among groups were tested with one-way analysis of variance followed by Tukey’s multiple range tests. A p-value <0.05 was considered statistically significant.
Results
Octanol alleviates the CCI-induced hyperalgesia
To investigate the effect of octanol on the development of neuropathic pain symptoms, we assessed the behavioral responses of mice with CCI. Thermal hyperalgesia was assessed using Hargreaves’s test. The results revealed that compared with the normal group, sciatic nerve ligation led to significant decreases in the paw withdrawal latency, and the latency reached its lowest point on the 14th day post-surgery and maintained at this value for the rest of testing period (p < 0.01). However, intraperitoneal injections of octanol at the dose of 20 mg/kg did not obviously affect the paw withdrawal latency in CCI mice (p > 0.05). By contrast, 40 mg/kg (p < 0.05) and 80 mg/kg (p < 0.05 on Day 14 and p < 0.01 on Day 21) octanol significantly rescued the decreased paw withdrawal latency dose-dependently (Figure 2(a)). Then, response toward mechanical hyperalgesia was also tested. The results demonstrated that compared with the normal group, the CCI group displayed significant mechanical allodynia (p < 0.01), whereas treatment with octanol at doses of 40 mg/kg (p < 0.05) and 80 mg/kg (p < 0.05 on Day 7 and p < 0.01 on Days 14 and 21) after CCI resulted in a significant suppression of mechanical allodynia compared to the CCI group in a dose-dependent manner (Figure 2(B)). These results show that octanol can effectively attenuate the CCI-induced thermal and mechanical hyperalgesia at doses of 20 mg/kg, 40 mg/kg, and 80 mg/kg. Octanol alleviates the CCI-induced hyperalgesia. (a) The thermal hyperalgesia was examined using Hargreaves’s test. (b) The mechanical allodynia was examined via paw withdrawal mechanic threshold. Data were expressed as mean ± standard deviation (n = 8 mice per group). Statistical signaling was determined by two-way analysis of variance followed by Bonferroni correction post hoc tests. **p < 0.01 compared with the normal group; #p < 0.05, ##p < 0.01 compared with the CCI group.
Octanol attenuates the CCI-induced increase in the SFI value
The results of walking track analysis demonstrated that compared with the normal group, the level of SFI was significantly reduced on Day 7 and reached the lowest level on Day 14 postoperatively (p < 0.01), whereas administration of octanol at the doses of 40 mg/kg (p < 0.05) and 80 mg/kg (p < 0.01) significantly reversed the suppressive effect of CCI operation on SFI value dose-dependently (Figure 3(a)). These results suggest that octanol can recover the sciatic functional loss. Octanol attenuates the CCI-induced increase in the SFI value. (A) The recovery of locomotive function was assessed by calculating the SFI. Data were expressed as mean ± standard deviation (n = 8 mice per group). Statistical significance was determined by two-way analysis of variance followed by Bonferroni correction post hoc tests. **p < 0.01 compared with the normal group; #p < 0.05, ##p < 0.01 compared with the CCI group.
Octanol alleviates the CCI-induced abnormal histopathological changes
As hematoxylin and eosin staining revealed, the sham and normal groups displayed normal structure of the sciatic nerve, and we could hardly observe Schwann cells and inflammatory cell infiltration. However, after CCI operation, inflammatory cell infiltration and proliferation of a large number of Schwann cells were observed. Additionally, inflammation-induced edema between the nerve fibers, rupture of myelin sheaths and loss of myelinated nerve fibers were also observed in the CCI group. After treatment with 20 mg/kg or 40 mg/kg octanol, we observed a dose-dependent decrease in the number of inflammatory cells and Schwann cells, but the nerve fibers are still separated in some areas. By comparison, in the 80 mg/kg octanol group, in addition to reduced inflammatory cells and Schwann cells, apparently normal nerve fascicle consisted mainly of myelinated nerve fibers and nuclei of the neurolemmocytes in endoneurial areas and regular appearance of the nerve fascicle were seen, but the morphology of sciatic nerve tissue structure did not return to normal (Figure 4(a)). Octanol alleviates the CCI-stimulated abnormal histopathological changes. (a) Histopathological changes were examined by hematoxylin and eosin staining. N = 4. Scale bar = 20 μm.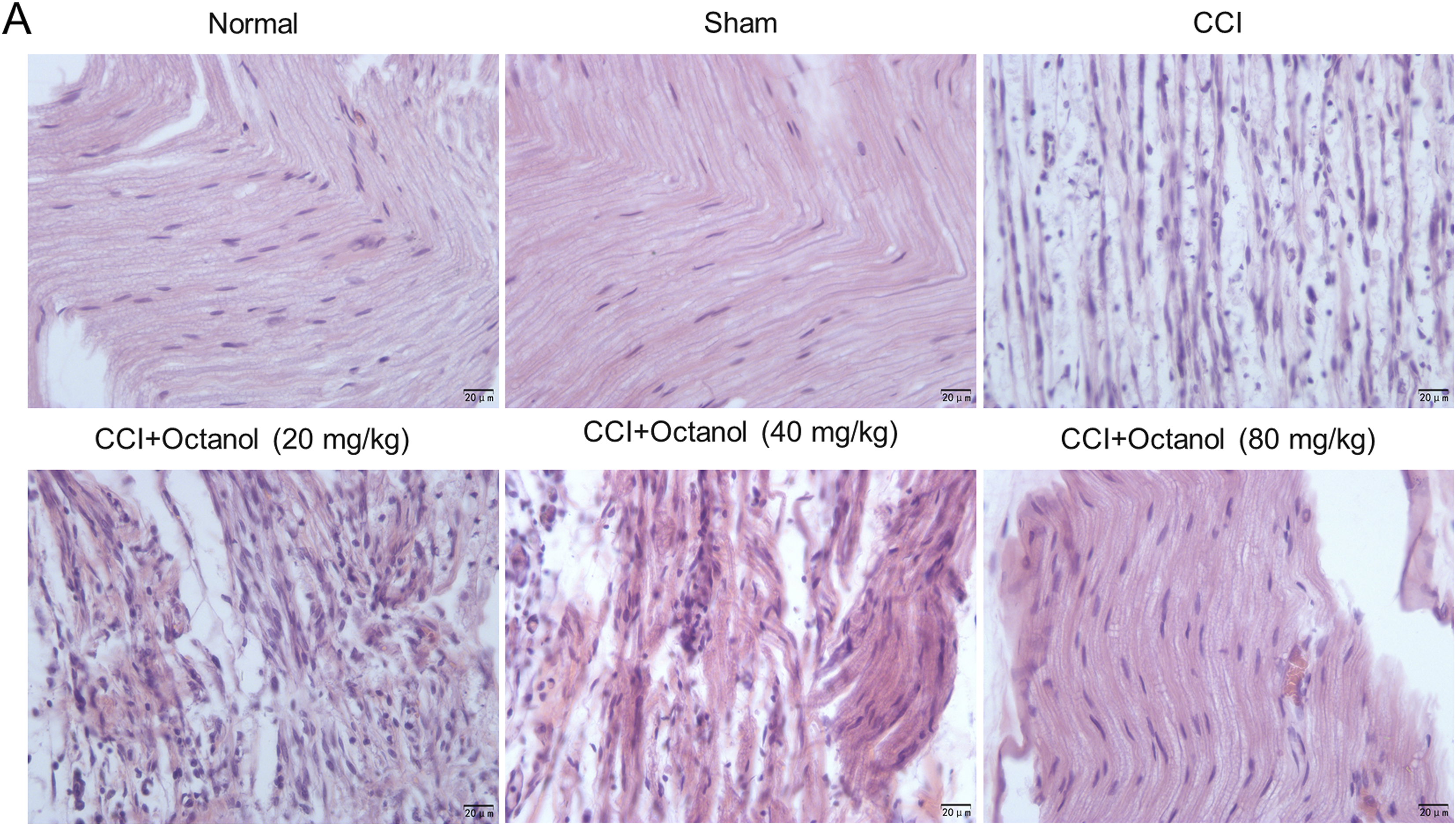
Octanol inactivates the Akt/mTOR pathway in mice with CCI
As shown by western blotting, compared with the normal group, the protein levels of phosphorylated Akt and phosphorylated mTOR were markedly elevated after CCI operation (p < 0.01), which were significantly reduced by 40 mg/kg (p < 0.05) and 80 mg/kg (p < 0.01) octanol dose-dependently in CCI mice (Figure 5(a)). The results of immunofluorescence staining revealed that the CCI group exhibited significantly higher levels of Akt and mTOR than the normal group (p < 0.001), whereas 40 mg/kg and 80 mg/kg octanol reversed the enhancing effect of CCI operation on the expression of Akt and mTOR in a dose-dependent manner (p < 0.01) (Figure 5(b) and (c)). These results indicate that octanol blocks the activation of Akt/mTOR signaling after CCI operation. Octanol abolished the enhancing effect of CCI on the phosphorylation of Akt and mTOR. (a) The protein levels of phosphorylated Akt and mTOR were measured by western blotting. (b)-(c) Immunofluorescence staining of Akt and mTOR. Data were expressed as mean ± standard deviation (n = 4 mice per group). Statistical significance was determined by one-way analysis of variance followed by Tukey’s multiple range tests. **p < 0.01, ***p < 0.001 compared with the normal group; #p < 0.05, ##p < 0.01, ###p < 0.001 compared with the CCI group.
Discussion
Peripheral neuropathy is featured by abnormal sensory abnormalities such as stimulus-independent persistent pain or sensory perception including hyperalgesia and allodynia. 29 There is no effective treatment for peripheral neuropathy, and currently available agents always have severe adverse effects. Therefore, a better understanding of the molecular mechanisms underlying neuropathic pain induced by peripheral nerve injury and development of novel effective therapeutic agents for treatment of chronic pain are urgent.30–32 In the current study, we investigated the roles of octanol in CCI-induced neuropathic pain and also detected the associated mechanisms of octanol pain relief.
Gap junctions are specialized intercellular communication channels. The functions of gap junctions, especially those constructed of connexin 43, are enhanced after nerve injury. 33 Accumulating evidence has suggested that inhibiting the function of gap junctions by decreasing connexin 43 expression may attenuate neuropathic pain.34,35 The gap junction inhibitor carbenoxolone has been found to relieve neuropathic pain induced by CCI by suppressing neuroinflammatory responses.20,33,36 Thus, we hypothesized that octanol, a gap junction inhibitor that has been confirmed to suppress gap junction-dependent cell-cell communication,37–39 also has the analgesic effect on CCI-induced neuropathic pain like the Gap junction inhibitor carbenoxolone. It is reported that octanol is an active ingredient of frankincense oil extract which has analgesic activities. 40 Inhibition of tetrodotoxin-resistant Na + currents can effectively suppress neuropathic pain. 41 Octanol is suggested to inhibit tetrodotoxin-resistant Na+ currents in dorsal root ganglion cells. 42 Here, we found that octanol alleviated the CCI-stimulated thermal hyperalgesia, mechanical hyperalgesia, and sciatic function loss. Additionally, administration of octanol also reduced the number of inflammatory cells and Schwann cells in mice with CCI and partially recovered the morphology of sciatic nerve tissue structure.
Akt, phosphorylating various substrates that participate in cell growth, survival, metabolism and proliferation, is involved in peripheral neuropathy pathogenesis. 43 The main target of Akt involved in cell growth is mTORC1, whereas mTORC2 regulates cell survival and metabolism through phosphorylation and activation of Akt. 44 Inactivation of Akt/mTOR pathway can attenuated neuropathic pain and pain related hypersensitivity after peripheral nerve injury.45,46 Previous studies have shown that inhibition of Akt and mTOR phosphorylation can result in connexin43 suppression.47,48 Here, we found that CCI induction led to the activation of Akt/mTOR signaling, which was similar to previous findings,49,50 whereas administration of octanol, a blocker of gap junctions containing connexin 43 proteins, blocked their activation in CCI mice.
There are limitations to this study. First, the major limitation is that our CCI-induced neuropathic model dose not duplicate the lesion phenocopy in females. Second, more evidence is needed to detect the function of the Akt/mTOR signaling. Third, more biological functions of octanol in peripheral neuropathic are required to be detect.
In conclusion, this study demonstrates that octanol can alleviate the CCI-induced peripheral neuropathy in mice by regulating the Akt/mTOR pathway. Octanol may serve as a potential agent for the treatment of peripheral neuropathic.
Footnotes
Acknowledgments
The authors appreciate the help of The Central Hospital of Wuhan, Tongji Medical College, Huazhong University of Science and Technology.
Author contributions
Biquan Deng and Hui Zou conceived and designed the experiments. Keli Hu, Hui Zou, Yunlu Liu, Achao Han and Biquan Deng carried out the experiments. Biquan Deng and Hui Zou analyzed the data. Biquan Deng, Keli Hu and Hui Zou drafted the manuscript. All authors agreed to be accountable for all aspects of the work. All authors have read and approved the final manuscript.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
