Abstract
Background
It is well known that estrogen is closely related to bone and joint tissue. Findings indicate that estradiol, injected during pregnancy, passes through the placental barrier and reaches the fetuses in utero where it exerts its action. Since trochlea appears well established in the prenatal period, however, whether trochlear dysplasia is related to estradiol exposure has not been confirmed, and the pathological process of estradiol exposure-induced trochlear dysplasia remains unclear. This study aimed to establish an estradiol exposure animal model in fetuses and to analyze the morphology of the femoral trochlear in neonatal rats.
Methods
30 pregnant Wistar rats provided by the local Animal Center were assigned randomly into three groups, a high dose estradiol injection group, a low dose estradiol injection group and a blank control group. Gross, cross-sectional observation, histological staining measurement and microcomputed tomography of the rat offspring were conducted to evaluate the morphological changes of the femoral trochlea.
Results
The incidence of trochlear dysplasia increased with the concentration of estradiol injection. Gross and cross-sectional observation showed a shallower trochlea groove in two groups with estradiol injection. Histological staining measurement indicated that the trochlear sulcus angle and trochlear sulcus depth were significantly different between the two groups with estradiol injection and the blank control group at 0,5 and 10 days after birth. Subchondral bone loss was observed in the two estradiol injection groups by micro-CT, and the bone loss was found to deteriorate over time.
Conclusion
In this study, estradiol exposure in fetuses had an adverse effect on intrauterine development and could induce trochlear dysplasia and bone loss in rat offspring. In addition, this study also showed that the higher concentration of estradiol injection in pregnant rats, the more incidence of trochlear dysplasia in rat offspring.
Background
Trochlear dysplasia belongs to a spectrum of anatomical morphological abnormalities in the distal femur.1,2 There is a close association of trochlear dysplasia with patellar instability, the relationship was first described by Brattstroem in 1964 3 and with subsequent literature demonstrating that the presence of trochlear dysplasia is up to 85% of patients with patellar instability.4,5 It has been reported that trochlear dysplasia causes deviations in patellofemoral biomechanics, resulting in instability and potential patellofemoral osteoarthritis in the long term. 6 At present, the etiology of trochlear dysplasia is unclear. It is reported that femoral trochlear dysplasia may be caused by early patellar dislocation in animal experiments. 7 However, it is indicated that the cartilaginous trochlear develops early in gestation, between 9 and 16 weeks. 8 Since the trochlea appears established well in the prenatal period, it is conceivable an intrauterine developmental process maybe the etiology of trochlear dysplasia. 9 Estrogen is c1losely related to the proliferation, differentiation and metabolism of cartilage tissue. 10 Mansson and Norberg 11 showed that hip dysplasia could be induced in young dogs by administration of estradiol. Considering the similarities between the hip joint and patellofemoral joint. We hypothesized that estradiol exposure might lead to morphological alterations in the femoral trochlear during intrauterine developmental process.
The objectives of the present study were to establish an estradiol exposure animal model in fetuses by estradiol injection to gestational rats and to analyze the pathological process of the trochlear dysplasia.
Methods
The present study was approved by the Animal Ethics Committee. 30 pregnant Wistar rats provided by the local Animal Center were assigned randomly into three groups (10 in each group). Estradiol is the most biologically active of the estrogens, the dosage of estradiol referred to Kasström’s study on estradiol-induced hip dysplasia. 12 High dose group: the pregnant rats were intramuscular injected with estradiol benzoate (1812101, Tianjin Jinyao Pharmaceutical Co. LTD, China), 200 μg/kg body weight. Low dose group: the pregnant rats were intramuscular injected with estradiol benzoate, 100 μg/kg body weight. Control group: blank control. After line breeding, observation of vaginal plug and spermatozoa in the vaginal smear was regarded as successful mating and recorded as gestational day 1. The pregnant rats were intramuscular injected with estradiol benzoate 7 days after embolization, and intramuscularly injected until the neonatal rats were born. The food intake and body weight of pregnant rats were calculated every day. 21 to 22 days after embolization, 92 neonatal rats were born in the high dose group, 94 neonatal rats were born in the low dose group, and 95 neonatal rats were born in the control group. All rats were fed by their mothers.
Gross observation
20 neonatal rats (female 10, male 10) in each group were euthanized by excessive intraperitoneal injection of pentobarbital sodium at 0, 5 and 10 days after birth respectively, and the left knees were selected for surgery. The skin and subcutaneous tissue were removed and the distal femur were carefully isolated and fixed in 5% paraformaldehyde overnight. The anatomic morphology and the size of the femoral trochlea was assessed visually by an observer who was blinded to the experiment. The morphological characteristics of the femoral condyles were further analyzed based on micro-CT images. The slice just the point of proximal to the posterior condyle was chosen to observe the cross-sectional of the femoral trochlea. The slice was the representative, it was the point of deepest trochlear groove in normal rat fetuses. At this level, all of the femoral trochlea were sectioned axially using a scalpel. 13 The width depth and the angle of the femoral trochlea were evaluated qualitatively and compared between the three groups.
Microcomputed tomography (micro-CT) analysis
The femoral trochleas were scanned via micro-CT (SkyScan Model 1076). The parameters of micro-CT were set at a 9-µm voxel size, 50 kV, and 800 µA. The region of interest (ROI) is indicted by two orange cylinders approximately 0.5 mm in diameter (starting from 0.20 mm distal to the growth plate) and was located transversely under the lateral and medial facets of the trochlea to analyze microstructural parameters Figure 1. The four following representative parameters were assessed to analyze the morphological characteristics of the femoral condyles, as described in previous reports
14
: average trabecular thickness (Tb. Th, µm), average trabecular number (Tb. N, 1/µm), bone mineral density (BMD), bone volume density (BV/TV, %). The region of interest (ROI) is indicted by two blue cylinders approximately 0.5 mm in diameter (starting from 0.20 mm distal to the growth plate) and was located transversely under the lateral and medial facets of the trochlea to analyze microstructural parameters.
Hematoxylin and eosin (HE) staining
After the gross observation, we performed the hematoxylin and eosin staining of the femoral trochlea. The specimens of the distal femur were soaked in 4% paraformaldehyde (pH = 7.40) overnight at 4°C and then immersed to 10% ethylene diamine tetraacetic acid at 4°C for approximately 1 month for decalcification. A concentration gradient of xylenes and alcohol was used for dehydration, in paraffin the specimens were embedded in paraffin for subsequent tissue staining. Next, tissue slices were cut into three 5-μm sections along the femoral axis to obtain the transverse images of the trochlear sulcus and the sections were stained with hematoxylin and eosin to show the subchondral bone and cartilage,15,16 the measurement of femoral trochlea morphology was observed through a microscope. The cartilaginous trochlear sulcus angle was defined as the angle of the deepest point of the trochlear connecting with the medial trochlear and the lateral trochlear cartilaginous surface at the same slice,
17
Figure 2. The method applied to the measurement of trochlear sulcus depth, width and trochlear sulcus angle is summarized in Table1. This work was performed by an experienced author blinded to condition. The representative sections of each rat were recorded by the microscope. This is a histological section of the middle trochlear sulcus. The angle ∠BAC is defined as trochlear sulcus angle, AC is the trochlear sulcus width, BD is the trochlear sulcus depth. Description of measurements.

Statistical analysis
The descriptive data were expressed as the mean ± SD, and SPSS 24.0 software was performed for statistical analysis. The Shapiro–Wilk (S-W) test was used for normal tests, one-way ANOVA was used for comparisons between the three groups, and the chi-square test was used to analyze the differences between sex. p-value <.05 was considered as statistical significance.
Results
Gross observation
All the rats developed normally in the three groups, the weight of the rats was similar at each stage of development, and there was no death in pregnant rats and neonatal rats in the experiment. At 0 days, the size of the femoral trochleas in the high dose group and low dose group were smaller than the control group. The trochlear grooves were shallower in high dose group and low dose group than in the control group. At 5 days after birth, the surface of the articular cartilage was visually smooth, the luster was bright in the low dose group and control group. But compared to control group, the high dose group and low dose group had relatively flat trochlear grooves. At 10 days after birth, there was no significant difference noted in the size of the femoral trochleas among the three groups. However, compared to the control group, the high dose group and low dose group had a decrease in the depth and an increase in the trochlear sulcus angle of the trochlear grooves. (Figures 3 and 4) Gross anatomy of the femoral trochlea: (a) 0 days of the high dose group; (b) 0 days of the low dose group; (c) 0 days of the control group; (d) 5 days of the high dose group; (e) 5 days of the low dose group; (f) 5 days of the control group; (g) 10 days of the high dose group; (h) 10 days of the low dose group; (i) 10 days of the control group. Cross-sections of the femoral trochlea: (a) 0 days of the high dose group; (b) 0 days of the low dose group; (c) 0 days of the control group; (d) 5 days of the high dose group; (e) 5 days of the low dose group; (f) 5 days of the control group; (g) 10 days of the high dose group; (h) 10 days of the low dose group; (i) 10 days of the control group.

Micro-CT analysis
The morphological characteristics of the femoral condyles was observed by micro-CT. The micro-CT assay suggested that the Tb.Th, Tb.N, BMD and BV/TV decreased in the estradiol injection groups compared to the time-matched control group. There were statistical differences in Tb.Th, Tb.N, BMD and BV/TV of micro-CT between the two estradiol injection groups and control group (p < .05). And there was no obvious statistical differences in the high-dose group and the low-dose group (Figure 5). There were statistical differences in Tb. Th, Tb.N, BMD and BV/TV of micro-CT between the two estradiol injection groups and control group (p < .05). And there was no obvious statistical differences in the high-dose group and the low-dose group.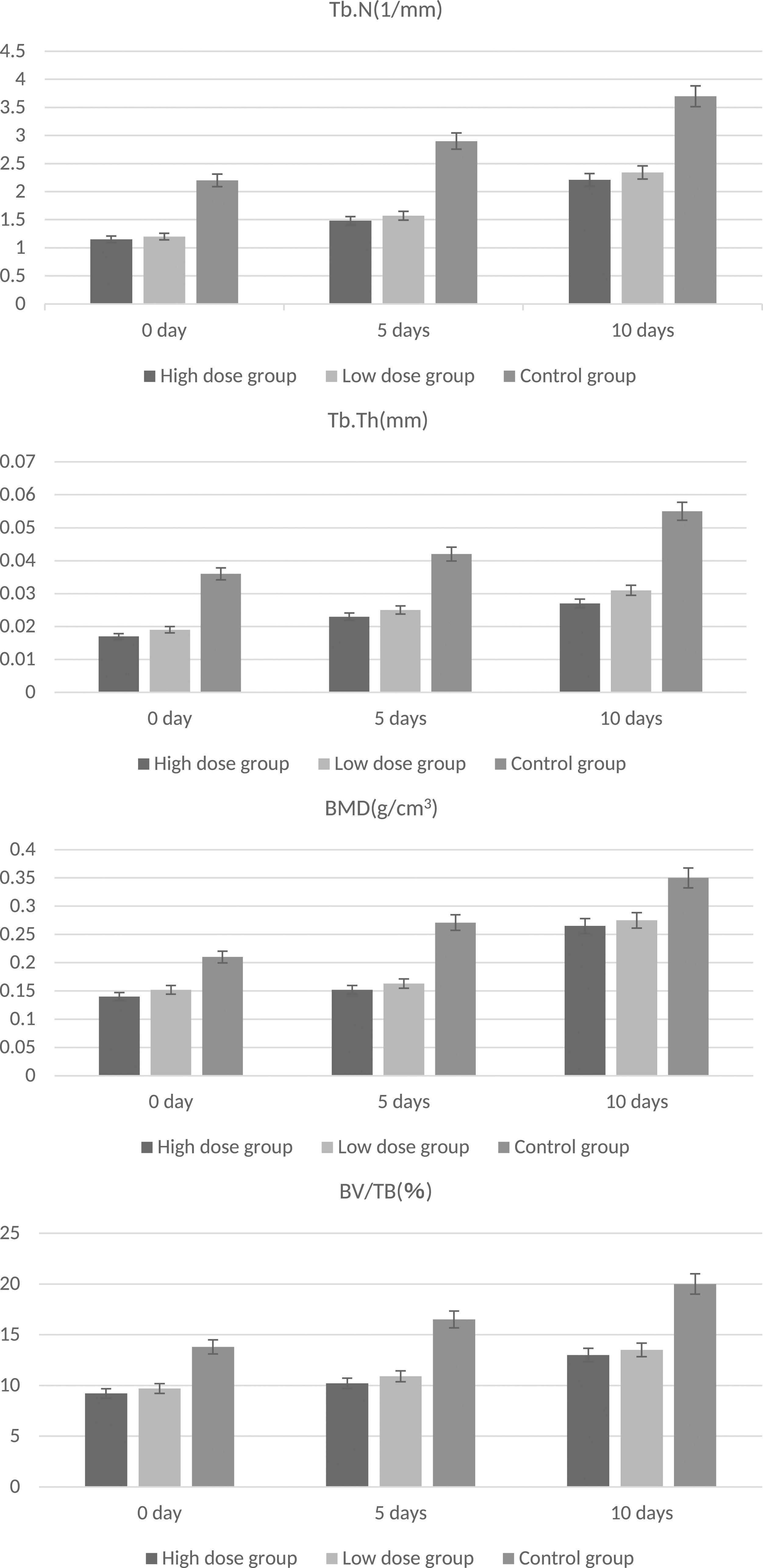
Histological analysis
Trochlear dysplasia in the estradiol exposure rats was defined as a 5° greater of trochlear sulcus angle than in the blank control rats. 18
Analysis of the pathological sections demonstrated that the arrangement and distribution of articular cartilage cells was orderly, the surface of the articular cartilage was smooth in the low dose group and control group. However, the surface of the articular cartilage was rough and arrange disorderly in high dose group. Compared with the control group, The trochlear groove was shallower in the two groups with estradiol injection (Figure 6). The average trochlear sulcus depth was shallower significantly in the two estradiol injection groups than in the control group. There were statistical differences in trochlear sulcus angle and trochlear sulcus depth between the two estradiol injection groups and control group (p < .05). No obvious difference was seen in the trochlear sulcus width between the two estradiol injection groups and control group (p > .05).
The variation trend of trochlear sulcus angle and trochlear sulcus depth in the two estradiol injection groups was similar at 0,5 and 10 days after birth. The mean degree of trochlear sulcus angle was greater significantly in the two estradiol injection groups than in the control group. The average trochlear sulcus depth was shallower significantly in the two estradiol injection groups than in the control group. There were statistical differences in trochlear sulcus angle and trochlear sulcus depth between the two estradiol injection groups and control group (p < .05). However, no obvious difference was seen in the trochlear sulcus width between the two estradiol injection groups and control group (p > .05), (Figure 7). Microscopic view of femoral trochlear by HE staining: (a) 0 days of the high dose group; (b) 0 days of the low dose group; (c) 0 days of the control group; (d) 5 days of the high dose group; (e) 5 days of the low dose group; (f) 5 days of the control group; (g) 10 days of the high dose group; (h) 10 days of the low dose group; (i) 10 days of the control group.
Compare the high-dose group with the low-dose group. The mean degree of trochlear sulcus angle in the high dose group was statistically greater than that in the low dose group (p < .05). The average trochlear sulcus depth was statistically shallower in the high dose group than in the low dose group (p < .05). Similar to the above, there was significantly difference in the trochlear sulcus width between the high dose group and low dose group (p < .05). The data show that the incidence of trochlear sulcus angle and trochlear sulcus depth increased with the concentration of estradiol injection.
The incidence of trochlear dysplasia between the high dose group, the low dose group and the sexual difference.

Discussion
The key findings of the current study were that estradiol exposure in fetuses could cause trochlear dysplasia and bone loss in the offspring of rats and the incidence of trochlear dysplasia increased with the concentration of estradiol exposure. The estradiol exposure may occur if estradiol was not properly metabolized during pregnancy.
Trochlear dysplasia which is characterized by a flattened and shallow trochlear groove is the most common knee disorders in adolescents and children with an incidence of 29 to 43 of 100 000, 19 and there is no consensus on the etiology of trochlear dysplasia. However, a number of evidence in the previous studies has revealed the correlation between trochlear dysplasia and the risk factors. 20 Trochlear dysplasia is recognized as a developmental condition of hereditary and congenital nature strongly influenced by environmental factors such as hormone levels and mechanical stress on the patella. 21 Øye et al. and Kohlhof et al. reported trochlear dysplasia occurs in utero 22,23 and the study of Yrjänä N showed that trochlear cartilage develops as early as 9 weeks of gestation . 8 Henricson 11 reported that hip dysplasia could be induced by the administration of estradiol. After this study, Gustafsson and Beling found that changes in growth pattern similar to those observed in hip dysplasia could be induced in the offspring of experiment dogs by estradiol injection during pregnancy. 24 Through the above research, given that femoral trochlea appears established well in the prenatal period, it is conceivable that estradiol injection during pregnancy causes trochlear dysplasia in the offspring.
Estrogen is known to affect bone metabolism and to regulate the balance between bone formation and resorption. 25 Estrogen indirectly inhibits growth hormone. 26 Studies on estrogen placental transfer in different species is limited. In guinea dog, rat and in human, 27 partial amounts of maternal estrogens can enter the fetoplacental circulation during pregnancy. Retarded growth of the femur was found in the offspring of dogs, which was injected with estradiol during the last trimester of pregnancy. The finding indicates that estradiol, injected during pregnancy, could pass through the placental barrier and reach the fetuses in utero where it exerts action. 28 Andren and Borglin studied newborn infants with congenital subluxation of the hips and found an increased urinary estrogen excretion as well as significant changes in the estrogen metabolism. 29 Studies of the effect of different doses of estradiol directly into the fetuses in utero, may shed some light on the different degrees of hip dysplasia. 24
In this study, an estradiol exposure model in fetuses was established by estradiol injection to gestational rats. The variation trend of trochlear sulcus angle and trochlear sulcus depth in the two estradiol injection groups was similar at 0,5 and 10 days after birth. There were statistical differences in trochlear sulcus angle and trochlear sulcus depth between the two estradiol injection groups and control group (p < .05). The trochlear sulcus angle was greater and trochlear sulcus depth was shallower in the two estradiol injection groups than in the control group. However, no obvious difference was seen in the trochlear sulcus width between the two estradiol injection groups and control group (P>.05). But in terms of the specific statistics, the trochlear sulcus angle was greater and trochlear sulcus depth was shallower in the high dose group than in the low dose group. In terms of the number of trochlea dysplasia, there were 53 of total 60 rats (88.3%) in the high dose group and there were 39 of total 60 rats (65.0%) in the low dose group with trochlear dysplasia. This indicated that estradiol exposure was associated with trochlear dysplasia, and the pathogenesis of estradiol-induced trochlear dysplasia was an ongoing process. In the study, high-dose estradiol injections caused a 23.3% increase in the prevalence of trochlear dysplasia (from 65% for low dose group to 88.3% for high dose group). This finding suggests that the severity of the pathological changes increases as the concentration of estradiol injection.
The relationship between estrogen and cartilage metabolism had been demonstrated. Estrogen has a direct effect on articular cartilage and the effect is dose-dependent. When estradiol level is at physiological dose, there is an advance relationship between estradiol and cartilage proliferation, but further increase of estradiol level will inhibit cartilage proliferation. 30 Excessive estradiol exposure can damage articular cartilage. It was found that the synthesis of vulcanized proteoglycan in articular cartilage of rabbits significantly decreased after estrogen administration. 31 In vitro culture, it has been found that estrogen can stimulate prostaglandin synthesis of articular cartilage. Talwar et al. 10 found that estrogen can reduce cartilage thickness by inhibiting chondrocyte proliferation and increasing chondrocyte maturation in organ culture. In normal pups the femoral trochlear and head consist of cartilage entirely during the first 2 weeks and are still largely cartilaginous at 6 weeks of age. It may the reason why the femoral trochlear is particularly sensitive to estrogen, which obviously exerts its action by inhibiting cartilage growth.
The morphological characteristics of the femoral condyles were further analyzed based on micro-CT images. Considering these studies, we speculated that estradiol exposure plays an essential role in bone loss in the development of the femoral trochlea. Subchondral bone loss might have been caused by the abrogation of osteogenesis. The study provides some insights into the mechanism of bone loss in trochlear dysplasia and may lay a theoretical basis for novel therapeutic options.
Any changes in surrounding tissues such as ligaments and muscles can affect the patellofemoral joint, whether congenital or posttraumatic, depending on the severity of the disorder. 32 At present, estradiol receptors have been found in tissues such as muscles and ligaments around the knee joint. 33 Estradiol exposure may have an effect on the structure and metabolism of muscles and ligaments, which in turn may interfere with knee relaxation and neuromuscular control, 34 which can further affect the coordination and balance of the patellofemoral joint, leading to patellar instability. Li et al. 35 found femoral trochlear dysplasia or flattening after patellar instability in growing rabbits. Estradiol exposure not only plays an important role in bone metabolism, but also is closely related to the muscles and ligaments around the patella, which has an high etiological correlation on the femoral dysplasia.
Apart from a direct effect of estradiol exposure on cartilage, soft tissues (joint capsule, ligaments and attachment muscles) around the joint are also involved. Estrogens can induce profound changes in tensile properties of connective tissues, which increase the diameter of the birth canal and facilitate parturition. 36 Collagen is important for joint stability and proper function, which is produced by fibroblasts, the quality and quantity of collagen can be affected by estrogen. The main function of collagen is to resist biological tension. Estrogen inhibits the activity of fibroblasts, leading to a decrease in the proportion of collagen in soft tissue. As a result, joint relaxation occurs and soft tissue balance is disrupted. It is usually acknowledged that there is a unique matching relation of the femoral trochlea and patella, which is the basis of its biomechanical function. 17 Given that stress stimulation plays a significant role in the development of subchondral bone and articular cartilage. 37 Based on the above analysis, in this study, we conceive that the matching relation of the femoral trochlea and patella was affected by the soft tissue balance, the mechanical stress stimulation of the patellofemoral joint was reduced, which exacerbated trochlea dysplasia.
The pathogenesis of estradiol-induced trochlear dysplasia can be described as a vicious circle, initiated by retarded growth of femoral trochlea and soft tissue imbalance, giving rise to trochlear incongruity - trochlear dysplasia - remodeling (flattening of the femoral trochlea) - more trochlear dysplasia. Such estradiol exposure could occur, either by an increased production in ovary and placenta, or by a normal production and an incapability of the fetuses to metabolize estrogens. This alerts that estrogen monitoring during pregnancy and in neonates, combined with imaging examination, is necessary. The purpose is to detect risk factors of trochlear dysplasia, and therefor early intervention.
There were several limitations to this study. We used estradiol injections to establish an estradiol exposure animal model rather than establishing a real spontaneous animal model, which made it difficult to completely reflect the situation of the human. This study has shown that trochlear dysplasia can be induced experimentally in rat offspring by serial estradiol injections of pregnant rats, but this is no proof that spontaneously occurring trochlear dysplasia was induced by estradiol. Second, the study included the observation of femoral trochlear development, moreover, further studies to include the morphological changes of patella could also be performed in order to get more information on patellofemoral joint. Third, a dynamic observation of femoral trochlear in animal models would have been more conclusive, but radiological methods such as CT or X-ray could not function well in such tiny tissue.
Conclusions
This investigation confirms the possibility that trochlear dysplasia in rat offspring is caused by estradiol exposure in gestational rats, and to our knowledge, the present study was the first to observe the pathological process of estradiol exposure-induced trochlear dysplasia. There was a positive linear correlation between the concentration of estradiol exposure and the incidence of trochlear dysplasia, moreover, estradiol exposure-induced trochlear dysplasia may persist after birth.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with re- spect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Medical science research subject of Hebei Provincial Health Commission (grant no. 20232031).
