Abstract
Objective
Local infiltration analgesia (LIA), adductor canal block (ACB), and infiltration between the popliteal artery and the capsule of the posterior knee (IPACK) are popular multimodal analgesia techniques used during total knee arthroplasty (TKA). This study aimed to explore the efficacy of adding the IPACK technique to ACB and LIA in patients undergoing TKA.
Methods
In this retrospective cohort study, patients who underwent primary unilateral TKA were divided into two groups based on their date of admission. Sixty-three patients underwent IPACK, ACB and LIA (IPACK group) during surgery, while 60 patients underwent ACB and LIA (control group). The primary outcome was the postoperative administration of morphine hydrochloride as a rescue analgesic. Secondary outcomes included time to first rescue analgesia, postoperative pain assessed using the visual analog scale (VAS), functional recovery assessed by knee range of motion and ambulation distance, time until hospital discharge, and complication rates.
Results
The two groups were similar in average postoperative 0-to-24-h morphine consumption (11.8 mg for the control group vs 12.7 mg for the IPACK group, p = .428) and average total morphine consumption (18.2 mg vs 18.0 mg, p = .983) during hospitalization. There were also no significant differences in the secondary outcomes.
Conclusions
The addition of IPACK to ACB and LIA did not provide any clinical analgesic benefits. Orthopedic surgeons and anesthesiologists are justified in using ACB and LIA without IPACK for TKA.
Keywords
Introduction
Total knee arthroplasty (TKA) is a painful orthopedic procedure. 1 Inadequate pain management can delay recovery and affect physical, emotional, and social well-being.2,3 Multimodal analgesia can enhance postoperative analgesia by combining techniques with drugs having different mechanisms of action.4,5 Local infiltration analgesia (LIA) and peripheral nerve block are the primary multimodal analgesia technique during TKA. 5
Local infiltration analgesia is performed by injecting drugs with different analgesic mechanisms that target local sensory nerve endings and allow retention of maximal muscle strength.6,7 Peripheral nerve block is another commonly used analgesic modality.8–10 Presently, adductor canal block (ACB) is commonly used for postoperative analgesia after TKA, which preserves quadriceps strength.11–14 However, many patients who undergo ACB still experience severe posterior knee pain because it blocks only the anteromedial sensory nerve of the knee and not the posterior or lateral sensory nerves.15–18 To solve this problem, researchers have used ultrasound-guided infiltration between the popliteal artery and capsule of the posterior knee (IPACK). The IPACK technique can provide significant analgesia to the posterior knee. 19 In recent years, ACB+IPACK has become a popular combination of analgesic techniques after TKA.20–22
At present, the analgesic efficacy of ACB+IPACK remains controversial. Some studies have reported that the addition of IPACK to ACB can significantly improve analgesic and functional outcomes following TKA.20,23,24 However, other studies have reported that the ACB + IPACK block does not provide clinically significant improvement compared with ACB alone.25,26 The difference in the results may be due to the differences in the multimodal pain regimens of each medical center. In some studies, LIA was performed,20,25 but some studies did not include LIA.23,26,27 We speculate that the administration of LIA may affect the analgesic effects of ACB + IPACK. However, current clinical evidence is unclear as to whether adding IPACK to ACB can further enhance analgesia when LIA is performed. Therefore, we conducted this retrospective cohort study to explore whether the addition of IPACK to ACB and LIA could further enhance analgesia after TKA.
Methods
The Clinical Trials and Biomedical Ethics Committee of our institution approved this study. Before surgery, written informed consent was obtained from all the participants to allow their anonymized clinical data to be analyzed and published for research purposes.
Patients
Patients who underwent primary unilateral TKA between March 2021 and October 2023 at our institution were screened for eligibility. Patients were required to meet all of the following inclusion criteria: (1) primary unilateral TKA due to knee osteoarthritis, (2) age between 18 and 80 years, and (3) American Society of Anesthesiologists functional status I to III.
Patients were excluded if they had any of the following: (1) abnormal liver function, defined as levels of alanine aminotransferase and aspartate aminotransferase more than 1.5 times the upper limit of the reference range; (2) diabetes mellitus with poor blood glucose control and, in the investigators’ opinion, a high risk of infection; (3) history of knee surgery; (4) neuromuscular dysfunction affecting the lower extremities; (5) severe coagulation disorder; (6) severely unstable knee that was incurable with ligamentous retention TKA; (7) knee flexion deformity ≥ 30° or varus-valgus deformity ≥ 30°; (8) allergies to the drugs used in this study; (9) opioid dependency (defined as more than 100 mg morphine equivalent of opioids used weekly to control pain before surgery for more than 3 months); or (10) inability to report pain using a visual analogue scale (VAS) or other communication difficulties.
The patients were divided into two groups (control and IPACK) based on the date of admission. Patients admitted before July 8, 2022 received LIA + ACB (control group), whereas patients admitted after July 8, 2022, received LIA + ACB + IPACK (IPACK group).
Surgery and perioperative management
One senior surgeon performed all the TKAs in the study. Under general anesthesia, a midline skin incision was made using the medial parapatellar approach. Cemented prostheses were used. From the skin incision until wound closure, a pneumatic tourniquet was maintained at 240 mmHg. No drains were used after suturing the wound.
On the day before the procedure, celecoxib (200 mg) and pregabalin (150 mg) were orally administered twice as preemptive analgesics. Thirty minutes before the incision, intravenous cefazolin (2 g) was administered to prevent infection. Thirty minutes before the induction of general anesthesia, the same senior anesthesiologist performed a peripheral nerve block, including ACB alone or ACB + IPACK.
ACB
A high-frequency linear array ultrasonic transducer (Anesus ME7; Mindray, Shenzhen, China) was used to scan the midportion of the thigh (half the distance between the inguinal crease and patella) to identify the adductor canal, superficial femoral artery, sartorius, adductor longus, and adductor magnus. The anterolateral hyperechoic structure of the artery (saphenous nerve and nerve to the vastus medialis) was used as the injection target. A 21-gauge, 100-mm needle (Pajunk, Geisingen, Germany) was introduced in-plane lateral to medial, then 20 mL of local anesthetics (0.2% ropivacaine + 2.0 μg/mL epinephrine) was injected after ensuring the correct placement of the needle using 3 mL of isotonic saline.
IPACK
IPACK was performed using the same ultrasonic transducer. The anesthesiologist identified the popliteal artery at the popliteal crease and moved cephalad just beyond the femoral condyles, at the level where the condyles merge with the shaft of the femur. The tibial and peroneal nerves were visualized superficially to the popliteal artery. After identifying the space between the femur and popliteal artery, the needle was advanced in-plane from the medial to lateral direction. The tip was positioned in the middle of the femur near the lateral border of the periosteum. Subsequently, 5–10 mL of local anesthetic was injected to ensure adequate spread to the lateral end of the femur. Upon withdrawing the needle, the anesthesiologist further injected the rest of the ropivacaine along the femur, infiltrating 5 mL incrementally into the area between the artery and femur and finishing at the medial edge of the femur. IPACK involved a total of 25 mL of local anesthetics (0.2% ropivacaine + 2.0 μg/mL epinephrine).
All patients received standard general anesthesia. After 100% oxygen inhalation, the following anesthetics were administered intravenously: midazolam, 2 mg/kg; propofol, 2 mg/kg; sufentanil, 0.3 μg/kg; and cis-atracurium, 0.2 mg/kg. During surgery, the surgeon performed periarticular LIA with a cocktail of 0.2% ropivacaine, 2.0 μg/mL epinephrine, and 0.1 mg/mL dexamethasone. Prior to prosthesis implantation, 20 mL of the cocktail was injected into the posterior aspect of the capsule, and 20 mL of the cocktail was used as an infiltration analgesic for the medial and lateral collateral ligaments. After implantation, the quadricep and retinacular tissues were infiltrated with 20 mL of cocktail; the adipose and subcutaneous tissues were infiltrated with 40 mL of the cocktail. Before releasing the pneumatic tourniquet, intravenous tranexamic acid (15 mg/kg) was administered to reduce blood loss; intravenous sufentanil (5 μg) and dexamethasone (10 mg), to prevent postoperative pain; and intravenous tropisetron (5 mg), to prevent postoperative nausea and vomiting.
Following the patients’ recovery from general anesthesia, they were transferred to a bed and an ice compress was placed around the incision. After surgery, the patients received oral celecoxib (200 mg) and pregabalin (150 mg) twice daily. If the patient was unable to tolerate the pain (defined as whenever the pain score at rest was ≥ 4 or during motion was ≥ 6), 10 mg of morphine hydrochloride was injected subcutaneously as a rescue analgesic. Intravenous tranexamic acid (15 mg/kg) was administered at 3 and 6 h postoperatively, and intravenous cefazolin (2 g) was administered at 12 h postoperatively.
Outcome assessment
All outcome assessments were performed by a postoperative care team blinded to the group allocation.
Primary outcome
The primary outcome was postoperative consumption of morphine hydrochloride as rescue analgesia.
Secondary outcomes
Secondary outcomes included the time to first rescue analgesia, postoperative pain assessed using the visual analog scale (VAS), functional recovery assessed by knee range of motion and ambulation distance, time to hospital discharge, and complication rates.
Postoperative pain was measured using a visual analog scale (VAS) ranging from 0 to 10. 28 Pain was measured at rest or during motion when the patient was asked to flex the knee as much as possible. Pain was measured at 3, 6, 12, 24, 36, and 48 h postoperatively.
Functional recovery of the knee was measured in terms of range of motion (ROM) and daily ambulation distance. ROM was measured using a protractor, three times per day, 6 h apart, and the best value was used as the day’s value. Ambulation was initiated on the morning of postoperative day (POD) 1. During hospitalization, after breakfast, the patients were asked to walk the longest possible distance in one attempt with a walking aid, and the distance was measured as the daily ambulation distance.
The time from end of surgery to hospital discharge was recorded. The discharge criteria were as follows: adequate pain control with oral pain medication, independent transfer, independent ambulation for at least 60 m, and the ability to climb stairs.
Postoperative complications were recorded. Complications included nausea, vomiting, venous thrombotic events, nerve damage, wound complications (wound oozing and delayed wound healing), and postoperative falls.
Statistical analysis
All statistical analyses were performed using the Statistical Product and Service Solutions (SPSS) 26.0 (IBM Corp., Armonk, NY, USA). The normality of the data was assessed using histograms and quantile-quantile plots. Continuous data are presented as means and standard deviations. Categorical data are presented as numbers or percentages. Intergroup differences in normally distributed data were assessed for significance using the Student’s t-test; differences in skewed and ordinal data, the Mann–Whitney U test; and differences in categorical data, Pearson’s Chi-squared or Fisher’s exact probability tests. The time to first rescue analgesia and time to hospital discharge were analyzed using Kaplan–Meier survival analyses29,30 and log-rank tests. Differences were considered statistically significant at p < .05.
Results
Patient characteristics
Baseline demographic and clinical characteristics of patients.

Values are expressed as mean ± standard deviation or n. ASA, American Society of Anesthesiologists; ROM, range of motion; VAS, visual analogue scale.
aStudent’s t test.
bPearson’s chi-squared test.
cMann-Whitney U test.
Primary outcome
Outcomes of postoperative rescue analgesia.

Values are expressed as mean ± standard deviation or n (%).
aMann-Whitney U test.
bPatients who did not receive rescue analgesia were excluded.
cKaplan-Meier method with log-rank test.
dPearson’s chi-squared test.
Secondary outcomes
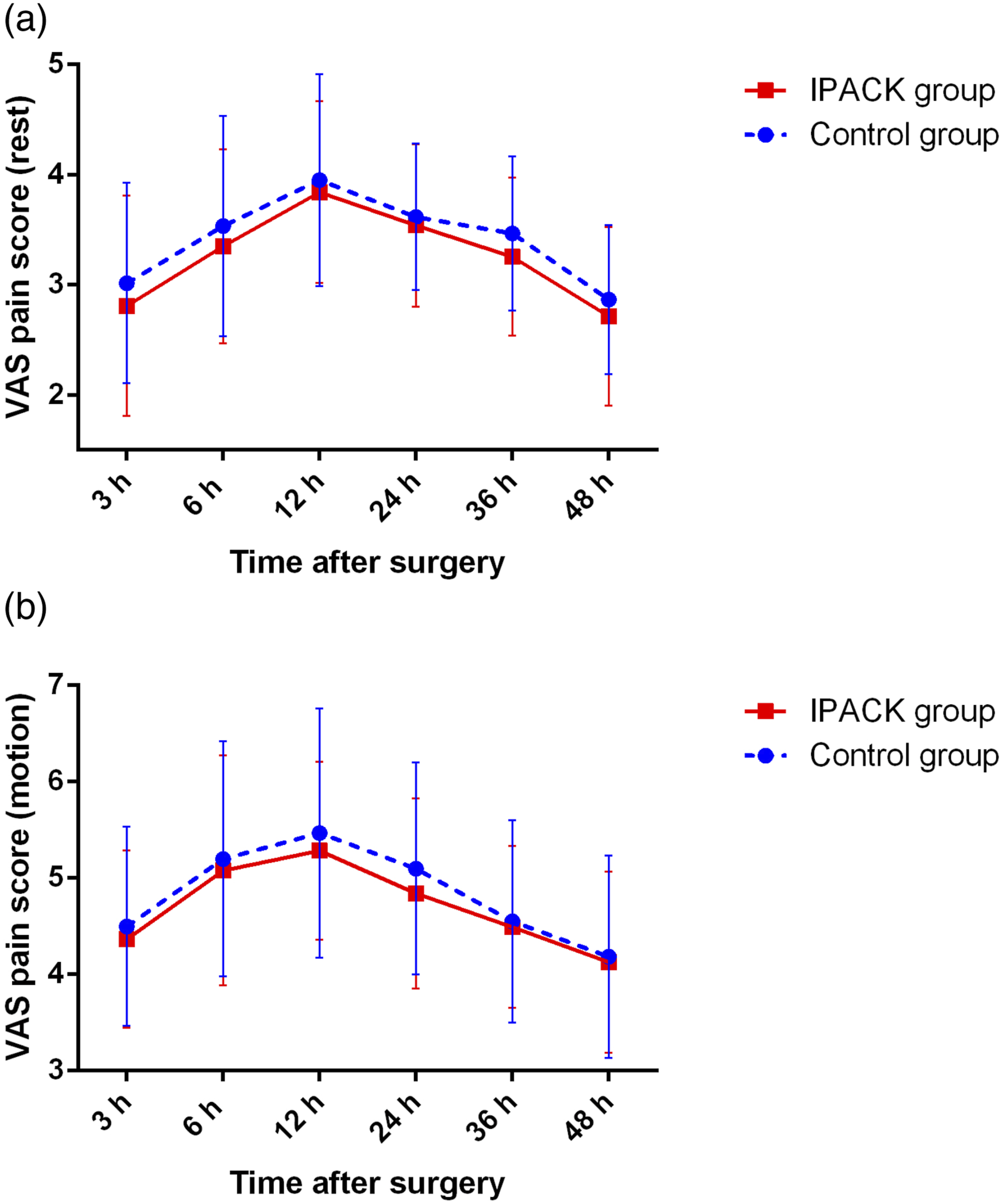
The time to first rescue analgesia did not differ between the two groups (average 12.7 h for control group vs 13.4 h for IPACK group, p = .478) (Table 2 and Figure 1), nor did the postoperative VAS pain scores at rest or during motion at any time point (Figure 2). In terms of functional recovery after surgery, the two groups showed similar knee range of motion and ambulation distance during hospitalization (Table 3). The two groups had a similar postoperative length of hospitalization (average 58.8 h for control group vs 60.4 h for IPACK group, p = .532) (Table 3 and Figure 3). The survival analysis function of the time to first rescue analgesia. The average postoperative VAS pain scores at rest (a) and during motion (b) of patients in both groups. The error bars indicate the standard deviation of the mean. Postoperative functional recovery. Values are expressed as mean ± standard deviation. ROM, range of motion. aMann-Whitney U test. bKaplan-Meier method with log-rank test. The survival analysis function of the time to hospital discharge.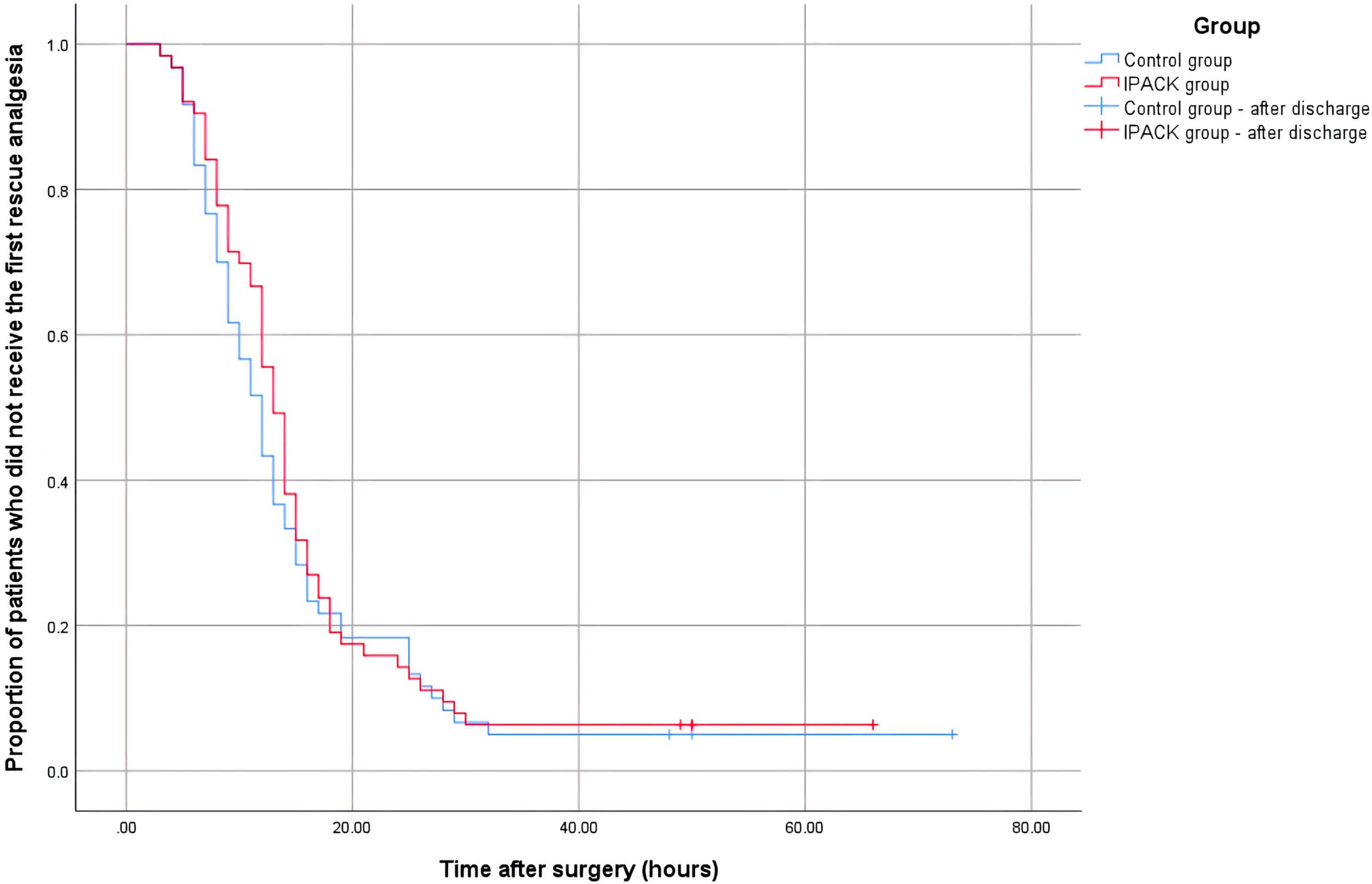


Postoperative complications.

Values are expressed as n (%).
aPearson’s chi-squared test.
Discussion
The ACB is currently the most commonly used regional analgesic technique for postoperative analgesia after TKA, and its analgesic efficacy is widely recognized. 25 Because ACB blocks only the anteromedial sensory nerve of the knee and not the posterior or lateral sensory nerves, 31 researchers have tried to use IPACK to solve this problem. The IPACK technique provides significant analgesia to the posterior compartment of the knee without compromising foot strength. 19 In recent years, the combination of ACB and IPACK has been the focus of research on multimodal pain protocols and regional analgesic techniques for TKA.20,23 However, the analgesic efficacy of combination therapy remains controversial. To date, few studies have specifically explored the efficacy of adding IPACK to ACB with LIA in patients undergoing TKA. Current research status prompted us to conduct this retrospective cohort study and found that there were no clinically evident analgesic benefits when adding IPACK to ACB in the presence of LIA.
Although the analgesic efficacy of ACB and IPACK remains controversial, researchers have attempted to summarize the existing clinical evidence. A previous systematic review and meta-analysis included seven randomized controlled trials (304 knees in the ACB + IPACK group and 305 knees in the ACB group) and reported that IPACK combined with ACB improved pain scores, morphine consumption, and functional abilities. 32 However, this systematic review and meta-analysis did not include whether LIA was performed. Another systematic review and meta-analysis evaluated the analgesic benefits of adding IPACK to ACB compared with ACB alone in the setting of LIA after TKA. 33 They found that adding the IPACK technique to ACB in the setting of periarticular LIA did not improve analgesic outcomes following TKA (four trials, 273 patients). In the absence of LIA, adding IPACK to ACB reduced pain for up to 24 h and enhanced functional recovery (eight trials, 631 patients). 33 Their conclusions support our results. However, the number of patients with LIA was relatively small. More high-quality, large-sample studies are required to validate our results.
A previous retrospective cohort study evaluated whether additional nerve blocks were superior to LIA in patients undergoing TKA with ACB. 34 That study enrolled 530 patients who underwent primary TKA. The patients were divided into two groups: Group A was treated with additional nerve blocks + ACB, and Group B was treated with LIA + ACB. Additional nerve blocks included IPACK, lateral femoral cutaneous nerve block, and obturator nerve block. These additional nerve blocks were administered alone or in combination. Their results suggested that additional nerve blocks do not provide superior pain relief compared with LIA in patients undergoing TKA under ACB. Their results also suggested that LIA + ACB could provide adequate analgesia in patients undergoing TKA.
In addition to the administration of LIA, the dosage of local anesthetics may affect the efficacy of ACB+IPACK.35–37 In previous studies, the concentration of ropivacaine used for ACB + IPACK block ranged from 0.2% to 0.5% (including 0.2%, 0.25%, 0.375%, and 0.5%), 35 whereas the volume for ACB + IPACK block ranged from 15 to 30 mL. 36 In the present study, 20 mL 0.2% ropivacaine was used for ACB and 25 mL 0.2% ropivacaine was used for IPACK. It is unknown whether other doses of local anesthetics used for ACB + IPACK blocks would yield the same results as those in our study. However, further studies are required to address this issue.
Our findings should be interpreted with caution because of several limitations. First, the lack of randomization and double-blinding are obvious limitations. In our study, patients were assigned to either the control or IPACK group according to the order of admission. This led to an inevitable bias, which could be reduced in a randomized controlled trial. Second, the number of patients included in the study was determined based on the number of patients admitted over the study period. Our sample size was relatively small, and may not have been sufficiently powered to evaluate the outcomes. Third, the postoperative follow-up of the patients was limited to the hospitalization period. Therefore, we were unable to assess the differences in outcomes and side effects after discharge. The effect of adding IPACK to ACB and LIA on long-term outcomes remains unknown. Therefore, long-term follow-up is warranted. Fourth, as mentioned above, our results are applicable only to the doses of the local anesthetics used. Different doses of local anesthetics used for ACB, IPACK, or LIA may lead to different conclusions.
Although there are several limitations, our study specifically explored the efficacy of adding IPACK to ACB with LIA for postoperative analgesia after TKA and provided new evidence for future clinical research and practice. Based on our study, prospective, blind, and sufficient powered randomized controlled trials can be conducted to verify our results in the future. In addition, researchers may consider exploring whether the dose of local anesthetic is a factor affecting the analgesic effects of ACB + IPACK.
Conclusions
In conclusion, when LIA has been included, the addition of the IPACK technique to ACB did not provide any clinical analgesic benefits. Orthopedic surgeons and anesthesiologists are justified in using ACB and LIA without IPACK for TKA.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
