Abstract
Background
Intra-articular injection of C-type natriuretic peptide (CNP) at the acute inflammatory stage suppressed fibrotic changes in the infrapatellar fat pad (IFP), articular cartilage degeneration, and persistent pain in a monoiodoacetic acid (MIA)-induced rat knee arthritis model. In this study, we administered CNP during the inflammation subsiding period to evaluate CNP effectiveness in knees with osteoarthritis (OA) pathology.
Methods
20 male Wistar rats were randomly divided into two groups. The rats received an intra-articular injection of MIA solution in the right knee to induce inflammation-induced joint degeneration. One group subsequently received an intra-articular CNP injection for six consecutive days from day 8, whereas another group received vehicle solution. Pain avoidance behavior tests and histological analyses were conducted to examine the therapeutic effects of CNP.
Results
The incapacitance test indicated that the percent weight on the ipsilateral limb decreased after MIA injection by day 4 and continued to decrease until the end of the experiment in the vehicle group, suggesting persistent pain in the knee. Intra-articular injection of CNP reversed the weight-bearing ratio on day 19. Histological evaluation showed that the CNP group had more residual fat tissue in the IFP and fewer calcitonin gene-related peptide-positive nerve endings compared to the vehicle group. CNP could not reverse articular cartilage degeneration.
Conclusions
Intra-articular injection of CNP after the IFP fibrosis onset had no significant effect on OA severity and extent. Nevertheless, CNP might be utilized therapeutically for OA treatment since it can alleviate persistent knee pain and inhibit structural changes in residual fat tissue.
Introduction
Knee osteoarthritis (OA) is a major musculoskeletal disorder affecting an aging society. Slow but irreversible structural changes such as osteophyte formation, joint space narrowing, degeneration of the articular cartilage and meniscus, and periarticular soft tissue fibrosis are usually observed in the knee joint with disease progression, and early proactive management is strongly recommended.1,2 The current conservative treatment for OA is based on symptom management (analgesics) with weight control and exercise to enhance knee joint stability.3,4 However, these are not designed to eliminate the underlying causes of OA, and it is often difficult to completely stop disease progression or improve the symptoms. To overcome these problems, disease-modifying osteoarthritis drugs (DMOADs), which are expected to inhibit or reverse the OA progression, are eagerly anticipated. 5
The infrapatellar fat pad (IFP) is fat tissue located in the anterior intracapsular region of the knee joint. Since it includes a certain number of free endings of sensory nerve fibers, pathological changes of the IFP, such as fibrosis and new vessel formation, have been considered to be related to persistent knee pain.6–8 We have demonstrated that fibrotic changes in the IFP after joint inflammation are closely correlated with persistent joint pain in a monoiodoacetate (MIA)-induced rat arthritis model.9–12 We further demonstrated that intra-articular injection of C-type natriuretic peptide (CNP), which has been reported to exert anti-inflammatory and anti-fibrotic effects, before the occurrence of tissue fibrosis, effectively alleviates persistent joint pain.13–15 In addition, we showed that CNP significantly inhibits articular cartilage degeneration after inflammation. 13 These data suggest that CNP may serve as a novel drug candidate (DMOAD) for controlling both the pathology and symptoms of OA. However, synovial hyperplasia and osteophyte formation are often observed at initial diagnosis. Thus, it seems difficult to apply initial anti-fibrotic treatment to prevent the onset of primary OA in actual clinical settings. Based on these considerations, in a rat MIA arthritis model, we administered CNP after IFP fibrosis had occurred and examined whether CNP could also reduce joint pain and inhibit articular cartilage degeneration. This study may be also essential to investigate the mechanism of action of CNP. If CNP could inhibit joint pain after IFP fibrosis, it would be helpful for speculation of the mechanism of action of CNP to confirm whether it is due to reversing fibrosis or suppressing inflammation.
We demonstrated that, although the effect was limited and the articular cartilage degeneration was not reversed, intra-articular CNP injection after the onset of IFP fibrosis can alleviate persistent knee pain and inhibit structural changes in the residual IFP. These data suggest that CNP might be utilized not only prophylactically, but also therapeutically for OA treatment.
Methods
Ethics
This study was approved by the Institutional Animal Care and Use Committee (IACUC) of this university (approval number A2022-104A). This study was reported in accordance with ARRIVE guidelines.
Materials
CNP, which comprises of the C-terminal 22 amino acid of human origin, was purchased from Peptide Institute Inc. (Tokyo, Japan). MIA and paraformaldehyde (PFA) were purchased from Sigma-Aldrich (St Louis, MO, USA). Ethylenediaminetetraacetic acid (EDTA), Fast Green FCF, and isoflurane were purchased from Wako Pure Chemical Industries, Ltd (Osaka, Japan). Safranin O was purchased from Chroma-Gesellschaft-Schmidt and Co (Stuttgart, Germany). Anti-calcitonin gene-related peptide (CGRP) antibody was purchased from Peninsula Laboratories LLC (San Carlos, CA, USA). Mayer’s hematoxylin solution, 1% eosin alcohol solution, and Masson stain solution B (Ponceau xylidine, acid fuchsin, and azophloxine solutions) were purchased from Muto Pure Chemicals Co, Ltd (Tokyo, Japan).
MIA-Induced rat arthritis model
Twenty male Wistar rats (Charles River, Tokyo, Japan) at 9−11 weeks of age and 310−350 g in weight were used. Rats were randomly divided into two groups, with 10 rats in each group; one group received CNP, whereas another group received a vehicle solution (phosphate-buffered saline [PBS]). Isoflurane was used to anesthetize rats (2% in oxygen, flow rate of 2 L/min). Under anesthesia, an intra-articular injection of MIA (1 mg in 30 μL PBS) was injected into the right knee joint, as described previously. 9 CNP and vehicle group animals were injected with CNP (1 µg in 20 μL of 5% glucose solution) and PBS (20 μL) into the right knee joint, respectively, for six consecutive days from eighth day after MIA administration. The rats were kept under a 12/12-h light/dark cycle with food and water ad libitum. On day 20, the rats were euthanized using carbon dioxide, and the right knee joints were dissected for histological and immunohistochemical evaluations. To confirm fibrosis, the knee joints of six rats were harvested on the eighth day independently and analyzed (Supplemental Figure S1).
Evaluation of pain avoidance behavior
Weight-bearing imbalance was evaluated at day 0 (pre-injection), and on days 1, 4, 7, 14, 18, and 19 after MIA injection using an incapacitance tester (Linton Instrumentation, Norfolk, UK) (Figure 1).10,11,13 The rats were acclimatized to the tester three times a week before MIA injection. The rats were placed in a clear plexiglass case for the first 5 min until they settled down with their left and right limbs on an independent force plate. Measurements were performed 100 times per measurement, and the percentage weight of the right hind limb (MIA side) was calculated. The percentage weight distribution of the ipsilateral hind limb was calculated as follows:
16
Schematic diagrams of in vivo experiments (a) Experimental plan (b) Schematic diagrams of sectioning. IFP fibrosis and inflammatory cell infiltration were evaluated in the mid-sagittal section, where the anterior cruciate ligament was visible (red line in the center and boxed in red in the left panel). After spacing 200 μm, medial cartilage was evaluated in the sagittal section (blue line in the center and boxed in red in the right panel).
[Weight distribution of ipsilateral hind limb rate (%) = ipsilateral weight/(ipsilateral weight + contralateral weight) × 100].
Tactile hypersensitivity of the hind paw was measured on the same day as the incapacitance test using von Frey Hairs (Touch Test Sensory Evaluator, North Coast Medical Inc. Morgan Hill, CA, USA) with weights ranging from 1 to 26 g. Rats were placed in a wire mesh cage for over 10 min until they settled down. The hair was applied to the central plantar surface of the hind paw from below the cage in ascending order starting with 1 g hair. Hair of a certain weight was pressed until it bent and held for 5 s. If the hind paw was lifted off the floor of the cage, this was considered a withdrawal response. A test was performed five times at 5 s intervals per session. If a withdrawal response occurred more than three times in a session, a one-rank lighter weight was recorded as the withdrawal threshold. If it did not, the next increased hair weight was applied. After determining the withdrawal threshold of the left side, the right side was tested in the same manner. The protocol was modified from that of a previous study.17,18 The pain avoidance behavior tests were conducted in a standardized manner without blinding after training with multiple people beforehand.
Histological and immunohistochemical examinations
The dissected knee joints were fixed in 4% paraformaldehyde at pH 7.4 for 1 week, demineralized in 20% EDTA in PBS (pH 7.4) for 3 weeks, and embedded in paraffin. Sagittal sections were cut to a thickness of 5 µm. The sections were de-paraffinized and stained with hematoxylin/eosin (H/E), Masson trichrome, or Safranin-O/fast green. The IFP inflammation score was used to semi-quantitatively evaluate the severity of synovial inflammation and fibrosis (Supplemental Table S1). Cartilage degeneration was evaluated using OA Research Society International (OARSI) score as previously reported.9,19 IFP fibrosis and inflammatory cell infiltration were evaluated in the mid sagittal section where the anterior cruciate ligament was visible. The medial cartilage was evaluated after a spacing of 200 µm (Figure 1).
Sections were deparaffinized in xylene, rehydrated in graded alcohol, and soaked in PBS for immunohistochemical staining of CGRP-positive nerve fibers. Endogenous peroxidases were quenched in methanol for 15 min using 0.3% hydrogen peroxidase. For optimal antigen retrieval, the sections were rinsed three times with PBS, fixed again in 4% PFA for 15 min, pretreated with Target Retrieval Solution (Dako, Glostrup, Denmark) in citrate buffer for 30 min at 98°C, and incubated for 20 min at room temperature. To avoid non-specific antibody binding, the sections were rinsed with PBS and blocked with 5% normal goat serum for 30 min at room temperature (Vector Laboratories, Burlingame, CA, USA). Sections were subsequently incubated with rabbit anti-CGRP polyclonal antibody (1:250 dilution in PBS containing 1% bovine serum albumin [BSA]; Peninsula Laboratories LLC, San Carlos, CA) in a humid chamber overnight at 4°C. As a negative control, normal rabbit serum was used as a primary antibody instead of anti-CGRP antibody (Supplemental Fig S2). Sections were rinsed with PBS and incubated for 30 min with biotinylated goat anti-rabbit IgG secondary antibody (Vector Laboratories). Vectastain ABC reagent (Vector Laboratories) was used to visualize the CGRP-positive nerve fibers. Hematoxylin was used to counterstain sections. The number of CGRP‐positive fibers was counted as previously described.10,11,13 Histochemical and immunohistochemical evaluations were performed by three independent raters in a blinded manner, and the Intraclass Correlation Coefficient (ICC) (2,1) were 0.77–0.93.20,21 The ICC values were classified as follows: values ≤0.5 indicated poor reliability, 0.5–0.75 indicated moderate reliability, 0.75–0.90 indicated good reliability, and 0.90 ≤ indicated excellent reliability. The ICC values of H/E stain for cell infiltration, Masson trichrome stain for IFP fibrosis, CGRP stain for CGRP-positive nerve fiber, and Safranin O/Fast Green Stain for cartilage degeneration were 0.86, 0.77, 0.86, and 0.93, respectively.
Statistical analysis
For statistical evaluations, easy R (EZR version 1.60) interface for R was utilized. 22 For comparison between the CNP group and the PBS group in the incapacitance test, von Frey hair test, and histochemical evaluation, the Shapiro-Wilk normality test was firstly used, followed by the T-test for those following the normal distribution, and the Mann–Whitney U test for those not following the normal distribution. The values were presented as means plus or minus the standard deviation (SD). Statistically significance was set at p < 0.05; 95% confidence interval (CI) was also represented in T-test.
Results
Intra-Articular injection of CNP after IFP fibrosis improves pain avoidance behavior in an MIA-Induced rat arthritis model
After extensive practice, a self-evaluation conducted prior to this experiment revealed an almost perfect success rate for intra-articular injection. Moreover, in the histological examination conducted after the animals were sacrificed, increased synovial cellularity was observed in all cases. Based on this finding, we believe that the success rate of intra-articular injection was close to 100%. There were no injection complications. Histological evaluations indicated extensive synovial hyperplasia with fibrotic lesions in the IFP on day 8 after MIA injection (Supplemental Figure S1), and CNP was administered on day 8 in this study. To evaluate time-course changes in pain avoidance behavior, an incapacitance test was conducted according to the time course indicated in Figure 1. A decrease in the percentage of weight-bearing on the ipsilateral limb was observed in both the CNP and vehicle groups by day 4 after MIA injection (Figure 2(a)). No clear difference was observed between the groups on day 14. However, the CNP group showed a mild increase in weight-bearing in the ipsilateral hind limb than that of the vehicle group on day 18 (Figure 2(a)). On day 19, a significant improvement was observed in weight-bearing in the CNP group (Figure 2(a)). The Von Frey hair test, which indicates the mechanical hypersensitivity of the hind paw, was also conducted on the same time course as that of the incapacitance test. The withdrawal threshold of the right hind paw in both groups gradually decreased by day 8 after the MIA injection. CNP did not increase the paw withdrawal threshold by day 19 (Figure 2(b)). Intra-articular injection of CNP improves weight-bearing ratio in rats. (a) Time-dependent changes in percent weight on the ipsilateral limb (incapacitance test). Intra-articular CNP injection improved weight-bearing on the ipsilateral limb on day 19. Asterisk indicates p < 0.001 by Student t-test. n = 10 in each group. (b) Time-dependent changes in tactile hypersensitivity of the ipsilateral limb. The mean difference from day 0 for each rat was compared. Numerical data are presented under each panel. n = 10 in each group.
Intra-Articular administration of CNP during the inflammation subsiding period reduced the severity of IFP fibrosis
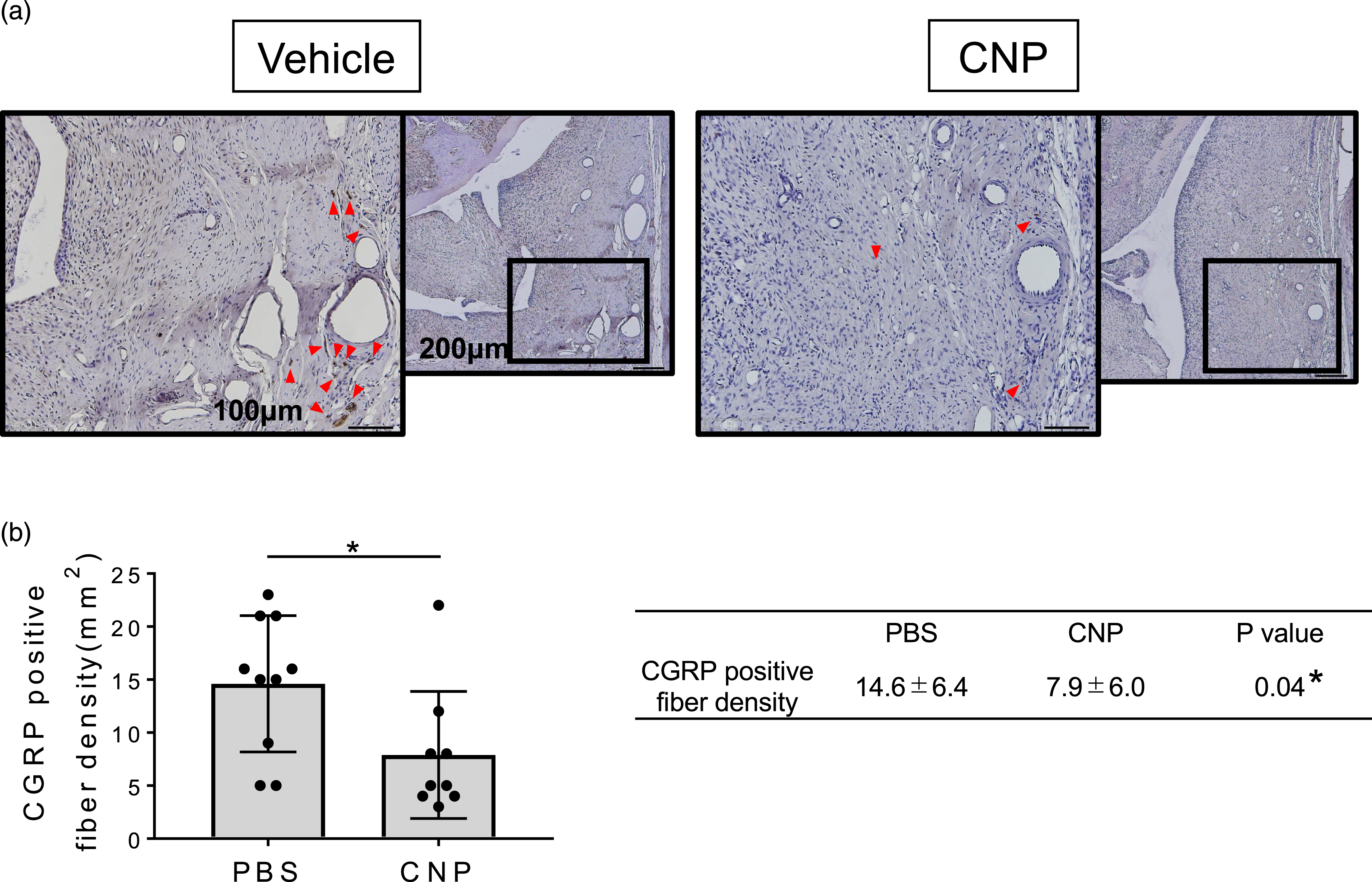
To verify whether the increase in weight-bearing after CNP administration during the inflammation subsiding period correlates with the suppression of IFP degeneration, inflammation, fibrosis, and invasion of CGRP-positive sensory nerve fiber endings were examined at day 20 after MIA administration. H/E staining indicated that in the vehicle group, almost all of the fat cells had already been replaced by fibroblastic cells, with an increase in cell density in the IFP region at day 20 (Figure 3(a), vehicle). In contrast, CNP administration during the inflammation subsiding period significantly suppressed the replacement of fat cells by fibroblastic cells, with a decrease in cell density in the IFP region (Figure 3(a), CNP). Histological grading analysis supported these results (Figure 3(b), cellularity score; vehicle vs. CNP; p = 0.03). Deposition of collagen fibers in IFP, which is indicated by blue staining during the Masson trichrome staining, was also reduced after CNP administration during the inflammation subsiding period (Figure 3(c)). Semi-quantitative IFP fibrosis grading analysis indicated that although the difference was not statistically significant, CNP administration reduced collagen fiber deposition in the IFP region (Figure 3(d), vehicle vs. CNP; p = 0.08). Immunohistochemical evaluation indicated that the number of CGRP-positive nerve fiber endings in the CNP group was significantly lower than that in the vehicle group (Figure 4, vehicle vs. CNP; p = 0.04). Histological evaluations of IFP at day 20 (H/E and Masson trichrome staining) (a) Representative images of IFP stained with H/E. Arrows indicate naïve IFP. More fat cells were observed in IFP in the CNP group. (b) Semi-quantitative evaluation of IFP cellularity according to the grading system described in Supplemental Table S1 at day 20. Dots represent each sample (n = 10 in each group). Asterisk indicates a p < 0.05 by Student t-test. (c) Representative photos of IFP stained with Masson trichrome. In the CNP group, less collagen fibers were deposited, and many fat cells remained in the IFP (arrows). (D) Semi-quantitative evaluation of IFP fibrosis according to the grading system described in Supplemental Table S1 at day 20. Fibrotic lesions tended to decrease in the CNP group (p = 0.07; PBS n = 10; CNP n = 8; Decreased n numbers in the CNP group due to a technical issue). CGRP-positive nerve fiber invasion was decreased by CNP administration (a) Arrowheads indicate CGRP-positive nerve fiber endings. The CNP group shows fewer CGRP-positive nerve fiber endings than that of the vehicle group. (b) Numbers per unit area of CGRP-positive nerve fiber endings in IFP regions are counted. Dots represent each sample. Asterisk indicates p < 0.05 by Student t-test (PBS n = 10; CNP n = 9; Decreased n numbers in the CNP group due to a technical issue).
Articular cartilage degeneration was not inhibited by intra-articular injection of CNP during the inflammation subsiding period
In our previous study, we showed that CNP administration during the acute inflammatory stage significantly inhibited articular cartilage degeneration on day 28.
13
To verify whether CNP administration during the inflammation subsiding period also has anti-catabolic effects on articular cartilage, histological evaluation using Safranin-O/fast green was performed. As shown in Figure 5, extensive loss of proteoglycan in the loading surface of the articular cartilage was observed in both groups (Figure 5(a)). OARSI grading was consistent with these results, as no significant difference was observed between the two groups (vehicle vs. CNP: 95%CI -6.85−2.20; p = 0.29) (Figure 5(b)). Articular cartilage degeneration was not inhibited by intra-articular CNP administration at the inflammation subsiding period (a) Safranin O staining indicated that both groups had extensive loss of proteoglycan on the loading surface of the articular cartilage. (b) OARSI histological grading of the articular cartilage. Dots represent each sample. No significant difference was observed between the two groups (p = 0.29; PBS n = 10; CNP n = 8; Decreased n numbers in the CNP group due to a technical issue).
Discussion
In a previous study, we reported that intra-articular CNP injection at the acute inflammatory stage suppressed IFP fibrosis, articular cartilage degeneration, and persistent knee pain in a rat model of arthritis. 13 However, patients diagnosed with OA often already have joint deformities on X-ray or MRI and knee pain at the initial examination. Thus, it seems difficult to intervene prophylactically in OA with CNP in clinical settings. To evaluate whether CNP is still effective in knees with advanced OA pathology, in this study, we administered CNP during the inflammation subsiding period. We showed that CNP administration alleviated persistent knee pain in rats. Histological analysis of the IFP showed a trend of improvement in the pathology of OA, although OA severity and extent were not improved. These data suggest that CNP might have the potential to be an effective drug not only prophylactically but also therapeutically for the treatment of knee OA.
Incapacitance tests indicated that administration of CNP during the inflammation subsiding period significantly improved weight-bearing on the affected knee on day 19. Since the rats suffered persistent knee pain from day 7 until the end of the experiment after intra-articular administration of 1 mg MIA (Figure 2(a)), 10 we consider that CNP has a palliative effect on persistent pain. In this experiment, we administered CNP daily from days 8–13 and observed increased weight-bearing on the affected knee not during days 8–13, but after day 14 (Figure 2(a)). These data suggest that the pain-relieving effects of CNP may not be directly or immediately mediated by the modulation of pain mediators, such as prostaglandins, although the mechanism has not been unveiled.
Abnormal nerve fiber density has often been suggested to be a cause of persistent pain.23–26 We have shown that CGRP-positive free nerve fiber endings were mainly accumulated in the surrounding region of small vessels invading the fibrotic lesions in the IFP. 13 Histochemical and immunohistochemical analyses showed that fibrotic lesions in the IFP tended to decrease in the CNP groups (p = 0.08, Figures 3(c) and (d)), and the density of CGRP-positive free fiber innervation into the IFP was significantly decreased at day 20 in the CNP group (p = 0.04, Figure 4). Although the molecular mechanisms underlying the antifibrotic effects of CNP remain unclear, obtained data suggest that CNP may alleviate persistent knee pain by suppressing fibrotic changes in the IFP. Das et al. 27 reported that cyclic GMP (cGMP)-dependent protein kinase (cGK), which is activated by natriuretic peptides, suppresses renal fibrosis by repressing the cell cycle dependent kinase (CDK) blockers p21Cip1 and p27 Kip1 in the kidney. It is still unclear whether CNP suppresses IFP fibrosis via a similar molecular mechanism. We will identify the target cells and target genes regulated by CNP in IFP in future studies.
Histochemical analysis also indicated that CNP had an anti-inflammatory effect on MIA-induced arthritis. As shown in Figures 3(a) and (b), the cellularity of the IFP was significantly decreased on day 20 in the CNP group. The intracellular cGMP-cGK pathway promotes dihydropyridine receptor-mediated calcium influx in CD4+ T lymphocytes, which induces the potent anti-inflammatory cytokine IL-4 in these cells. Reciprocally, inhibition of guanylate cyclase reduces Th2 cytokine production associated with T-cell receptor activation. 28 Since the presence of CD4-positive T lymphocytes has been reported in the stromal vascular fraction of IFP, 29 we hypothesize that at least some of the anti-inflammatory effects of CNP are mediated by Th2 cells that reside in the IFP.
Compared with our previous reports, 13 a subtle anti-catabolic effect on articular cartilage was observed in the CNP group in this study (Figure 5). Krejci et al. 30 reported that fibroblast growth factor (FGF) 2-induced expression of matrix-remodeling molecules, including Matrix Metallopeptidase 13 (MMP13), was partially antagonized by CNP. Our previous work on gene expression analyses showed that proteoglycan-degrading enzymes, ADAMTS-4, ADAMTS-5, and MMP13 were downregulated in inflammation-induced IFP by CNP on day 8. 13 However, no anabolic effect of CNP on the articular cartilage has been observed. A previous study reported that the activation of cGK suppresses IGF-1-induced proteoglycan synthesis in chondrocytes. 31 Thus, we believe that the reason why we did not observe an anti-catabolic effect on articular cartilage in this study is that the substrate-degrading enzymes were already activated at the initial administration of CNP, and no anabolic effect of CNP was observed on articular cartilage. On the other hand, Peake et al. 32 demonstrated that endogenous CNP released via mechanical loading showed anabolic effects in ex vivo model. This suggests that the anabolic effects of CNP may be elicited under certain conditions. Further examination is necessary in the future.
This study had several limitations. First, we performed histological analyses at only one time point, that is, at the end of the experiment (day 20). Thus, it remains unclear whether the therapeutic effects of CNP observed in this study were due to the inhibition of disease progression or the recovery of OA pathophysiology. Second, a detailed study may be conducted to optimize the frequency and dosage of CNP administration to obtain better results in animal studies. Third, no comparison with other antinociceptive/anti-inflammatory drug used in clinical practice has been conducted. To use CNP in a clinical practice, it needs to show at least non-inferiority to currently used drugs for OA symptoms relief.
Conclusion
Intra-articular injection of CNP after the onset of IFP fibrosis had no significant impact on the severity and extent of OA. However, it can alleviate persistent knee pain and prevent structural changes in residual fat tissue. CNP might have the potential to be an effective drug not only prophylactically but also therapeutically for the knee OA treatment.
Supplemental Material
Supplemental material - Therapeutic effect of C-type natriuretic peptide on persistent pain in a rat knee arthritis model
Supplemental Material for Therapeutic effect of C-type natriuretic peptide on persistent pain in a rat knee arthritis model by Shoichi Hasegawa, Jae-Sung An, Jun Hino, Yusuke Amano, Yusuke Nakagawa, Kazumasa Miyatake, Hiroki Katagiri, Tomomasa Nakamura, Ichiro Sekiya, Hideyuki Koga and Kunikazu Tsuji in Journal of Orthopaedic Surgery
Supplemental Material
Supplemental material - Therapeutic effect of C-type natriuretic peptide on persistent pain in a rat knee arthritis model
Supplemental Material for Therapeutic effect of C-type natriuretic peptide on persistent pain in a rat knee arthritis model by Shoichi Hasegawa, Jae-Sung An, Jun Hino, Yusuke Amano, Yusuke Nakagawa, Kazumasa Miyatake, Hiroki Katagiri, Tomomasa Nakamura, Ichiro Sekiya, Hideyuki Koga and Kunikazu Tsuji in Journal of Orthopaedic Surgery
Supplemental Material
Supplemental material - Therapeutic effect of C-type natriuretic peptide on persistent pain in a rat knee arthritis model
Supplemental Material for Therapeutic effect of C-type natriuretic peptide on persistent pain in a rat knee arthritis model by Shoichi Hasegawa, Jae-Sung An, Jun Hino, Yusuke Amano, Yusuke Nakagawa, Kazumasa Miyatake, Hiroki Katagiri, Tomomasa Nakamura, Ichiro Sekiya, Hideyuki Koga and Kunikazu Tsuji in Journal of Orthopaedic Surgery
Footnotes
Acknowledgements
The authors thank Ms Ami Maehara and Ms Miyoko Ojima for their technical support of this study. The authors also thank Ms Miho Okada and Ms Masayo Tsukamoto for their administrative contributions.
Authors’ contributions
Substantial contributions to study conception and design: SH, JH, KT
Substantial contributions to acquisition of data: SH, YA
Substantial contributions to analysis and interpretation of data: SH, JA, JH, YA, YN, KM, HK1(Hiroki Katagiri), TN, IS, HK2(Hideyuki Koga), KT
Drafting the article or revising it critically for important intellectual content: SH, KT
All authors read and approved the final manuscript.
Final approval of the version of the article to be published: SH, JH, KT
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Japan Society for the Promotion of Science (JSPS) to KT (21K09196).
Ethics approval and consent to participate
The Institutional Animal Care and Use Committee of the Tokyo Medical and Dental University approved this study (approval no. A2022-104 A). All animal experiments were conducted per institutional guidelines.
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Competing interests
The authors have no competing interests to declare that are relevant to the content of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
