Abstract
Ovarian cancer remains the most lethal gynecologic malignancy. This is due to lack of effective screening, diagnosis predominance in late stage of disease, a high recurrence rate after primary therapy, and poor treatment response in platinum-resistant tumor. Thus, unique biomarkers, predictive of individual disease course, and prognosis are urgently needed. The aim of our study was to assess the clinicopathological significance of plasma, peritoneal fluid, and tumor tissue levels of mesothelin in epithelial ovarian cancer patients. Plasma and peritoneal fluid levels of mesothelin were measured by enzyme-linked immunosorbent assay. Tissue expression of MSLN was evaluated using quantitative real-time polymerase chain reaction. Preoperative plasma mesothelin levels were significantly higher in epithelial ovarian cancer patients in comparison to the patients with benign tumor and controls. There have been noticed significant differences in the plasma mesothelin levels based on International Federation of Gynecology and Obstetrics stage, grade, and histology type. No significant changes were observed between Kurman and Shih type I versus type II epithelial ovarian cancer. Interestingly, peritoneal fluid mesothelin levels revealed significant differences based on both grade and Kurman and Shih–type epithelial ovarian cancer. There were no relevant changes in the mesothelin level in peritoneal fluid between different stages and histology types compared to benign tumor. MSLN expression level in tumor tissue was significantly higher based on stage, grade, and Kurman and Shih–type epithelial ovarian cancer than in the benign masses. In addition, data showed significant higher MSLN expression in endometrioid tumors compared to benign masses and serous tumors. Plasma, peritoneal fluid, and tumor tissue levels of mesothelin positively correlated with level of CA125. Low mesothelin concentrations in plasma were also associated with prolonged patient survival. More importantly, we revealed that plasma mesothelin level was correlated with both peritoneal fluid mesothelin level and tumor MSLN expression. This study highlights that plasma mesothelin level may be a useful noninvasive biomarker surrogate for local tumor mesothelin status in monitoring of epithelial ovarian cancer patients.
Introduction
Epithelial ovarian cancer (EOC) is the leading cause of death from gynecologic cancer among women in developed countries. 1 Thus, ovarian cancer is commonly known as a “silent killer” due to the lack of specific symptoms until advanced International Federation of Gynecology and Obstetrics (FIGO) stages (III/IV) of disease when 75% of the cases are diagnosed and 5-year survival rates are less than 30%.2,3 Despite the modern management, introduction of improved surgical techniques, and combination chemotherapy and targeted therapies, the overall survival (OS) rates for these patients have not been significantly improved. 4 The main prognostic parameters for ovarian cancer are the tumor stage, the histological type, the degree of malignancy, and the residual tumor after debulking surgery. However, these factors present an incomplete picture of the complex tumor biology of EOC. Multiple efforts have been made to improve survival rates through early screening methods based on serum cancer antigen 125 (CA125) concentrations and transvaginal ultrasound. Although the CA125 and transvaginal ultrasound are currently the two main techniques used to diagnose ovarian cancer, they lack both sensitivity and specificity for early detection of disease. Moreover, some histological types of EOC, such as mucinous cystadenocarcinoma, do not express CA125. In addition, CA125 would increase in some benign conditions, such as uterine myoma, endometriosis, and pelvic inflammatory disease. 5 Thus, the identification of new biomarkers, predictive of individual disease course and prognosis, would be extremely useful.
Mesothelin (MSLN) was described for the first time by I Pastan and M Willingham at the National Cancer Institute (NCI) over 20 years ago.6,7 It is a glycophosphatidylinositol (GPI)-linked cell surface protein physiologically expressed in mesothelial cells that line the pleura, peritoneum, and pericardium. The MSLN gene encodes a precursor protein of 71 kDa that is processed to a 31 kDa shed protein named megakaryocyte potentiating factor (MPF) and a 40 kDa membrane-bound protein, MSLN. 8 MSLN gene has two minor spliced forms of the major MSLN transcript (variant 1; NM_005823) that encode two alternative proteins termed variant 2 (NM_013404) and 3 (AF18095). The MSLN variant 1 is the predominant mature messenger RNA (mRNA) species expressed by both normal and tumor cells.9,10 A soluble form of MSLN (soluble MSLN-related protein (SMRP)) has also been revealed in the sera of patients with solid tumors. 6
The physiological function of MSLN is poorly known; however, no detectable abnormalities were demonstrated in reproduction, tissue development, and blood cell count in a MSLN knock-out murine model. 11 MSLN was reported to be highly expressed in mesothelioma (∼100%) and several types of malignant tumors, including pancreatic (80%–85%), lung (60%–70%), ovarian (60%–65%), gastric (50%–55%), colon (40%–45%), breast (25%–30%), endometrial cancer (20%–25%), and cholangiocarcinoma (60%–65%). 12 Although many studies have examined the possible function of MSLN, its role in cancer is still not entirely clear and may be cancer-type specific. 11 A few preclinical and clinical studies showed that MSLN is involved in cell proliferation and anoikis prevention, and its downregulation promotes drug-induced apoptosis and chemosensitivity.13–18 Paradoxically, data revealed that MSLN expression positively correlated with longer survival in gastric cancer. 19 Notwithstanding, in some cases, MSLN expression has been associated with increased tumor aggressiveness and poor clinical outcome and thus can be a potential tumor marker both in diagnosing and monitoring cancer progression. 7 Besides, due to unique combination of high expression in different solid tumors and its complete lack of vital normal tissues, MSLN is being widely desirable for tumor-selective toxic payload delivery and tumor immunotherapy approaches. 20
Numerous research has investigated serum MSLN as a diagnostic marker for EOC;21–29 however, very few studies have investigated tumor MSLN gene expression in EOC, and to date, no report has addressed its level in peritoneal fluid (PF) of ovarian cancer patients. In this study, we comprehensively investigated tumor MSLN gene expression and the plasma and PF MSLN level in EOC patients with different clinicopathological features including FIGO staging (FIGO I–IV), Shimizu–Silverberg grading (grades I–III), histological type of tumor (serous, mucinous, endometrioid, and undifferentiated), and Kurman and Shih type of EOC (types 1 and 2). Moreover, we assessed the correlation between level of MSLN and prognostic factors, that is, age, menopausal status, CA125, and body mass index (BMI), as well as we investigated MSLN prognostic value in EOC patients. Finally, we investigated plasma and serum MSLN levels and tumor MSLN expression to indicate whether there exists a correlation between them in patients with ovarian cancer.
Materials and methods
Ethic statement, standard protocol approvals, registrations, and patient consents
The study was approved by the Medical University of Lublin Ethics Committee. All samples were collected with signed informed consent from patients or healthy volunteers, and all related procedures of investigation were conducted in accordance with the ethical standards. From 2012 to 2016, 97 women with EOC were enrolled in this study in I Chair and Department of Oncological Gynaecology and Gynaecology (Independent Public Clinical Hospital No. 1, Medical University of Lublin, Poland). Exclusion criteria for study cohort included a history of previous malignancies, chemotherapy, or radiation therapy prior to surgery and autoimmune, allergic, and infectious diseases. In some cases, this requirement reduced the number of sample draws. In addition, 20 women without any definite gynecological disease (healthy donors (HD)) and 39 women with serous cystadenoma (SC; benign tumor) were also enrolled.
Clinical and molecular characteristic of patients
Clinical stage was determined according to FIGO classification. 30 Tumors were graded and classified according to Silverberg 31 grading system by two independent gynecological pathologists. The reference group consisted of 39 patients with benign tumors (histology: SC), with no evidence of malignancies or pelvic adhesions. Pre-existing clinical information, including age, menopausal status, clinical stage and grade, treatment history, and CA125 biomarker level, was collected from clinical and operative notes and discharge summary that were deposited in a centralized database. The BMI was calculated using mathematical online calculator (https://www.nhlbi.nih.gov/health/educational/lose_wt/BMI/bmi-m.htm). Data about the OS of EOC patients were obtained from the Document Personalization Center of the Ministry of Internal Affairs and Administration, Department of Protection of Confidential Information in Warsaw. The clinicopathological characteristics of the study cohort are summarized in Table 1.
Patient baseline characteristics.
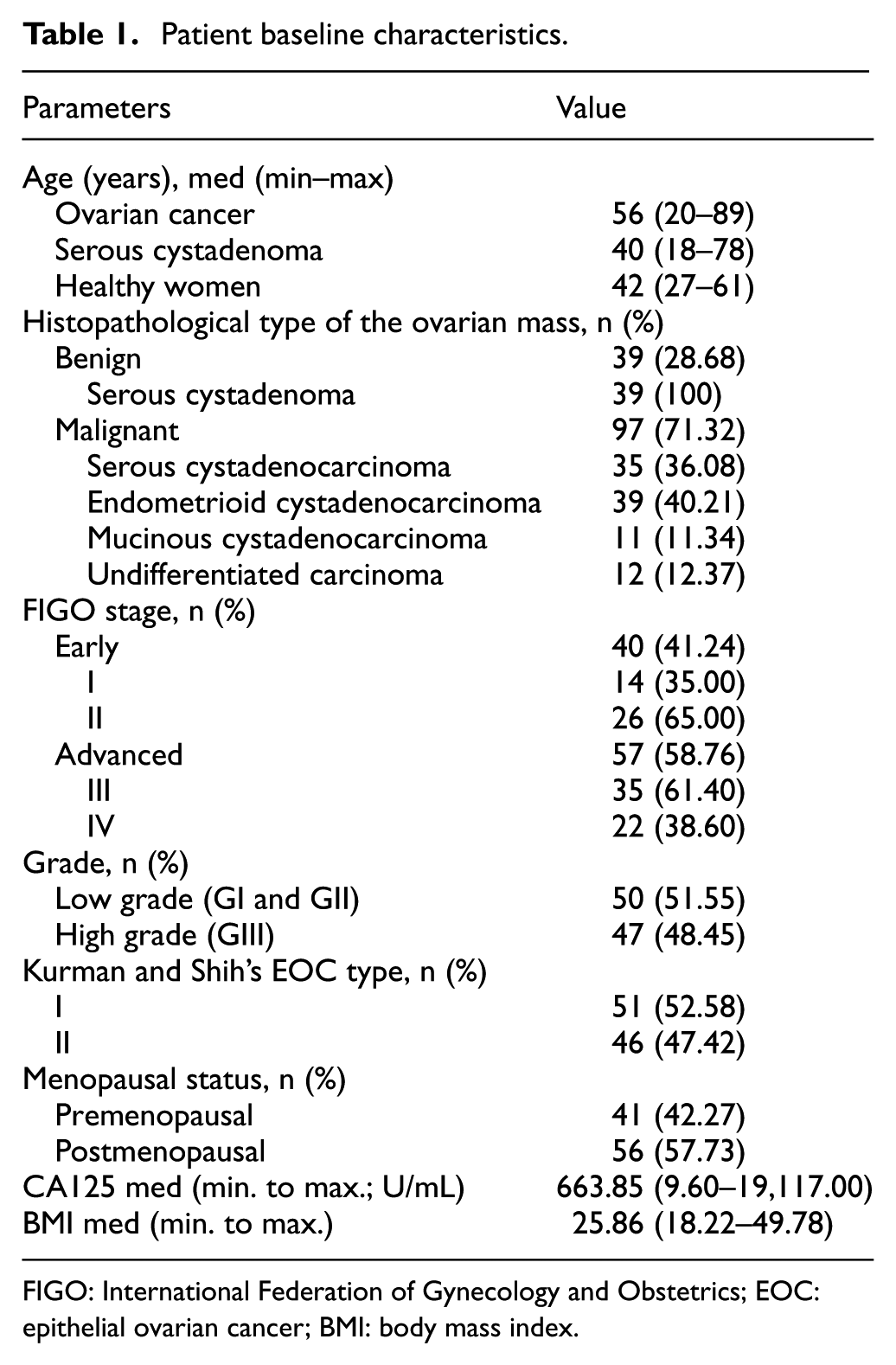
FIGO: International Federation of Gynecology and Obstetrics; EOC: epithelial ovarian cancer; BMI: body mass index.
Peripheral blood, PF and tumor tissue sample collection
Peripheral blood (PB) and PF were taken into heparinized tubes (sodium heparin). Venous blood samples were collected before the surgical procedure of all patients. Fresh PF was obtained during the operation, and fragments of neoplastically changed tissue, sized at least 1 cm3, without necrotic areas, were collected during the surgical procedure. Plasma and PF samples were rendered cell free by centrifugation at 1500 r/min for 10 min and stored at −80°C before being tested by enzyme-linked immunosorbent assay (ELISA). Tumor tissues (TT) were placed in RNAlater and stored at −80°C before being tested by quantitative real-time polymerase chain reaction (qPCR).
ELISA
MSLN concentrations in plasma and PF were determined by immunoassay kit (Research and Diagnostic Systems, Minneapolis, MN, USA) following the manufacturer’s protocol. Plate absorbance was read on an ELX-800 plate reader (BioTek Instruments, Inc, USA) and analyzed using Gen5™ (BioTek Instruments, Inc). Concentrations of MSLN (pg/mL) were calculated by interpolation from a standard curve.
RNA extraction and reverse transcription
Total RNA was extracted from TT (malignant n = 36 and benign n = 10) using AllPrep DNA/RNA/Protein Mini Kit (Qiagen, Venlo, Netherlands) according to manufacturer’s manual. From each sample, 1 μg of total RNA was reverse transcribed to 20 μL of complementary DNA (cDNA) using the high-capacity RNA-to-cDNA kit (Life Technologies). For qPCR, 1 μL of cDNA was used.
Real-time polymerase chain reaction (qPCR)
qPCR was performed using a CFX96 Touch™ Real-Time PCR Detection System (Bio-Rad Laboratories, Hercules, CA, USA) and was analyzed in CFX™ Manager Software (Bio-Rad) according to TaqMan technology. The analysis was performed using TaqMan® primer/probe sets (Thermo Fisher Scientific, Waltham, MA, USA) specific for glyceraldehyde-3-phosphate dehydrogenase (GAPDH; Hs03929097_g1) and MSLN (Hs00245879_m1) genes. All qPCR reactions were performed in triplicates in a final volume of 50 μL, including a 1 μL cDNA sample, 25 μL TaqMan® Gene Expression Master Mix (Thermo Fisher Scientific), 21.5 μL nuclease-free water (Thermo Fisher Scientific), and 2.5 μL TaqMan® GAPDH or MSLN probes (Thermo Fisher Scientific). After 5 min of initial denaturation at 95°C, cDNA was amplified in 50–60 cycles (denaturation for 15 s at 95°C and annealing for 60 s at 60°C). Reactions for all samples were run in three repetitions. The CT values (the number of cycles needed for amplification-generated fluorescence to reach a specific threshold of detection) obtained for each qPCR were used to calculate relative quantification (RQ) by the following formula: ΔCT = CTtarget (MSLN) − CThousekeeping (GAPDH). Next, 2–ΔCt value was calculated for each sample. 2–ΔCt of individual patients in ovarian tumors group was then normalized to the average 2–ΔCt value in benign tumor patients and represented as x-fold change.
Statistical analysis
Data were presented as medians with the interquartile ranges. The Mann–Whitney U test and one-way analysis of variance (ANOVA) were applied to the results of statistical comparison between the studied groups. Spearman’s rank test was used to assess the relationship between levels of MSLN and clinicopathological characteristics. Kaplan–Meier method and differences in survival curves were calculated using the logrank test; p value less than 0.05 was considered statistically significant. All statistical analyses were performed using Statistica 10.0/GraphPad Prism 5 software.
Results
Plasma MSLN level in EOC patients in relation to clinicopathological features
The concentrations of MSLN in the plasma of patients with different clinicopathological characteristic of EOC, SC (benign tumor) group, and healthy individuals are presented in Table 2.
The clinicopathological items, median plasma and peritoneal fluid mesothelin concentrations of MSLN in benign and malignant lesions in patients.
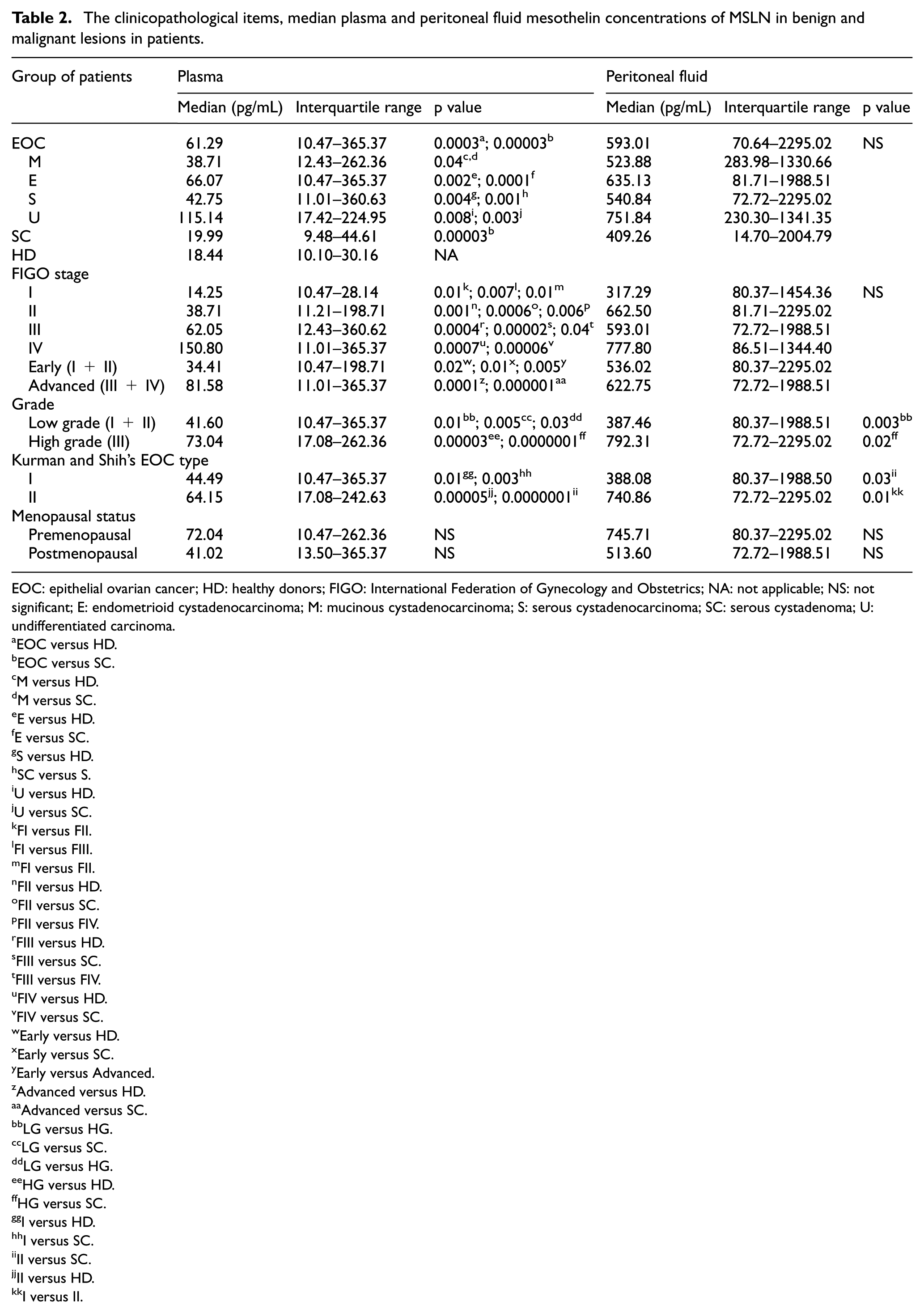
EOC: epithelial ovarian cancer; HD: healthy donors; FIGO: International Federation of Gynecology and Obstetrics; NA: not applicable; NS: not significant; E: endometrioid cystadenocarcinoma; M: mucinous cystadenocarcinoma; S: serous cystadenocarcinoma; SC: serous cystadenoma; U: undifferentiated carcinoma.
EOC versus HD.
EOC versus SC.
M versus HD.
M versus SC.
E versus HD.
E versus SC.
S versus HD.
SC versus S.
U versus HD.
U versus SC.
FI versus FII.
FI versus FIII.
FI versus FII.
FII versus HD.
FII versus SC.
FII versus FIV.
FIII versus HD.
FIII versus SC.
FIII versus FIV.
FIV versus HD.
FIV versus SC.
Early versus HD.
Early versus SC.
Early versus Advanced.
Advanced versus HD.
Advanced versus SC.
LG versus HG.
LG versus SC.
LG versus HG.
HG versus HD.
HG versus SC.
I versus HD.
I versus SC.
II versus SC.
II versus HD.
I versus II.
MSLN levels detected in the plasma of women with EOC were found to be significantly higher than those with benign ovarian disease (p = 0.00003) and control group (p = 0.0003). No significant changes were observed between MSLN concentration in control group and benign tumor (p > 0.05; Figure 1(a)).

Box plots of plasma levels of mesothelin in (a) healthy controls (n = 20), benign tumor (n = 25), and EOC patients (n = 56) (b and c) by FIGO stage; (d) histology type; (e) Kurman and Shih type; and (f) grade. Boxes indicate the 25th to 75th percentiles. The horizontal lines within the boxes are the median plasma levels and the whiskers indicate the minimum and maximum values. The p values indicate statistical significance of differences between each study group.
Plasma samples from women with different FIGO stages of EOC contained significantly higher concentrations of MSLN compared with samples from women with SC (vs stage II (p = 0.0006), stage III (p = 0.00002), and stage IV (p = 0.00006), respectively) and the HD (vs stage II (p = 0.001), stage III (p = 0.0004) and stage IV (p = 0.0007, respectively). No significant differences of MSLN level were observed in patients with FIGO stage I compared to SC patients and HD (p > 0.05). The plasma MSLN levels also differ significantly between different FIGO stages of EOC (stage I vs stage II (p = 0.01), III (p = 0.007) and IV (p = 0.01); stage II vs stage IV (p = 0.006); and stage III vs stage IV (p = 0.04), respectively; Figure 1(b)). The concentrations of MSLN in stage II were lower compared to stage III; however, the differences were not significant (p > 0.05; Figure 1(b)). In addition, plasma samples from women with early stage (FIGO I and II) and advanced stage (FIGO III and IV) of EOC contained significantly higher concentrations of MSLN compared with samples from women with SC (p = 0.01 and p = 0.000001, respectively) and HD (p = 0.02 and p = 0.0001, respectively). The significant difference in plasma MSLN level was also revealed between early and advanced FIGO stages (p = 0.005; Figure 1(c)) in EOC cohort.
Plasma MSLN levels from patients with different histological type of EOC were significantly elevated compared with both HD (HD vs mucinous (p = 0.04), endometrioid (p = 0.002), serous (p = 0.004), and undifferentiated type (p = 0.008)) and SC patients (SC vs mucinous (p = 0.04), endometrioid (p = 0.0001), serous (p = 0.001), and undifferentiated type (p = 0.003)); however, there were no statistically significant differences between different histological types (p > 0.05; Figure 1(d)).
The concentrations of MSLN were significantly higher in EOC type I and type II Kurman and Shih compared to both HD (p = 0.01 and p = 0.00005, respectively) and SC (p = 0.003 and p = 0.0000001, respectively) groups. The level of MSLN in type I tends to be lower compared with type II; however, the differences were not significant (p > 0.05; Figure 1(e)).
As shown in Figure 1(f), the plasma MSLN level was significantly higher in low-grade (I and II) and high-grade (III) EOC patients compared to women with SC (p = 0.005 and p = 0.0000001, respectively) and HD (p = 0.01 and p = 0.00003, respectively). Besides, the significant difference in MSLN level was also observed between low- and high-grade (p = 0.03) tumors. However, we do not observe changes in level of plasma MSLN between premenopausal and postmenopausal women with EOC (p > 0.05; Table 2).
PF MSLN level in EOC patients in relations to clinicopathological features
The concentrations of MSLN in the PF of patients with different clinicopathological characteristic of EOC and benign tumor patients are presented in Table 2.
The level of MSLN in EOC patients tend to be higher compared to women with benign tumor; however, the differences were not significant (p > 0.05) (Figure 2(a)). We did not observe significant changes in the MSLN levels in PF based on FIGO stage (Figure 2(b) and (c)) and histological type (Figure 2(d)) and compared to benign tumor. Interestingly, PF MSLN levels revealed significant changes between both Kurman and Shih type I and type II EOC (p = 0.01) and between benign tumor and type II EOC (p = 0.03) (Figure 2(e)). In addition, there were significant differences in MSLN level between SC patients and high-grade (III) EOC (p = 0.02) as well as between high-grade and low-grade (I and II) tumor samples (p = 0.003; Figure 2(f)). However, no significant changes were observed between MSLN level in women with benign tumor compared to patients with Kurman and Shih type I (Figure 2(e)) and low-grade (Figure 2(f)) (I and II) EOC, respectively (p > 0.05). Moreover, similarly to MSLN in plasma samples, we did not observe changes in the level of PF MSLN between premenopausal and postmenopausal patients with EOC (p > 0.05; Table 2).

Box plots of peritoneal fluid levels of mesothelin in (a) benign tumor (n = 24) and EOC patients (n = 71) (b and c) by FIGO stage, (d) histology type, (e) Kurman and Shih type, and (f) grade. Boxes indicate the 25th to 75th percentiles. The horizontal lines within the boxes are the median peritoneal fluid levels and the whiskers indicate the minimum and maximum values. The p values indicate statistical significance of differences between each study group.
Plasma and PF MSLN levels correlate with serum CA125 biomarker
The statistical data of the correlation of MSLN concentrations in plasma and PF with prognostic factors, that is, menopausal status, age, CA125, and BMI, are detailed in Table 2. Plasma and PF levels of MSLN positively correlated with concentrations of preoperative CA125 in serum (p = 0.002; R Spearman = 0.46, p = 0.02; R Spearman = 0.31; Figure 3(a) and (b), respectively). There were no significant correlations between plasma and PF MSLN levels in relation to age and BMI (p > 0.05; Table 2).

Correlations between levels of mesothelin and CA125 in EOC patients: (a) level of plasma mesothelin versus CA125 in serum (n = 42) and (b) level of PF mesothelin versus CA125 in peritoneal fluid (PF) (n = 53).
Relations of MSLN gene expression level to clinicopathological characteristic of patients
The relationships between clinicopathological items and expression levels of MSLN were further evaluated. A summary of the correlations between the expression level of MSLN gene and clinical characteristics of the patients is shown in Table 3.
The levels of MSLN expression in relation to clinicopathological features in epithelial ovarian cancer.

EOC: epithelial ovarian cancer; FIGO: International Federation of Gynecology and Obstetrics; NS: not significant; E: endometrioid cystadenocarcinoma; M: mucinous cystadenocarcinoma; S: serous cystadenocarcinoma; SC: serous cystadenoma; U: undifferentiated carcinoma.
EOC versus SC.
SC versus E.
E versus S.
Early versus SC.
Advanced versus SC.
LG versus SC.
LG versus HG.
HG versus SC.
I versus SC.
II versus SC.
The MSLN mRNA expression levels were significantly higher in EOC tissue compared to benign masses (p = 0.02; Figure 4(a)). In addition, tissue samples from women with early stage (FIGO I and II) and advanced stage (FIGO III and IV) of EOC expressed significantly higher level of MSLN compared with samples from women with SC (p = 0.03 and p = 0.04; respectively). No significant changes were observed between MSLN expression level in early stage and advanced stage of EOC (p > 0.05; Figure 4(b)).

MSLN gene expression levels in (a) benign tumor (n = 10) and EOC patients (n = 36) (b and c) by FIGO stage, (c) grade, (d) histology type, (e) Kurman and Shih type, and (f) correlation with CA125. Box plots ((a)–(d)) indicate the 25th to 75th percentiles. The horizontal lines within the boxes are the median tissue levels and the whiskers indicate the minimum and maximum values. The p values indicate statistical significance of differences between each study group.
Besides, we observed significant differences in MSLN expression level between SC patients and both low-grade (I and II) and high-grade (III) (p = 0.04 and p = 0.01, respectively) EOC as well as between high-grade and low-grade (I and II) TT (p = 0.04; Figure 4(c)).
Furthermore, the level of MSLN expression in patients with different histology type of EOC tends to be higher compared to women with benign tumor; however, the significant differences were observed only in mucinous type (p = 0.01). Interestingly, we observed significantly higher MSLN expression in endometrioid type compared to serous type of EOC (p = 0.02; Figure 4(d)).
The expression level of MSLN was significantly higher in EOC type I and type II Kurman and Shih compared to SC (p = 0.03 and p = 0.04, respectively); however, no significant changes were observed between MSLN expression level in type I and type II EOC (p > 0.05; Figure 4(e)). Besides, similar to MSLN level in both plasma and PF samples, we did not observe changes in level of MSLN expression in relation to menopausal status of EOC patients (p > 0.05; Table 3).
Moreover, TT levels of MSLN positively correlated with concentrations of serum CA125 (p = 0.002; R Spearman = 0.53; Figure 4(f)). Similar to plasma and PF samples, there were no significant correlations between TT MSLN levels and both age and BMI of EOC patients (p > 0.05; data not shown).
Elevated plasma MSLN level is related with poor survival in EOC patients
Next, the level of MSLN was evaluated if it could also be a prognostic factor for the 5-year OS of EOC patients. The MSLN levels of women operated from 2012 to 2014 were enrolled in this analysis.
Using the Kaplan–Meier product-limit method, we calculated the probable factors for predicting 5-year OS for EOC patients. When the cut-off value was set as the median, Kaplan–Meier analyses showed that only high MSLN levels in plasma of pretreatment EOC patients were significantly associated with worse 5-year OS compared with low MSLN levels (logrank test, p = 0.03; Figure 5). Neither PF nor TT levels of MSLN were significantly correlated with patient survival (data not shown).

Kaplan–Meier 5-year overall survival curves for patients with EOC (n = 51) based on mesothelin levels in plasma (red line: values above median; blue line: values below median). Statistical analysis of prognostic survival was performed by the logrank test (p = 0.03).
Plasma MSLN level was correlated with both PF MSLN level and tumor MSLN expression
Finally, we investigated the correlation between plasma and PF MSLN levels and tumor MSLN expression to assess whether plasma MSLN level may reflect a patient’s tumor MSLN status.
Plasma levels of MSLN positively correlated with concentrations of MSLN in PF (p = 0.0001; R Spearman = 0.60; Figure 6(a)) and MSLN expression in TT (p = 0.03; R Spearman = 0.54; Figure 6(b)). There were no significant correlations between level of MSLN in PF and TT (p > 0.05; data not shown).

The relationship between levels of mesothelin in plasma, peritoneal fluid, and tumor tissue in EOC patients. (a) Level of plasma versus peritoneal fluid mesothelin (n = 40) and (b) level of plasma versus tumor tissue mesothelin (n = 16).
Discussion
Ovarian cancer biomarkers for miscellaneous applications, such as early detection, prognosis monitoring, and prediction of therapeutic response—in particular, for targeted therapy—remain an important unmet clinical need. In this study, we comprehensively analyzed MSLN expression in TT by qPCR and we assessed both plasma and for the first time PF MSLN level using ELISA in EOC patients in terms of its clinicopathological relevance.
We found concentrations of MSLN in the plasma to be significantly elevated in women with EOC compared with both benign ovarian SC patients and healthy women. Besides, we did not see any elevated plasma MSLN levels in patients with benign ovarian SC, which seems to be consistent with its noninvasive nature. This is in agreement with previous findings which assessed serum MSLN level in ovarian cancer patients.5,21 MSLN could be a predictor for disease severity in women with EOC. In this study, we found that higher preoperative MSLN plasma levels in patients with EOC significantly correlated with advanced FIGO stages, higher grade, elevated concentrations of CA125 in serum, and reduced 5-year OS, which are consistent with a previous report, thus confirmed engagement of MSLN in tumor invasion.5,32 However, we did not demonstrate the significant correlation between plasma MSLN level and age, menopausal status, and BMI in ovarian cancer patients. Similar results were published in another study, whereby levels of serum MSLN did not associate with personal factors including age and BMI. 33
According to the Shih and Kurman 34 classification, EOC can be divided into type I and type II with distinct genetic and clinicopathologic characteristics. Type I includes low-grade endometrioid, mucinous, serous, and clear cell carcinomas, which are less aggressive, and better prognosis. In contrast, type II contains mostly high-grade undifferentiated and serous carcinomas which evolve rapidly and are strongly aggressive.35,36 Although we observed the tendency to elevate MSLN concentrations in type II compared with type I, the differences were not statistically significant. Our results confirm previous observations that MSLN was secreted to the circulation by the mesothelial cells of the peritoneum affected by the tumor cells. The elevated serum levels of MSLN represented the phenomenon of tumor progression. Concerning the peritoneal dissemination/metastatic spread, genes associated with the malignant features of cancer cells, that is, decreased intercellular adhesion, increased cell to matrix adhesion, and resistance to apoptosis, might be pivotal for the identification of potential biomarkers of ovarian cancer.
Our current data revealed that the plasma MSLN concentrations of all EOC patients with various histological types increased when compared to those in healthy individuals or women with benign ovarian tumors. However, the levels of MSLN in various histological types of EOC were not significantly different, which is consistent with hitherto findings.5,37 It seems that MSLN could be used as a general marker for the screening of ovarian cancer patients. The previous study showed that the serum MSLN levels of ovarian cancers of the mucinous type increased compared with the other histological types, that is, serous or endometrioid. Therefore, MSLN seems to be more effective than CA125 in detecting ovarian cancers of mucinous type, according to this survey. 5 However, our data demonstrated that the median of plasma MSLN concentration in mucinous cystadenocarcinoma was the lowest compared with other histological types, that is, endometrioid, serous, and undifferentiated, and no such correlation for the mucinous type of cancer was found. This is in accordance with results presented in another study. 38 It is worth notifying that data revealed that MSLN cannot serve alone as a biomarker for the detection of ovarian cancer.22,39 However, MSLN and CA125 as a combined marker provided greater sensitivity for early ovarian cancer diagnosis. Thus, data suggest that MSLN might be used in combination with CA125 and/or HE4 to achieve greater sensitivity. 22
In contrast to the extensive research that has been conducted for serum MSLN as a new diagnostic marker for ovarian cancer,21–29 to our knowledge, there have been no reports regarding MSLN level in PF. Our findings imply that the PF MSLN levels did not differ significantly among patients with benign and EOC, although levels of MSLN in the PF samples were nearly 10-fold higher compared with plasma samples. Moreover, there were no significant changes between benign tumor and both FIGO stages and histological types of EOC. Besides, we did not observe differences between the level of PF MSLN in different FIGO stages and histological types. These data led us to surmise that high MSLN production in the peritoneal cavities may be characteristic features for both benign and malignant diseases. Notwithstanding, similar to plasma samples, PF MSLN levels in patients with EOC positively correlated with CA125 level; however, we did not demonstrate the significant correlation between PF MSLN level and other personal factors, that is, age, menopausal status, and BMI. In contrast to plasma MSLN level, the low PF MSLN concentration was not associated with better prognosis in EOC patients. Interestingly, we demonstrated significantly elevated level of PF MSLN in type II compared with type I Kurman and Shih and in the high grade compared with low grade of EOC. The reason for this finding is not well understood. However, these data led us to speculate that highly elevated PF MSLN production in the peritoneal cavities of aggressive ovarian cancer patients (type II Kurman and Shih and high-grade EOC) may have an important role in the peritoneal dissemination. Recent report indicates that inhibition of immunosuppressive interleukin (IL)-10 in the tumor microenvironment (TME) can restore MSLN chimeric antigen receptor (CAR) T cell activity in pancreatic carcinoma. 40 In previous studies, we demonstrated considerably elevated IL-10 levels in the PF in women with malignant ovarian tumors. 41 We have also shown that monocyte-derived dendritic cells (Mo-DCs) generated from type II Kurman and Shih of OC secreted higher levels of IL-10 compared to the cells collected from women with type I. 36 It seems probable that substantial reversal of immunosuppression can be realized by blocking IL-10 in the TME, which may allow to increase cytotoxicity of MSLN-engrafted CAR T cells and enhance the potential for their using in clinical practice. It is worth noting that previous results demonstrated that tumor cells undergo MSLN/CA125-dependent cell adhesion in the mesothelial cells of peritoneum and confirmed that MSLN and CA125 mediate cell attachment. 42 Another study revealed that this MSLN/CA125 interaction may also play a pivotal role in peritoneal metastasis of ovarian cancer. 43 In this perspectives, the potential for future research in the field of new therapeutic approaches aiming to limit peritoneal dissemination of ovarian cancer cells is tremendous.
We further investigated the MSLN gene expression in benign and malignant masses. Many studies assessed the MSLN expression by immunohistochemistry (IHC),37,43–46 and there have been only a few reports regarding tumor MSLN expression by qPCR.15,47 We demonstrated that high tumor MSLN expression was observed in EOC compared to benign tumor and that it was correlated with the clinical grade. We did not observe significant differences between early and advanced clinical stages as well as between type I and type II Kurman and Shih EOC. Tissue expression of MSLN in women with different histology type of EOC tends to be higher compared to patients with benign tumor; however, the significant differences were observed only in mucinous tumors. Interestingly, we noticed significantly elevated expression of MSLN in endometrioid type compared to serous type of EOC. Similar to plasma and PF samples, MSLN expression levels in patients with EOC positively correlated with CA125 level; however, we did not demonstrate the significant correlation between MSLN gene expression and other prognostic factor, that is, age, menopausal status, and BMI as well as survival outcomes. MSLN expression and its relation to clinicopathological factors in EOC remain controversial. Previous data reported that 55% of ovarian serous carcinomas express MSLN, and high MSLN level was associated with a favorable prognosis. 44 However, another study revealed that more than 75% of high-grade serous carcinomas showed positive staining for MSLN but revealed no prognostic significance with disease-specific survival. 45 Furthermore, recent data reported that ∼70% EOC and ∼90% serous histotype tumors showed high MSLN expression. Besides, high MSLN expression was prognostic for OS in the entire cohort and not for OS in the serous subgroup. 37 Although these three reports used the same anti-MSLN monoclonal antibody (i.e. clone 5B2), discrepancies are noticeable. These results thus need to be interpreted with caution. Moreover, previous results 47 showed significant correlations of MSLN gene expression level with tumor stage and grade, as well as shorter OS. However, they presented MSLN expression in all FIGO stages separately as well as demonstrated significantly worse OS in advanced stage of EOC group; hence, there may be the cause of partial discrepancies in data results presented by our group, where we presented level of MSLN expression in early (I and II) and advanced (III and IV) FIGO stage as well as we assessed OS in the whole EOC cohort.
The identification of precise and feasible EOC biomarkers is urgently needed. Plasma/serum factors can be assessed longitudinally and noninvasively, thus they are potentially an ideal source of such biomarkers. To determine whether a relationship exists between the levels of MSLN in three TMEs in patients with EOC, we investigated circulating MSLN level in serum and PF and tumor MSLN expression. We demonstrated that plasma MSLN level was correlated with both PF MSLN level and tumor MSLN expression. Although analysis showed no significant correlation between level of MSLN in PF and TT, our data suggest that the plasma MSLN level may noninvasively predict the local tumor MSLN status, and its level is a potential surrogate marker for tumor MSLN expression in EOC patients. These results were in agreement with previous studies which revealed a relationship between tumor MSLN expression assessed by immunochemistry and the serum MSLN level measurement by ELISA in EOC patients. 37 The present results mostly demonstrate the potential of monitoring of the local tumor MSLN status noninvasively through measurements of plasma/serum MSLN.
The limitations of our study need to be addressed. First, in view of currently insurmountable difficulties in obtaining normal PF, we acquired it from women with SC as a reference fluid developing in the absence of malignant process. Second, due to the hardship in obtaining normal ovarian tissue and limited availability of ovarian TT, we analyzed MSLN gene expression in small patient cohort with malignant and benign tumors. Third, we were unable to investigate a correlation between level of MSLN and progression-free survival (PFS). This factor was primarily because we did not have clinical data of patients with recurrent ovarian cancer. Of note, in some patients, clinical data (i.e. BMI, CA125) were not obtained due to the shortages of these information in database.
A comprehensive prospective study to assess the correlation of plasma, PF, and tumor MSLN with clinicopathological features of patients in a large cohort is warranted. A robust detection method based on molecular profiles for ovarian cancer has not yet been established because the disease exhibits a wide range of morphological, clinical, and genetic variations during the course of tumor progression. Owing to these cellular and molecular characteristics, identifying appropriate treatments for the disease is a major clinical challenge. In conclusion, we report herein the differences in clinicopathological significance of plasma, PF, and TT levels of MSLN in EOC patients. Notwithstanding, our data suggest a potential application of the MSLN as a diagnostic and prognostic biomarker and in tumor-evolution monitoring in patients with ovarian tumors. What is important, we revealed a new clinicopathological relevance of MSLN in PF of EOC patients, as well as we indicated a potential application of the plasma MSLN level as a noninvasive biomarker for real-time monitoring of patients with ovarian cancer. In this perspective, future studies are needed, which may be essential to managing patients better and to reveal potential new disease pathomechanisms and therapeutic options.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Signed informed consent was obtained from all individual participants included in the study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Medical University of Lublin MNsd127 grant. The paper was developed using the equipment purchased within the Project “The equipment of innovative laboratories doing research of new medicines used in the therapy of civilization and neoplastic diseases” within Operational Program Development of Eastern Poland 2007–2013, Priority Axis I Modern Economy, Operations I.3 Innovation Promotion.
