Abstract
Objectives:
Never-smokers may be a distinct subgroup among patients with advanced non-small cell lung cancer, appearing to benefit less from immunotherapy than smokers. We report results from never-smokers enrolled in the Italian cohort of the nivolumab expanded access program in pre-treated patients with advanced squamous non-small cell lung cancer.
Materials and methods:
Nivolumab (3 mg/kg every 2 weeks for ≤24 months) was available on physician request. Efficacy data included objective tumor response, date of progression, and survival information. Safety was monitored.
Results:
Overall, 371 patients received at least one dose of nivolumab, including 31 never-smokers (8%). Objective response rate, disease-control rate, and median overall survival were 23%, 45%, and 12.1 months (95% confidence interval: 3.7–20.4), respectively, in never-smokers, and 18%, 47%, and 7.9 months (95% confidence interval: 6.2–9.6), respectively, in the overall expanded access program population. Any-grade and grade 3–4 treatment-related adverse events were reported in 12 (39%) and 3 (10%) never-smokers, respectively, and in 109 (29%) and 21 (6%) patients, respectively, in the overall expanded access program population. Grade 3–4 treatment-related adverse events in non-smokers were increased transaminases (n = 2; 6%) and diarrhea (n = 1; 3%). Treatment-related adverse events led to treatment discontinuation in 4 non-smokers (17%) and in 26 patients (9%) overall.
Conclusion:
Pre-treated never-smokers with advanced squamous non-small cell lung cancer in this Italian expanded access program demonstrated efficacy and safety that were consistent with those in the overall expanded access program population and clinical trials. These results suggest that a proportion of never-smoker patients with squamous non-small cell lung cancer may be responsive to immunotherapy. Other factors, such as the tumor mutational load and the status of programmed death-ligand 1, anaplastic lymphoma kinase, and epidermal growth factor receptor, might play a potential key role.
Introduction
Non-small cell lung cancer (NSCLC) accounts for 85% of newly diagnosed cases of lung cancers, with squamous NSCLC comprising 20%–30% of these.1,2 For many years, docetaxel has been the standard of care for advanced squamous NSCLC following failure of first-line systemic therapy, despite providing limited clinical benefit. 2 Because squamous NSCLC is less likely than non-squamous NSCLC to be driven by specific gene mutations, molecularly targeted agents, such as inhibitors of epidermal growth factor receptor (EGFR) or anaplastic lymphoma kinase (ALK) that have improved survival in non-squamous NSCLC, are ineffective or rarely indicated in squamous NSCLC.3,4
Results of the randomized phase III CheckMate 017 study demonstrated that nivolumab, a fully human programmed death 1 (PD-1) immune checkpoint inhibitor antibody, provided long-term survival benefit in patients with previously treated advanced squamous NSCLC. 5 This trial, conducted in patients after failure of first-line platinum-based chemotherapy, found a 41% reduction in the risk of death with nivolumab compared with docetaxel (hazard ratio (HR) 0.59; 95% confidence interval (CI): 0.44–0.79; p < 0.001); median overall survival (OS) was 9.2 months with nivolumab compared with 6.0 months with docetaxel. Similar benefit was also reported in patients with previously treated advanced non-squamous NSCLC who were treated with nivolumab in the randomized phase III CheckMate 057 study. 6 These results led to the approval of nivolumab in the United States and the European Union for locally advanced or metastatic squamous and non-squamous NSCLC with disease progression during or after prior chemotherapy.7,8
Nearly a quarter of all cases of lung cancer (mostly NSCLC) occur in never-smokers, and these have been proposed as a distinct disease entity based on clinical characteristics and possible etiologic factors. Lung cancer in never-smokers is more common in females, is often driven by specific actionable gene mutations (e.g.
An international nivolumab expanded access program (EAP) in advanced squamous NSCLC allowed patients who were unable to take part in local nivolumab clinical trials to gain access to treatment before it became commercially available. EAPs have relatively broad entry criteria and thereby mimic routine clinical practice more closely than does the controlled clinical trial setting. An analysis of preliminary data from the entire Italian cohort of patients with squamous NSCLC enrolled in the nivolumab NSCLC EAP has been reported elsewhere. 20 Here, we present preliminary efficacy and safety data from the subgroup of never-smokers in this Italian cohort.
Materials and methods
Patients
Patients aged 18 years or older with stage IIIB or IV squamous NSCLC that had been histologically or cytologically confirmed at the participating institution were eligible for enrollment. All patients had disease progression or recurrence during or after one or more prior systemic treatments for advanced or metastatic disease. Patients who developed recurrent disease within 6 months of completing platinum-based adjuvant, neoadjuvant, or definitive chemoradiation therapy for locally advanced disease were also eligible. Patients with treated central nervous system metastases that had been stable for at least 2 weeks were eligible, provided that they did not require corticosteroids or were on a stable or decreasing dose of no more than 10 mg prednisone daily (or an equivalent). In addition, patients were required to have an Eastern Cooperative Oncology Group (ECOG) performance status of 2 or less; have completed prior treatment with chemotherapy, tyrosine kinase inhibitors, tumor vaccine, cytokines, or palliative radiotherapy at least 2 weeks before beginning nivolumab therapy, with resolution of all adverse events to baseline or stabilization; have adequate hematologic, hepatic, and renal function; and have life expectancy of at least 6 weeks.
Patients were excluded if they had active, known, or suspected autoimmune disease, with the exception of type 1 diabetes mellitus, residual hypothyroidism due to an autoimmune condition requiring hormone replacement therapy, or psoriasis not requiring systemic treatment. Patients with carcinomatous meningitis, prior therapy with any drug specifically targeting T-cell co-stimulation or checkpoint pathways, symptomatic interstitial lung disease, HIV infection, or prior treatment in any nivolumab clinical trial were also excluded.
The assessment of
Study design and treatment
Nivolumab was made available on physician request through the EAP. The EAP guidelines were approved at each participating center, and participating physicians had to comply with generally accepted good clinical practice and ethical standards. All patients provided written, informed consent to their participation in the program.
Nivolumab 3 mg/kg was administered intravenously every 2 weeks for up to 24 months. Treatment could be discontinued due to disease progression, unacceptable toxicity, or withdrawal of consent. Although no reductions in nivolumab dosing were allowed, the treatment could be delayed in the event of toxicity. Nivolumab treatment could be continued beyond progression if, in the opinion of the investigator, the patient was deriving clinical benefit in the absence of rapid disease progression, the study drug was tolerated, performance status was stable, and treatment would not delay any intervention to prevent serious complications of disease progression.
Assessments
The EAP had no prespecified endpoints, but investigators were encouraged to make every effort to document the objective disease progression. Efficacy data collected included investigator-assessed objective tumor response, date of progression, and survival information.
Safety was monitored by continuous assessment of adverse events, physical examination, ECOG performance status, hematology and clinical chemistry tests, and thyroid function tests according to local regulations and standards of care. Adverse events were graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events version 4.0, with the causal relationship to treatment determined by the investigators.
Statistical analysis
All patients who received at least one dose of nivolumab were included in both the efficacy and safety analyses. Never-smokers were defined on the basis of patient-reported information. Objective response rate (ORR), disease control rate (DCR; defined as the combined rates of complete response, partial response, and stable disease), progression-free survival (PFS), and OS were evaluated. PFS and OS were estimated using the Kaplan–Meier method, and 95% CIs were derived using the Greenwood formula of asymptotic variance. PFS was calculated as the time between the start of nivolumab treatment and any evidence of progressive disease, or death, whichever occurred first.
Results
Patients and treatment
Between April and September 2015, 371 patients were enrolled in the EAP at 96 centers in Italy and received at least one dose of nivolumab. Among the 31 patients (8%) who were never-smokers, 3 had tumors with adenosquamous histology. In the never-smoker group and overall EAP cohort, 39% and 80% were male, respectively; the median age (range) was 66 (31–82) and 68 (31–91) years; 61% and 64% had ECOG performance status 1–2; and 68% and 56% received at least two prior therapies (Table 1).
Baseline patient characteristics.
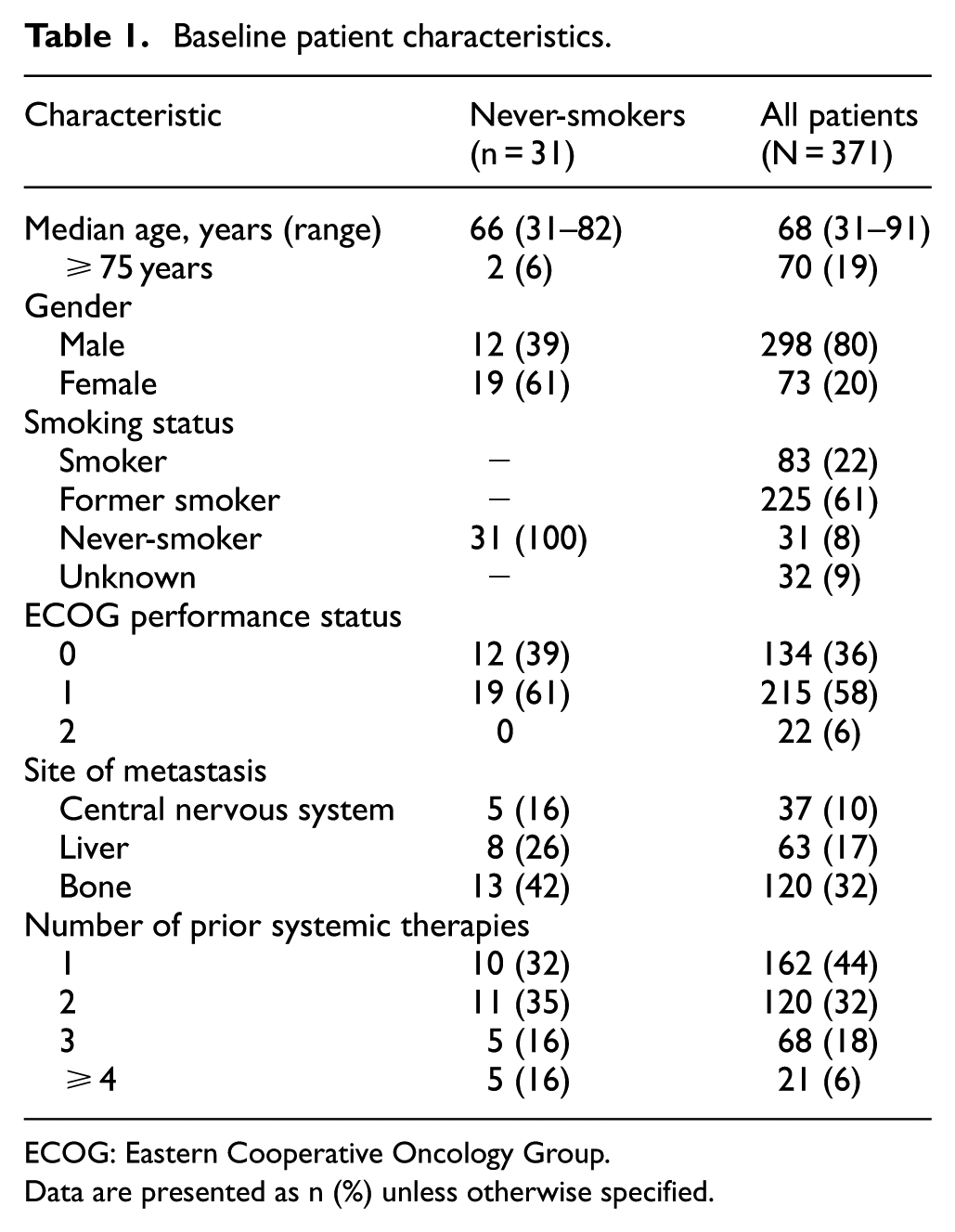
ECOG: Eastern Cooperative Oncology Group.
Data are presented as n (%) unless otherwise specified.
Overall, patients received a median of six doses of nivolumab (range, 1–22), with a median follow-up of 7.1 months (range: 0.1–16.4). The median number of nivolumab doses among never-smokers was 6 (range, 1–21), and the median follow-up was 7.0 months (range, 1.1–13.6). In total, 24 never-smokers (77%) had discontinued treatment at the time of analysis mostly because of progressive disease (n = 13; 54%) or death (n = 6; 25%; Table 2).
Summary of treatment discontinuations.

Data are presented as n (%).
Efficacy
Among never-smokers, the ORR was 23%, which included seven (23%) partial responses. An additional seven patients (23%) had stable disease, and the DCR for the never-smokers group was 45% (Table 3). In the overall EAP population, the ORR was 18%, including 4 complete responses (1%) and 63 partial responses (17%), an additional 108 patients (29%) had stable disease, giving a DCR of 47% (Table 3). Among 14 never-smokers with available
Response to treatment.
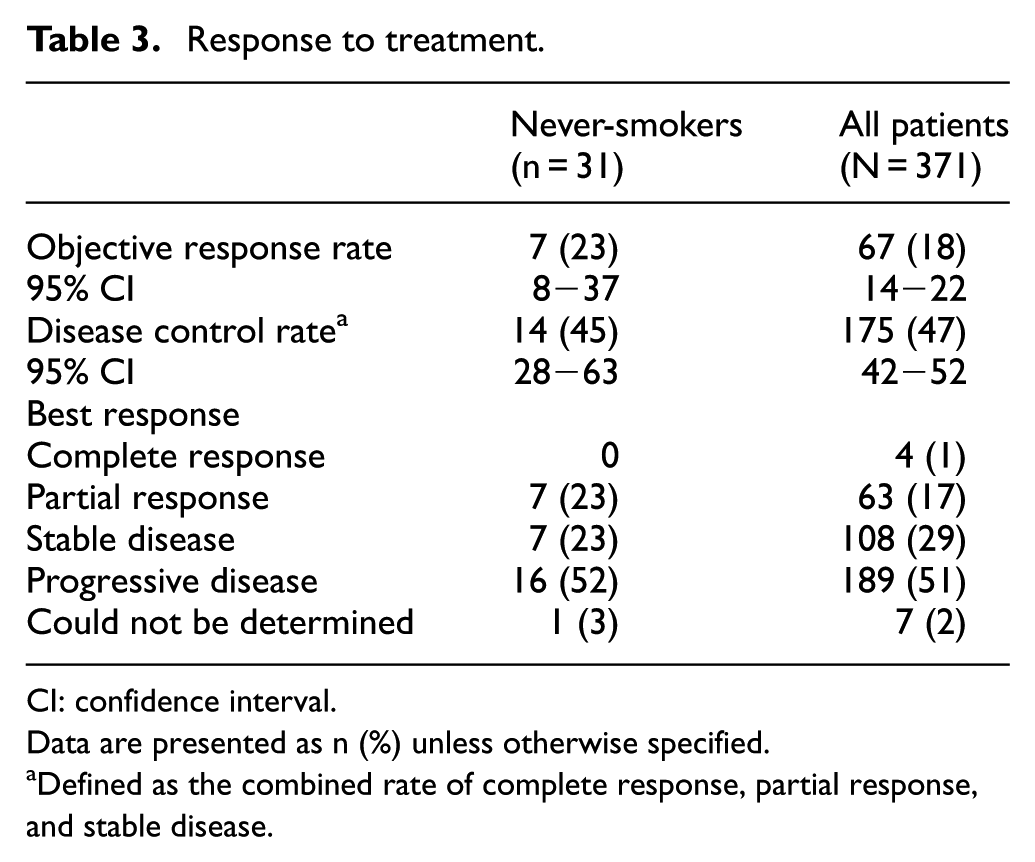
CI: confidence interval.
Data are presented as n (%) unless otherwise specified.
Defined as the combined rate of complete response, partial response, and stable disease.
The three patients with
No data were available on the PD-L1 status.
For never-smokers and the overall EAP population, the 1-year OS rates were 51% and 39%, respectively, while the median OS was 12.1 months (95% CI: 3.7–20.4) and 7.9 months (95% CI: 6.2–9.6; Figure 1(a)). The 1-year PFS rates were 30% and 27% for never-smokers and the overall EAP population, respectively, while the median PFS was 3.5 months (95% CI: 0.0–7.1) and 4.2 months (95% CI: 3.4–5.0; Figure 1(b)), respectively.

Kaplan–Meier estimates of overall survival (a) and progression-free survival (b).
Safety
Any-grade and grade 3–4 treatment-related adverse events occurred in 12 (39%) and 3 (10%) never-smokers, respectively, compared with 109 (29%) and 21 patients (6%) in the overall EAP population (Table 4). Among never-smokers, the most common (≥5%) treatment-related adverse events of any grade were fatigue/asthenia (13%; n = 4), rash (13%; n = 4), pain (10%; n = 3), anemia (6%; n = 2), diarrhea (6%; n = 2), elevated transaminases (6%), and pyrexia (6%; n = 2). Grade 3–4 treatment-related adverse events in never-smokers comprised increased transaminases (n = 2; 6%) and diarrhea (n = 1; 3%). No treatment-related deaths were reported.
Treatment-related adverse events occurring in ≥1% of patients.

Data are presented as n (%).
Of 24 never-smokers who discontinued nivolumab treatment, 4 (17%) did so as a result of adverse events, compared with 26 of 281 patients (9%) in the overall EAP population (Table 2). Discontinuations due to treatment-related adverse events occurred in 8% (2/24) of never-smokers and 5% (14/281) of the overall EAP population. Treatment-related adverse events leading to discontinuation among never-smokers were hepatic toxicity and bilateral exophthalmos (n = 1, each), while those among the overall EAP population were pneumonitis (n = 3); diarrhea, hepatotoxicity, and skin toxicity (n = 2, each); and bilateral exophthalmos, dyspnea, headache, paralytic ileus, and psoriasis (n = 1, each). Treatment-related adverse events of immune etiology (“select adverse events”) were managed using protocol-defined toxicity management algorithms.
Discussion
Never-smokers with advanced squamous NSCLC in the Italian cohort of this EAP demonstrated benefits with nivolumab therapy comparable to those in the overall EAP population and largely mirrored the efficacy observed in the nivolumab arm of the phase III CheckMate 017 trial in patients with advanced squamous NSCLC (median OS, 9.2 months; ORR, 20%; DCR, 49%). 5 Contrasting with results from controlled studies showing that never-smokers with advanced NSCLC as a whole seem to benefit less from immunotherapy than smokers,6,13,15 our data suggest that a proportion of never-smokers with squamous NSCLC may be responsive to nivolumab. However, the patient populations of the nivolumab studies in which response or survival differed according to smoking history comprised exclusively non-squamous NSCLC, as in the phase III CheckMate 057 trial, or had greater numbers of patients with non-squamous than squamous histology, as well as relatively small numbers of never-smokers.6,12,13,15,20–22
In this study, no data were available on the PD-L1 status because at the time in which it was conducted, the assessment of PD-L1 was not a standard practice in Italy and it was not an inclusion criterion of the EAP.
Interestingly all four never-smokers who tested positive for recognized NSCLC driver mutations (three with
Safety was comparable between never-smokers and the overall EAP population and was consistent with results previously reported for the nivolumab arm in CheckMate 017. 5 Most adverse events were mild to moderate, and the proportion of EAP never-smokers who had grade 3–4 treatment-related adverse events (10%) was comparable to that found in the overall EAP population (6%), as well as in the nivolumab arm of CheckMate 017 (7%). The proportion of EAP never-smokers who discontinued therapy due to treatment-related adverse events (8%) was also similar to that in the overall EAP population (5%) and the nivolumab arm of CheckMate 017 (3%).
This report presents the most extensive real-world clinical experience with nivolumab in never-smokers with advanced squamous NSCLC in current practice to date, and the proportion of never-smokers in the Italian EAP cohort of previously treated patients with advanced squamous NSCLC was comparable to that reported in the nivolumab arm of CheckMate 017 (8% vs 7%).
5
In summary, nivolumab provided clinical activity coupled with a manageable safety profile in never-smokers. While the study was limited by small patient numbers and its observational design, the efficacy and safety profiles of nivolumab among never-smokers without driver mutations in a real-world setting were consistent with those obtained in the broader population of patients (including former and current smokers) with squamous NSCLC, including those in controlled clinical trials.
5
Among patients with advanced NSCLC, never-smokers are of particular interest, because as a group, they appear to derive less benefit from immunotherapy versus smokers, who have a higher mutational load.6,13,15 Although further study is needed to clarify the association between nivolumab response and smoking history, these results suggest that never-smokers with squamous NSCLC, especially those without driver mutations, may be a distinct patient subgroup. Other factors such as the assessment of the tumor mutational load and the status of PD-L1,
Supplemental Material
Supplemental_Information – Supplemental material for Nivolumab in never-smokers with advanced squamous non-small cell lung cancer: Results from the Italian cohort of an expanded access program
Supplemental material, Supplemental_Information for Nivolumab in never-smokers with advanced squamous non-small cell lung cancer: Results from the Italian cohort of an expanded access program by Marina Chiara Garassino, Lucio Crinò, Annamaria Catino, Andrea Ardizzoni, Enrico Cortesi, Federico Cappuzzo, Paola Bordi, Luana Calabrò, Fausto Barbieri, Antonio Santo, Giuseppe Altavilla, Francesca Ambrosio, Enrico Mini, Enrico Vasile, Floriana Morgillo, Alessandro Scoppola, Carmelo Bengala, Alessandro Follador, Natale Tedde, Diana Giannarelli, Giuseppe Lo Russo and Fabiana Vitiello in Tumor Biology
Footnotes
Acknowledgements
We thank all of the patients and their families, as well as the investigators who participated in the Italian cohort of the nivolumab EAP for NSCLC (![]() ). All drafts of this manuscript were prepared with medical writing and editorial assistance, provided by Wendy Sacks, PhD, and Emilia Raszkiewicz of StemScientific, an Ashfield Company, funded by Bristol-Myers Squibb.
). All drafts of this manuscript were prepared with medical writing and editorial assistance, provided by Wendy Sacks, PhD, and Emilia Raszkiewicz of StemScientific, an Ashfield Company, funded by Bristol-Myers Squibb.
Contributorship
M.C.G. and G.L. R. for study concept and design. D.G. for statistical analysis. All authors for acquisition of data, analysis and interpretation of data, drafting of the manuscript, critical revision of the manuscript for important intellectual content, study supervision. All authors contributed to the decision to submit this manuscript for publication.
Declaration of conflicting interests
We wish to confirm that there are no significant conflicts of interest associated with this publication that could have influenced its outcome. M.C.G. declares consultancies from Astra Zeneca, Roche, Boehringer Ingelheim, BMS, MSD, Eli Lilly, Novartis, Bayer, Pfizer, Sanofy, Italfarmaco. L.C. received fees for speakers’ bureau participation from AstraZeneca, Bristol-Myers Squibb, and Novartis. A.A. received honoraria from Boehringer Ingelheim, Bristol-Myers Squibb, and Lilly and served in a consulting/advisory role for Boehringer Ingelheim, Bristol-Myers Squibb, GlaxoSmithKline, Lilly, and MSD Oncology. F.C. has received honoraria from BMS, Roche, Pfizer, Takeda, and AstraZeneca. E.V. declares consultancies from Eli Lilly, Boehringer Ingelheim, Astra Zeneca, Celgene, Italfarmaco. All the other authors have no conflict of interest to declare (financial, professional, or personal).
Ethical approval
We confirm that any aspect of the work covered in this manuscript that has involved human patients has been conducted with the ethical approval of all relevant bodies and that such approvals are acknowledged within the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Bristol-Myers Squibb.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
