Abstract
Introduction
Incorrect positioning is one of the main factors for glenoid component loosening in reverse shoulder arthroplasty and component placement can be challenging. This study aimed to assess whether Patient-Specific Instrumentation (PSI) provides better guide pin positioning accuracy and is superior to standard guided and freehand instrumentation methods in cases of glenoid bone deformity.
Materials and Methods
Based on the Walch classification, five different scapula types were acquired by computed tomography (CT). For each type, two different surgeons placed a guide pin into the scapula using three different methods: freehand method, conventional non-patient-specific guide, and PSI guide. Each method was repeated five times by both surgeons. In these experiments, a total of 150 samples of scapula models were used (5 × 2 × 3 × 5 = 150). Post-operative CT scans of the samples with the guide pin were digitally assessed and the accuracy of the pin placement was determined by comparison to the preoperative planning on a three-dimensional (3D) model.
Results
The PSI method showed accuracies to the preoperative plan of 2.68 (SD 2.10) degrees for version angle (p < .05), 2.59 (SD 2.68) degrees for inclination angle (p < .05), and 1.55 (SD 1.26) mm for entry point offset (p < .05). The mean and standard deviation errors compared to planned values of version angle, inclination angle, and entry point offset were statistically significant for the PSI method for the type C defected glenoid and non-arthritic glenoid.
Conclusion
Using the PSI guide created by an image processing software tool for guide pin positioning showed advantages in glenoid component positioning over other methods, for defected and intact glenoid types, but correlation with clinical outcomes should be examined.
Keywords
Introduction
Reverse shoulder arthroplasty (RSA) is generally successful although different complications can occur. The most common complications in RSA are instability, infection, and glenoid loosening.1–3 Glenohumeral instability in RSA can be caused by malpositioning of the joint prosthesis components. Any misplacement of the component in distance or angle can cause instability.4–6 In conventional methods, the identification of anatomical landmarks and accurate positioning of the implant baseplate are difficult to achieve when there are deformities in the glenoid cavity. 7 For instance, the implant inferior version and tilt angle have shown 16- and 12-degree variations, respectively, in traditional surgery methods. 8 Conventional methods of implantation are not able to restore the retroversion angle in cases with severe glenoid deformity.9,10 Correct placement of the glenoid implant, which is one of the main goals of preoperative planning, is crucial for prosthesis durability and better shoulder function,11,12 component placement can be challenging because of the variable anatomy of the scapula, joint contractures, bone loss in the glenoid, and lack of reliable anatomic landmarks after previous operation. 13
Preoperative planning and use of patient-specific guides can help improve implant positioning14–19 and reduce the risk of complications resulting from malpositioning. Software can be of great help in preoperative planning and creating patient-specific guides. Viewing a three-dimensional (3 D) model of the anatomy of a patient and their specific condition can help a surgeon to determine different treatment solutions
The purpose of this study was to evaluate and compare the accuracy of the PSI in different glenoid types under the hypothesis that PSIs designed using preoperative planning software would provide better guide pin positioning than standard guided and freehand instrumentation methods, especially in cases of glenoid bone deformity.
Material and methods
Scapula bone models
This study used computed tomography (CT) scans of five different scapula types of real patients. As most glenoid deformities are related to osteoarthritis, the Walch classification was used to build the study groups. Based on the Walch classification, 28 one scapula each was type A2, B2, C, and D, and one scapula had an intact or non-arthritic glenoid. Based on the Favard classification, 29 the same scapula bones were categorized as E0 (non-arthritic glenoid), E1 (Walch types A2, B2, and D), and E2 (Walch type C). High-resolution imaging was performed with a pitch of less than one during CT scanning using Aquilion 16; Toshiba Medical Systems, Japan. The CT scan parameters were as follows: 120 kV, 0.75-mm thick axial slices, a pixel spacing of 0.53, and an image matrix of 512 × 512. A total of 150 full-size physical scapula models were manufactured using the 3 D-printing rapid prototyping method. Each scapula bone model was manufactured 30 times to be operated on by three different methods and by two different shoulder surgeons with a minimum experience of 5 years in shoulder arthroplasty (KB, MK).
3D planning with PSI software (ShoulderART)
The present research developed a new software tool for shoulder arthroplasty planning by the authors.
An explanation of the 3 D planning with the software is presented here and the software tool’s development stages are explained in more detail in a study by the same authors. 30 In reverse shoulder arthroplasty, the PSI guide is used intra-operatively to direct the Kirschner wire which itself is used as the guide for the baseplate location and orientation. The software provides the surgeon with proper tools to preoperatively make adjustments to achieve the best implant position.
The software is designed to first import the patient’s data captured using CT scan with predefined specifications, in DICOM format. Next preprocessing is performed on the imported data to improve the quality of the images and apply segmentation of the bones from other tissues. Afterward detection of Scapula bone and its features are performed. At this point, the software possesses the required information to perform the next steps in designing the PSI guide and providing the user with proper information. The software presents two-dimensional (2 D) and 3 D images of the scapula on the graphical user interface (GUI) to allow the surgeon to observe scapular anatomy (Figure 1). To facilitate 3 D preoperative planning, different types and sizes of the implant can be chosen by the surgeon to be displayed on the bone model. Afterward, the surgeon can precisely adjust the implant position and orientation. Parameters such as version, inclination, amount of bone removal, and baseplate seating percentage are presented to the surgeon to help them make better decisions. After the surgeon is satisfied with the implant position and orientation, the software generates a patient-specific surgical guide model so that the surgeon can perform a final check. This guide model is then saved and used for rapid prototyping. The process of planning and creating the guide’s computer model can take up to 15 minutes based on the user’s experience and the patient’s anatomy. The graphical user interface of the ShoulderART software.
The guide automatically created by the software has a central cylinder with a hole that orientates guide pin insertion during operation. This central cylinder hole has the exact surgeon-defined location and angles for the entry point location. The rest of the guide consists of a body that provides enough surface area to firmly sit on the glenoid surface, using the coracoid base as a reference. The body of the guide covers both sides of the coracoid base to create a unique mounting position that sits securely in place (Figure 2). (Left) Implant model located into the 3 D space, (Center) patient-specific guide model created by the PSI software at position and orientation desired by the surgeon, (Right) Glenoid reamed at the height adjusted by the surgeon for best sitting.
Experiment method
The scapula bone models of the selected types were manufactured for the experiment from polylactic acid (PLA) using 3 D printing technology. The models were labeled and coded. For each group, the experiment was performed using three different methods, and each method was repeated five times by two different surgeons. The experiment was performed 30 times for each group, making a total of 150 tests.
The 3 D planning was performed for each scapula by the surgeons and the same planning was used for all methods. The first method was the freehand technique in which the surgeon does not use any guide for positioning the guide pin for the glenoid baseplate. The surgeon first marked the inferior circle of the glenoid surface and determined the entry point of the guide pin without any helping device.
In the second method, the surgeon used an RSA conventional glenoid guide with a predefined angle to guide the guide pin. The surgeon placed the guide flush to the inferior rim of the glenoid cavity. This guide position and the central hole with a 10° inclination allows the possibility of the appropriate placement of the central guide pin.
The third method used the guide created by the PSI software. It had an exact position for the guide pin and sat tightly on the glenoid area in a specific predefined position.
For the experiment, the models were held by fixtures connected to a table. For the freehand and the conventional guide methods, models were covered by aluminum foil and only the glenoid cavity of the model was visible to the surgeon during the testing to simulate the surgeon’s view of the scapula bone in a real operation (Figure 3). The different scapula models were randomly mixed and the surgeons were blinded to the randomization. Operation using the PSI guide was performed after the other methods and other sections of the scapula were not covered as the guide can only sit at a specific predefined position (Figure 4). During the experiment, all models were recoded to be distinguished based on the surgeon and the method. The sequence of the experiment is described in Figure 5. (Left) Scapula model covered by aluminum foil, mounted on the fixture, (Right) Experiment setup, surgeon inserting the guide pin with the freehand method. (Left) Inserting guide pin using a general guide, (Center and Right) ShoulderART PSI guide mounted on the glenoid. The sequence of experiment performed.


Measurement method
Post-operative CT scans of the models and guide pins were obtained using the same CT scan protocol used for the software. These CT images were used to create 3 D models of the bones with the guide pin in place inside the bone. The 3 D models of each sample were registered in 3 D space to match each other exactly (Figure 6). Then, using the Friedman
31
and Maurer
32
methods, the version and inclination of the 3 D models were measured digitally. For the measurement of version angle, the registered 3 D models are projected in the axial plane. The version angle was found as the angle between the line connecting anterior and posterior edges of the glenoid and the line of K-wire inserted. For the measurement of inclination angle, the registered 3 D models are projected in the coronal plane. The inclination angle was measured as the angle between the line connecting the uppermost point of the glenoid and the lowermost point of the glenoid and the line of K-wire inserted. The position of the guide pin entrance to the glenoid surface was also measured in reference to the planned entry point. The mean value, the standard deviation of the version, and inclination and entry point locations were measured for each sample operated on by each method. (Left) All 30 samples of a specific group bone model registered. The deviation in the angles and entry point can be observed, (Right) Same group samples operated only by using PSI guide.
The measurement results were used for two types of evaluation. First, the reliability and precision of the patient-specific guide in positioning the guide pin on different glenoid types were assessed. Second, the PSI method was compared to the other two conventional methods.
For the evaluation of reliability and precision, the actual experimental results were compared to the values obtained in the preoperative planning.
When comparing the PSI method to the other methods, the results were evaluated to determine if the patient-specific guide had advantages over conventional methods.
The version and inclination angle errors were defined as the absolute values of the differences between the planned and post-operative angles. The error for the entry point offset was defined as the absolute value of the distance of the post-operative entry point in reference to the planned entry point.
Statistical analysis
IBM SPSS Statistics for Windows, version 24.0 was used to conduct all statistical analyses. Analysis of variance (ANOVA) and two-way ANOVA were employed for all comparisons. Duncan’s post-hoc tests were used to differentiate the subgroups. A significance level of 0.05 was used to determine statistically significant results. For the sample size of 50 for each group, assuming ANOVA at a significance level of 0.05, a power calculation of 0.78 was obtained for our study.
Results
The real values of the glenoid native version angle, planned version angle, and post-operative achieved version angle for each type of scapula only for the PSI method.
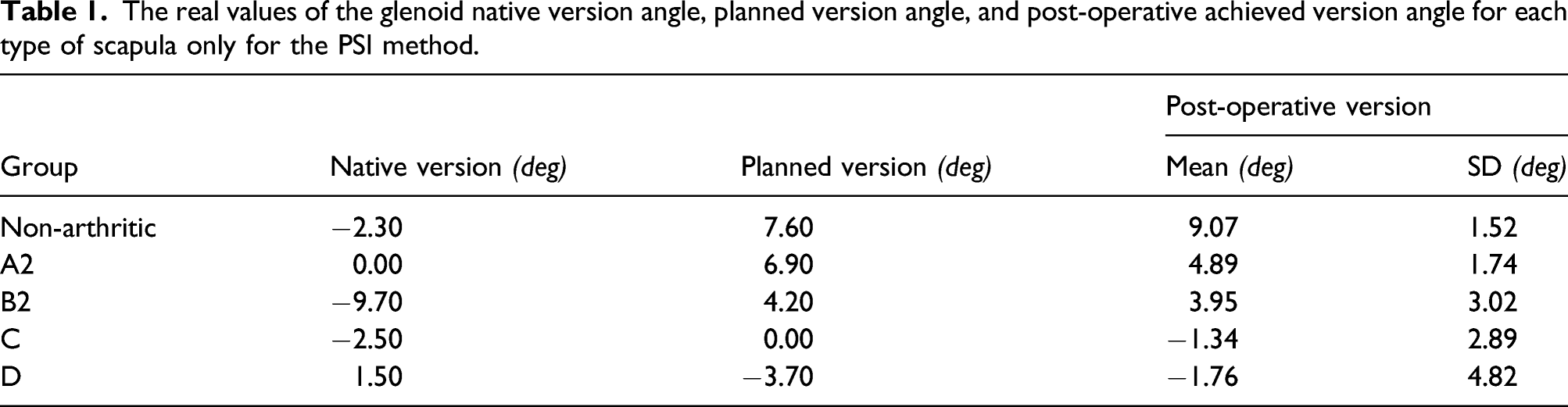
The real values of the glenoid native inclination angle, planned inclination angle, and post-operative achieved inclination angle for each type of scapula only for the PSI method.

The deviation from the plan (error) values between planned and post-operative version angle, inclination angle, and entry point offset for the PSI method.
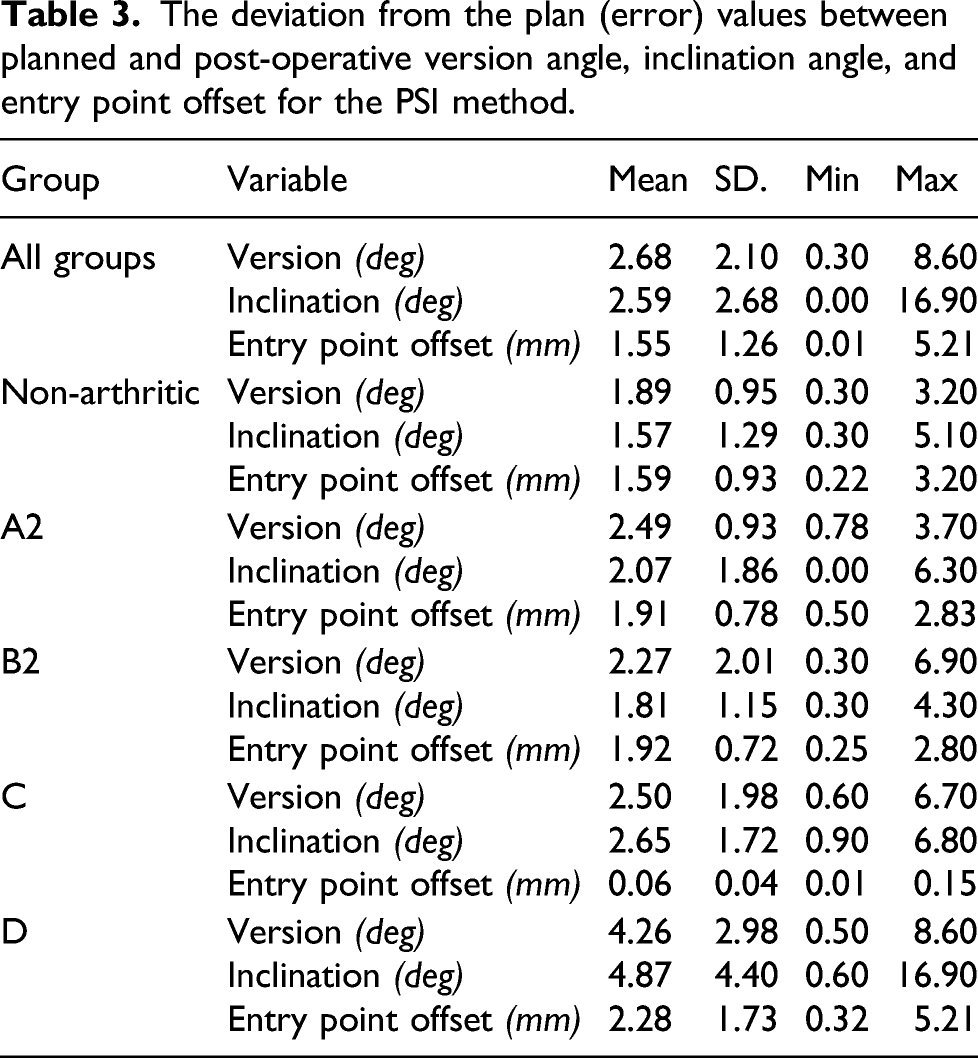
Version angle, inclination angle, and entry point offset deviation from the plan (error) values, for each method, for all groups collectively and each group separately.

When all the groups were analyzed together, the PSI method was better than the other methods. The version angle deviation from the plan for PSI (2.68° ± 2.10°) was significantly better than those of the other methods (conventional guide: 6.44° ± 4.62°, freehand: 6.86° ± 4.43°, p = .035). The inclination angle deviation from the plan for PSI (2.59° ± 2.68°) was also significantly better than those of the conventional guide (6.31° ± 4.45°) and freehand (7.44° ± 4.76°) (p < .05). Finally, the entry point offset deviation from the plan for PSI (1.55 mm ± 1.26 mm) was also significantly better than those of the other methods (conventional guide: 2.29 mm ± 1.74 mm, freehand: 2.17 mm ± 1.48 mm, p < .05).
The quantification of the significance of differences observed between all three methods for all groups collectively.

The version angle of the PSI method was significantly better than those of the conventional guide and freehand methods (p = .031). The PSI method was significantly better than the conventional guide method (p = .033) but was in the same subgroup as the freehand method (p = .015) for the inclination angle.
The version angle deviation from the plan for PSI (1.89° ± 0.95°) in the non-arthritic group was significantly better than that for the other methods (conventional guide: 9.76° ± 5.29°, freehand: 9.8° ± 4.24°, p < .05). The inclination angle deviation from the plan for PSI in the non-arthritic group (1.57° ± 1.29°) was better than that of the other methods (conventional guide: 4.45° ± 3.19°, freehand: 9.71° ± 6.16°, p = .008).
The version angle deviation from the plan for PSI in the type C group (2.5° ± 1.98°) was significantly better than those of the other methods (conventional guide: 8.6° ± 5.39°, freehand: 9.9° ± 4.29°, p = .001). The inclination angle deviation from the plan for PSI in the type C group (2.65° ± 1.72°) was significantly better than those of the other methods (conventional guide: 7.47° ± 3.47°, freehand: 9.49° ± 3.4°, p = .001). The entry point offset deviation from the plan for PSI in the type C group (0.064 mm ± 0.04 mm) was better than those of the conventional guide (0.14 mm ± 0.05 mm) and freehand (0.10 mm ± 0.05 mm), (p = .025).
The quantification of the significance of differences observed between all three methods for each group.
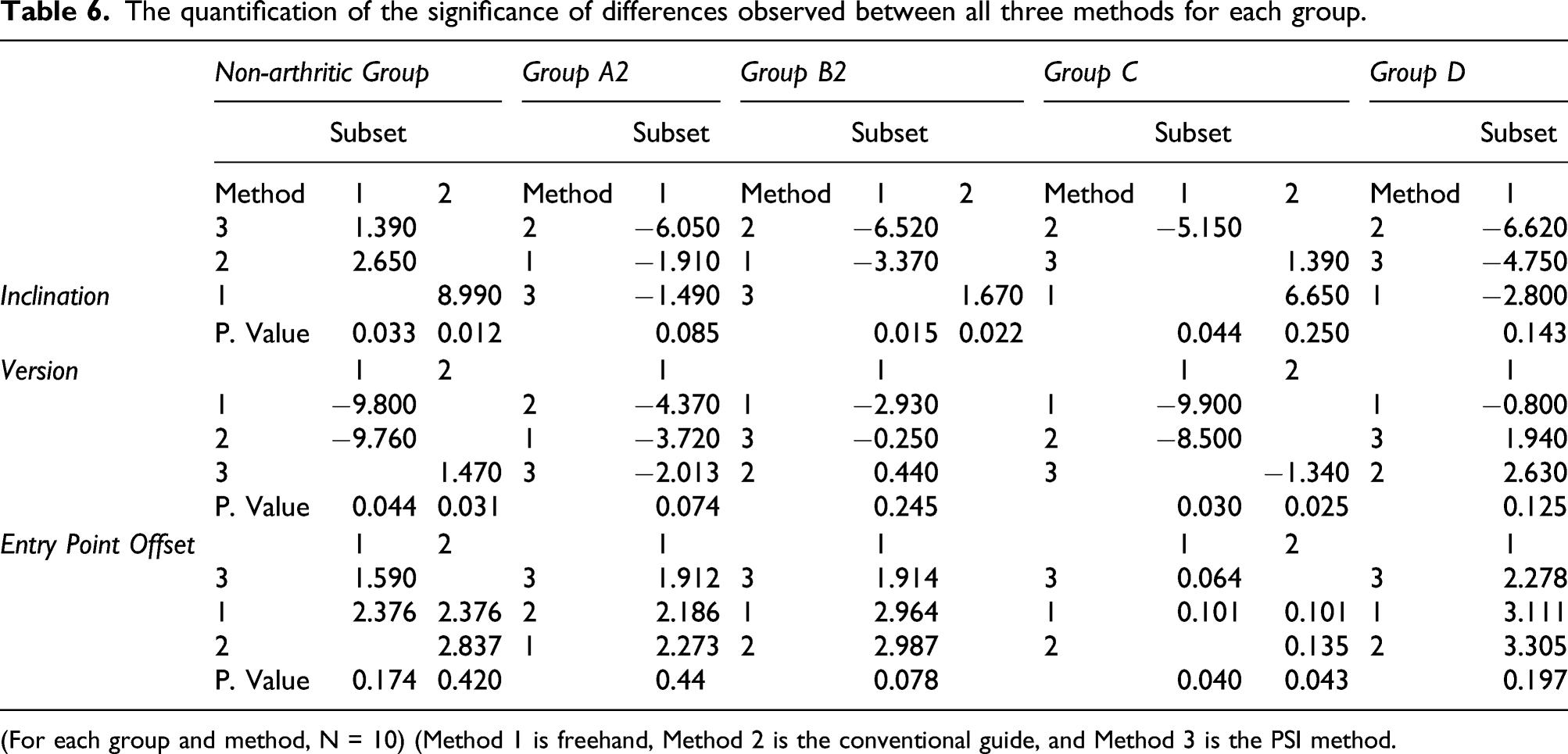
(For each group and method, N = 10) (Method 1 is freehand, Method 2 is the conventional guide, and Method 3 is the PSI method.
For the non-arthritic group, the version angle of the PSI method was significantly better than those for the conventional guide and freehand methods (p = .044). The PSI method was significantly better than the freehand method (p = .012) but was in the same subgroup as the conventional guide method (p = .033) for the inclination angle.
For the type C group, the PSI method had a significantly better inclination angle than that of the conventional guide method (p = .044) but was in the same subgroup as the freehand method (p = .250). The PSI method was significantly better than the conventional guide method (p = .040) but was in the same subgroup as the freehand method for entry point offset (p = .043). The PSI method was significantly better than the other two methods for the version angle (p = .025).
No statistically significant differences were observed between the two surgeons in terms of the deviation from the plan for version angle, inclination angle, and entry point offset.
Discussion
This study used the results of a 3 D patient-specific guide created by 3 D preoperative planning for the evaluation of the accuracy and reliability of the PSI for guide pin positioning as well as for the comparison of the PSI method to conventional methods.
First, the accuracy and reliability of the PSI were tested by comparing the version angle, inclination angle, and entry point offset between the actual post-operative results and the planned values using the PSI method.
As shown in Tables 1, 2, and 3, the post-operative values of the PSI method were very close to the planned values and the patient-specific guide achieved the planned values with good accuracy.
The results were also evaluated for the comparison of the PSI method to conventional methods. As shown in Tables 4, 5, and 6, when all types are considered together, the error values of the PSI method showed a better mean and standard deviation than those of the other methods.
Comparisons of error values between different types showed better mean and standard deviation values for the PSI for all groups, except for four values, with significant differences in the non-arthritic and type C groups. The exceptions were the mean and standard deviation of the version angle, the standard deviation of the inclination angle, and the standard deviation of the entry point offset in the type D group and the standard deviation of the entry point offset in the non-arthritic group.
The standard deviation of the data for different glenoid types and how a patient-specific guide reduced the chance of extreme malpositioning are shown in Figure 7. Only version angle, inclination angle, and entry point offset in the type D group and entry point offset in the non-arthritic glenoid type group showed higher standard deviation values. Comparison of results of three different methods for each group’s version angle, inclination angle, and guide pin entry point distance.
While the PSI guide showed mean version and inclination angle values and standard deviations very close to the planned values, the mean values of the other methods did not always agree with the planned values. This observation demonstrates how surgeons can make different decisions about implant positioning when they are provided more visual information about the scapula. These results showed that patient-specific guides can help to reduce the risks of surgery complications resulting from deciding the wrong positioning of the guide pin without sufficient visual preoperative data.
Other studies have also shown the accuracy of using patient-specific guides in shoulder arthroplasty. Levy et al. 13 reported a translational accuracy of 1.2 ± 0.7 mm, inferior tilt accuracy of 1.2° ± 1.2°, and glenoid version accuracy of 2.6° ± 1.7° in their evaluation of the accuracy of patient-specific planning and a patient-specific drill guide for glenoid baseplate placement in RSA. Walch et al. 17 showed a mean entry point position error of 1.05 mm, a mean inclination angle error of 1.42, and an average error in the version angle of 1.64°. In our study, the entry point offset accuracy was 1.55 mm ± 1.26 mm, the inclination angle accuracy was 2.59° ± 2.68°, and the version angle accuracy was 2.68° ± 2.10° for all samples. The other studies mentioned above showed better overall accuracy when all types of deformities are considered, but this study is to the knowledge of the authors, the first study that evaluates the accuracy between different glenoid types.
Other techniques have also been used to improve the accuracy of the guide pin or glenoid component positioning in shoulder arthroplasty. Similar to PSI, navigation systems showed better accuracy than those of standard methods.8,33–35 While PSI requires time for preoperative planning and guide manufacturing, navigation systems can be more complex and require extra effort and time for calibration, reference frame positioning, and real-time tracking of components.8,33–35 Augmented reality is a new surgical assistance method that could improve RSA outcomes by providing visual information at the time of surgery and enabling remote interactions between surgeons.36,37
This study evaluated the accuracy of the PSI method in guide pin positioning and found that PSI was more beneficial than conventional guide and freehand instrumentation methods in cases with glenoid bone deformity. The results of this study demonstrated the guide pin positioning accuracy of PSI and showed that, for non-arthritic glenoid type and defective glenoid types C, B2, and A2, PSI was more accurate than other methods, although the results were statistically meaningful for non-arthritic glenoid and defective glenoid type C.
This study had some limitations. Although 30 samples from each type of scapula were tested, future research should test more glenoid types to further evaluate the accuracy and performance of the method. The scapula models used for the experiment did not have any soft tissue coverage as would be present in actual surgical interventions. Cadaveric models can be used in future studies.
Conclusion
Evaluation of the results of experimenting PSI glenoid guide pin positioning in RSA for different glenoid deformity types showed advantages in accuracy and reliability compared to non-PSI methods. This study used a PSI preoperative planning software and experimented on five different types of glenoid types. The correlation of these outcomes can be examined more by in Vivo studies.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
