Abstract
The aim of this study was to explore the role of NOX4 in the biology of the normal endometrium and endometrial cancer. NOX4 plays a key role in other adenocarcinomas and has been implicated in the pathogenesis of diabetes and obesity, which are important risk factors for endometrial cancer. NOX4 expression was assessed in 239 endometrial cancer and 25 normal endometrium samples by quantitative real-time polymerase chain reaction, in situ hybridization, and immunohistochemistry. DNA methylation of the NOX4 promoter was determined by means of MethyLight PCR. Data were correlated with clinicopathological parameters and analyzed in the context of diabetes and body mass index. In the normal endometrium, NOX4 microRNA expression was significantly higher in the secretory transformed compared with proliferative endometrium (p = 0.008). In endometrial cancer specimens, NOX4 expression did not differ between diabetic and non-diabetic patients, but was the highest in patients with a body mass index ≤ 26 (p = 0.037). The lowest NOX4 expression was found in carcinosarcomas (p = 0.007). High NOX4 expression predicted poorer clinical outcome with regard to overall survival, especially in non-diabetic patients and those with a body mass index > 20. Independent prognostic significance of NOX4 transcripts was retained in type I endometrial cancer and was the most meaningful in patients with a body mass index > 20. No prognostic impact was shown for NOX4 promoter methylation in endometrial cancer. For the first time, we demonstrate that NOX4 plays a considerable role in the cycle-dependent changes in the normal endometrium and in the biology of endometrial cancer.
Introduction
Endometrial cancer (EC) is the leading cancer of the female genital tract in Europe and North America. 1 Metabolic syndrome, which is closely associated with obesity, type 2 diabetes, and hypertension, is the most relevant risk factor for EC.2–4 Luo et al. 5 showed that intentional weight loss of more than 5% already leads to a significant risk reduction for EC of approximately 40%. Although metabolic conditions are undoubtedly related to EC, the underlying molecular mechanisms governing their exact influence on the pathogenesis of that cancer are poorly understood.
The membrane-associated, multi-unit enzyme family of NADPH oxidase (NOX) includes hydrogen peroxide (H2O2)–producing proteins and is an important source of reactive oxygen species (ROS). NOX4 is regulated by the cytokine transforming growth factor beta 1 (TGF-β1) and hormones like insulin and angiotensin II, as well by hypoxia, hyperoxia, and vascular injury.6,7 ROS modulates various cellular processes such as proliferation, apoptosis, migration, differentiation, survival, and insulin signaling in normal as well as malignant cells.6,8 ROS imbalance promotes tumor growth and progression by inflammation- and hypoxia-mediated oxidative DNA damage and tissue injury.9–11
Dysregulation of NOX4-derived ROS has been implicated in tumor neoangiogenesis via the induction of hypoxia-inducible factor-1α (Hif-1α) expression which in turn enhances vascular endothelial growth factor (VEGF) expression.12,13 Furthermore, elevated ROS production caused by NOX4 was reported to impair the cytotoxic function of tumor-infiltrating lymphocytes. 14 In renal cancer cells, elevated NOX4 expression was found to confer resistance to platinum, vincristine, and etoposide through the modulation of pro- and antiapoptotic signaling, whereas the inhibition of NOX4 increased the sensitivity of these drugs. 15
Meitzler et al. 9 demonstrated high NOX4 expression in a number of malignomas including prostate cancer. Ductal prostate carcinomas share several morphological and endocrinological similarities with EC. 16 Both are steroid hormone dependent and develop from glandular epithelium embedded in a broad steroid-reactive stromal environment. Evolution of both tumors is believed to be strongly influenced by the interplay between the stromal and the cancerous components. The elevated production of TGF-β1 by the normal secretory epithelium, which stimulates prostate cancer cell proliferation by inducing the NOX4–H2O2 cascade, is considered the inducing stimulus for this stromal remodeling. Targeting NOX4 by means of specific inhibitors such as GKT137831 has recently been taken into consideration as a potential treatment strategy for prostate and other cancers. 17
Importantly, there is evidence to show that the use of the antidiabetic drug metformin has a protective effect against cancer in general, but particularly against endometrial and prostate cancers.18,19 A recent in vitro study showed that metformin can decrease cell survival by decreasing ROS production, partly through down-regulation of NOX4 protein expression. 20 Moreover, NOX4 is involved in the inhibition of insulin signaling by inhibiting the phosphorylation of the insulin receptor. 6
To the best of our knowledge, there is as yet no study investigating the relevance of NOX4 in the physiological menstrual cycle–dependent changes in the normal endometrium and in the biology of EC. In this study, we therefore explored NOX4 expression at the messenger RNA (mRNA) level by quantitative real-time polymerase chain reaction (qRT-PCR) and in situ hybridization (ISH) and at the protein level by immunohistochemistry (IHC), and assessed DNA methylation of the NOX4 promoter in the normal endometrium and EC.
Material and methods
Patients and samples
Endometrial tissue specimens from 239 patients with EC (age at diagnosis: 37–93 years; median age: 69 years) obtained at primary surgery and control tissue from 25 patients (age: 32–51 years; median age: 42 years; 13 patients with endometrial tissues in the proliferative phase and 12 patients with secretory transformed endometrial tissues) undergoing hysterectomy for non-malignant reasons were collected and processed at the Department of Obstetrics and Gynecology of the Medical University of Innsbruck, Austria, as described recently. 21 Cases for this study were randomly selected on the basis of the availability of fresh frozen tissues and have not been stratified for known preoperative or pathological prognostic factors. Since the tissues used in this study are from patients diagnosed between 1989 and 2015, not from all patients a written informed consent is available. But in accordance with the Austrian law, the study was reviewed and approved by the Ethics Committee of the Medical University of Innsbruck (reference no. 1233/2017) and conducted in accordance with the Declaration of Helsinki. The median observation period was 5.8 years. Table 1 shows all clinicopathological features.
Association between NOX4 mRNA expression and DNA methylation with clinicopathological features in 239 endometrial cancer patients.

mRNA: messenger RNA; PMR: percentage of methylated reference; IQR: interquartile range; FIGO: International Federation of Gynecology and Obstetrics; BMI: body mass index; n.r.: not reached; PCTH: polychemotherapy.
Classical multifactor risk assessment (as defined in section “Methods”).
Adjuvant polychemotherapy.
Bold values represents significance level of p < 0.05.
RNA extraction and cDNA synthesis
Total cellular RNA isolation and reverse transcription were performed as previously described. 21 In brief, RNA was extracted from the tumor specimens using the TRI reagent (Sigma-Aldrich, Seelze, Germany) according to the manufacturer’s instructions. Reverse transcription of RNA was performed in a final volume of 20 µL containing 1× Reverse Transcription Buffer, 40 U of rRNasin RNase Inhibitor (Promega, Madison, WI, USA), 10 mM dithiothreitol, 200 U of M-MLV Reverse Transcriptase (Life Technologies, Carlsbad, CA, USA), 5 mM random hexamers (Life Technologies), and 400 ng of total RNA. The samples were first incubated at 65°C for 5 min and then quick-chilled on ice. After adding the M-MLV enzyme, the samples were incubated at 25°C for 10 min and at 37°C for 50 min, followed by a period of 15 min at 70°C to inactivate the reverse transcriptase enzyme.
qRT-PCR
Primers and probes for the TATA box-binding protein (TBP; a component of the DNA-binding protein complex transcription factor II D (TFIID) as an endogenous RNA control) were used according to Bieche et al. 22 We used TBP as a housekeeping gene in this EC study, since we observed nearly constant TBP Ct values within the cancer and the control tissues. Primers and probes for NOX4 were purchased from Applied Biosystems (Foster City, CA, USA; Applied Biosystems Assay ID: Hs00418356_m1). qRT-PCR was performed as recently described. 21 We used TBP as a housekeeping gene, since we observed nearly constant TBP Ct values within the 239 cancer tissues and the 25 control tissues. Briefly, polymerase chain reactions (PCRs) were performed using a QuantStudio™ 6 Flex Real-Time PCR System (Applied Biosystems) with a total volume of 20 μL reaction mixture containing 5 μL of each appropriately diluted RT sample (standard curve points and patient samples), 10 μL TaqMan Fast Universal PCR Master Mix (Applied Biosystems), 900 nM of each primer, and 250 nM of the probe. The thermal cycling conditions comprised an initial denaturing step at 95°C for 10 min and 40 cycles at 95°C for 15 s and at 65°C for 1 min. Each experiment included a standard curve with five cDNA concentrations, a control sample (OVCAR3 cell line), 40 patients, and no template control. The standard curves were generated using serially diluted solutions of standard cDNA derived from EC patients. Real-time PCR assays were conducted in duplicate for each sample, and the mean value was used for calculation.
DNA isolation
Genomic DNA from pulverized, quick-frozen specimens was isolated using the DNeasy Tissue Kit (Qiagen, Hilden, Germany).
Bisulfite modification and MethyLight PCR
Bisulfite modification was performed using the EZ DNA Methylation-Gold Kit (Zymo Research, Orange, CA, USA) according to the manufacturer’s instructions. MethyLight analysis was performed and the percentage of methylated reference (PMR) values was calculated, as described previously. A CpG island with a length of 393 bp was identified using the EMBOSS Newcpgreport program (https://www.ebi.ac.uk/Tools/seqstats/emboss_newcpgreport/). Primers and probes in this region were determined with the assistance of the computer program Primer Express, version 2.0.0 (Applied Biosystems), to produce a 105-bp PCR amplicon (located at +30 to +134 relative to the transcription start site of NOX4 transcript variants 1 and 4). Genomic DNA not treated with bisulfite (unmodified) was not amplified with the primers (data not shown). The primer sequences were as follows: NOX4 forward 5′-CGA ATT TTC CGA ACG CCC-3′, NOX4 reverse 5′-GAT TGG TGT AGT TTG GGT CGC-3′, and NOX4 TaqMan probe 5′FAM-AAC TCC CAC CCG CAC CGA ATC AAC T-3′-BHQ1.
NOX4 RNA ISH
To analyze the location of NOX4 mRNA expression, RNA ISH was performed using the RNAscope 2.5 HD Red kit (Advanced Cell Diagnostics, Inc., Newark, CA, USA) and the RNAscope® Probe- Hs-NOX4C2 (Advanced Cell Diagnostics, S.r.l., Milan, Italy) according to the manufacturer’s instruction. In brief, 5 μm formalin-fixed paraffin-embedded (FFPE) primary tumor sections were pretreated with heat (1 h at 60°C) and deparaffinized in xylene, followed by dehydration in ethanol. Slides were incubated for 10 min in hydrogen peroxidase solution, rinsed in distilled water, and boiled in RNAscope target retrieval solution for 15 min. After drying over night at room temperature, the slides were treated with RNAscope Protease Plus for 30 min at 40°C in a hybridization oven. After washing with distilled water, the tumor sections were hybridized with RNAscope Probe- Hs-NOX4C2 for 2 h at 40°C. A horseradish peroxidase–based signal amplification system was then bound to the target probes followed by color development with diaminobenzidine. Positive staining was identified as brown, punctuate dots. A PPIB Positive Control Probe and a dapB Negative Control Probe were used on the tissues to verify a high positive control signal and a negative control background (Advanced Cell Diagnostics, S.r.l.).
NOX4 IHC
IHC was performed using the VECTASTAIN ABC Elite kit (Vector Laboratories, Burlingame, CA, USA) with 3,3′-diaminobenzidine (DAB) as a substrate and anti-NOX4 mAb (47-6) at a dilution of 1:750 after heat-induced epitope retrieval in citrate buffer (Vector Laboratories) as recently described. 9 47-6 mAb was a gift from Pidder Jansen Dürr (Research Institute for Biomedical Aging Research, University of Innsbruck, Innsbruck, Austria).
Statistical analysis
To test for statistical significance between two groups or more than two groups, the non-parametric Mann–Whitney U-test or Kruskal–Wallis test was applied. The time from diagnosis of the primary tumor to histopathological confirmation of recurrence or metastases was defined as progression-free survival (PFS) and the time from diagnosis to death from any cause or to the last clinical inspection was defined as overall survival (OS). Survival analyses were performed by means of univariate Kaplan–Meier analyses and multivariable Cox analyses. Patients were dichotomized into low- and high-mRNA expression groups or low- and high-DNA methylation level groups using the optimal cut-off value calculated with Youden’s index. 23 A p-value less than 0.05 was considered statistically significant. The SPSS statistical software (version 20.0.0; SPSS Inc., Chicago, IL, USA) was used for all statistical analyses.
Results
NOX4 expression on the transcriptome level
In 25 normal endometrial samples, the median value of NOX4 mRNA levels was 0.54 (Q1: 0.44; Q3: 0.81; Table 1). In secretory transformed endometrial tissue, NOX4 mRNA expression was significantly higher (median: 0.81 (Q1: 0.52; Q3: 0.88)) than in samples obtained during the proliferative phase (median: 0.48 (Q1: 0.35; Q3: 0.59); p = 0.004; Figure 1(a)).

NOX4 mRNA expression: (a) control tissues in proliferative (n = 13) and secretory (n = 12) phases in comparison to EC tissues stratified according to tumor grade (G1, n = 43; G2, n = 121; G3, n = 58; G3 carcinosarcoma, n = 15); (b) cancer tissues stratified according to the body mass index (BMI) ≤ 26 (n = 94) and >26 (n = 126).
In 239 EC specimens, NOX4 mRNA expression levels with a median value of 0.68 (Q1: 0.40; Q3: 1.18) were determined (Table 1). In comparison to all normal endometrial tissues, no statistically significant difference in expression was observed in the cancer tissue (Table 1). After a stratification of the control tissues according to the menstrual stage and of the tumor tissues according to the tumor grade, a significantly higher NOX4 mRNA expression was found in high-grade EC tissues in comparison to control tissues in the proliferative phase (grade 2, p = 0.003; grade 3, p = 0.012; Figure 1(a)). In the EC cohort, no associations between clinicopathological characteristics like FIGO (International Federation of Gynecology and Obstetrics) stage, grading, histology, histological type, patient age (median: 68.8 years) at diagnosis, body mass index (BMI) or diabetes, and NOX4 mRNA levels were found. Only a tendency of a higher NOX4 expression in serous papillary EC was observed (Table 1). A subgroup analysis revealed a higher NOX4 mRNA expression in high-grade EC tissues in comparison to carcinosarcomas (grade 2, p = 0.003; grade 3, p = 0.018; Figure 1(a)). We identified an association between NOX4 mRNA expression levels and myometrial invasion (p = 0.048; Table 1). Tumors from patients with a myometrial invasion ≥ 50% of the entire thickness of the myometrium exhibited the highest NOX4 expression (median: 0.77 (Q1: 0.50; Q3: 1.34); Table 1).
In total, 30 (12.5%) study patients suffered from diabetes, 87% of which (26/30) had EC. Nonetheless, no relationship between NOX4 expression in the cancers and diabetes could be identified. Regarding the patients’ BMI and NOX4 expression in cancers, we were able to elucidate that patients with a BMI > 26 had tumors with a significantly lower NOX4 expression (median: 0.65 (Q1: 0.40; Q3: 1.08)) than did patients with a BMI ≤ 26 (median: 0.83 (Q1: 0.50; Q3: 1.40); p = 0.037; Figure 1(b)). In contrast, cancers from patients with a BMI ≤ 20 exhibited a clear although not statistically significant trend to higher NOX4 expression (median: 1.32 (Q1: 0.31; Q3: 1.89)), as depicted in Supplementary Figure S1. In fact, 53.8% of patients with a BMI ≤ 20 were affected by high NOX4-expressing cancers, whereas this was the case in only 22.7% of the patients with a BMI > 20 (p = 0.011). No difference in the histological distribution according to the BMI of the patients was observed.
Cancers were dichotomized in high- and low-NOX4 mRNA–expressing groups using a cut-off value of 1.20 arbitrary units, which corresponds to the 76th percentile of the whole examined cohort as determined with Youden’s index. High NOX4 mRNA expression was associated with poorer OS in the univariate survival analysis (p = 0.021; Table 2 and Figure 2). This proved especially true for non-diabetic patients (p = 0.011) and patients with a BMI > 20 (p = 0.019; Table 2). However, in neither subgroup analyzed were NOX4 mRNA transcripts predictive for disease-free survival (DFS; Table 2).
Univariate survival analysis (analysis in all 239 EC patients).

CI: confidence interval; FIGO: International Federation of Gynecology and Obstetrics; mRNA: messenger RNA; BMI: body mass index; n.r.: not reached.
The optimal cut-off points for NOX4 mRNA expression were calculated with Youden’s index referring to the overall survival data. NOX4 mRNA expression ≤ optimal cut-off (76th percentile) is referred to as low and that greater than the optimal cut-off (76th percentile) as high.
The significance level (p) was determined by log-rank test.
Bold values represents significance level of p < 0.05.

Kaplan–Meier survival analysis: (a) probability of OS in all ECs and (b) probability of OS in type I ECs.
Similar results were revealed in the subgroup of 208 type I EC patients using a cut-off value of 0.83 arbitrary units, which corresponds to the 57th percentile of this subgroup. High NOX4 mRNA expression was associated with poorer OS in the univariate analysis (p = 0.016; Table 3 and Figure 2(b)). Again, this was observed only in non-diabetic patients (p = 0.025) and patients with a BMI > 20 (p = 0.032; Table 3). However, no prognostic impact was observed in type II EC (data not shown). In the Cox regression model, the prognostic relevance of NOX4 mRNA expression with regard to OS was confirmed only for the subgroup of type I EC patients (p = 0.027) and for the cohort of patients with a BMI > 20 (p = 0.032; Tables 4 and 5).
Univariate survival analysis (separate analysis for the subgroup of 208 type I EC patients).
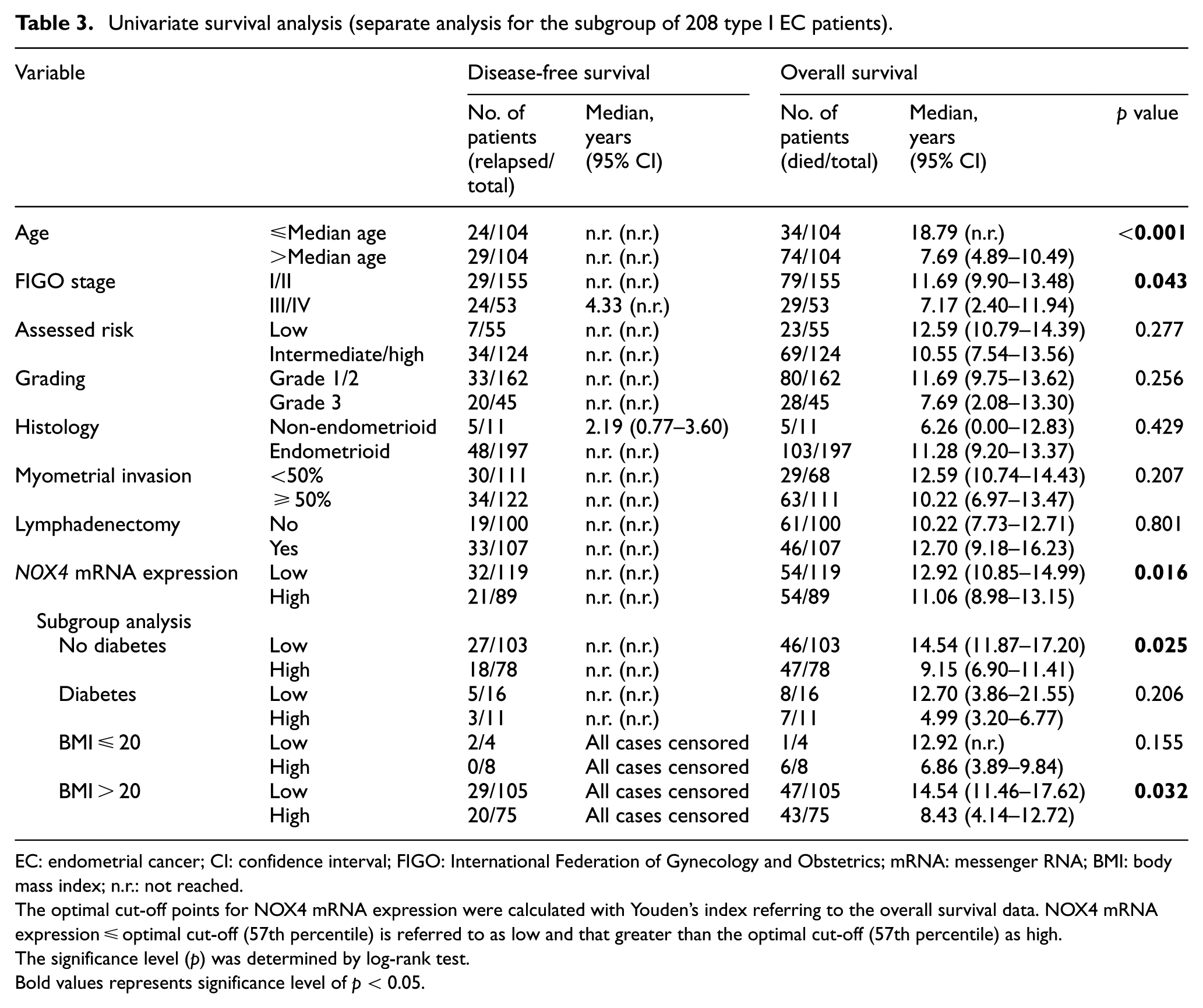
EC: endometrial cancer; CI: confidence interval; FIGO: International Federation of Gynecology and Obstetrics; mRNA: messenger RNA; BMI: body mass index; n.r.: not reached.
The optimal cut-off points for NOX4 mRNA expression were calculated with Youden’s index referring to the overall survival data. NOX4 mRNA expression ≤ optimal cut-off (57th percentile) is referred to as low and that greater than the optimal cut-off (57th percentile) as high.
The significance level (p) was determined by log-rank test.
Bold values represents significance level of p < 0.05.
Multivariate analysis (analysis in all 239 EC patients).

EC: endometrial cancer; RR: relative risk; CI: confidence interval; FIGO: International Federation of Gynecology and Obstetrics; BMI: body mass index.
Bold values represents significance level of p < 0.05.
Multivariate analysis (analysis in the subgroup of 208 type I endometrial cancer patients).

RR: relative risk; CI: confidence interval; FIGO: International Federation of Gynecology and Obstetrics; mRNA: messenger RNA; BMI: body mass index.
Bold values represents significance level of p < 0.05.
NOX4 RNA ISH
In the normal endometrium, we separately examined the RNA expression of NOX4 in the proliferative and the secretory phase and in atrophic endometrium. A very faint NOX4 expression was observed in the stroma of proliferative endometria, whereas the glandular epithelium showed absolutely no NOX4 expression (Figure 3(a)). A strong expression was found in the secretory phase endometrium where it was observed as well in the stroma as in the epithelium (Figure 3(b)). Stromal expression was most prominent in the condensed stroma surrounding the glands (Figure 3(b)). Finally, in atrophic endometrium, only small foci of positivity were seen in the superficial epithelium (Figure 3(c)).

NOX4 in situ hybridization in normal endometrial tissues. (a) Proliferative phase endometrium with very faint stromal NOX4 expression and no expression in proliferative glands. (b) Secretory phase endometrium showed a strong NOX4 expression in both compartments. The condensed stroma surrounding the glands is marked by an arrow, the glandular epithelium by a triangle. (c) Atrophic endometrium. The superficial epithelium is indicated by a triangle. (d) dapB negative control.
In EC tissues, NOX4 RNA was expressed in cancer cells as well as in the surrounding stroma and in the endothelium of small vessels. In ECs, a strong expression of NOX4 was seen especially in the stroma and occasionally a low expression was observed in cancer cells (Figure 4(a)). In serous papillary cancers, NOX4 exhibited a notable focal expression predominantly observed in cancer cells (Figure 4(b)).

NOX4 RNA in situ hybridization and NOX4 immunohistochemistry in EC tissues. (a) In EC, NOX4 RNA was highly expressed in the stromal cells and occasionally found in cancer cells predominantly located at the epithelial–stromal interface. Close-up picture with a fivefold magnification. (b) In serous papillary EC, NOX4 RNA expression was mostly observed in carcinoma cells. Close-up picture with a twofold magnification. (c) Cytoplasmic NOX4 expression in endometrioid cancer cells together with a higher expression in the surrounding stromal cells. Close-up picture with a fivefold magnification. (d) The NOX4 protein expression in serous papillary EC was the strongest in the tumor cells. Close-up picture with a twofold magnification. (e) IHC isotype control for EC. (f) IHC isotype control for serous papillary EC; epithelial cells are indicated by a triangle and stromal cells by an arrow.
NOX4 IHC
IHC confirmed the intra-tissue NOX4 localization obtained by ISH. An even cytoplasmic NOX4 staining was seen in EC. However, the staining was distinctly stronger in the surrounding stromal cells (Figure 4(c)). Figure 4(d) illustrates the predominant NOX4 expression in cancer cells of serous carcinomas. Staining heterogeneity was frequently observed in serous papillary carcinoma. In this tumor type, NOX4 staining was predominant in the cancer cells as compared to the lower immunostaining in the tumor stroma.
NOX4 DNA methylation
Virtually, no methylation was detected in the healthy control endometria compared to cancer samples where sparse methylation of the NOX4 promoter was found in 224/239 (93.7%) (median PMR value: 0.31 (Q1: 0.06; Q3: 0.80); p < 0.001; Table 1).
The highest NOX4 methylation was found in ECs (median PMR value: 0.37 (Q1: 0.09; Q3: 0.79)) as compared with the other histological subtypes (median: 0.11 (Q1: 0.02; Q3: 1.11); p = 0.037). The highest identified degree of NOX4 DNA methylation was furthermore in tumors with no myometrial invasion (p = 0.035; Table 1).
No association between NOX4 promoter methylation and NOX4 mRNA expression was observed.
In neither subgroup analyzed was NOX4 promoter methylation found to be of prognostic relevance.
Discussion
NOX4 has been shown to play a crucial role in the carcinogenesis and progression of a number of malignant tumors where the epithelial–stromal interplay is believed to be of special importance (e.g. pancreas and prostate cancer).9,10,20 Furthermore, NOX4 was implicated in several pathophysiologic steps of insulin resistance and diabetes, which represents one of the most important risk factors, especially for type I EC. 24 Moreover, NOX4 expression was found to be influenced by metformin, which in addition to its antidiabetic properties is also known to protect against a number of cancer entities, including EC. 18 It is also known that NOX4 is crucially implicated in ROS generation in adipocytes, which shows particularly high levels of ROS in obesity. 25 All this motivated us to assess the expression of NOX4 by means of qRT-PCR in the normal endometrium samples, matched according to the menstrual cycle phase, and in 239 ECs. Furthermore, we wanted to elucidate the intra-tissue distribution of NOX4 at the mRNA as well as the protein level in normal and malignant specimens by means of ISH and to explore the degree of DNA methylation of the NOX4 promoter.
One of the most intriguing results of our work was that NOX4 is predominately expressed in the secretory transformed normal endometrium. As marked progesterone receptivity characterizes the endometrium during the secretory phase, we are tempted to speculate that the expression of NOX4 could be controlled by this steroid hormone. 26 In the glandular epithelium of the normal endometrium, progesterone has in fact been shown to enhance the expression and secretion of TGF-β1 that is recognized as a key stimulator of NOX4 expression. 27
This may stand in contrast to our findings that in type I EC high NOX4 expression is independently linked to adverse clinical outcome in terms of OS, considering that the progestin effects on the normal endometrium are regarded to induce cell differentiation and to protect against EC. In addition, qRT-PCR and ISH showed that NOX4 expression was particularly strong in serous ECs, a subtype that generally does not exhibit significant progesterone receptivity. All this indicates a completely different regulation of NOX4 expression in malignant endometrial tissues in comparison to normal endometrial tissues. This is corroborated by the findings that high TGF-β1 expression in EC promotes invasion and metastasis and deteriorates survival. 28
Furthermore, in malignant tumors, NOX4 is highly implicated in neoangiogenesis via ROS-mediated VEGF upregulation 29 and therefore indirectly confers decisive advantages for growth and progression to cancers. Probably, this pro-angiogenic effect may be one important cause of the poor survival of patients with EC showing high NOX4 expression. In this context, there are two rationales for the determination of NOX4 transcripts in EC tissue: First, NOX4 could be a novel direct therapeutic target for abrogating neoangiogenesis in EC and other malignant tumors. With this in mind, it has been shown that the dual inhibitor of both NOX1 and NOX4 (GKT137831) was able to attenuate hypoxia-induced pulmonary vascular cell proliferation. 30 Second, NOX4 tumor expression could be a useful biomarker for reliably predicting the response to antiangiogenic drugs such as bevacizumab.
We also analyzed NOX4 mRNA expression in cancer tissues according to patient BMI and diabetic metabolic state. We were unable to reveal a difference in NOX4 expression in cancers between diabetic and non-diabetic patients. This is in disagreement with the in vitro data showing that high glucose levels in healthy proximal tubule cells of the kidney increase NOX4 expression via increased TGF-β1. 31 However, in this work, a certain inverse relationship between NOX4 mRNA expression and BMI was identified. At present, we are unable to explain the significantly higher NOX4 expression in the EC of patients with BMI values ≤ 26.
We are aware that the main findings of this work are based on pre-evaluated cut-off values obtained in a training set of patients and in fact require validation in a larger test cohort. Another limitation of our study is the fact that qRT-PCR is not an ideal method for the analyses in a tissue type which is so heterogeneous like EC. Therefore, we combined the quantitative NOX4 mRNA expression analysis with RNA ISH and IHC.
Conclusion
We conclude by stating that NOX4 may play a considerable role in secretory transformation of the normal endometrium. In type I ECs, high NOX4 expression is independently related to poorer OS and appears to be influenced by BMI, but not by the diabetic metabolic status of these patients.
Supplemental Material
Supplementary_Figure_S1 – Supplemental material for NADPH oxidase 4 expression in the normal endometrium and in endometrial cancer
Supplemental material, Supplementary_Figure_S1 for NADPH oxidase 4 expression in the normal endometrium and in endometrial cancer by Christine Degasper, Andrea Brunner, Natalie Sampson, Irina Tsibulak, Verena Wieser, Hannah Welponer, Christian Marth, Heidi Fiegl and Alain Gustave Zeimet in Tumor Biology
Footnotes
Acknowledgements
We thank Inge Gaugg, Martina Fleischer, Brigitte Greiderer-Kleinlercher, and Annemarie Wiedemair for excellent technical assistance and Georg Goebel from the Department of Medical Statistics, Informatics and Health Economics, Medical University of Innsbruck for his statistical support. H.F. and A.G.Z. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study was reviewed and approved by the Ethics Committee of the Medical University of Innsbruck (reference no. 1233/2017) and conducted in accordance with the Declaration of Helsinki.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the Verein zur Krebsforschung in der Frauenheilkunde, an association which is exclusively financed by donation funds for cancer research in female malignancies.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
