Abstract
On 30 September 2021, Viet Duc University Hospital in Northern Vietnam, reported a severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) Delta outbreak, leading to an 18-day lockdown and repeated testing of all on-site individuals. At this time, SARS-CoV-2 was not yet circulating at scale among the general public and COVID-19 vaccination status varied among staff, patients, and caregivers. We estimated vaccine effectiveness (VE), attack rates (ARs), and adjusted risk ratios (aRRs) to identify factors associated with infection among infection-naïve patients, caregivers, and staff using multivariable regression analysis. Among 630 at-risk individuals, there were 131 (21%) polymerase chain reaction (PCR)-confirmed COVID-19 cases including three deaths. The overall VE was 78%. Hospital staff and people who were fully vaccinated had a significantly lower risk of SARS-CoV-2 infection (P < .05). Our analysis shows the importance of being fully vaccinated in reducing the risk of SARS-CoV-2 infection in hospital settings.
Keywords
What We Already Know
COVID-19 outbreaks in health care settings can disrupt care.
The Delta variant is highly transmissible and pathogenic and dominated Vietnam’s 2021 wave.
COVID-19 vaccine effectiveness data in infection-naïve hospital populations, especially from low-middle-income countries, is limited.
What This Article Adds
Provides rare vaccine effectiveness estimates for infection-naïve patients, caregivers, and staff from a large hospital outbreak.
Shows that being fully vaccinated halves the risk of infection compared to being unvaccinated or partially vaccinated in health care settings.
Links lower infection risk for hospital staff to higher vaccination rates and adherence to infection control measures.
Introduction
Coronavirus disease (COVID-19), an infectious disease first reported in December 2019 in China, has quickly transformed into a global pandemic, infecting more than 775 million people and causing more than 7 million deaths as of August 4, 2024. 1 The occurrence of COVID-19 outbreaks within hospitals can severely affect the well-being of health care workers (HCWs) and result in high morbidity and mortality among patients. Consequently, such outbreaks may also lead to a depletion of institutional capacity, including staff and other resources, necessary to deliver comprehensive clinical care. 2 In addition, nosocomial COVID-19 outbreaks have the potential to spread to the general public, as seen in Italy in February 2020, where the nation-wide epidemic was triggered by an outbreak in a hospital, which caused infections to several HCW and other inpatients. 3
Vietnam reported the first two COVID-19 cases in January 2020 and gained considerable success in containing the spread of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) during the initial phase of the pandemic, during which the dominant variants remained less transmissible without significant community transmission until the introduction of the Delta variant in late April 2021.4,5 Between June to December 2021, the highly transmissible Delta variant quickly became dominant, resulting in a rise in infections and fatalities predominantly in the South of Vietnam.6,7 Since the introduction of the COVID-19 vaccination campaign in early March 2021,8,9 nine COVID-19 vaccines have been approved for emergency use in Vietnam: A2D1222 (AstraZeneca); Comirnaty (Pfizer-BioNTech); Spikevax (Moderna); Vero-Cell; Abdala; Covaxin; Hayat-Vax; Janssen; and Sputnik-V. 10 Most previous studies have examined the effectiveness of COVID-19 vaccination in hospital settings in high-income countries, including South Korea, Germany, Singapore, and Israel.11 -14 Not only in Vietnam but also in other low-and-middle-income settings in the region, protective effects from COVID-19 vaccination among infection-naïve population in hospital settings have not yet been assessed.
On 30 September 2021, Viet Duc University Hospital (VDUH) in Hanoi, the largest surgical hospital in Northern Vietnam, reported an outbreak of SARS-CoV-2. 15 At the time, there was still very limited community transmission in the Northern and Central provinces, and overall only 8.5% of the population in Vietnam had received 2 doses of COVID-19 vaccine. 7 We aimed to assess vaccine effectiveness (VE) among patients, caregivers, and hospital staff in this outbreak, and to identify factors associated with SARS-CoV-2 infection in this population.
Methods
Study Design and Data Sources
This was a retrospective analysis using outbreak investigation data from VDUH, the Centers for Disease Control and Prevention (CDC) of the five provinces Hanoi, Hai Duong, Hung Yen, Nam Dinh, and Ha Tinh, between September 27 and October 28, 2021. Vaccination data were extracted from the National COVID-19 vaccination database of Vietnam. Data on co-morbidities were extracted from hospital records where this information was recorded.
Study Setting
Viet Duc University Hospital in Hanoi, the largest surgical referral hospital in Northern Vietnam, handles more than 300 visits, 800 consultations, and 180 operations daily. It has a 1500-bed capacity and 2200 staff. 16 At the time of the outbreak, VDUH was following the Ministry of Health guidelines, which included SARS-CoV-2 screening for all patients and caregivers at admission and discharge.17,18
Case Definition
In this study, COVID-19 cases and close contacts were defined as per recommendations by the Vietnam Ministry of Health dated 30 July 2021. 19 Specifically, a confirmed COVID-19 case was defined as an individual who received a positive test result for SARS-CoV-2 on real-time reverse transcription polymerase chain reaction (RT-PCR) using nasopharyngeal swabs. A person was considered a close contact of a COVID-19 case if they had come into contact within 2 meters or had been in the same enclosed space with COVID-19 case(s) during the infectious period of the COVID-19 case. Specifically, the infectious period of symptomatic COVID-19 cases was defined as starting 3 days prior to their date of symptom onset until the start date of quarantine, while in asymptomatic cases, it was starting 14 days prior to date of sampling used until the start date of quarantine. 19
Study Variables
The COVID-19 RT-PCR test result (positive/negative) of all individuals related to the VDUH outbreak was the outcome variable. The explanatory variables included demographic information (age, gender), classification (hospital staff, patient, caregiver, community member), and participants’ vaccination status. People who had received two COVID-19 vaccine doses, of which the last dose was more than 14 days prior to the start of the VDUH outbreak (September 27, 2021), were classified as “fully vaccinated.” Individuals with only one COVID-19 vaccine dose or two doses but who had the second dose less than 14 days before 27 September 2021, were classified as “partially vaccinated.” People who had not received any vaccine by 27 September 2021 were classified as “unvaccinated.”
For COVID-19 cases, we also obtained data on the date of symptom onset (if symptomatic), date of sampling for RT-PCR testing, and presence of symptoms at the time of testing (Yes/No).
Statistical Analysis
Data were incorporated into Microsoft Excel for cleaning and then imported into STATA 17.0 (StataCorp LLC, USA) for analysis. Descriptive statistics were employed, presenting frequencies and percentages for categorical variables, and medians along with interquartile ranges for continuous variables as they were not normally distributed. Attack rates (ARs) were computed by dividing the number of COVID-19 cases by the at-risk population (all COVID-19 cases and close contacts). 20 In this study, VE was estimated in relation to potential risk factors. To calculate the VE, we used the following formula: VE (%) = (1 − RR) × 100. To calculate the risk ratio (RR), we divided the AR of fully vaccinated individuals by the AR among people unvaccinated or partially vaccinated. 20
Chi-square and Fisher-Exact tests were used during univariable analysis to select potential variables for the multivariable analysis. Variables with a P value of less than .05 in the univariable analysis were incorporated in the multivariate regression analysis. In the multivariable regression analysis, variables with a P < .05 were considered significant factors associated with COVID-19 infection.
Ethical Approval
This study was approved by the Science and Medical Delegated Ethics Review Committee of the Australian National University (Protocol 2022/459) and received exemption by the Vietnam National Institute of Hygiene and Epidemiology as a part of routine activities monitoring the COVID-19 emergency response in Vietnam.
Results
Outbreak Description and Response
On September 30, 2021, a 50-year-old caregiver in the VDUH Oncology Department tested positive for SARS-CoV-2. In response, 4577 individuals present on the hospital premises at that time were placed under immediate lockdown and underwent same-day RT-PCR testing. The lockdown lasted for 18 days, halting elective surgeries, and included repeated universal testing on 1, 3, and 6 October. A dedicated COVID-19 unit was established on 6 October, and VDUH reopened on 18 October. The outbreak resulted in 131 confirmed cases, with the last case detected on October 11, and three deaths on October 13, 18, and 27, 2021 (Supplementary 1).
Virus Sequencing
We analyzed the sequences from 21 of the SARS-CoV-2 samples taken from COVID-19 cases associated with the VDUH cluster (Supplementary 2). The phylogenetic tree showed that all Viet Duc samples were clonal and belonged to the “Vietnamese” sub-lineage AY57 of the Delta variant. The results also indicated that our sequences were in the same Delta sub-lineage that was dominant in Vietnam in 2021 and differed from circulating lineages in other countries of Southeast Asia at that time. In addition, VDUH sequences clustered together, characterized by mutation ORF1a: S4106R. There was an observation that a sample from Ha Nam province, south of Hanoi (#1348 taken on October 7, 2021) was closely related to other samples from the VDUH outbreak, suggesting that the outbreaks in VDUH and Ha Nam province shared a common source, though the exact transmission chain remained unclear. Compared to Delta variants elsewhere, none of the mutations that significantly changed the immune escape of SARS-CoV-2 were found in this outbreak.
Population Characteristics
Table 1 illustrates the characteristics of all individuals related to the VDUH outbreak. Case finding identified 131 PCR-confirmed COVID-19 cases, including 10 hospital staff (8%), 53 patients (40%), 59 caregivers (45%), and 9 others (7%) in the community. Contact tracing identified 499 close contacts, who all tested negative for SARS-CoV-2. Among the total of 630 individuals at risk, the median age was 43 years old (interquartile range [IQR]: 31-58), 52% were women and nearly half of them (44%) were aged 18 to 39 years. None of them reported a history of COVID-19 prior to the outbreak. Of the identified COVID-19 cases, 58% were men and only 8% were fully vaccinated, compared to 34% among close contacts. Caregivers and community members accounted for the highest proportion among COVID-19 cases (52%), followed by patients (40%) and hospital staff (8%). At the time of testing, 77% of these COVID-19 cases were asymptomatic. There were three deaths among the confirmed SARS-CoV-2 patients, all of whom had co-morbidities and were unvaccinated.
Characteristics of All Population at Risk in the VDUH Outbreak.
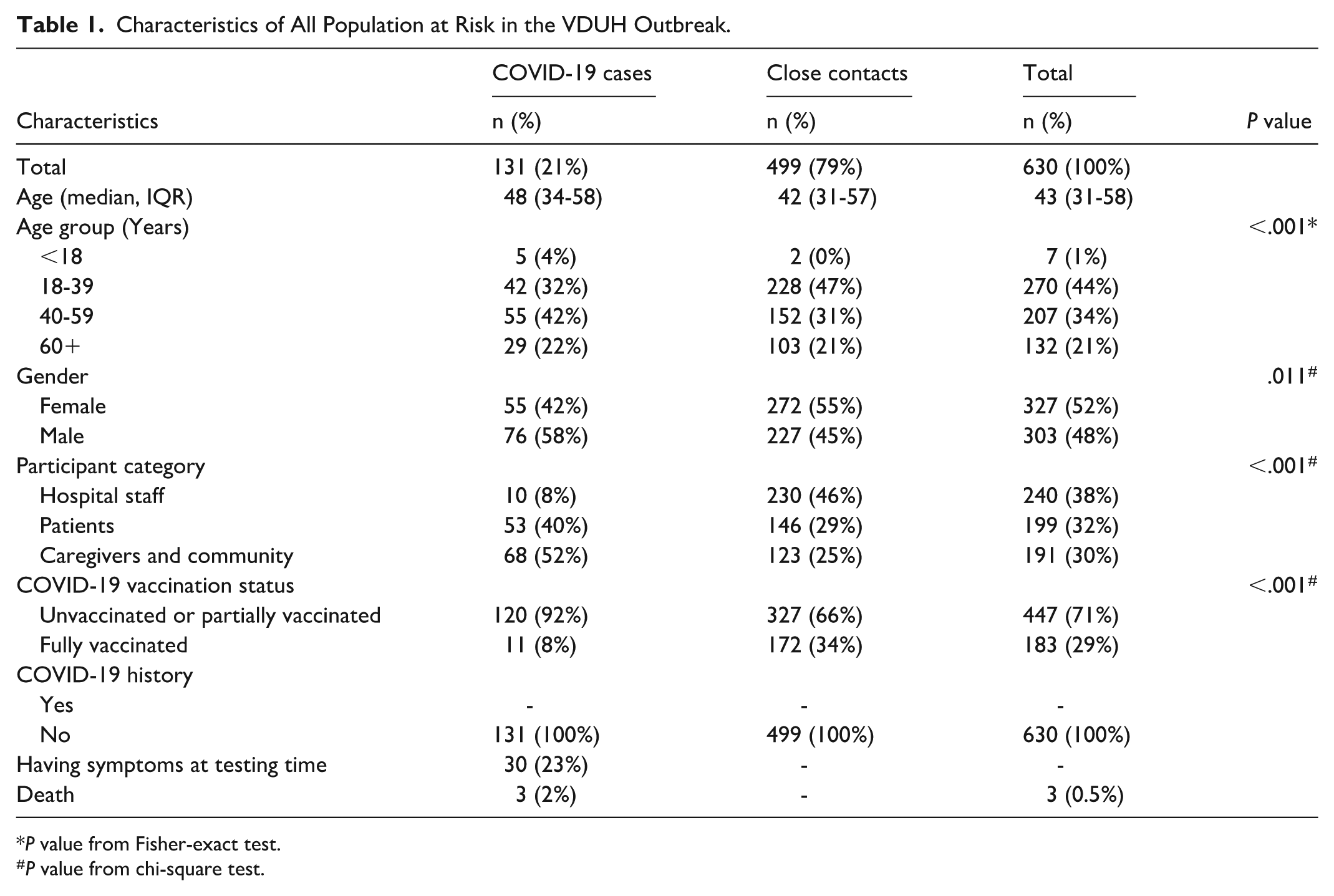
P value from Fisher-exact test.
P value from chi-square test.
Attack Rate
The overall AR was 21% (95% confidence interval [CI]: 18-24). People under 18 years of age had a higher AR than the other groups (71%; 95% CI: 29-96). Men had a higher AR (25%; 95% CI: 20-30) than women (17%; 95% CI: 13-21). The AR among hospital staff (4%; 95% CI: 2-8) was lower than among patients (27%; 95% CI: 21-33) and among caregivers and the community (36%; 95% CI: 29-43). Participants who were fully vaccinated at the start of the outbreak had a substantially lower AR (6%; 95% CI: 3-11) than unvaccinated or partially vaccinated people (27%; 95% CI: 23-31) (Supplementary 3).
Factors Associated With SARS-CoV-2 Infection
Table 2 shows factors associated with the risk of contracting SARS-CoV-2 during the VDUH outbreak. People under 18 years old had a two-fold higher risk of contracting SARS-CoV-2 than those aged 18-39 (aRR 2.17; 95% CI: 1.20-3.91; P < 0.05). Hospital staff had one-fifth the risk of contracting SARS-CoV-2 compared to patients (aRR 0.20; 95% CI: 0.09-0.42; P < 0.001). People who were fully vaccinated had half the risk of SARS-CoV-2 infection compared to people who were unvaccinated or partially vaccinated (aRR 0.51; 95% CI: 0.28-0.95) P < 0.05).
Univariable and Multivariable Analysis of the Effects of Factors on COVID-19 Infection in VDUH Outbreak.
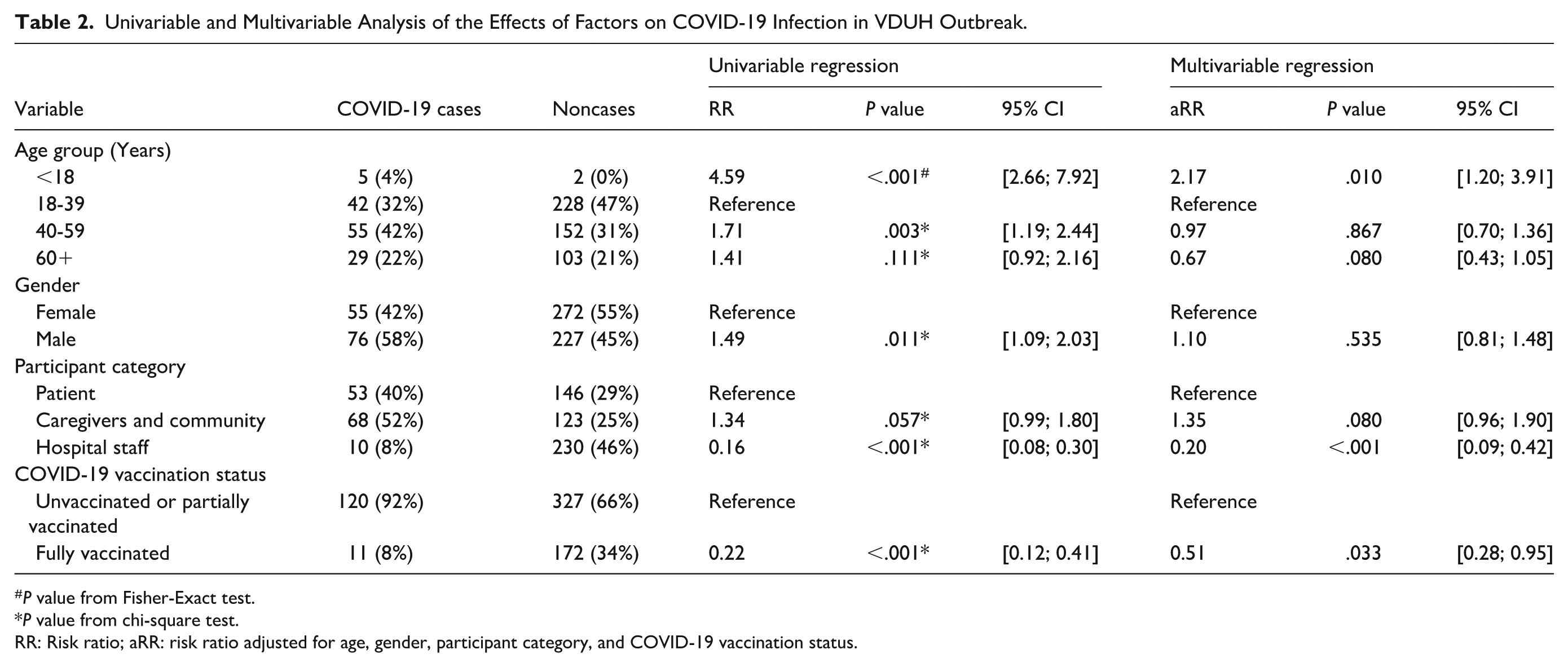
P value from Fisher-Exact test.
P value from chi-square test.
RR: Risk ratio; aRR: risk ratio adjusted for age, gender, participant category, and COVID-19 vaccination status.
Vaccination Coverage
Vaccination coverage varied among different study population groups at the time of the VDUH outbreak. At that time in Vietnam, COVID-19 vaccines were not available for individuals under 18 years old, so that entire group was unvaccinated. Regarding age groups, the highest rate of full vaccination was among people aged 18-39 (49%), followed by 40 to 59 (22%), and 60 years old and above (5%). The proportion of full vaccination among women (37%) was higher than that among men (21%). Hospital staff had the highest fully vaccination coverage (63%) compared to those among patients and caregivers/community, which were 5% and 12%, respectively (Supplementary 4).
Vaccine Effectiveness
Table 3 shows VE by age, gender, and participant category. The overall VE was 78% and was statistically significant (95% CI: 59 to 88; P < 0.05). VE was found to offer statistically significant levels of protection for people aged 18-39 with 82% (95% CI: 59 to 92; P < 0.05), people aged 40-59 with 72% (95%CI: 26 to 89; P < 0.05), women with 79% (95% CI: 52 to 91; P < 0.05), men with 73% (95% CI: 36 to 89; P < 0.05), caregivers and community with 77% (95% CI: 12 to 94; P < 0.05). This metric was found to be statistically non-significant among individuals aged 60 and above (25%; 95% CI: −362 to 88; P = .748), patients (−13%; 95% CI: −201 to 58; P = .805), and hospital staff (12%; 95% CI: −205 to 74; P = .845).
Vaccine Effectiveness (VE) of COVID-19 Vaccines Among Study Participants.
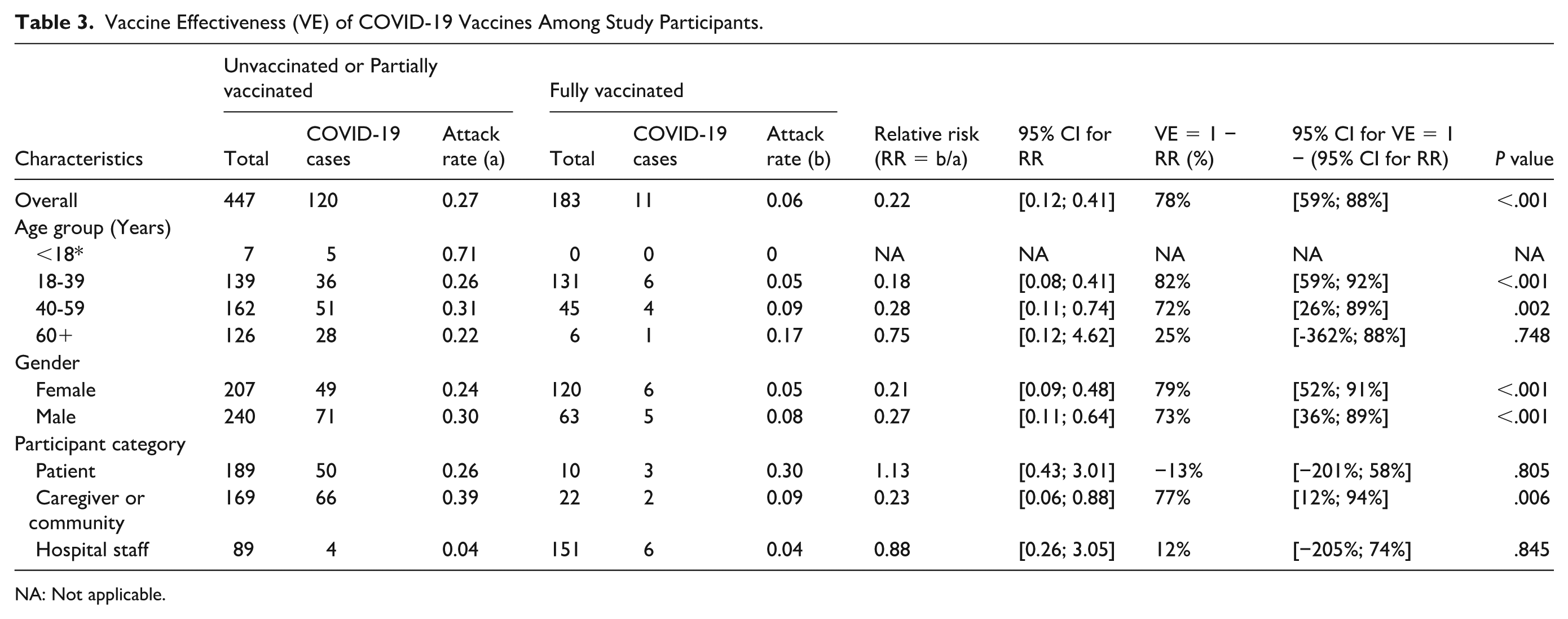
NA: Not applicable.
Discussion
Our analysis highlights the protective effects of full vaccination in reducing the risk of contracting infection with the SARS-CoV-2 Delta variant among infection-naïve populations in a hospital setting, with an observed overall VE of 78%. Hospital staff and people who were fully vaccinated had a significantly lower risk of SARS-COV-2 infections.
The overall AR observed in our study was 21%. This was lower than the AR found in a nosocomial cluster due to the Delta variant in Singapore during a post-vaccination period (30.4%). 11 The AR among hospital staff in our study was 4%, similar to the research by Lim et al 11 in a SARS-CoV-2 Delta variant outbreak in a major acute care hospital in Singapore (3.9%) with 29.4% of participants having at least one dose of COVID-19 vaccination. Also conducted in the post-vaccination era during a Delta variant outbreak, a study from Israel found a higher AR in exposed staff (10.6%). 21 The disparities between studies could be attributed to the nature of pre- or post-vaccination outbreak in each study, different populations, different COVID-19 variants, and infection control measures applied in different countries.
From the calculation of ARs and multivariable analysis, we found that staff working in the hospital had a substantially lower risk of SARS-CoV-2 infection than patients and caregivers. This result is compatible with findings from one study in Israel, where the AR in hospital staff and patients were 10.6 and 23.7%, respectively. 21 This may be explained by hospital staff being more likely to follow infection control measures such as wearing personal protective equipment, keeping a safe distance, and performing hygiene practices regularly, than others. Moreover, HCWs received priority for vaccination against COVID-19 in Vietnam during that time, 9 so their infection risk should be lower than the patients and caregivers, who largely remained unvaccinated and partially vaccinated.
The results from our study showed that individuals under 18 years of age had a significant higher risk of infecting SARS-CoV-2 than those aged 18 to 39 (aRR 2.17; 95% CI: 1.20-3.91; P < .05). This could be due to the fact that prior to the start of this outbreak, COVID-19 vaccines were not available for children and adolescents in Vietnam, so all of them were unvaccinated. Earlier research suggested that the Delta variant was more transmissible compared to Alpha variant and it led to increased risk of SARS-CoV-2 infection among children, especially if unvaccinated.22,23 One study in the United Kingdom during the time of the Delta variant concluded that when compared to people over 65 years old (most of whom had received full COVID-19 vaccination), predominantly unvaccinated children aged five to 12 had a five-fold higher risk of infection. 22 Data from the CDC in the USA showed that since the rise of Delta variant, COVID-19-associated hospitalization witnessed a 10-fold rise in very young children (0-4 years, all of them were not eligible for vaccination), and a 10-fold increase in adolescents (12–17 years) who were not vaccinated compared to those who were. 23 These findings imply that COVID-19 vaccination can help reduce the risk of infection and hospitalization among children and adolescents.
In our study, the sequences of the VDUH outbreak were identified as Pango lineage AY.57 with Delta’s signature mutation ORF1a: P380S. No additional mutations in the Spike protein region were found which may have influenced the infectivity, pathogenicity of SARS-CoV-2 virus or immune escape.24,25 The overall effectiveness of full vaccination among all types of COVID-19 vaccine was 78% (95% CI: 59-88), which is consistent with the result from one large-sample analysis examining effectiveness of two doses of mRNA vaccine (79%; 95% CI: 77-81) against symptomatic infection during the Delta period at urgent care centers in New York city. 26 Another test negative case-control study among 8153 patients in the United States found a higher VE of the two-dose mRNA-1273 against Delta variant infection with 86.7% (95% CI: 84.3-88.7). 27 Different study designs and different settings may be the reasons for the discrepancy between our study and others.
Our multivariable analysis showed that individuals who were fully vaccinated (aRR 0.51; 95% CI: 0.28-0.95) had a significantly lower risk of SARS-CoV-2 infection compared to those unvaccinated or partially vaccinated (P < .05). This finding is in line with existing literature that revealed the protective effects of vaccination in lowering infection risks in clinical settings. According to the research in a hospital in Singapore, having at least one vaccination dosage was associated with 79% lower odds of infection (adjusted odds ratio 0.21, 95% CI: 0.05-0.95, P < .05). 11 Another study in Germany assessed the effect of COVID-19 vaccine on tertiary-care hospital cluster events involving patients and employees. The results from their analysis showed that the number of SARS-CoV-2 clusters decreased with increasing vaccination rates. 13
To our knowledge, this study was the first analysis of COVID-19 VE against SARS-CoV-2 Delta variant in an infection-naïve population in hospital settings in a low- or middle-income setting. The major strength of this analysis was incorporating different data sources to examine infection risks and VE in different population groups present during the SARS-CoV-2 outbreak.
This analysis has several limitations. First, data were collected during an emergency response rather than through a planned study, likely compromising quality and potentially leading to over- or under-estimation of findings. Self-reported COVID-19 history without antibody confirmation likely underestimated asymptomatic infections, affecting reported rates. We also lacked data on hospital staff’s adherence to infection prevention measures, possibly overestimating infection risks. Second, vaccination data from the National COVID-19 vaccination database may not have been fully updated, leading to information bias. Missing or duplicated records could misestimate vaccination status, affecting perceived VE. Third, we lacked information on antibody levels among vaccinated individuals, estimating immune response based on time since the last vaccine dose. This likely underestimated true variability, particularly in sick patients. In addition, we could not specify the types of vaccines administered, limiting effectiveness comparisons. The small number of infected staff also necessitates cautious interpretation, and further studies with larger sample sizes are needed for more precise conclusions.
Conclusion
Our analysis showed high VE against infection and that being fully vaccinated reduced the risk of contracting SARS-CoV-2 Delta variant by half among an infection-naïve population in a hospital setting. We recommend all hospitals in Vietnam promote the completion of vaccination schedules among all persons accessing medical facilities to protect vulnerable populations.
Supplemental Material
sj-docx-1-aph-10.1177_10105395251360136 – Supplemental material for Preparing for Future Pandemics: Vaccine Effectiveness Against SARS-CoV-2 Variant in a High-Risk Hospital Environment—A Case Study From Vietnam
Supplemental material, sj-docx-1-aph-10.1177_10105395251360136 for Preparing for Future Pandemics: Vaccine Effectiveness Against SARS-CoV-2 Variant in a High-Risk Hospital Environment—A Case Study From Vietnam by Hien Thi Nguyen, Luong Huy Duong, Thai Quang Pham, Than Huu Dao, Kieu-Anh Thi Nguyen, Huyen Thi Nguyen, Nghia Duy Ngu, Duong Nhu Tran, Kim-Nhung Thi Le, Tung Son Trinh, H. Rogier van Doorn, Florian Vogt and Khanh Cong Nguyen in Asia Pacific Journal of Public Health
Footnotes
Acknowledgements
The authors acknowledge the National Steering Committee for COVID-19 Prevention and Control, Ministry of Health, National Institute of Hygiene and Epidemiology, Viet Duc University Hospital, Centers for Disease Control of 5 provinces including Hanoi, Hai Duong, Hung Yen, Nam Dinh, and Ha Tinh, and Oxford University Clinical Research Unit for providing them the original data. They appreciate all efforts from VDUH staff and relevant stakeholders in implementing contact tracing, case management, and control measures in this outbreak. This work was conducted as part of the Master of Applied Epidemiology program at the Australian National University in collaboration with National Institute of Hygiene and Epidemiology, Vietnam. HTN was a trainee of the program and received funding from the ASEAN-Australia Health Security Fellowship by the Commonwealth Department of Foreign Affairs and Trade.
Ethical Approval
This analysis was reviewed and approved by the Science and Medical Delegated Ethics Review Committee of the Australian National University (Protocol 2022/459) and was exempted by the Vietnam National Institute of Hygiene and Epidemiology as a part of the efforts in investigating the COVID-19 outbreak and conducting emergency response activities.
Author Contributions
Conceptualization, HTN (Hien Thi Nguyen), KCN and FV; methodology, HTN (Hien Thi Nguyen), FV and KCN; validation, LHD, TQP, NDN, and DNT; formal analysis, HTN (Hien Thi Nguyen), TST, and FV; investigation, LHD, THD, KATN, and KNTL; resources, LHD, TQP, NDN, DNT, and HRD; data curation, HTN (Hien Thi Nguyen) and KCN; writing—original draft preparation, HTN (Hien Thi Nguyen); writing—review and editing, LHD, FV, KCN, TQP, TST, and HRD; visualization, HTN (Hien Thi Nguyen) and TST; supervision, FV and KCN; project administration, H.T.N. (Huyen Thi Nguyen). All authors have read and agreed to the published version of the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article and its supplementary information files.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
