Abstract
Background
Treatment of large segmental bone defects is still a major clinical challenge, and bone grafting is the main method. The development of novel bone graft substitutes will help solve this problem.
Methods
Porous bioceramics hydroxyapatite (HA) scaffolds coated with different ratios of HA/β-tricalcium phosphate (β-TCP) were prepared by 3D printing. The scaffolds were sampled and tested in large segmental bone defect rabbit models. X-ray, micro-computed tomography (CT), hematoxylin and eosin (HE) staining, Van-Gieson staining, and type I collagen staining were performed to find the best scaffolds for large segmental bone defect treatment.
Results
The average length, diameter, compressive strength, and porosity of the bioceramics scaffolds were 15.05 ± 0.10 mm, 4.98 ± 0.06 mm, 11.11 ± 0.77 MPa, and 54.26 ± 5.38%, respectively. Postoperative lateral radiographs suggested the scaffold group got better bone healing and stability than the blank group. Micro-CT showed new bones grew into the scaffold from the two ends of the fracture along the scaffold and finally achieved bony union. The new bone volume around the scaffolds suggested the 3:7 HA/β-TCP-coated bioceramic scaffolds were more favorable for the healing of large segmental bone defects. The results of HE, Van-Gieson, and type I collagen staining also suggested more new bone formation in 3:7 HA/β-TCP-coated bioceramic scaffolds.
Conclusion
3:7 HA/β-TCP-coated porous bioceramics scaffolds are more conducive to the repair of large bone defects in rabbits. The results of this study can provide some reference and theoretical support in this area.
Keywords
Introduction
Clinically, a large segmental bone defect is defined as a bone defect whose length exceeds 1.5 times the diameter of the tubular bone. 1 It is mainly caused by trauma, infection, tumor, and other bone diseases and often cannot heal spontaneously. 1 Owing to the low regenerative potential of a bone, large bone defect treatment has been a major problem in clinical practice. 2 Currently, the main clinical treatment for large segmental bone defects is bone grafting, including autologous, allogeneic, and artificial bone grafts. 3 To date, autologous bone is still the best material for bone grafting, with non-immunogenic properties and good promotion of bone healing, but its use is very limited due to the problems of secondary damage and limited bone sources. In contrast, allogeneic bone grafting is far less effective than autologous bone grafting due to its lack of osteoblasts and biologically active substances but has significant limitations in clinical application due to its high price, potential immune rejection, disease transmission, infection, and limited bone source. 4 Additionally, available artificial bones still cannot completely replace autologous and allogeneic bones in terms of bone conductivity, inducibility, and biomechanics. Therefore, it is urgent to develop bone graft substitutes that can meet the needs of large bone defect treatment.
With the progress of biomaterial science, biological scaffolds with good biocompatibility and osteogenesis have been discovered, mainly including metals and their alloys,5,6 high molecular material, 7 bioceramics, 8 biomacromolecule, 9 etc. Bioceramics with stable physical and chemical properties, excellent biocompatibility, corrosion resistance, and non-toxic side effects have attracted much attention and have been widely used in bone tissue engineering.10,11 At present, the main biological ceramic materials used in clinical practice are hydroxyapatite (HA), 12 β-tricalcium phosphate (β-TCP), 13 and biphasic calcium phosphate ceramics (BCP). 14 Among them, HA is similar to natural bone in inorganic composition, with high strength and biocompatibility, but pure HA bone has poor conductivity, insufficient fatigue resistance in the physiological environment, slow degradation rate, and poor transplantation effect.15,16 β-TCP has good biocompatibility and bone conductivity but poor biomechanical strength, making it difficult to support bones during large bone defect treatment, and a too fast degradation rate to match the growth rate of new bones.17,18 Thus, it is also difficult to meet the requirement of bone scaffolds. As to BCP, which is composed of HA and β-TCP, the biomechanical strength, degradation rate, and osteoconductivity are determined by the HA:β-TCP ratio. In the treatment of large segmental bone defects, high biomechanical strength of the scaffold is required to support the broken bone ends. Therefore, when using BCP to make scaffolds for large bone defects, a higher percentage of HA is required to meet the biomechanical needs, but this sacrifices the degradation rate and osteoconductive properties of the scaffold. In this study, we used HA to prepare scaffold embryos that could meet the biomechanical requirement of bone scaffolds. Then, the surface of the scaffold embryos was coated with HA/β-TCP, and the ratio was adjusted according to the bone healing time.
In the production of bone scaffolds, 3D printing technology has the advantages of mature technology, high precision, personalization, and efficient preparation.19,20 The use of 3D printing technology to prepare tissue engineering bone scaffolds with highly connected porous structure, strong mechanical properties, and excellent biocompatibility has been widely recognized and applied, with great application value and potential in the field of bone defect repair.21–23
Therefore, in this study, three biomimetic porous bioceramic scaffolds coated with commonly used HA:β-TCP ratios were prepared by 3D printing technology, and the effect of different scaffolds in treating large segmental bone defects was evaluated in New Zealand white rabbits. The optimal HA:β-TCP coating ratio for bone healing was screened to provide some reference and theoretical basis for its future clinical application.
Methods
Preparation of porous bioceramic scaffolds
An impregnation solution containing HA (Maihai Material Genome International Research Institute, China, Item No. 20200508) and β-TCP powder (Maihai Material Genome International Research Institute, China, Item No. 20200508) was prepared with mass ratios (m:m) of 7:3, 5:5, and 3:7. The prepared HA/β-TCP mixed powder and normal saline according to the mass ratio of 5:2 were prepared and stirred evenly to form the impregnation solution.
An aqueous solution of 8% polyvinyl alcohol (Sigma Aldrich ( Shanghai) Trading Co., LTD., China, Item No. Mkck 4857) was added to the HA powder (m:m = 1.07:1) and stirred in a thermostatic magnetic mixer (Jilin Province Sedna Pharmaceutical Technology Co., LTD., China) at room temperature to form a well-mixed bioprinting ink.
Bioceramics 3D printer (PCPrinter MF150, Xi 'an Dot Cloud Biotechnology Co., LTD., China) was used for bio-ink printing and forming. The nozzle diameter was 60.0 μm, the printing speed was set to 10.0 mm/s, and the filling rate was 42.0%. Finally, a semi-finished model of porous bioceramics bone scaffold with an external dimension of ∅5.0 mm × 15.0 mm was completed. The model was freeze-dried in a silicone oil in situ lyophilizer (Ningbo Xinzhi Lyophilization Equipment Co., LTD., SCIEN T Z-20F, China) for 24 h and then sintered in a high-temperature box furnace (Hefei Koji Material Technology Co., LTD., KSL-1700X, China) for 4 h at 1300°C to obtain the dried porous bioceramics bone scaffold.
Finally, the printed scaffold was placed in the impregnation solution and impregnated at room temperature for 5 min each time three times. The drying temperature of the scaffold was set at 120°C for 6 h, and porous bioceramic bone scaffolds coated with HA/β-TCP in different ratios were obtained.
Sampling test of basic scaffold performance
Five scaffolds were randomly sampled from the 32 scaffolds prepared. Their appearance, diameter, length, compressive strength, and porosity were detected.
Appearance
The appearance of the surface coating coverage of the scaffolds was assessed by a naked-eye and electron microscope (Phenom proX, Frontier Scientific Instruments (Shanghai) Co.).
Diameter
The diameter and length of the scaffolds were measured using a vernier caliper (Electronic digital caliper 605A (Ⅰ), HaErBin Tools Electrical Co., LTD., Harbin, China).
Porosity measurement
According to GB/T 1966-1996, the Archimedes principle was used to measure the porosity of porous hydroxyapatite bone embryo body/biphasic calcium phosphate-coated composite scaffold. First, the dry weight of the sample was measured and recorded as m1. The sample was then placed into a beaker infused with distilled water until the sample was submerged. Then the beaker was heated to boiling in a constant temperature drying oven, and the boiling state was kept for 2 h, so that the distilled water was completely penetrated into the void of the sample. After cooling to room temperature, the sample was again immersed in distilled water with a hydrophobic line suspension and weighed and recorded as m2. Finally, the sample was removed from the water and the water on the surface was removed with filter paper, then, the sample was weighed again and recorded as m3, according to the following formula to calculate the porosity of the porous scaffolds rate:
Compressive strength
According to GB/T 1448-2005, the specimens were dried at 37°C for 24 h. The two ends of the scaffolds were smoothed with a diamond. After measuring the compressed area, the specimen was placed vertically on a mechanical testing machine to determine its compressive strength with a loading speed of 1 mm/min until the cylinder ruptured, and the ultimate load at the time of specimen destruction was recorded to calculate the ultimate compressive strength:
Experiments on animals
A total of 36 5-month-old male New Zealand white rabbits weighing 3.2 ± 0.5 kg (provided by DILEPU Biotech Medical Co., LTD., License No.: SCXK [Shanxi] 2019-002) were randomly divided into four groups (nine rabbits in each): blank group, 7:3 scaffold group, 5:5 scaffold group, and 3:7 scaffold group. The right forelimb of each animal was subjected to a large bone defect model at the upper 1/3 of the radius. Scaffolds coated with a corresponding HA:β-TCP ratio were implanted in the rabbits at the bone defect sites in each group (no scaffolds were implanted in the blank group). The operation procedure was as follows. Celazine hydrochloride (4.0 mg/kg) (Jilin Huamu Animal Health Products Co., LTD., China, 2 mL:0.2 g/branch) and 1% pentobarbital (30.0 mg/kg) (China Pharmaceutical Group Shanghai Chemical Reagent Company, China, 100 mg/branch) were injected into the ear vein for anesthesia. After the right forelimb was partially shaved and the skin was prepared for disinfection, the skin and subcutaneous fascia were cut, the muscles and periosteal were removed, and the radius was exposed. A 15-mm bone segment was measured distal to the upper 1/3 of the radius and marked. The 15-mm bone segment was cut off with a swing saw, and the corresponding HA/β-TCP proportional coating scaffold was implanted into the bone defect area of the radius. A silk thread (2-0) was used to fix the bone scaffold to the surrounding soft tissue. We only performed the bone removal procedure in the blank group rabbits. After irrigating the surgical area with normal saline, the wound was closed by layer-by-layer suturing. After recovery from anesthesia and with a good response, the experimental animals were sent back to the animal room and fed routinely. 12 × 104 U penicillin G sodium (Sichuan New Brilliant Animal Pharmaceutical Co., LTD., four million units/branch) was intramuscularly injected in each rabbit once a day for three consecutive days after the operation to prevent wound infection.
At 4, 8, and 12 weeks after the operation, three experimental animals in each group were selected. Lateral radiography of the operative side of the radius (Siemens Digital X-ray machine: XP/XP Hybrid, Germany) was performed first to observe the growth of callus in the bone defect area in each group. Then, the experimental animals were sacrificed using excessive ketamine hydrochloride (Tianjin Damao Chemical Reagent Co., LTD., China, 2 mL:0.l g branch), and the forelimbs of the operative side were dissected. The soft tissues around the ulna and radius were appropriately removed, and the ulna and radius were removed and fixed with a formalin fixation solution for 24 h. After fixation, the gross specimens were scanned by micro-CT (160 kV microfocus industrial CT AX2000, Germany). The X-ray tube voltage (V) and current (I) were set to 80 kV and 200 mA, respectively. After scanning each sample to obtain 450 microlayer sections, VG Studio MAX software was used to reconstruct the image. The implanted scaffold was shown in blue, and the new bones in the scaffold were shown in yellow. Considering that the scaffold extended outward into a circular sleeve area with a wall thickness of 0.5 mm, the new bone volume around the scaffolds in the bone defect area was composed of new bone volume inside and outside the scaffold. Meanwhile, bone volume/total volume (BV/TV), bone surface/bone volume (BS/BV), trabecula thickness (TbTh), trabecula number (TbN), trabecula seperation (TbSp) were also measured inside and outside the scaffold, respectively.
After finishing the micro-CT, the transverse section of the middle of the implanted scaffold and the longitudinal continuous tissue section at the bone junction of the scaffold were taken for hematoxylin and eosin (HE), type I collagen, and Van-Gieson staining. The bone healing of each group was compared.
HE staining
The radius of the bone defect repair was taken at 4, 8, and 12 weeks after surgery, and the specimens were dehydrated in gradient ethanol, embedded in paraffin (Wuhan Junjie Electronics Co., LTD., JB-P5, China), cured, then cut into 10-μm-thick sections using a hard tissue slicer (Leica SP 1600, Shanghai Leica Instruments Co., LTD., China), and stained. The procedure was as follows. (1) Sections were stained with hematoxylin staining solution (Servicebio, China) for 8–10 min, washed with tap water, differentiated with differentiation solution, washed with tap water, returned to blue with blue return solution, and rinsed with running water. (2) Sections were sequentially dehydrated into 85% and 95% gradient alcohol for 5 min each and stained in eosin staining solution (Servicebio, China) for 8–10 min (3) After the dehydration, the sections were placed in clean xylene for 1–5 min and sealed with neutral gum. Then, the sections were observed and photographed under an optical microscope (Nikon E100, Japan).
Type I collagen staining
The radius of the bone defect repair was taken at 4, 8, and 12 weeks after surgery, and the specimens were dehydrated in gradient ethanol, embedded in paraffin (JB-P5, Wuhan Junjie Electronics Co., LTD., China), cured, and then stained by cutting into 10-μm-thick sections using a hard tissue sectioning machine (Leica SP 1600, Shanghai Leica Instruments Co., LTD., China). The operation steps were as follows. (1) Paraffin sections were dewaxed. (2) Using ready-to-use pepsin repair solution for antigen repair, the sections were incubated at 37°C for 20 min, followed by washing with phosphate-buffered saline (PBS) (pH 7.4) for 5 min three times. (3) Using volume fraction 3% hydrogen peroxide solution to block endogenous peroxidase, the sections were incubated at room temperature for 25 min, followed by washing with PBS (pH 7.4) for 5 min × three times. (4) Then, 3% bovine serum albumin (BSA) was added, and the sections were incubated at room temperature for 30 min (5) Rabbit anti-COL-1 primary antibody (ratio 1:500, Servicebio) was added dropwise, and the sections were incubated overnight at 4°C, followed by washing with PBS (pH 7.4) for 5 min three times. (6) Horseradish-peroxidase (HRP)-labeled goat anti-rabbit IgG secondary antibody (ratio 1:200, Servicebio) was added dropwise, and the sections were incubated for 50 min at room temperature, followed by washing with PBS (pH 7.4) for 5 min three times. (7) Finally, sequential steps of DAB staining, hematoxylin re-staining for cell nuclei, dehydration and sealing were executed. The sections were observed and photographed under a light microscope.
Van-Gieson staining
The radius of the bone defect repair was taken at 4, 8, and 12 weeks after surgery, and the specimens were dehydrated in gradient ethanol, embedded in paraffin (Wuhan Junjie Electronics Co., LTD., JB-P5, China), cured, and then stained by cutting into 10-μm-thick sections using a hard tissue sectioning machine (Leica SP 1600, Shanghai Leica Instruments Co., LTD., China). For Van-Gieson (VG) staining solution (Servicebio, China) preparation, 9 mL of VG staining solution B was added to 1 mL of VG staining solution A and mixed. Then, the VG staining solution was stained for 3 min, quickly washed with water, and quickly dehydrated in three cylinders of anhydrous ethanol. Afterward, transparent sealing was performed using clean xylene for 1–5 min and neutral resin sealing. An optical microscope (Nikon E100, Japan) was used for observation and photography.
Statistical analysis
The experimental data were expressed as
Results
Appearance and implantation of the scaffold
The general appearance of the scaffold prepared according to the preparation process is shown in Figure 1(a), which was a porous white scaffold. The electron microscope scan results suggested that the scaffold had uniform fiber arrangement and voids. The upper picture of Figure 1(b) shows the surface morphology of the scaffold without impregnated HA/β-TCP mixture coating, which was relatively smooth, and the lower picture of Figure 1(b) shows the surface morphology of the scaffold with impregnated HA/β-TCP mixture coating, which had a rough surface and was conducive to cell attachment. Figure 1(c) shows the precise osteotomy of the radius in the experimental animal during the operation. Figure 1(d) shows the length of the osteotomized bone segment and the selection of a scaffold of the same length during the operation. Figure 1(e) shows the photograph of a scaffold implanted in the right radial defect area of the rabbit, and the scaffold was fixed to the surrounding soft tissue using a 2-0 silk suture to avoid its displacement. The porous bioceramic bone scaffold was implanted into a large segmental bone defect of the rabbit radius. (a). Macroscopic view of the porous bioceramic bone scaffold. (b). Scanning electron microscopy of porous bioceramic bone scaffold: the upper part shows the appearance of the uncoated scaffold, and the lower part shows the appearance of the coated scaffold. (c). A model of a large segmental bone defect of the right rabbit's radius. (d). The appearance of the resected bone and the proposed scaffold. (e). Photograph of the scaffold implanted into the defective area of the right rabbit's radius.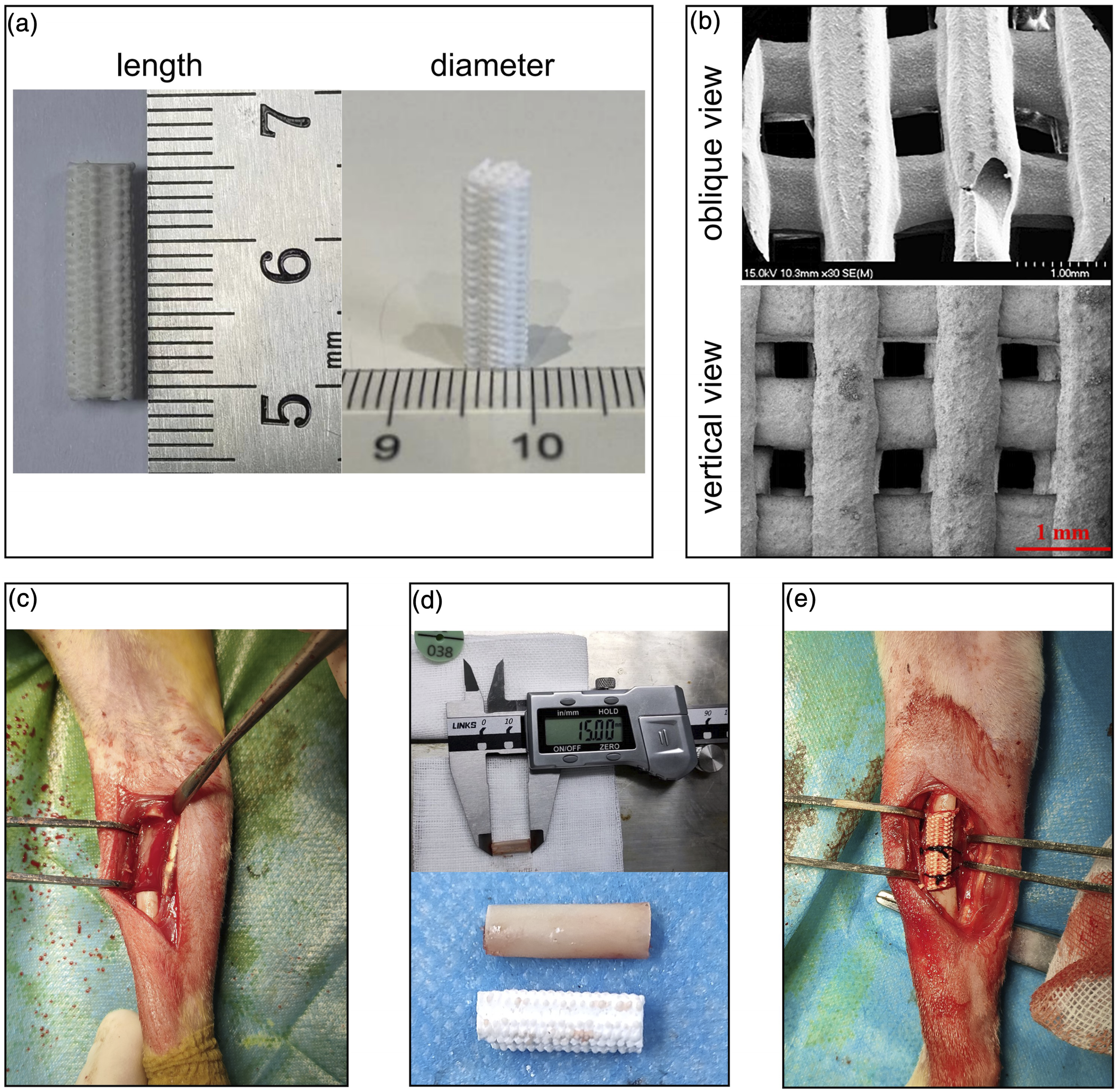
Parameters of the sampled scaffolds
Some parameters of the sampled scaffolds.

Basic information about experimental animals
A total of 36 animals were enrolled in this study and randomly divided into four groups. All enrolled animals had no unnatural death during surgery and experiments. All the experimental animals recovered from anesthesia within 30 min after the operation. On the first day after the operation, they were in good condition and could eat freely, but the affected limb was lame and had reduced activity. Within 2 weeks after the operation, all the animals could walk normally, the incision was healed, and no local infection was observed.
X-ray results
The X-ray results of the bone defect sites in the four groups are shown in Figure 2. Four weeks after the operation, there was no obvious callus formation at both ends of the bone defects in the blank group and in the 7:3 and 5:5 coating groups. The interfaces between the broken ends of bone defects and the scaffolds were clearly visible, and there was no obvious callus formation as well. Meanwhile, the interfaces between the broken ends of the bone defects and the scaffolds were clearly visible in the 3:7 coated group, but a trace of callus at the broken ends was observed in this group (Figure 2(a)–2(d)). Eight weeks after the operation, a small amount of callus was observed at the broken ends in the blank group. Although a small amount of callus was also observed at the broken ends, the interfaces between the broken ends and the scaffolds were still clearly visible in the 7:3 and 5:5 coated groups. In contrast, the interfaces between the broken ends and the scaffolds in the 3:7 coated group became blurred and were wrapped and covered by callus (Figure 2(e)–2(h)). 12 weeks after the operation, some calli were formed at both ends of the bone defect in the blank group, but there was no connection between the two fracture ends. In the 7:3 coated group, obvious callus formation was observed at 4 and 8 weeks, but there were still some obvious radiolucent areas at the interface between the bone defect ends and the scaffold, and the amount of local callus was small. In the 5:5 and 3:7 coated groups, the interface between the bone defect ends and the scaffold was blurred, both were covered by callus, and there were no obvious radiolucent areas (Figure 2(i)–2(l)). Comparing the osteogenesis of the scaffold groups with that of the blank group, we could see that the amount of callus around the fracture ends in the blank group was very small within 8 weeks, and only a small amount of scattered callus was formed in the bone defect area at 12 weeks, which was disorganized and located between the fracture ends, not growing from both sides of the fracture ends to each other, with the original fracture ends being sclerotic and atrophied. In sum, the osteogenesis of each scaffold group was significantly better than that of the blank group, with callus growing from the broken ends in opposite directions, and the broken ends and the scaffolds were tightly connected at 12 weeks, suggesting a good osteoconductivity of the scaffold. Hence, the osteogenesis of the scaffold groups was significantly better than that of the blank group, considering that the use of artificial bone implants, such as HA or β-TCP, had been proven to facilitate fracture healing.24,25 Therefore, our following study would not discuss the osteogenesis in the blank group but would focus on comparing the osteogenesis capacity between different coating scaffolds. X-ray film examination results of bone defect sites in each group at different time points after the operation ( n = 9 for each group). Fracture healing of the blank group (a), 7:3 coating group (b), 5:5 coating group (c), and 3:7 coating group (d) by X-ray 4 weeks after the operation ( n = 3 for each group at 4 weeks). At 8 weeks after the operation, the bone healing of the blank group (e), 7:3 coating group (f), 5:5 coating group (g), and 3:7 coating group (h) were examined by X-ray ( n = 3 for each group at 8 weeks). At 12 weeks after the operation, the bone healing of the blank group (I), 7:3 coating group (j), 5:5 coating group (k), and 3:7 coating group (l) were examined by X-ray (n = 3 for each group at 12 weeks).
Results of micro-CT examination
Micro-CT 3D imaging was used to explore the osteogenesis around the scaffold (Figure 3). The images were reconstructed using VG Studio MAX software, with the implanted scaffold in blue and the new bones inside the scaffold in yellow. The middle section of the scaffold was selected for comparison both in transverse and longitudinal sections. Figure 3(a)–3(c) shows that there was little callus in the middle cross-section of the scaffold in the 7:3 and 3:7 coated groups 4 weeks after the operation, while a small amount of callus was seen in the 5:5 coated group. In the longitudinal section, the callus in the bone defect area grew from the broken ends, and more callus was observed in the 5:5 and 3:7 coated groups. Three-dimensional reconstruction of the bones showed that more callus was observed in the 5:5 and 3:7 coated groups than in the 7:3 coated group. Figure 3(d)–3(f) shows that there was a small amount of callus in the middle cross-section of the three different scaffold groups 8 weeks after the operation, with the highest amount of callus in the 3:7 coated group and the lowest amount of callus in the 7:3 coated group. In the longitudinal cross-section, scattered new bone tissue was seen between the two broken ends in the 5:5 and 3:7 coated groups, suggesting that the new bones might have formed bony connections in the scaffolds. While no callus was observed in the middle of the longitudinal section of the scaffold in the 7:3 coated group, suggesting that bone growth was slower in this group. The result of the 3D reconstruction of the bones also showed that there was more callus in the 5:5 and 3:7 coated groups than in the 7:3 coated group, especially in the 3:7 coated group, in which the new bone tissue was denser. Figure 3(g)–3(i) shows that there was much callus in the middle cross-section of the three scaffold groups 12 weeks after the operation, and the pores in the scaffold were basically filled with callus in the 5:5 and 3:7 coated groups, while there were still gaps in the 7:3 coated group. In the longitudinal section, more calli filled the pores in the scaffolds in the 5:5 and 3:7 coated groups 12 weeks after the operation than 8 weeks after the operation, and the bone volume was denser. Meanwhile, more callus was observed in the 3:7 coated group. The result of the 3D reconstruction of the bones showed that the scaffolds were filled with callus, and the broken ends in the three groups all achieved bone connection, while the 7:3 coated group had sparser callus and more voids compared with the 5:5 and 3:7 coated groups. The results of micro-CT of bone defect sites in each group at different time points after the operation. The osteogenesis in the bone defect sites in the 7:3 coated group (a), 5:5 coated group (b), and 3:7 coated group (c) examined by micro-CT 4 weeks after the operation ( n = 3 for each group at 4 weeks). The osteogenesis in the bone defect sites in the 7:3 coated group (d), 5:5 coated group (e), and 3:7 coated group (f) examined by micro-CT 8 weeks after the operation ( n = 3 for each group at 8 weeks). The osteogenesis in the bone defect sites in the 7:3 coated group (g), 5:5 coated group (h), and 3:7 coated group (i) examined by micro-CT 12 weeks after the operation ( n = 3 for each group at 12 weeks).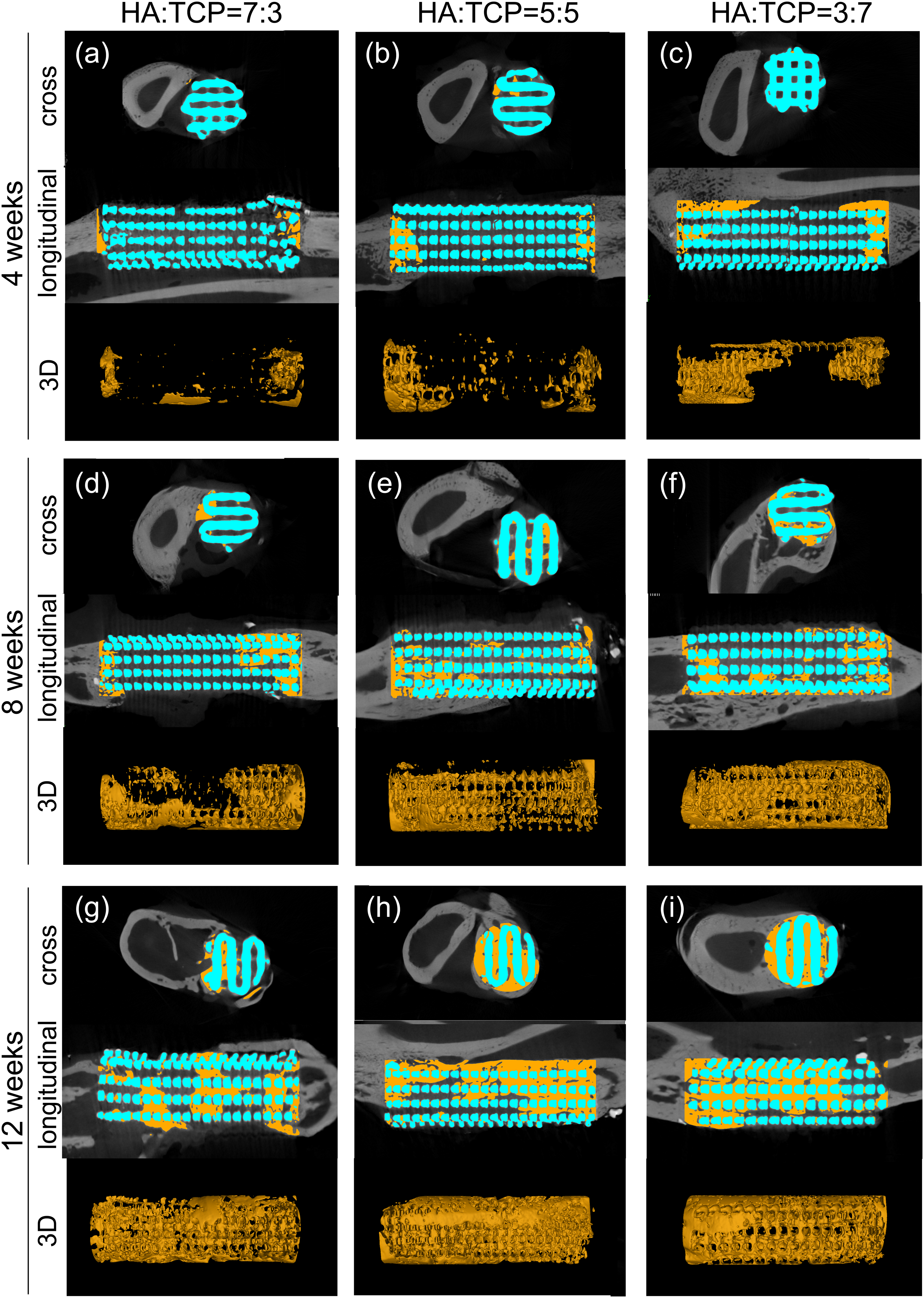
In each group, the mean volume of new bone around the scaffold was 57.2 mm3, 84.3 mm3, and 137.4 mm3 at 4 weeks, 137.2 mm3, 183.5 mm3, and 232.0 mm3 at 8 weeks, and 189.9 mm3, 240.7 mm3, and 312.0 mm3 at 12 weeks in the 7:3, 5:5, and 3:7 coating groups, respectively (Figure 4(a) and 4(b)). At the same time point, the new bone volume in each group increased significantly with the increase of β-TCP content. The new bone volume of the three groups was analyzed by a multiple repeated statistical data measurement method. The result showed that the new bone volume of the three groups was statistically different (p < .0001). Tukey's multiple comparisons test was used to compare the difference between the groups at the same time point. The new bone volume around the scaffolds between the 7:3 and 3:7 coating groups was significantly different at 4 weeks (adjusted p value = .0245), while no significant difference was found between the other groups. Meanwhile, the same result was observed at 8 weeks (adjusted p value = .0133). At 12 weeks, significant differences were found in the new bone volume around the scaffolds between any two of the groups (adjusted p value = .006 between 5:5 and 7:3 coating groups, adjusted p value = .0104 between 3:7 and 7:3 coating groups, and adjusted p value = .037 between 3:7 and 5:5 coating groups) (Figure 4(b)). Meanwhile, significant differences were also found in intra- and extra-scaffold BV/TV, TbN and extra-scaffold TbSp among the three groups (Figure 4(c), (e), (h), (j), (l)). While no significant difference was found in BS/BV and TbTh (Figure 4(d), (f), (g), (i), (k)). Post hoc test (Tukey's multiple comparisons test) of the BV/TV, TbN and TbSp indicated significant differences between 3:7 and 7:3 coating groups at 12 weeks (adjusted p value <.05). Comparison of the new bone volume, BV/TV, BS/BV, TbN, TbTh and TbSp among the three groups ( n = 9 for each group). (a). The definition of new bones around the scaffolds. The yellow part represents the new bones in the scaffolds, and the light blue part represents the new bones outside the scaffolds ( the round sleeve with the wall thickness of 0.5 mm extending outward as the external analysis area of the scaffold ); (b). Comparison of the new bone volume among the three groups; Comparison of intra-scaffold BV/TV (c), BS/BV (d), TbN (e), TbTh (f), TbSp (g) among the three groups; Comparison of extra-scaffold BV/TV (h), BS/BV (i), TbN (j), TbTh (k), TbSp (l) among the three groups. (Significance levels: NS, not statistically significant; *, p < 0.05; **, p < 0.01; ***, p < 0.001; ****, p < 0.0001).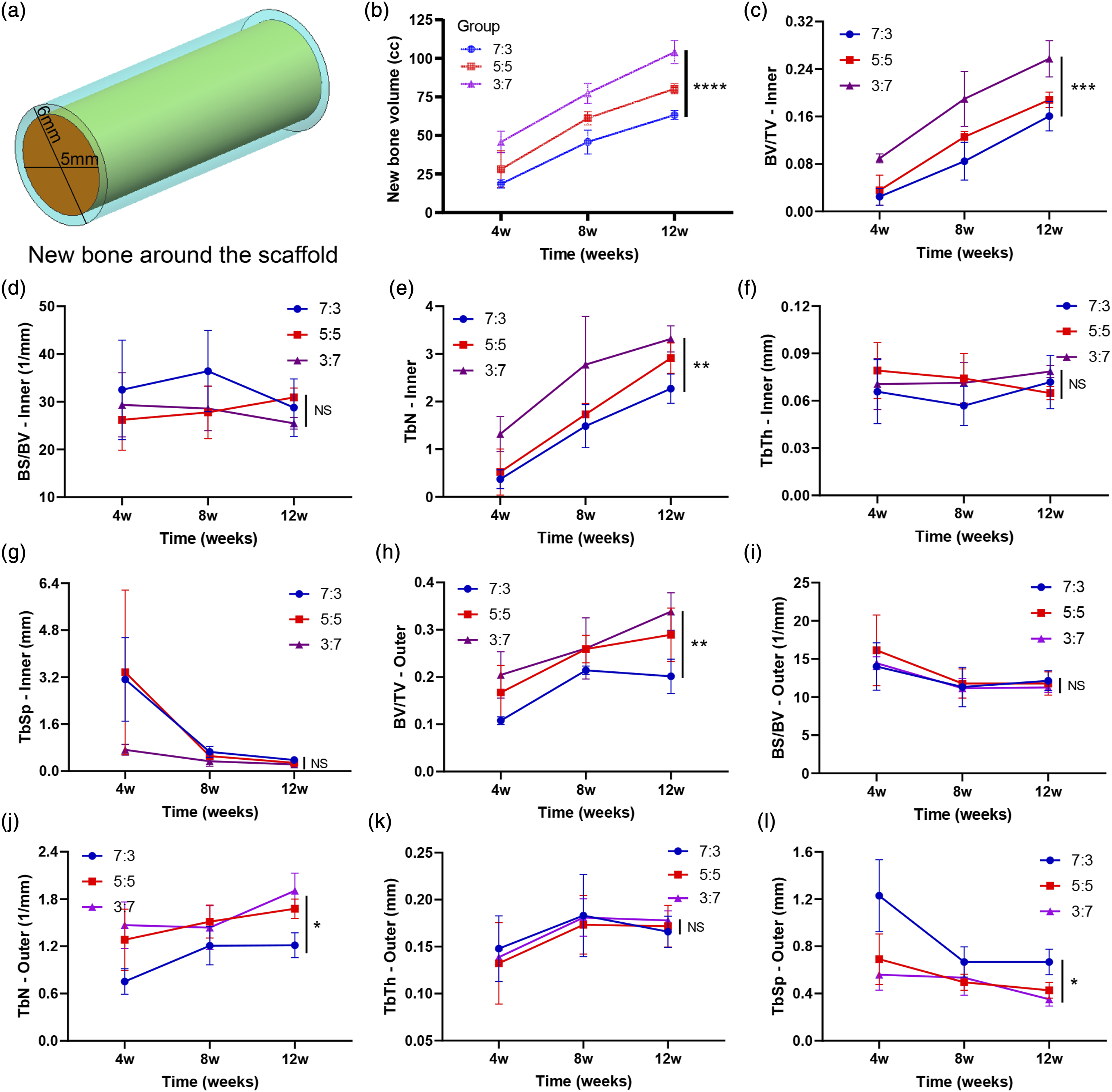
Staining of hard tissue sections
Four weeks after the operation, scattered new tissue could be observed in the scaffold-bone contact interface of each group in the longitudinal sections of HE staining, but with few basophilic stained nuclei, which suggested the newly formed tissue was mainly composed of an extracellular matrix with few cells (Figure 5(a), (e), (i)). Collagen I staining showed that a small amount of collagen I accumulated (dark brown) in the scaffold gap, suggesting active osteogenesis at the site. However, more collagen I in the 5:5 coated group of scaffolds was observed (Figure 5(b), (f), (j)). VG staining (the longitudinal section) showed that new bone tissue grew into the scaffolds from the broken ends, and no bone tissue grew into the middle section of the scaffold at 4 weeks (Figure 5(c), (g), (k)). The staining results of bone tissue sections of each scaffold group 4 weeks after the operation ( n = 3 for each group). Results of HE staining (a), collagen I staining (b), VG staining of longitudinal sections (c), and VG staining of cross-sections (d) in the 7:3 coated group 4 weeks after the operation. Results of HE staining (e), collagen I staining (f), VG staining of longitudinal sections (g), and VG staining of cross-sections (h) in the 5:5 coated group 4 weeks after the operation. Results of HE staining (i), collagen I staining (j), VG staining of longitudinal sections (k), and VG staining of crosssections (l) in the 3:7 coated group 4 weeks after the operation.
Eight weeks after the operation, more tissue grew into the scaffold gaps in square shapes than 4 weeks after the operation in each group by HE staining, but the newly formed tissue was still dominated by extracellular matrix, and some blue-stained nuclei could be seen (Figure 6(a), (e), (i)). Darker, brown-stained collagen I was observed in the scaffolds of the 7:3 and 5:5 coated groups, while less brown staining was seen in the 3:7 coated group (Figure 6(b), (f), (j)). Meanwhile, at 8 weeks, more new bone tissue was grown into the scaffold from the fracture ends in longitudinal sections of VG staining. In the cross-sections of VG staining, a small amount of new bone tissue was observed in the 7:3 coated group. More new bone tissue was observed in the 5:5 coated group, accounting for nearly 1/2 of the cross-sectional area of the scaffold. In the 3:7 coated group, it accounted for more than 1/2 of the cross-sectional area of the scaffold. In sum, more callus was observed in the 5:5 and 3:7 coated groups. However, less collagen I was observed in the 3:7 coated group than in the 7:3 and 5:5 coated groups, which might be due to the quality of section making since we could see the displaced tissue strips and disorganized tissue in the section. Finally, we believe that the difference in the volume of new bone around the scaffold between different groups could be measured more accurately by micro-CT. The results of section staining could only reflect the osteogenesis of a specific section of the scaffolds and could not reflect the overall osteogenesis around the scaffolds. The staining results of bone tissue sections of each scaffold group 8 weeks after the operation ( n = 3 for each group). Results of HE staining (a), collagen I staining (b), VG staining of longitudinal sections (c), and VG staining of cross-sections (d) in the 7:3 coated group 8 weeks after the operation. Results of HE staining (e), collagen I staining (f), VG staining of longitudinal sections (g), and VG staining of cross-sections (h) in the 5:5 coated group 8 weeks after the operation. Results of HE staining (i), collagen I staining (j), VG staining of longitudinal sections (k), and VG staining of crosssections (l) in the 3:7 coated group 8 weeks after the operation.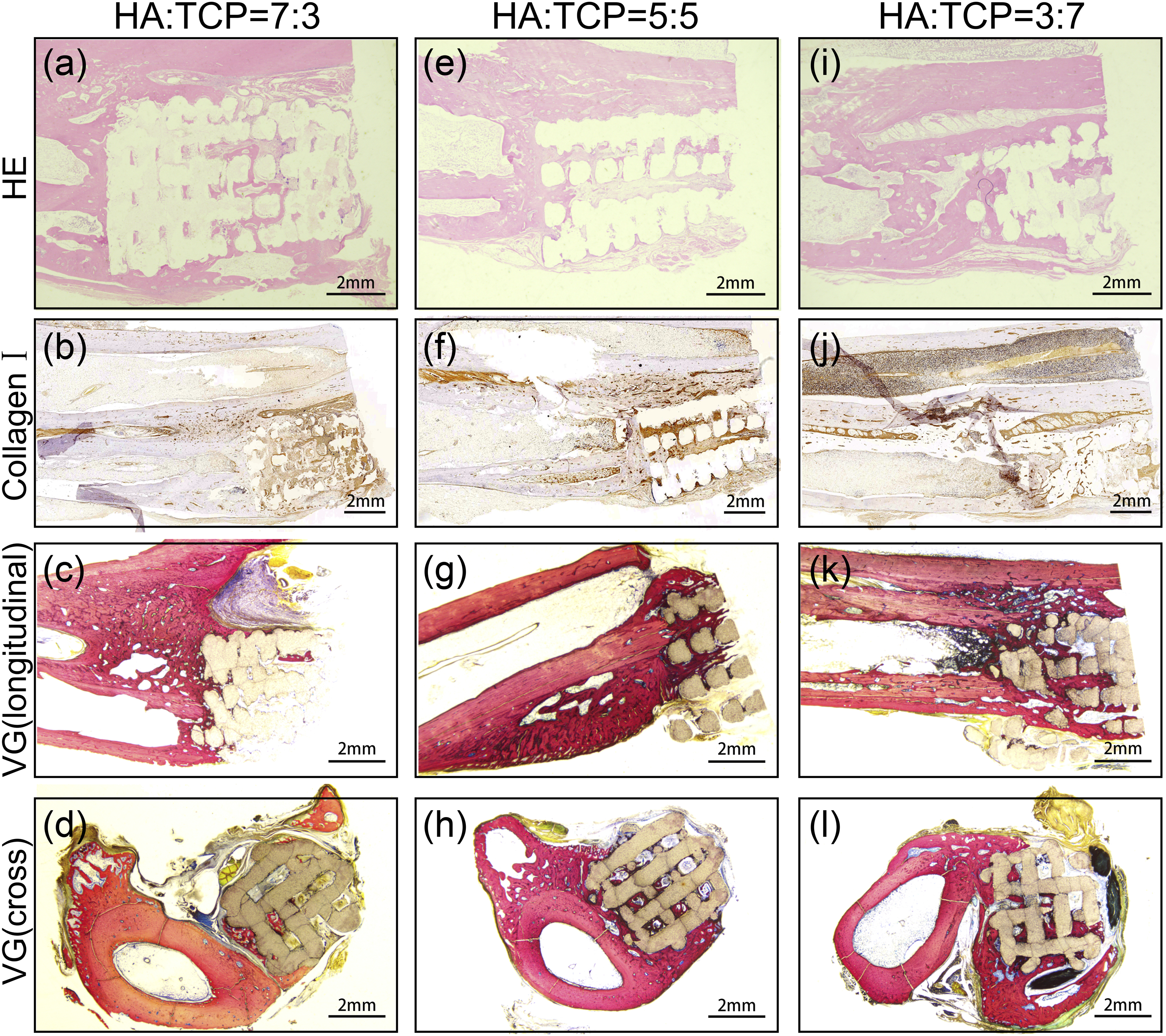
12 weeks after the operation, HE staining of longitudinal sections of the scaffold-bone contact interface in each group showed that the gap in scaffolds was filled with newly formed tissue showing a square, red-stained area (Figure 7(a), (e), (i)). Collagen I staining showed that the 7:3 coating group had more collagen I near the bone segment of the scaffold, indicating that local osteogenesis may still be active, while the other two groups had no local collagen I accumulation, indicating that osteogenesis near the bone segment of the scaffold was completed, and the osteogenic activity was decreased (Figure 7(b), (f), (j)). The staining results of bone tissue sections of each scaffold group 12 weeks after the operation (n = 3 for each group). Results of HE staining (a), collagen I staining (b), VG staining of longitudinal sections (c), and VG staining of cross-sections (d) in the 7:3 coated group 12 weeks after the operation. Results of HE staining (e), collagen I staining (f), VG staining of longitudinal sections (g), and VG staining of cross-sections (h) in the 5:5 coated group 12 weeks after the operation. Results of HE staining (i), collagen I staining (j), VG staining of longitudinal sections (k), and VG staining of crosssections (l) in the 3:7 coated group 12 weeks after the operation.
VG staining of the longitudinal section showed that there were still obvious gaps in the proximal end of the scaffold in the 7:3 coated group, which were not filled with new bone tissue, while the proximal end of the scaffold in the remaining two groups was basically filled with bone tissue (Figure 7(c), (g), (k)). In the middle cross-sections of the scaffold, there was still not much bone tissue in the middle cross-section of the scaffold in the 7:3 coated group, while the new bone tissue in the middle cross-section of the scaffold in the 5:5 coated group grew over most of the cross-section of the scaffold. Additionally, the new bone tissue in the 3:7 coated group grew over the whole cross-section of the scaffold and wrapped around the scaffold from the periphery(Figure 7(d), (f), (l)).
Discussion
The ideal bone scaffold should have a three-dimensional porous structure, suitable biodegradability, and biocompatibility and provide a large surface-to-volume ratio and appropriate biomechanical strength.26,27 Studies have shown that HA/β-TCP has good bone conductivity, and HA/β-TCP coating is effective in improving the bioactivity of scaffolds.28,29 In this study, we used 3D printing technology to prepare biomimetic scaffolds for large bone defects with HA as the body and different proportions of HA/β-TCP mixture as the surface coating. Then, the effect of the scaffolds in promoting large bone defect healing was evaluated in New Zealand white rabbits to find the optimal HA/β-TCP coating ratio for large bone defect treatment in rabbits, which might be a potential method for the treatment of large bone defects. Our findings could provide some reference and theoretical support for the clinical treatment of large bone defects.
Zhao MD et al. have found that a 15-mm bone defect exceeded the critical size for self-repair in rabbits. 30 Therefore, in this study, we established a 15-mm large segment bone defect in the right radius of the New Zealand white rabbit. According to the literature,31–33 the porosity of human cancellous bone is 40%–95%, and the compressive strength is 10–25 MPa. The porosity and compressive strength of the scaffolds prepared in this study were similar to those of normal cancellous bone, which met the basic requirements of scaffolds for large bone defects.
In animal experiments, New Zealand white rabbits with almost the same body weight and age were selected to minimize the bias, and they were randomly divided into the blank group, 7:3 coated scaffold group, 5:5 coated scaffold group, and 3:7 coated scaffold group (HA/β-TCP). All New Zealand white rabbits underwent the same operation procedure with the same bone defect length and site. The blank group did not receive scaffold implantation, while the 7:3, 5:5, and 3:7 coated scaffold groups (HA/β-TCP) were implanted with corresponding HA/β-TCP ratio-coated scaffolds.
Lateral radiographs of the affected ulnar radius taken at different times after surgery showed that although there was osteogenesis in the bone defect area of the blank group, the osteogenesis was obviously disorganized and did not grow from the broken ends towards the bone defect area but rather from the remaining periosteum or osteocytes, with scattered new bone islands in the defect area. In contrast, in scaffold implantation groups, new bones could fuse with and grow along the scaffold. At 12 weeks, the gaps between the fracture ends and the scaffolds in different scaffold groups were filled with new bones, and stability was established. However, in the blank group, there was no bony connection between the fracture ends, the new bones were rare and disorganized, and no stability was established. Therefore, compared with the blank group, the scaffold group had obvious advantages in repairing large bone defects. This is consistent with the result of the metal bone scaffolds coated with HA/β-TCP in repairing bone defects. 34
Radiographs between different scaffold groups after the operation showed that the 3:7 coated group was superior to the other two groups in the new bone volume. Micro-CT could further investigate the osteogenesis of the scaffold in cross-sections and longitudinal sections, and the results clearly indicated that new bones in the scaffold group grew into the scaffold from both sides of the broken ends and along the scaffold in opposite directions, eventually filling the inside and periphery of the scaffolds to form a new healing bony. Besides, we found significantly more new bone tissue within the scaffolds in the 5:5 and 3:7 coated groups (Figure 3). Meanwhile, the new bone volume around the scaffolds was calculated using CT 3D imaging. A significant difference in new bone volume was found among the three groups. There was no statistical difference between the 7:3 and 5:5 coating scaffold groups in the new bone volume at 4 and 8 weeks, whereas a statistical difference was found between the 7:3 and 3:7 groups. At 12 weeks, the amount of new bone volume around the scaffold increased incrementally with the increase in β-TCP ratio in the coating of the scaffolds, suggesting that 3D bioceramic scaffolds coated with 3:7 HA/β-TCP ratio were more favorable for the healing of large segmental bone defects. The results of intra- and extra-scaffold bone volume fraction (BV/TV), TbN and TbSp at 12 weeks also suggested the same conclusion that the 3:7 HA/β-TCP coated scaffolds were more conducive to bone growth. The possible reasons for this were as follows. First, β-TCP had a better ability to promote bone growth than HA; therefore, the higher its ratio, the better the promoting effect of bone healing. Second, the degradation rate of 3∶7 HA/β-TCP ratio coating was closer to the bone healing time of New Zealand white rabbits. The results obtained by HE staining and VG staining were consistent with those obtained by X-ray and micro-CT, demonstrating that the 3D bioceramic scaffold in the 3:7 coating group was more conducive to the healing of large bone defects in New Zealand white rabbits. Additionally, type I collagen staining showed that the accumulation of type Ⅰ collagen in the scaffold was more obvious in the 3:7 coating group, indicating that the osteogenesis in the scaffold was more active. Currently, there is no definitive consensus regarding the optimal ratio of HA/TCP in Biphasic Calcium Phosphate (BCP) for bone defect repair.35,36 Sung Eun Kim and Kyeongsoon Park have suggested that BCP-based materials may exhibit superior bone conduction properties and enhanced proliferation and differentiation of osteogenic cells at specific HA/TCP ratios. 37 Prisana Pripatnanont et al., in their treatment of rabbit calvarial defects using 9:1 and 8:2 HA/TCP scaffolds, found that scaffolds with a higher HA proportion had more favorable therapeutic effects. 38 Conversely, Christian Schopper and colleagues implanted 5:5 and 3:7 HA/TCP bone scaffolds into sheep rib defects and observed no statistically significant difference in new bone volume between the two groups. 39 In a bone defect model involving beagle dogs, Pil-Young Yun and his team discovered that, compared to 2:8 HA/TCP bone scaffolds, 3:7 HA/TCP scaffolds exhibited more pronounced new bone formation and space maintenance. 40 Moreover, several other studies have also suggested that BCP scaffolds with a higher TCP ratio may have a more favorable impact on promoting new bone formation.41–44 In this study, our findings also indicate that, in comparison to scaffolds with a lower TCP ratio, a 3:7 HA/β-TCP bone scaffold is more advantageous for repairing large segmental bone defects. It's worth noting that certain metrics, such as bone volume fraction, exhibited no statistically significant differences between the 5:5 and 3:7 groups. In conclusion, our findings indicate that the 3D-printed 3:7 HA/β-TCP bioceramic scaffolds outperformed the 7:3 and 5:5 scaffolds in the treatment of large bone defects in New Zealand white rabbits.
At present, the treatment of large bone defects is still a clinical difficulty, and the preparation of appropriate artificial bone to replace the autologous and allogeneic bones for the treatment of large bone defects is also a hot spot and direction of current research. Our 3D printed biomimetic scaffolds are made of absorbable biomimetic materials, which have been partially used in some fields of clinical practice and are familiar to doctors. This scaffold is very close to clinical application and has high translational value, which can be widely used in the treatment of fracture, bone defect, tumor, deformity,and other diseases, as well as in regenerative medicine. In this study, the disadvantages of HA, β-TCP, and BCP bioceramic scaffolds were improved, and 3D-printed bioceramic scaffolds with good biomechanical properties and suitable degradation rates were prepared. The 3D printing technology used in this study could adjust the shape and 3D structure of the scaffold according to the needs to meet the needs of different shapes and porosity and could also completely simulate the original bone morphological characteristics. Nowadays, 3D bioprinting technology is mature and even can use cells to print organs.45–47 In the field of bone scaffolds, some researchers have used tissue engineering combined with 3D printing technology to introduce osteoblasts and growth factors into the construction of scaffolds to create new scaffolds that are conducive to bone growth, close to in vivo microenvironment, and can achieve good results.23,48–51 Peng Liu, et al. 52 also reported a bioactive porous biocomposites coated magnesium alloy implant for bone rejuvenation and identified its biocompatibility and the ability to promote bone rejuvenation in vitro and in vivo. Li Cuidi et al. 53 summarized the current popular technologies, methods and shortcomings of regenerative medicine, which were useful for development of bone graft substitutes and bone regeneration. Therefore, combining bioprinting and tissue engineering methods to improve the performance of our biomimetic scaffold is also a direction for our next research.
This study also had some limitations. First, this study involved a limited sample size and had a relatively short duration. To gain a more comprehensive understanding of bone function after bone defect healing, it is necessary to conduct longer-term observations and research. Second, the specific mechanism by which HA/β-TCP (3:7) coating was more conducive to osteocyte growth was not clarified. Third, the effect of HA/β-TCP coating thickness on the results was not considered.
In conclusion, 3D porous bioceramic scaffolds coated with a 3:7 HA/β-TCP ratio were more favorable for the repair of large segmental bone defects in New Zealand White rabbits and could be used as a potential material for bone tissue regeneration and repair. Our findings provide some reference and theoretical support for the treatment of large segmental bone defects in humans.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Jiangxi Province of China under grant no. 81860384.


