Abstract
Purpose
Radiological imaging in Dedifferentiated liposarcoma (DDLPS) often shows the coexistence of fatty and non-fatty solid components; however, it has been shown that when fatty components were not identified on magnetic resonance imaging (MRI), the diagnosis of DDLPS would not have been diagnosed if immunohistochemical (IHC) staining had not been performed. The aim of this study was to investigate the pattern of MRI and relationship between MRI and IHC findings in DDLPS.
Methods
We retrospectively reviewed the cases of 25 patients with DDLPS. To identify the MRI spectrum of DDLPS, tumors were classified into the following four categories based on MRI findings: I = a well-defined fatty mass and juxtaposed well-defined non-fatty mass, II = a non-fatty component within a predominantly fatty mass, III = a focal fatty component within a large non-fatty mass, and IV = a non-fatty mass with atypical MRI findings. IHC staining for CDK4, MDM2, and p16 were evaluated.
Results
Category IV tumor was the most common tumor in this population. Of the 22 patients who underwent IHC staining, MDM2, CDK4, and p16 were positive in 21, 20, and 19 patients, respectively. MDM2 was positive in all 11 patients with category IV tumors; CDK4 and p 16 were positive in 10 and eight patients, respectively. There was no difference of survival between the patients with category I, II and III and category IV.
Conclusions
DDLPS without fatty components on MRI scans was mostly found. We recommend IHC staining to screen for DDLPS even if the tumors in STS cases have a non-fatty component.
Keywords
Introduction
Liposarcoma (LPS) is the most commonly diagnosed sarcoma, accounting for 9.8%–18% of soft tissue sarcoma (STS) cases. 1 According to the 2020 World Health Organization, LPSs can be categorized into four subtypes: (1) atypical lipomatous tumor/well differentiated liposarcoma (ALT/WLPS), (2) dedifferentiated liposarcoma (DDLPS), (3) myxoid types, and (4) pleomorphic types (2). Except for ALT/WLPS, the American Joint Committee on Cancer (AJCC) can be applied. 2 DDLPS usually presents as a large, painless mass. Radiological imaging often shows the coexistence of lipomatous and non-lipomatous solid components. Dedifferentiation occurs in up to 10% of WLPS cases and the risk of dedifferentiation is significantly higher for deep-seated (particularly retroperitoneal) lesions and significantly lower for lesions in the extremities. 3 DDLPS is associated with local recurrence in at least 40% of cases. Distant metastases are observed in 15–20% of cases, with an overall mortality rate of 28–30% at 5 years follow-up.3,4 At an advanced stage, chemotherapy should be considered. Accurate diagnosis is important because first-line treatment is decided based on the histological diagnosis. Generally, pazopanib is not administered to patients with DDLPS.5,6 Magnetic resonance imaging (MRI) scans of DDLPS tumors typically show well-defined non-lipomatous masses with ALT/WLPS components.7–10 DDLPS is easily diagnosed with scans showing these typical results; however, it has been shown that when WLPS components were not identified on MRI scans, DDLPS would not have been diagnosed if immunohistochemical (IHC) staining had not been performed. ALT/WLPS and DDLPS share a genomic profile characterized by amplified sequences of the 12q14-15 chromosomal region, which contains two amplicons of interest: MDM2 and CDK4.11,12 MDM2 and CDK4 IHC expression correlates with gene amplification and plays an important role in differentiating ALT/WLPS and DDLPS from other types of STS. Furthermore, p16 immunostaining has also been effective in diagnosing ALT/WLPS and DDLPS.13,14 The purpose of this study was to investigate the pattern of MRI and relationship between MRI and pathological (including immunohistochemical) findings in patients with DDLPS.
Materials and methods
We retrospectively reviewed the cases of 25 patients with DDLPS between February 2013 and September 2019 at author’s affiliated hospital. We excluded the retroperitoneum DDLPS from this study. The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Institutional Review Board of author’s affiliated hospital. Patient consent was waived due to retrospective observational study and an “opt-out” option was permitted, where the patients had an opportunity to deny participation in the study. The patients included 16 men and nine women with a mean age of 70 years (range, 44–94 years). All patients underwent MRI. We analyzed T1-weighted images (T1WI) with or without fat suppression, T2-weighted images (T2WI), and/or gadolinium-enhanced T1WI. MRI was obtained using 3.0-T scanners: Ingenia (Phillips Healthcare, Tokyo, Japan) and 1.5-T scanners: Achieva (Phillips Healthcare, Tokyo, Japan).
To identify the MRI spectrum of DDLPS, tumors were classified into the following four categories based on MRI findings: I = a well-defined fatty mass and juxtaposed well-defined non-fatty mass, II = a non-fatty component within a predominantly fatty mass, III = a focal fatty component within a large non-fatty mass, and IV = a non-fatty mass with atypical MRI findings (Figure 1).
15
All images were assessed by three orthopedic surgeons (T.N, K.A, and T.H) who were blinded to patient outcomes and all clinicopathologic data. The two orthopedic surgeons (T.N and K.A) are orthopedic oncologists who are officially certified by the Japanese Orthopaedic Association. The orthopedic surgeons are General Clinical Oncologists who are officially certified by the Japanese Board of Cancer Therapy. MRI scans showing the four mass categories; (a) Category I. A well differentiated liposarcoma along with a well-defined nonfatty mass; (b) Category II. A non-fatty component within a well differentiated liposarcoma; (c) Category III. A focal fatty component within a large non-fatty mass; and (d) Category IV. A non-fatty mass (right; T1WI, right; T2WI).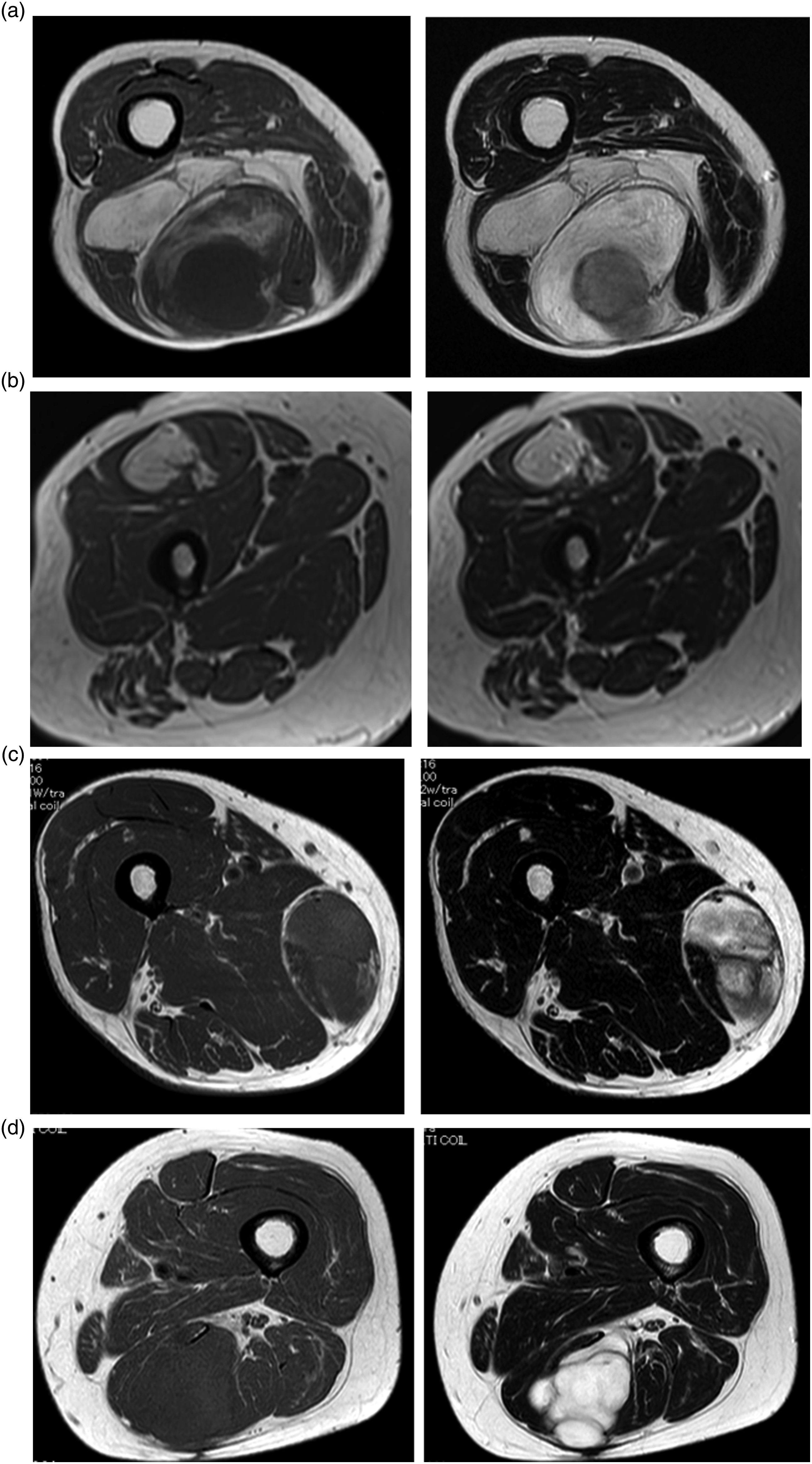
All tumors were histopathologically confirmed as DDLPS after wide excision (n = 21), needle biopsy (n = 3), or incision biopsy (n = 1). Histopathological examinations were performed for all cases. Examinations for 22 of the 25 patients were supplemented with IHC staining for CDK4, MDM2, and p16. IHC staining was not performed for the remaining three patients because the diagnosis of DDLPS was apparent even without IHC staining. To evaluate the relationship between MRI findings and oncological outcomes, we divided the patients into two groups according to the presence/absence of a fatty component and calculated the 5 years overall survival (5Y-OS) and 5 years event-free survival (5Y-EFS) using the Kaplan-Meier method. All statistical analyses were performed with the EZR graphical user interface (Saitama Medical Center, Jichi Medical University, Saitama, Japan) for R (R Foundation for Statistical Computing, Vienna, Austria), which is a modified version of R Commander designed to add statistical functions frequently used in biostatistics. Survival curves were generated using the Kaplan-Meier method and compared using the log-rank test.
Results
Patient’s characteristics
The characteristics of the patients with dedifferentiated liposarcoma.

AJCC; American Joint and cartilage committee, M; Male, F; Female, Cx; chemotherapy, CIRT; Carbon ion radiotherapy, Rx; Radiotherapy.
MRI and IHC findings (Table 1)
MRI findings revealed that the tumor category was I (Figure 1(a)) in six cases (24%), II (Figure 1(b)) in two cases (8%), III (Figure 1(c)) in six cases (24%), and IV (Figure 1(d)) in 11 cases (44%); evidently, category IV tumor was the most common tumor in this population. Of the 22 patients who underwent IHC staining, MDM2, CDK4, and p16 were positive in 21, 20, and 19 patients, respectively. MDM2 was positive in all 11 patients with category IV tumors; CDK4 and p 16 were positive in 10 and eight patients, respectively.
Clinical outcome
Of the 25 patients, 21 underwent surgical resection, and four of these received perioperative chemotherapy whereas one received adjuvant radiotherapy. Two of the four patients who did not undergo resection received palliative radiotherapy alone, whereas the other two received palliative chemotherapy. By the final follow-up, only 19 of the 25 patients were alive. For all patients, 5Y-OS and 5Y-EFS were 66.4% (95% confidential intervals (CI): 38.3–84%, Figure 2) and 53.2% (95% CI: 30–71.8%), respectively. Five patients developed local recurrence and eight patients developed metastasis. When we divided the patients into two groups according to the presence of the fatty component, the 5Y-OS was 72.5% (95% CI: 34.9–90.7%) for patients in the fatty component group (category I, II and III) and 55.6% (95% CI: 14.1–83.8%) for patients in the non-fatty component group (category IV) (p = 0.42, Figure 3). The 5Y-EFS was 51.6% for patients in the fatty component group (category I, II and III) and 57.3% for patients in the non-fatty component group (category IV) (p = 0.73). There was no significant between-group difference in terms of 5Y-OS and 5Y-EFS. Of the 13 patients with relapse, 10 received systemic chemotherapy at the advanced stage. For first-line treatment, six were administered eribulin, two were administered doxorubicin, one was administered gemcitabine with docetaxel, and one was administered pazopanib. We prescribed pazopanib to one patient because doxorubicin was administered as eribulin and trabectedin were not approved. Kaplan Meier curve showing the overall survival for 25 patients with DDLPS. Kaplan Meier curves showing the overall survival according to the existence of a fatty component on MRI scans (A: non-fatty component (category IV), B: Presence of a fatty component (category I, II and III)).

Discussion
DDLPS are previously characterized by the coexistence of well differentiated and poorly differentiated, non-fatty areas, in portions of the same tumor.8,15–17 However, in this study, we analyzed the MRI scans of patients with DDLPS and found that 44% of the patients had non-fatty masses without any fatty component. The final diagnosis was made upon IHC evaluation of these patients. The present study used the four DDLPS morphological categories described by Yun et al. 15 They reported seven of 14 patients with a total 18 samples with DDLPS (38.9%) had non-fatty masses without any fatty component. Thus, without IHC evaluation, 40% of DDLPSs might have been misdiagnosed as spindle cell sarcomas, such as fibrosarcoma and undifferentiated pleomorphic sarcoma (UPS). They also did not include DDLPS at retroperitoneum. Hong et al. 17 categorized retroperitoneal DDLPS into four groups. Tumors were classified into the following four categories based on MRI findings. Category I = Nonfatty component within predominant fatty mass, Category II = focal fatty component within large nonfatty mass, III = well-defined fatty mass and well-defined nonfatty mass, IV = two masses with predominantly nonfatty component. Surprisingly, all 15 patients with DDLPS had a fatty component. Therefore, the categorization by Yun may be useful for DDLPS at musculoskeletal system.
However, in the present study, we can’t find the difference of survival between the patients with fatty component (category I, II and III) and without fatty component (category IV). We should need to evaluate the meaning of the categorization by Yun in a large cohort. In this study, we emphasize that 44% of patients had non-fatty masses without any fatty component. We also consider the accurate diagnosis of DDLPS to be important. Before the approval of new drugs such as pazopanib, trabectedin, and eribulin, this issue would have been irrelevant as treatment was the same for varying types of STS (DDLPS, fibrosarcoma, UPS, etc.). Doxorubicin, with or without ifosfamide, was administered for the treatment of all types of STS. However, we now use the aforementioned new drugs and they each have specific characteristics. It has been reported that translocation-related sarcoma responds well to trabectedin treatment. Trabectedin has shown good response to myxoid liposarcomas. 18 Results from one multicenter study showed that patients with LPS were less likely to achieve a long-term response regardless of the pazopanib treatment.5,6 The other report described that there was a significant difference in PFS between patients with L-sarcoma (LMS and LPS) and those with non-L-sarcoma following treatment with eribulin. 19 These findings indicate that correct diagnosis of STS histopathology is very important for effective treatment. In the present study, eribulin was mainly administered in cases with advanced stage DDLPS if doxorubicin was administered for perioperative chemotherapy or if the patients were elderly. Cytogenetically, WLPS and DDLPS characteristically harbor supernumerary ring and/or giant chromosomes containing the amplification and overexpression of MDM2 and CDK4 cell cycle oncogenes. 13 p16 is an important cell cycle regulator. Overexpression was reported in patients with WLPS and DLPS, although its precise role remains unknown. 20 Thway et al. 13 reported that the trio of CDK4, MDM2, and p16 was expressed in 70% of patients with WLPS and DLPS, and the expression of at least two markers was noted in 95% of the cases. In the present study, we observed that the trio was expressed in eight of the 11 patients (72.7%) that had a non-fatty component alone, with all (100%) expressing at least two markers. Therefore, IHC staining for these proteins is a reliable tool for the accurate diagnosis of STS, although this must be validated further.
There were some limitations. First, lack of standardization in the MRI acquisition protocol was limitation in this study, although we think the MRI qualities sufficient to evaluate the DDLPS with fatty and non-fatty components. Furthermore, only 25 patients were very small and this is a limitation in this study, although there was largest number of the patients for the analysis of MRI in patients with DDLPS at musculoskeletal system. We should validate it in a large cohort.
In conclusion, we recommend IHC staining to screen for DDLPS even if the tumors in STS cases have a non-fatty component without the WLPS component. Approximately 40% of the patients with DDLPS may have non-fatty masses without any fatty component.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
