Abstract
Interleukin-13 receptor alpha 2 is one of the subunits of transmembrane receptor for interleukin-13. The aim of this study was to investigate the prognostic value of interleukin-13 receptor alpha 2 expression in invasive breast cancer. Interleukin-13 receptor alpha 2 expressions were assessed by immunohistochemistry in tissue microarrays of 1283 invasive breast cancer samples, and associations between these expressions and clinicopathological variables and clinical outcomes were investigated. Interleukin-13 receptor alpha 2 expression was observed in 138 (10.8%) samples, and found to be associated with positive estrogen receptor (p < 0.001) and progesterone receptor (p < 0.001) and with the luminal subtype (p < 0.001). No significant association was found between interleukin-13 receptor alpha 2 expression and other clinicopathological variables including age, tumor size, lymph node metastasis, histologic types, histologic grade, HER2 status, Ki-67 labeling index, or tumor-infiltrating lymphocytes levels. Patients with interleukin-13 receptor alpha 2 expression tended to have poorer disease-free survival, but the difference was not statistically significant (p = 0.069). Subgroup analysis showed luminal breast cancer patients positive for interleukin-13 receptor alpha 2 expression had significantly poorer disease-free survival (p = 0.018) than luminal breast cancer patients negative for interleukin-13 receptor alpha 2 expression. However, no association between interleukin-13 receptor alpha 2 expression and clinical outcome was observed in HER2-positive and triple-negative subgroups (p = 0.574 and p = 0.936, respectively). Multivariate analysis showed interleukin-13 receptor alpha 2 expression was an independent poor prognostic factor for luminal breast cancer (p = 0.03). This study shows interleukin-13 receptor alpha 2 expression could be a useful prognostic marker for selecting patients with luminal breast cancer likely to follow a clinically aggressive course despite receiving systemic therapy.
Introduction
Inflammatory cytokines are important components of tumor microenvironments and are linked to tumor growth. Interleukin-13 (IL-13) is a key immunoregulatory and anti-inflammatory cytokine in tumors and is produced by various immune cells such as T, B, mast, natural killer, and dendritic cells. 1 Many human solid tumors have been reported to express IL-13 receptor (IL-13R), which consists of three different subunits: IL-13R alpha 1 (IL-13Rα1), IL-13R alpha 2 (IL-13Rα2), and IL-4R alpha (IL-4Rα). IL-13 binds to IL-13Rα1, recruits IL-4Rα to form a functional receptor complex (so-called Type II IL-4R), and activates the signal transducer and activator of transcription 6 (STAT6) pathway to promote proliferation and apoptosis resistance. 1,2 IL-13 can also bind to transmembrane monomeric IL-13Rα2 with high affinity (∼50 times higher than that of IL-13Rα1) to activate the activator protein 1 (AP-1) pathway, induce transforming growth factor β (TGFβ) and, thus, increase metastasis. 2,3 A soluble IL-13Rα2 decoy receptor has also been reported to bind IL-13, but this receptor did not contain the transmembrane signaling domain, and resulted in alternative splicing. 4 IL-13Rα2 can inhibit signaling through the STAT6 pathway by sequestering IL-13 from IL-13Rα1/IL-4Rα complex or by physically blocking docking of STAT6 with this receptor complex. 5,6
IL-13Rα1/IL-4Rα and IL-13Rα2 are expressed at low levels on nonlymphoid cells and upregulated on tumor cells. 7,8 Furthermore, IL-13Rα2 upregulation has been reported to be associated with invasion, metastasis, and poor prognosis in colorectal, breast, and gastric cancer. 9–11 In orthotopic animal models of human pancreatic and ovarian cancer, IL-13Rα2 overexpression in tumor cells was found to be associated with increased metastasis and reduced survival. 12,13 Recently, we studied IL-13Rα1 expression in tumor cells of invasive breast cancer (IBC) samples and demonstrated that IL-13Rα1 expression was significantly associated with aggressive phenotypes and reduced survival. 14 However, little is known about the prognostic impact of IL-13Rα2 expression in breast cancer.
In this study, we evaluated IL-13Rα2 expression by immunohistochemistry in IBC samples and sought to identify relations between these results and clinicopathological features and survival to determine the prognostic value of IL-13Rα2 expression.
Materials and methods
Selection of cases and clinicopathological information
A total of 1283 IBC surgical samples (1283 patients) obtained by resection between 1995 and 2007 at Yeungnam University Hospital were included in this study. The patients concerned received standard radiotherapy or adjuvant systemic therapy (hormone therapy or chemotherapy) after surgery. However, those that received neoadjuvant chemotherapy were excluded. Tissue microarrays (TMAs) were constructed using a Quick-Ray® Manual Tissue Microarrayer (Unitma, Seoul, Korea) and Quick-Ray recipient blocks (Unitma), as previously described. 15 A pair of 2-mm-diameter tumor tissue cores was retrieved from each case and then transferred to a recipient block.
Clinicopathological information, that is, age, tumor size, lymph node status, histologic subtype, lymphovascular invasion, histological grade, Ki-67 labeling index, and tumor-infiltrating lymphocyte (TIL) level 16 were retrospectively collected by reviewing medical records and whole section hematoxylin and eosin-stained slides. Estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2) statuses were obtained from our previous study. 17 Molecular subtypes were defined as follows: luminal (ER and/or PR+/HER2−), HER2-positive (any ER/any PR/HER2+), and triple-negative (ER−/PR−/HER2−). IL-13Rα1 expression status was available in 1126 cases from our previous study. 14
Postoperative follow-ups ranged from 1 to 238 months (median, 117 months). Information on cause of death was obtained from medical records and the microdata service system provided by Statistics Korea (http://mdis.kostat.go.kr). Overall survival (OS) was defined as time from surgical resection to death from any cause or last follow-up. Disease-free survival (DFS) was defined as the time from surgical resection to locoregional recurrence, distant metastasis, death, or last follow-up. A patient with no event (death, recurrence, or metastasis) or follow-up loss was considered as a censored case.
The study was approved beforehand by the Institutional Review Board of Yeungnam University Hospital (YUMC 2017-12-005), which waived the requirement for informed consent.
Immunohistochemical evaluation of IL-13Rα2 expression
IL-13Rα2 expression was estimated immunohistochemically using 4-μm-thick sections taken from TMA slides and an automated Benchmark platform (Ventana Medical Systems, Tucson, AZ, USA) according to the manufacturer’s instructions. Briefly, slides were treated with protease for 4 min, and then incubated with anti- IL-13Rα2 mouse monoclonal antibody (1:1000, Abcam, Cambirdge, MA, USA) for 60 min at 37°C. Stainings were visualized using an UltraView universal DAB detection kit (Ventana Medical Systems).
Immunohistochemical expressions of IL-13Rα2 were interpreted semi-quantitatively by two pathologists (Y.K.B. and H.J.K.) under a multiheaded microscope. Immunoreactivity scores (IRSs) were calculated based on staining intensities and extents, as previously described. 17 Staining intensities were assessed as follows: negative (0), weakly positive (1), moderately positive (2), or strongly positive (3), and staining extents were defined as the proportions of positive tumor cells showing cytoplasmic staining and classified as follows: 0% (0), 1%–25% (1), 26%–50% (2), 51%–75% (3), or >75% (4). IRSs were calculated by multiplying intensity and extent scores (minimum score 0 and maximum score 12). For the statistical analysis, a case was classified as positive for IL-13Rα2 expression when its IRS was ≥ 1.
Statistical analysis
Statistical analysis was performed using IBM SPSS Version 23.0 for Windows (IBM Co., Armonk, NY, USA). The chi-square test or Fisher’s exact test was used to evaluate associations between IL-13Rα2 expression and clinicopathological variables. Survival curves were plotted using the Kaplan–Meier method and the log-rank test was used to determine the significances of survival differences. A Cox proportional hazards model was used to compare hazard ratios (HRs) during univariate and multivariate analyses. Variables found to be significant by univariate analyses were subjected to multivariate analysis. Adjusted HRs and associated 95% confidence intervals (CIs) were estimated for each variable. All tests were two-sided, and p values of <0.05 were considered statistically significant.
Results
Patient demographics
Ages at diagnosis of the 1283 patients ranged from 20 to 86 years (mean, 48 years) and tumor sizes from 0.5 to 11.0 cm (mean, 2.3 cm). A total of 648 (50.5%) patients had an invasive tumor of ≤ 2.0 cm (pT1), and the other 635 patients had an invasive tumor of >2.0 cm (pT2 in 583; pT3 in 47; pT4 in 5). Axillary lymph node metastasis was found in 601 (47%) patients and lymphovascular invasion in 662 (51.6%). In three patients, neither sentinel lymph node biopsy nor axillary lymph node dissection was performed. Histological grades were 1 in 214 (16.7%), 2 in 372 (29.0%), and 3 in 697 (54.3%) patients. In all, 759 (59.2%) underwent mastectomy, and 524 (40.8%) breast-conserving surgery. ER was positive in 859 (67.0%) and PR was positive in 743 (57.9%). HER2 overexpression or gene amplification was present in 257 (20.0%). Seven cases did not have a Ki-67 labeling index documented in pathology reports. As regards molecular subtypes, 756 (58.9%) were classified as luminal, 257 (20.0%) as HER2-positive, and 270 (21.0%) as triple-negative. IL-13Rα1 expression results were available in 1126 cases from our previous study. 14 Among them, 589 (52.3%) were classified as positive expression.
For adjuvant chemotherapy, 908 (70.8%) patients received anthracycline-based chemotherapy and 208 (16.2%) received non-anthracycline chemotherapeutic regimens. The remaining 167 (13.0%) patients did not receive chemotherapy. Hormone therapy using tamoxifen or aromatase inhibitors was performed in 876 (68.3%) and radiation therapy was performed in 632 (49.3%). During follow-up (mean 121 months after surgery), recurrence/metastasis occurred in 196 (15.3%) patients, and at last follow-up 156 (12.2%) deaths had occurred.
IL-13Rα2 expression and its clinical significance
Non-neoplastic ductal epithelial cells within tumor cores were negative or weakly positive for IL-13Rα2 expression. However, stromal inflammatory cells exhibited strong immunoreactivity for IL-13Rα2. Immunostaining intensities for IL-13Rα2 in tumor cells varied from case to case, weak in 55 (4.3%), moderate in 69 (5.4%), and strong staining in 14 (1.1%) cases, whereas they were similar in individual cases (Figure 1). IL-13Rα2 IRSs were as follows: 0 in 1145 (89.2%) patients, 1 in 31 (2.4%), 2 in 60 (4.7%), 3 in 5 (0.4%), 4 in 16 (1.2%), 6 in 12 (0.9%), 8 in 5 (0.4%), 9 in 5 (0.4%), and 12 in 4 (0.3%). Positive IL-13Rα2 (IRS ≥ 1) expression was observed in 138 (10.8%) patients (Figure 2).

Representative IL-13Rα2 expressions in breast cancer tumor cells. IL-13Rα2 immunoreactivities in breast cancer samples were rated as (a) negative, (b) weak, (c) moderate or (d) strong. Some stromal inflammatory cells (red arrows) were positive for IL-13Rα2, whereas non-neoplastic ductal epithelial cells (black arrows) were negative or weakly positive.

Distribution of IL-13Rα2 immunoreactivity scores (IRSs) in the study cohort. IRSs (0–12) are calculated by multiplying intensity score (0–3) and extent score (0–4).
IL-13Rα2 expression was found to be significantly associated with ER (p < 0.001) and PR (p < 0.001) positivity, and luminal subtype (p < 0.001), but not with clinicopathological variables, that is, age, tumor size, lymph node metastasis, histologic grade, HER2, Ki-67, TIL levels, and IL-13Rα1 expression in all breast cancers. IL-13Rα2 expression was positive in 110 (14.6%) of 756 luminal breast cancers, 22 (8.6%) of 257 HER2-positive breast cancers, and 6 (2.2%) of 270 triple-negative breast cancers (p < 0.001). When correlations between IL-13Rα2 expression and clinicopathological variables were analyzed in different molecular subtypes, IL-13Rα2 expression was significantly associated with high histologic grade in luminal breast cancers, and high TIL level in HER2-positive breast cancers (Table 1).
Correlations between IL-13Rα2 expression and clinicopathological parameters in 1283 invasive breast cancers and their molecular subtypes.
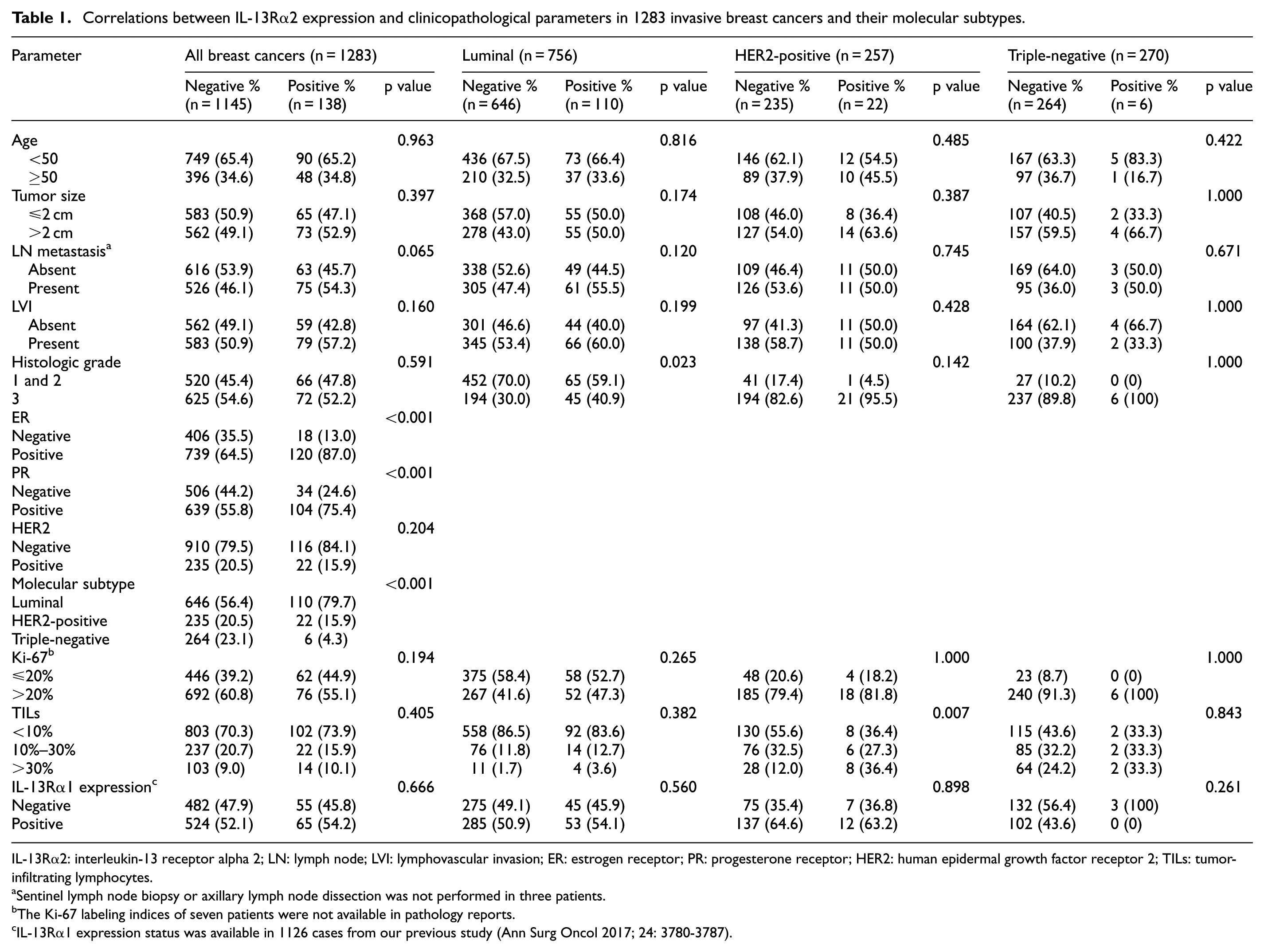
IL-13Rα2: interleukin-13 receptor alpha 2; LN: lymph node; LVI: lymphovascular invasion; ER: estrogen receptor; PR: progesterone receptor; HER2: human epidermal growth factor receptor 2; TILs: tumor-infiltrating lymphocytes.
Sentinel lymph node biopsy or axillary lymph node dissection was not performed in three patients.
The Ki-67 labeling indices of seven patients were not available in pathology reports.
IL-13Rα1 expression status was available in 1126 cases from our previous study (Ann Surg Oncol 2017; 24: 3780-3787).
Patients with an IL-13Rα2 expressing tumor showed a tendency to have poorer DFS than those with a non-IL-13Rα2 expressing tumor, but the difference was not statistically significant (p = 0.069, Figure 3(a)). However, there was no relation observed between IL-13Rα2 expression and OS (p = 0.767, Figure 3(b)). Subgroup analysis was performed to explore possible associations between IL-13Rα2 expression and prognosis by molecular subtype. Of the 756 luminal breast cancer patients, those with an IL-13Rα2 expressing tumor had significantly shorter DFSs (p = 0.018, Figure 3(c)). However, no such relation was observed for HER2-positive (p = 0.574, Figure 3(d)) or triple-negative subtypes (p = 0.936, Figure 3(e)). There was no survival difference according to staining intensity or IRS among positive cases for IL-13Rα2 expression.

Kaplan–Meier survival curves according to IL-13Rα2 expression status. (a) Disease-free survival (DFS) and (b) overall survival curves in all breast cancer patients, (c) DFS in luminal breast cancer patients, (d) DFS in HER2-positive breast cancer patients, and (e) DFS in triple-negative breast cancer patients.
Multivariate analyses showed IL-13Rα2 expression independently predicted DFS in luminal breast cancer patients (HR, 1.637; CI, 1.048-2.558; p = 0.03), along with lymph node status (p = 0.008), histologic grade (p = 0.001), and hormone therapy status (p = 0.046; Table 2).
Univariate and multivariate analyses of clinicopathological variables affecting disease-free survival in luminal breast cancer (n = 756).

CI: confidence interval; IL-13Rα2: interleukin-13 receptor alpha 2; BCS: breast conserving surgery.
We also evaluated survival differences with respect to the combined expression of IL-13Rα1 and IL-13Rα2 to explore synergistic effect of both receptors on patient survival of luminal breast cancer. The DFS rate was lowest in the IL-13Rα1+/IL-13Rα2+ group, moderate in the IL-13Rα1+/IL-13Rα2– and IL-13Rα1–/IL-13Rα2+ groups, and highest in the IL-13Rα1–/IL-13Rα2– group (p = 0.004, Figure 4). Both the positive cases showed a threefold higher risk of recurrence or metastasis (95% CI, 1.567−5.701; p = 0.001) than both the negative cases.

Kaplan–Meier survival curves for disease-free survival according to combined expression patterns of IL-13Rα1 and IL-13Rα2.
Discussion
In this study, we found IL-13Rα2 was overexpressed in 10.8% of IBC samples, and that its expression was significantly associated with the luminal subtype but not with the HER2-positive or triple-negative subtype. Furthermore, IL-13Rα2 expression independently predicted poor prognosis for the luminal subtype, but not for the HER2-positive or triple-negative subtype. These results suggest that IL-13Rα2 plays important roles in cancer cell survival and progression in luminal breast cancers.
Several studies have evaluated the clinical significance of IL-13Rα2 expression using human cancer samples. Papageorgis et al. 11 reported that high IL-13Rα2 expression was associated with the basal rather than the luminal subtype, and with poor prognosis for high-grade tumors but not for low-grade breast tumors, whereas we observed IL-13Rα2 expression was more frequently observed in the luminal subtype and not significantly associated with histologic grade. Furthermore, we found the association between IL-13Rα2 expression and poor prognosis was independent of histologic grade. However, Papageorgis et al. 11 performed a meta-analysis of gene expression microarray date obtained during the Memorial Sloan Kettering Cancer Center (MSKCC) primary breast tumor cohort study and determined tumor subtypes based on the expressions of keratins (basal, keratin 5/17 or luminal, keratin 8/18), that is, irrespective of ER, PR, or HER2 status. They also reported a marginal association between IL-13Rα2 expression and ER-negative tumors but no such association for PR or HER2 status. On the other hand, Kawakami et al. 18 reported IL-13Rα2 mRNA was expressed in MCF-7 cells (a luminal A breast cancer cell line), but not in triple-negative (BT-20 and MDA-MB-231), luminal B/HER2-positive (ZR-75-1), or HER2-positive (SK-BR-3) breast cancer cell lines. Their study supports our findings that most of the breast cancers that express IL-13Rα2 are luminal subtypes.
Kioi et al. 19 reported that IL-13Rα2 expression (determined immunohistochemically) was observed in 34% of ovarian cancer (75% of clear cell carcinoma samples) and strong IL-13Rα2 expression was associated with poor prognosis, whereas normal ovarian tissues and benign ovarian tumors did not express IL-13Rα2 or only expressed it at low levels. Lin et al. 10 reported immunohistochemical IL-13Rα2 expression in gastric cancer cells independently predicted OS, after adjusting for lymph node metastasis, depth of invasion, and distant metastasis. Barderas et al. 9 observed moderate or high IL-13Rα2 expression in 66.3% (53/80) of human colorectal cancer samples, but no or weak expression in adjacent normal tissue samples. Furthermore, they found IL-13Rα2 expression was significantly associated with late stage (advanced T stage, lymph node involvement, and presence of metastasis) and poor prognosis. As was reported in studies of ovarian and colorectal cancers, 9,19 we observed normal breast epithelium displayed no or weak IL-13Rα2 expression, and that IL-13Rα2 was mainly expressed in breast cancer cells. These results suggest IL-13Rα2 plays an important role during tumor progression.
Fujisawa et al. 12,13 demonstrated that IL-13 induces the expressions and productions of matrix metalloproteinases (MMPs) by signal transduction through IL-13Rα2 in vitro. In the earlier in vivo study using an orthotopically implanted human pancreatic cancer mouse model, mice with IL-13Rα2-positive cancer exhibited more metastases to lymph nodes and liver and poorer prognoses than mice with IL-13Rα2-negative cancer. 12 In the later study using a human ovarian cancer mouse model, it was found that IL-13 regulates invasion and metastasis of ovarian cancer cells through IL-13Rα2 via the ERK/AP-1 (extracellular signal regulated kinase/activator protein-1) signaling pathway. 13 Furthermore, as was reported in the earlier study on the pancreatic cancer mouse model, mice with an IL-13Rα2-positive tumor exhibited more metastases to lymph nodes and peritoneum and died earlier than mice with an IL-13Rα2-negative tumor. In a mouse model of human colorectal cancer, silencing of IL-13Rα2 in tumor cells reduced liver homing ability and increased survival compared with mice injected with IL-13Rα2-expressing tumor cells. 9 These studies reveal the role of IL-13Rα2 involving in invasion and metastasis of cancer. In fact, IL-13Rα2 targeting therapies using cytotoxin or monoclonal antibodies have been used to kill tumor cells or prevent cancer metastasis. 2
We first observed the expression of another subunit of IL-13R, IL-13Rα1. Based on our previous 14 and present studies, we have found that IL-13Rα1 is more frequently expressed with high intensity (IRS ≥ 8) than IL-13Rα2 (IRS ≥ 1) in breast cancer (52.3% vs 10.8%). Our studies support that most of the biologic effects of IL-13 are mediated by the STAT6 signaling pathway by its binding to IL-13Rα1 of the type II IL-4R. 2,20 IL-13Rα1 expression was significantly associated with clinicopathological parameters of aggressive phenotypes (presence of lymphovascular invasion and lymph node metastasis, invasive carcinoma, not otherwise specified and micropapillary subtypes, HER2 positivity, and high Ki-67). 14 In this study, although IL-13Rα2 expression was positively associated with hormone receptor status, it was not found to be significantly associated with any other clinicopathological variable. Furthermore, the expressions of IL-13Rα1 and IL-13Rα2 were not significantly related. These observations support that the roles of IL-13Rα1 and IL-13Rα2 during tumor progression involve different signaling pathways. Based on our previous 14 and present studies, we have found that IL-13Rα1 and IL-13Rα2 expression are independent predictors of poor DFS in breast cancer, especially in HER2-negative and luminal breast cancers. In addition, combined expression patterns of IL-13Rα1 and IL-13Rα2 showed more additive effect on patient survival than IL-13Rα1 or IL-13Rα2 alone in luminal breast cancer. Prediction of clinical outcome is still an unresolved issue in breast cancer, therefore, IL-13Rα2, as well as IL-13Rα1, may play a role as a prognostic biomarker in addition to traditional prognosticators in luminal breast cancer.
Summarizing, this study shows IL-13Rα2 expression is significantly associated with poor outcome in patients with luminal breast cancer, and suggests IL-13Rα2 could be a potential prognostic marker for selecting patients with luminal breast cancers likely to follow a clinically aggressive course despite receiving systemic therapy.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Medical Research Center Program (2015R1A5A2009124) through the National Research Foundation of Korea (NRF), Ministry of Science, ICT and Future Planning.
