Abstract
Background:
Painful diabetic peripheral neuropathy impairs quality of life and can be difficult to treat.
Objective:
To discuss current treatment recommendations for painful diabetic peripheral neuropathy.
Study design:
Literature review.
Methods:
Systematic review of the literature discussing treatment of painful diabetic peripheral neuropathy. Existing treatment guidelines were studied and compared.
Results:
Painful diabetic peripheral neuropathy occurs in about one in six people with diabetes. This condition impairs quality of life and increases healthcare costs. Treatment recommendations exist, but individual patient therapy can require a trial-and-error approach. Many treatment options have adjuvant benefits or side effects which should be considered prior to initiating therapy. Often, a combination of treatment modalities with various mechanisms of action is required for adequate pain control. Adequate medication titration and a reasonable trial period should be allowed.
Conclusion:
The treatment of painful diabetic peripheral neuropathy can be challenging, but effective management can improve patient’s quality of life.
Clinical relevance
Painful diabetic peripheral neuropathy impairs quality of life and can be difficult to treat. Many treatment options have adjuvant benefits or side effects which should be considered prior to initiating therapy. Often, a combination of treatment modalities with various mechanisms of action is required for adequate pain control.
Background
Diabetes affects more than 25 million people, in the United States representing greater than 11% of the adult population. 1 Up to two-thirds of people with diabetes develop peripheral neuropathy, with an increasing prevalence with longer disease duration; 15%–25% of diabetic neuropathies are painful.2 –6 Patients with painful neuropathies have healthcare costs three times greater than age matched controls. 7 Diabetic peripheral neuropathy (DPN) and its sequelae, including ulceration and loss of limbs, cost the United States between 5 and 15 billion US dollars per year. 8 Additionally, painful DPN significantly affects multiple measures of quality of life and is highly associated with depression, anxiety, and sleep disturbances.2,9 –11
Developing an individualized treatment plan for a person with painful DPN is challenging. 12 Choosing a treatment can be difficult because of a lack of comparative trials of effective treatments13,14 and limited or conflicting evidence for some treatments. 13 Once a treatment is initiated, a patient can experience incomplete pain relief13,15 or suffer from the high side-effect profiles seen with many of the effective treatments.13,16
Over the past decade, the treatment of painful peripheral neuropathies has evolved, and several treatment algorithms have been established (Figure 1).16 –20 Each of these guidelines considers treatment efficacy and adverse effects. The Canadian and the International Association for the Study of Pain Neuropathic Pain Special Interest Group (NeuPSIG) guidelines include ease of use and cost-effectiveness, while the NeuPSIG and the American Academy of Neurology (AAN) also consider quality-of-life outcomes. One of the guidelines is strictly for painful DPN, 20 and three others include specific subgroup recommendations for DPN.16,18,19
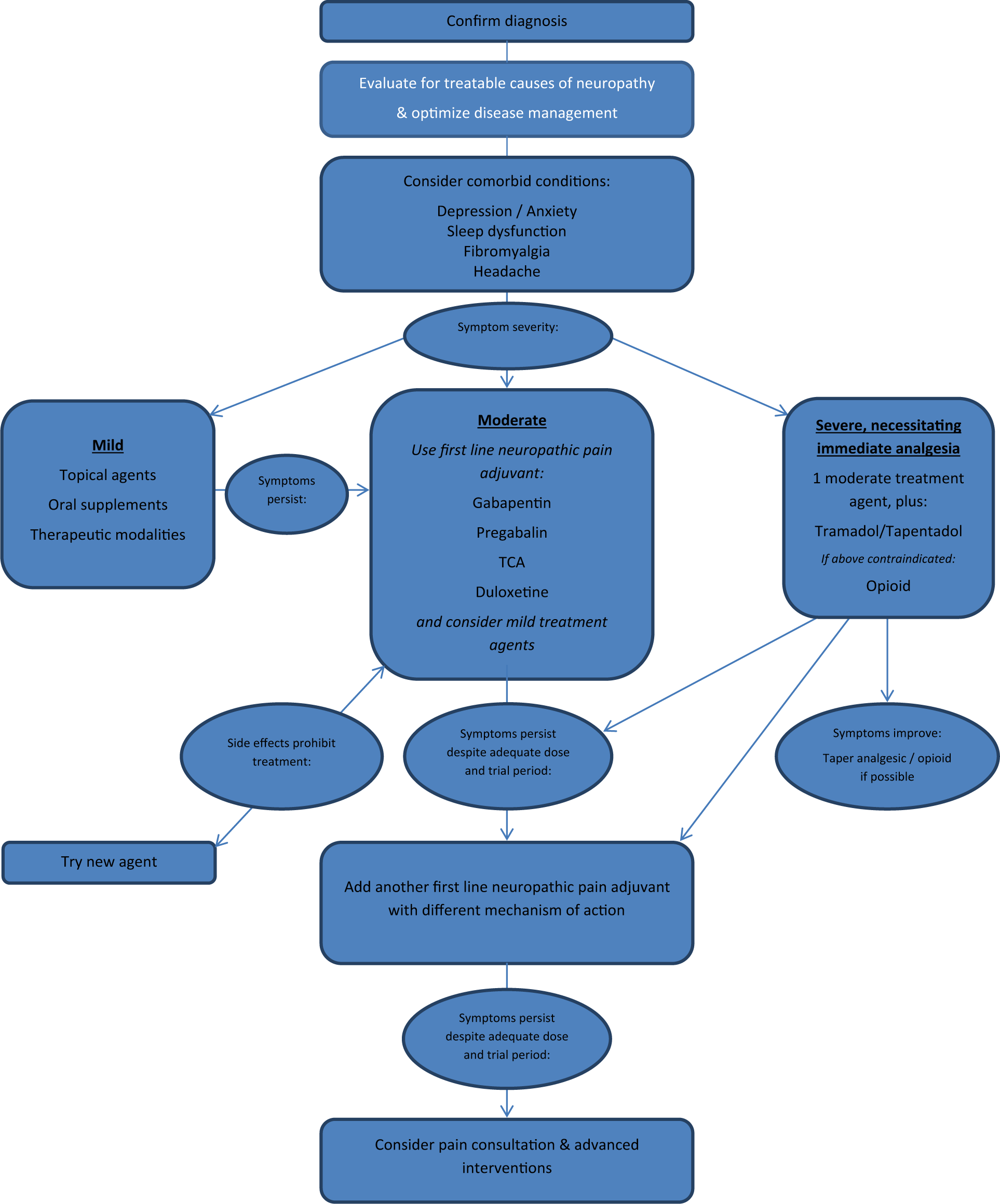
Treatment algorithm for painful diabetic peripheral.
Disease management
The first step in the management of painful DPN is treating the cause of neuropathy, the diabetes. There is evidence that tight glucose control, particularly in the early stages of diabetes, can delay progression of painful DPN. 21 A recent Cochrane review showed a relative risk of developing DPN of 0.35 for patients who undergo intensive glucose control compared to conventional treatment (95% confidence interval (CI) = 0.23–0.53; p < 0.00001; I 2 = 0%; 1203 participants; three trials). 22 Neuropathic pain symptoms may improve with tight glycemic control; however, if this occurs, the reversal of symptoms is limited and the primary goal of disease treatment is to prevent or slow progression. Comorbid conditions such as depression, anxiety, sleep impairment, and renal dysfunction can affect pain and should also be addressed.11,13,23,24
Medications
Careful consideration of several patient factors can improve success in medical therapy of painful DPN. First, the patient’s medical comorbidities should be considered. Limiting factors such as pulmonary, cardiovascular, renal, or hepatic impairment should be identified as these may influence medication choice. In certain patients, physiologic testing (such as electrocardiogram (EKG) prior to initiating tricyclic antidepressants (TCAs)) or laboratory testing (such as creatinine or liver function tests) may be required prior to medication initiation. Next, comorbid conditions which might benefit from the same medication used to treat the painful DPN, such as insomnia, depression, fibromyalgia, or headache, should be noted as treating two processes with one medication would be preferable to initiating separate treatments. Finally, the medication side-effect profile should be considered, and the risk-to-benefit ratio weighed.
Neuropathic pain medications rarely resolve pain. However, even a modest to moderate decrease in the patient’s reported pain level can be clinically meaningful and improve a patient’s quality of life. An improvement of 30% or 2 points on a 0–10 Likert scale has been found to be clinically meaningful to patients. 25 The practitioner and the patient should discuss expectations and treatment goals to maximize outcomes. When a neuropathic pain medication is tolerated and partially effective, but insufficient to improve function, a second agent with a distinct mechanism of action should be added. Many studies show an additive benefit when combining medications from different classes.26–28 Most treatment guidelines recommend adding an additional agent rather than transitioning from one agent to another, particularly if some benefit was achieved from the initial medication.18,19
TCAs
Mechanism
TCAs prevent the reuptake of norepinephrine and serotonin; antagonize N-methyl-
Additional benefits
TCAs are approved for the treatment of depression by the US Food and Drug Administration (FDA), though dosing needed for therapeutic benefit in painful DPN is usually lower than that required for the treatment of depression. 29 Additional off-label uses include insomnia, chronic myofascial pain, fibromyalgia, migraine headache, irritable bowel syndrome, and smoking cessation.30 –32
Side effects
The most common TCA side effects are dry mouth, decreased gastrointestinal (GI) motility, orthostatic hypotension, weight gain, urinary retention, and cognitive dysfunction. 33 Sedation is common and nighttime dosing may improve sleep and limit daytime drowsiness. Tolerance to side effects develops, and a low starting dose with gradual up-titration is recommended. 19 The most severe side effects are cardiac dysrhythmias, and an EKG to screen for conduction system disease in elderly or at-risk patients is recommended prior to initiation of therapy.34,35 As with other antidepressants, there is a slightly increased risk of suicidal thoughts or actions in young adults. 36 TCAs confer a risk of serotonin syndrome when used with other medications that affect serotonin (other antidepressants, tramadol, tapentadol, some antiemetics) and should be used with caution at lowered goal dosages when used in combination with other serotonergic agents. TCAs can also lower the seizure threshold. 37
DPN evidence
All of the medication treatment guidelines include the TCAs as first-line medications unless contraindicated, with one exception. 20 TCAs have robust evidence supporting analgesic efficacy in painful neuropathies compared to placebo. 12 Head-to-head trials have shown equal analgesic efficacy to gabapentin19,26,38
Serotonin–norepinephrine reuptake inhibitors
Mechanism
Serotonin–norepinephrine reuptake inhibitors (SNRIs) affect the balance of these central nervous system neurotransmitters, which is thought to promote anti-nociception and reduce pro-nociception. 39 While the primary mechanism of action is similar to TCAs, dosing is generally less limited by anticholinergic, antiadrenergic, or muscarinic side effects.
Additional benefits
Duloxetine is a balanced SNRI (blocking serotonin and norepinephrine equally) that is approved by the FDA for painful DPN, fibromyalgia, chronic musculoskeletal pain, depression, and anxiety.40,41 A potential advantage of duloxetine is that the therapeutic dosages are similar for each condition such that it can be used to treat two comorbid conditions without dosage adjustment. Other SNRIs, including venlafaxine, may have similar neuropathic pain treatment effects, but have not been as rigorously studied and are only FDA approved for depression and anxiety.
Side effects
Common SNRI side effects include GI upset, headache, and insomnia. Unique side effects with duloxetine are hyperhidrosis and a possible increase in bleeding risk.42,43 Side effects are typically less bothersome than those seen with TCAs. 41 As with other antidepressants, there is a slightly increased risk of suicidal thoughts or actions in young adults. 36 Like TCAs, SNRIs also decrease the seizure threshold, and their serotonergic effect gives a risk of serotonin syndrome. 44
Evidence
Two of the above guidelines list SNRIs as first-line treatment options.16,18 Studies have suggested similar efficacy of duloxetine to amitriptyline, with a slightly better side-effect profile. 45
Calcium channel blockers: gabapentin and pregabalin
Mechanism
Gabapentin and pregabalin block the presynaptic calcium channel at the α2δ ligand, inhibiting release of excitatory neurotransmitters. Additionally, they may antagonize NMDA receptors associated with pain signaling.46,47
Additional benefits
Pregabalin is FDA approved for the treatment of painful DPN, post-herpetic neuralgia, central neuropathic pain, and fibromyalgia. 48 Gabapentin is FDA approved for post-herpetic neuralgia but is also commonly used for other neuropathic pain states. 49 Other benefits of the gabapentanoids are improved sleep 50 and possibly anxiolysis. 51
Side effects
Common gabapentin and pregabalin side effects include cognitive slowing, drowsiness, dizziness, and peripheral edema. 17 Pregabalin can cause euphoria in a very small percentage of patients, which may be a consideration in patients with psychiatric comorbidities. 52 As both gabapentin and pregabalin are renally excreted, dosage adjustment is required in the setting of renal impairment. There are minimal significant medication interactions. As with antidepressants and other antiepileptic medications, there is a slightly increased risk of suicidal thoughts or actions in young adults. 49
Evidence
Gabapentin has been shown to be as efficacious as the TCA nortriptyline. 26 Gabapentin or pregabalin are recommended as first-line medications in the majority of the above guidelines17 –20 with one guideline giving preference to pregabalin over gabapentin or antidepressants, 20 and another as a second choice if TCAs are contraindicated. 16 One guideline recommends earlier-generation anticonvulsants first; however, gabapentin and pregabalin’s minimal drug interactions and reduced requirement for monitoring can be advantages over other anticonvulsants. 53
Opioids, including tapentadol and tramadol
Mechanism
Opioid medications bind to µ and other opioid receptors on neuronal cell membranes, modulating descending inhibitory and ascending excitatory pain mechanisms. Their primary analgesic targets are in the spinal cord and elsewhere in the central nervous system, but receptors throughout the body are responsible for analgesia and opioid-related side effects.
Tapentadol is a partial µ agonist with a norepinephrine reuptake inhibitory effect. 54 Tramadol is also a partial µ receptor agonist and has both serotonin and norepinephrine reuptake inhibition.55,56
Additional benefits
The main therapeutic benefit of opioid medications is analgesia. Opioid medications should not be used for insomnia, anxiolysis, or other indications.
Tapentadol is FDA approved for painful DPN. 57 It is postulated that the combined µ receptor activation and norepinephrine reuptake inhibition of tramadol and tapentadol offers additional benefit compared to the single-mechanism agents.58,59
Side effects
A primary concern in the prescription of opioid medications is their potential for misuse. Opioids activate the brain’s mesolimbic reward center, resulting in feelings of pleasure which can lead to craving and compulsive use. 60 Opioids are the most commonly misused prescription medication in the United States and are responsible for the majority of prescription drug–related acute hospital admissions and drug treatment center admissions. 61 Tolerance and physical dependence, resulting in the need for gradually increasing dosage as well as withdrawal symptoms if the medication is decreased or discontinued, are an inherent physiologic response to the medication and should not be confused with addiction. Addiction involves craving and compulsive use despite harm. 62
The most serious side effect of opioid medications is respiratory depression. The effect can be additive with other substances such as benzodiazepines and alcohol. Other side effects include nausea, vomiting, pruritus, constipation, sedation, and dizziness. Overdose or accumulation of breakdown products can produce myoclonus and contribute to sedation and respiratory depression. Over time, immune and endocrine dysfunction, including hypotestosteronism, occur. Another potential issue with chronic opioid use is opioid-induced hyperalgesia, a state of increased sensitivity to pain and worsening of pain while receiving opioids. This can be particularly problematic in patients who already struggle with pain issues. 63 Opioids should be used with caution in renal and hepatic impairment, particularly with extended or continuous release formulations.64,65
Tramadol and tapentadol can contribute to serotonin syndrome and can lower the seizure threshold. They should also be used with caution in patients with renal and hepatic impairment and in those receiving other medications with serotonergic effects.66–68
Evidence
There is evidence that opioid medications are effective in painful DPN,69 –71 including tramadol55,56 and tapentadol. 72 Notably, most studies were carried out with extended or continuous release formulations.55,69,70,72 Opioids are included in the treatment algorithms as later-tier choices due to issues with long-term use and risks. 20 Their later-tiered position in the treatment algorithms is large part related to the potential adverse effects and chronic use risks inherent with opioids. However, they may be useful in appropriately selected and monitored patients. In a crossover trial for post-herpetic neuralgia, patients preferred extended release morphine to nortriptyline. 73
There are multiple established guidelines for opioid prescribing risk mitigation. 74 Similar to other painful DPN treatments, an opioid trial should include pre-determined goals for pain and functional improvement. A treatment agreement should be agreed upon and maintained, and regular screening for aberrant use, abuse, misuse, and diversion should be completed, including urine drug screening to confirm appropriate medication and breakdown products and to screen for other medication or substance use that could increase the patient’s treatment-related risk. 74
Topical agents
Topical agents carry the advantage of limited systemic side effects, making them excellent first-line or adjuvant pain medications. Topical creams and gels can cover a wide area. Patches can be less practical given their limited size and the typical distribution of DPN involving the feet.
Topical capsaicin, including high-dose patches (Qutenza)
Mechanism
Capsaicin is an active component of chili peppers. It binds to the TRPV1 heat-activated calcium channel and causes it to open at normal body temperature. Prolonged activation of the receptor results in the depletion of neurotransmitters, particularly substance P, and reduces the ability of neurons to transmit pain signals. Standard capsaicin creams come in concentrations of 0.025%–0.25%. They must be applied multiple times per day for several weeks before analgesic effects become significant. Qutenza is a capsaicin patch with an 8% concentration. It is applied after appropriate local analgesia with topical lidocaine and left in place for 60 minutes. A single application of the Qutenza patch may provide up to 3 months of pain relief.
Side effects
Skin irritation is the most common side effect and many patients do not tolerate capsaicin treatment.75,76 During and after administration of the 8% capsaicin patch, pain can be significant (like a severe sunburn) and may require medical management with opioids for a number of days following application. 77 Extreme caution must be used to avoid contact of any concentration of capsaicin with the eyes or mucous membranes.
Evidence
A meta-analysis of studies reported a pooled 2.35 odds ratio in favor of capsaicin over placebo (95% CI = 1.48–3.22). 12 High-dose capsaicin was most effective in patients with post-herpetic and human immunodeficiency virus–related neuropathies. 78 Qutenza is FDA approved for post-herpetic neuralgia.
Lidocaine, including patches
Mechanism
Lidocaine blocks voltage-gated sodium channels, which stops propagation of action potentials in neurons. 79 A 5% concentration patch is available.
Side effects
With 12 h of continuous wear of the 5% patch, only 3% of the total dose is absorbed, making systemic side effects unlikely. 80 Skin irritation is the most common side effect. 80
Evidence
The lidocaine 5% patch is FDA approved for post-herpetic neuralgia. The patch has been found to reduce pain and improve quality of life in DPN. 81 Lidocaine patches were found to give a similar degree of benefit as pregabalin to patients with DPN, as well as additional benefit when used in conjunction with pregabalin. 82
Other topical agents
Menthol
Menthol transiently activates TRPM8 ion channels, causing an endogenous cooling-induced analgesia. 83 There are no randomized controlled trials for neuropathic pain; case reports and small studies have shown some benefits.84,85
Amitriptyline, ketamine and baclofen
These medications are typically compounded in a cream or pluronic lecithin organogel base for topical application. Studies show no systemic toxicities, but have mixed results, at best, for the treatment of neuropathic pain.86 –89
Other supplements
Alpha-lipoic acid
A lipophilic antioxidant, alpha-lipoic acid (ALA), has been shown to improve nerve blood flow and distal conduction and to reduce oxidative stress. 90 The primary side effect is nausea. Both oral (at a dosage of 600 mg per day) and intravenous supplementation have been shown to improve symptoms of painful DPN.91,92
Acetyl-l -carnitine
Acetyl-
Prescribing recommendations for medications for painful diabetic peripheral neuropathy.

SSRI: selective serotonin reuptake inhibitor; SNRI: serotonin and norepinephrine reuptake inhibitors; TCA: tricyclic antidepressant.
Based on information from Dworkin et al. 18 .
Therapeutic modalities
Acupuncture
Acupuncture has been shown to be effective for painful DPN in small trials without placebo control.97 –99
Transcutaneous electrical nerve stimulation
Transcutaneous electrical nerve stimulation (TENS) delivers an electrical current via electrodes on the skin arranged about the affected area. The intensity, frequency, and pattern of stimulation can be adjusted by the patient or therapist. Several studies including sham treatment arms have demonstrated benefit in painful DPN.100 –102
Cognitive behavioral therapy
Cognitive behavioral therapy (CBT) replaces dysfunctional thoughts and behaviors with positive ones and has been found effective in multiple pain states.103 –105 One randomized pilot study showed benefit for patients with painful DPN as compared to typical management. 106
Advanced interventions
In addition to medications, several interventions are gaining interest in the treatment of painful neuropathies. Interventional treatments have previously been described as “invasive procedures involving delivery of drugs into targeted areas, or ablation/modulation of targeted nerves.” 13 Recently, the NeuPSIG compiled evidence for interventional treatments in the management of neuropathic pain, including a subgroup analysis for painful DPN in combination with other painful peripheral neuropathies.
Neuromodulation
Neuromodulation involves the application of electrical current to neural structures to modulate neural transmission. The most common method of neuromodulation for pain is dorsal column stimulation, also known as spinal cord stimulation (SCS). Small electrodes are implanted in the epidural space and connected to a battery-operated pulse generator implanted in the flank or abdominal wall. Prior to implantation, a trial of stimulation with temporary percutaneous electrodes is undertaken to ensure effective pain relief. Several non-randomized case series have demonstrated pain relief of peripheral neuropathy even in medically resistant cases.107 –109 A single study with a crossover-type design showed benefit compared to sham stimulation. 108
For isolated mononeuropathies, placement of a traditional stimulator lead immediately adjacent to the affected nerve has been described. 110
Intrathecal drug delivery systems
Intrathecal drug delivery systems (IDDS) use a surgically implanted pump attached to a small, flexible catheter tunnelled into the cerebrospinal fluid (CSF). Morphine and ziconotide (a selective N-type voltage-gated calcium channel blocker) are FDA-approved medications for intrathecal infusion.111,112
There are currently no studies showing evidence for intrathecal drug delivery specifically for neuropathic non-cancer pain. Given concentrated drug delivery into the CSF, intrathecal systems may be useful when systemic side effects from oral analgesics or adjuvants are limiting.
Factors that increase the risk of IDDS implantation in diabetic patients include impaired wound healing, relative immunosuppression increasing the risk of infection, and comorbid cardiovascular or renal disease. If an intrathecal drug delivery system is being considered in a diabetic patient, it is recommended that their glucose control is optimized prior to implantation. 113 Patients with an intrathecal system need to be able to maintain regular follow-up for system refills and adjustments.
Conclusion
While painful DPN can prove difficult to treat, there are a variety of therapeutic options, including behavioral therapies, physical modalities, topical and oral medications, and implantable devices. There are several key concepts for developing a treatment approach.
Tight glycemic control is important to delay onset and progression of painful DPN. Consideration of comorbid conditions including sleep disturbance, depression, and other pain states is important both to minimize the pain of DPN itself and when selecting the most appropriate medical therapy. Many painful DPN treatments can also address other comorbid conditions. Furthermore, some medication side effects could exacerbate preexisting problems and should be avoided.
Managing expectations is vital when starting or changing medications. Most treatments are considered successful if they provide 30%–50% improvement or at least 2 points on a 10-point Likert scale. For some patients, this improvement will be worthwhile, while others may weigh the benefits less favorably relative to the side effects of the medication.
Medication side effects can be a barrier to treatment success. A frank conversation discussing common or serious side effects should be had prior to starting a new treatment. Many of these treatment side effects can be improved over time due to progressive tolerance; these can be mitigated at the start of treatment by gradual up-titration of the medication dose. Each medication should be maintained at the lowest effective dose.
For patients who have had prior medication trials to treat their painful DPN, it is important to ensure that their trial was adequate in dosage achieved and duration of treatment (several weeks at goal dosage). It is common that a prior trial was inadequate and that a retrial may be appropriate.
Finally, an important strategy to the management of painful DPN is multimodal therapy. Many studies show benefit from combination therapies. This strategy is likely effective due to the additive effects of pain relief from multiple mechanisms of action. It is important to carefully consider the individual treatments to avoid unpleasant or life-threatening side effects, such as serotonin syndrome with combinations of high-dose TCAs and tramadol. Topical medications, behavioral strategies, and physical modalities can be particularly beneficial in combination with other treatments due to their negligible side-effect profiles.
Footnotes
Author contribution
All authors contributed equally in the preparation of this manuscript.
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
