Abstract
Myeloid-derived suppressor cells are a heterogeneous population of immature myeloid cells that exhibit immunosuppressive activity (they block the proliferation and activity of both T cells and natural killer cells). In addition to their role in suppressing immune responses, myeloid-derived suppressor cells directly stimulate tumor cell proliferation, metastasis, and angiogenesis. In the area of gynecological cancer, increased numbers of circulating myeloid-derived suppressor cells or tumor-infiltrating myeloid-derived suppressor cells have been detected, and the increased frequencies of myeloid-derived suppressor cells are associated with a poor prognosis. Thus, the successful myeloid-derived suppressor cells depletion may hold the key to maximizing existing anti-cancer therapies and improving the prognosis of gynecological cancer. In this review, we summarize current knowledge regarding myeloid-derived suppressor cells biology, clinical significance of myeloid-derived suppressor cells, and the potential myeloid-derived suppressor cells–targeting strategies in gynecological cancer.
Introduction
An increasing body of evidence from murine and human studies suggests that the immunological microenvironment plays a significant role in cancer progression, and treatments that activate the immune system have emerged as a promising cancer treatment strategy. 1 However, as immunotherapy has become a viable treatment for cancer, it has become evident that current immunotherapies face barriers that limit their clinical efficacy. 1
Intratumoral immunosuppression has been described in certain types of cancer, including gynecological cancer.2,3 There are a diverse range of mechanisms underlying the suppression of anti-tumor immunity. However, recent investigations have suggested that myeloid-derived suppressor cells (MDSC) play key roles in immunosuppression of tumors.1,4,5
MDSC are emerging as an important mediator of immunosuppression in tumors, which can blunt responses to immunotherapy.1,4–6 Although MDSC were described as natural suppressor cells in 1970, 7 it took until 2007 for the term MDSC to be established. 8 Since then, an increasing amount of information regarding the biology and clinical significance of MDSC in various pathological conditions has been reported.
MDSC are a heterogeneous population of immature myeloid cells (IMC). They promote tumor growth by inducing tumor cell immunological anergy and tolerance. MDSC exhibit immunosuppressive activity (they block the proliferation and activity of both T and natural killer (NK) cells).1,4–6 In addition, MDSC can directly stimulate tumor cell proliferation, metastasis, and angiogenesis, all of which can limit the potency of current therapeutic interventions.1,4–6 Thus, MDSC are now regarded as an important therapeutic target for cancer treatment as well as a predictive biomarker of treatment outcomes.
In this review, we summarize current knowledge regarding MDSC biology and the role of MDSC in gynecological cancer. We also discuss the utility of the number of MDSC as a predictive marker and highlight how MDSC can be targeted therapeutically in patients with gynecological malignancies.
Definition of MDSC
MDSC are defined as myeloid cells that are distinct from terminally differentiated mature myeloid cells (i.e. macrophages, dendritic cells (DC), or neutrophils). MDSC can be subdivided in two subsets: monocytic myeloid-derived suppressor cells (M-MDSC), which are morphologically and phenotypically similar to monocytes, and polymorphonuclear (PMN-MDSC (also known as granulocytic MDSC), which are morphologically similar to neutrophils.1,4–6
In mice, MDSC were historically characterized by their expression of glutathione reductase (Gr-1) and CD11b myeloid lineage differentiation markers (CD11b+ Gr-1+ cells). However, as Gr-1 is not a single molecule, but rather is a combination of the lymphocyte antigen (Ly)6C and Ly6G molecules, MDSC can be more accurately identified based on these markers (M-MDSC are Ly6Chi Ly6G–, and PMN-MDSC are Ly6Clo Ly6G+).4–6
No definitive markers of human MDSC have been established yet. Although several combinations of antigens have been reported to define human MDSC,1,4–6 it is increasingly accepted that human MDSC are positive for CD11b and CD33, and negative for the human leukocyte antigen-D–related (HLA-DR) and lineage markers (CD3, CD13, CD19, CD56); CD11b+ CD33+ HLA-DR− Lin−. 5 Like murine MDSC, human MDSC can also be divided into two groups: PMN-MDSC, which generally express CD15 (CD14– CD15+ cells), and M-MDSC, which express CD14 (CD14+ CD15– cells).4–6 In addition to the abovementioned markers, it is generally accepted that the assessment of their ability to inhibit T cells is also the “gold standard” for defining MDSC.4–6
Functions of MDSC
Immunosuppressive functions of MDSC
MDSC (both PMN-MDSC and M-MDSC) have the ability to suppress both innate (NK cells) and adaptive (T cells) immune responses (Figure 1). However, whether the mechanisms responsible for immune suppression differ between M-MDSC and PMN-MDSC remain unclear.1,4–6

Functions of MDSC in the TME.
MDSC mainly exert their suppressive effects by removing L-arginine, an essential amino acid for T-cell differentiation, from the tumor microenvironment (TME) via the production of arginase-1 (Arg-1). The depletion of L-arginine subsequently causes the downregulation of CD247 (the ζ-chain of the T-cell receptor) expression in T cells. As CD247 is a subunit of the NK receptors NKp46, NKp30, and TcγIII in NK cells, the depletion of L-arginine also downregulates the expression of CD247 in NK cells, leading to the inhibition of T cell and NK cell proliferation. 9 MDSC also produce reactive oxygen species (ROS) and nitric oxide (NO), which lead to nitration of signaling molecules downstream of the FcγRIIIA, resulting in the inhibition of T cells and NK cells activities. 10 NO produced by MDSC also nitrate STAT1, leading to the diminished interferon response in T cells and NK cells. 11 Moreover, MDSC induce regulatory T-cell (Treg) expansion, which promotes tumor progression via the secretion of interleukin (IL)-10 and transforming growth factor (TGF)-β.1,4–6,12
Non-immune activities of MDSC
In addition to suppressing host immune functions within the TME, MDSC also promote cancer progression by stimulating tumor angiogenesis and enhancing tumor cell invasion and metastasis (Figure 1). These processes are regulated by MDSC-derived mediators, including vascular endothelial growth factor (VEGF), basic fibroblast growth factor (bFGF), Bv8, and matrix metalloproteinase (MMP)-9, all of which are essential for tumor angiogenesis and cancer cell invasion. 13 Moreover, recent reports suggested that MDSC stimulate metastatic activity of cancer cells by facilitating epithelial-to-mesenchymal transition (EMT) or by creating “premetastatic niches.”1,4–6 Importantly, other studies have indicated that MDSC might induce “stemness,” which might be associated with resistance to chemotherapy or radiotherapy, in certain cancers. 2
MDSC generation and recruitment
In healthy individuals, IMCs developed from common myeloid progenitor cells differentiate into mature non-suppressive myeloid cells such as DCs, macrophages or granulocytes.1,4–6,14 Under pathological conditions such as infection, inflammation, or cancer, the differentiation of IMCs was directed away from mature non-suppressive cells into those with suppressive cells, MDSC. Previous studies have suggested that up to a 10-fold increase in the number of MDSC can occur. 14 The mechanisms that are involved in the generation of MDSC and their subsequent recruitment into the TME have not been fully elucidated. However, previous studies have suggested that they involve various factors (Figure 2).

Development of MDSC.
MDSC generation
MDSC are generated in the bone marrow (Figure 2). It has been suggested that MDSC are generated from IMC whose differentiation to mature granulocytes, macrophages, or DC has been blocked.1,4–6,14 The processes responsible for MDSC generation and the inhibition of IMC differentiation into mature myeloid cells involve signaling mediators, such as tumor necrosis factor α (TNF-α), macrophage colony-stimulating factor (M-CSF), granulocyte colony-stimulating factor (G-CSF), granulocyte monocyte colony-stimulating factor (GM-CSF), VEGF, prostaglandin E2 (PGE2), and the ILs (IL-1β, IL-10, and IL-6).
As mentioned previously, under normal conditions, MDSC are mainly generated in the bone marrow. However, in tumor-bearing mice, a previous study showed that circulating MDSC arrest in the spleen, accumulate in the splenic marginal zones and periarteriolar lymphatic sheaths, migrate to the red pulp, and proliferate within the subcapsular red pulp. 15 Thus, the number of MDSC can be reduced by splenectomy.16,17
It has been reported that PMN-MDSC undergo expansion in most patients with solid tumors. However, in certain cancers, such as melanoma, multiple myeloma, and prostate cancer, the M-MDSC population is more prevalent.4,5,18 The mechanisms responsible for such preferential expansion are unknown, but they are probably associated with the differences in the factors that predominate in each type of cancer.
MDSC recruitment
The recruitment of MDSC to the TME is known to be mediated by interactions between tumor-derived chemokines and their receptors on MDSC (Figure 2). Previous studies have shown that tumor-derived chemokines, such as chemokine C-C motif ligand 2 (CCL2), chemokine C-X-C motif ligand 5 (CXCL5), CXCL12, and their receptors (C-C chemokine receptor (CCR)2, CCR5, and C-X-C chemokine receptor (CXCR)4) contribute to MDSC recruitment to tumors.4,5,19 It has been suggested that these factors are not specific to particular types of cancer and have a high degree of redundancy, which helps to maintain a constant supply of MDSC to the TME.
MDSC activation
The precise mechanisms responsible for MDSC activation have not been fully investigated; however, recent studies have suggested that MDSC are activated via the stimulation of various mediators in the TME, including IL-6, IL-1β, TNF-α, interferon (IFN)-γ, PGE2, G-CSF, GM-CSF, M-CSF, and TGF-β. These mediators trigger the activation of signaling pathways, including the Janus kinase/signal transducer and activator of transcription (JAK/STAT) pathway, phosphoinositide 3-kinase/AKT8 virus oncogene cellular homolog/mammalian target of rapamycin (PI3K/AKT/mTOR) pathway, Ras/mitogen-activated protein kinase (RAS/MAPK) pathway, and the SMAD pathway, which are known to play central roles in cell survival, proliferation, and differentiation. Among these signaling molecules, STAT3 seems to play a key role in the activation of MDSC; that is, it stimulates Arg production by binding directly to the Arg-1 promoter. 20 Hypoxia-inducible factor (HIF)-1α has emerged as another important mediator of MDSC functions. According to a recent report, HIF-1α promotes the immunosuppressive activity of tumor-infiltrating MDSC by upregulating inducible nitric oxide synthase (iNOS) and Arg expression and downregulating nicotinamide adenine dinucleotide phosphate (NADPH) oxidase complex expression. 21
The fate of MDSC in tumor-bearing hosts
At tumor sites, M-MDSC can differentiate into immunosuppressive tumor-associated macrophages or DC.22,23 Another study showed that MDSC in mice bearing bone metastases were capable of differentiating into osteoclasts in an NO-dependent manner. 24 These osteoclasts are capable of bone resorption and are involved in the bone remodeling required for bone metastases. Moreover, in a preclinical study, MDSC have been shown to differentiate into fibrocytes and myofibroblasts in the lungs, which suggests that they contribute to the establishment of lung metastases. 25
Clinical significance of MDSC in cancer patients
The frequency of MDSC as a prognostic indicator or a biomarker of tumor progression and responsiveness to therapy
Increased numbers of circulating MDSC have been detected in patients with various types of cancer.1,4,6 Moreover, increased frequencies of tumor-infiltrating MDSC or MDSC in the peripheral blood are associated with the clinical stage, tumor burden, and shorter survival in various types of cancer, including lung cancer, renal cell carcinoma, breast cancer, and colorectal cancer.1,4–6,26 Moreover, circulating MDSC were found to predict worse responses to cytotoxic chemotherapeutic agents.4,26,27 In a study of breast cancer that were treated with neo-adjuvant chemotherapy, although the difference was not statistically significant, the number of circulating PMN-MDSC at the end of chemotherapy was lower in patients who achieved complete or near complete response than in those who did not show responses to chemotherapy. 28 Resistance to the tyrosine kinase inhibitor (TKI) sunitinib was also shown to be associated with high numbers of MDSC in the peripheral blood in cancer patients. 29 Moreover, recent studies have suggested that MDSC are an important predictor of the response to immunotherapy, including ipilimumab, a monoclonal antibody (mAb) against cytotoxic T-lymphocyte–associated protein 4,1,30 and the anti-programmed cell death 1 mAb nivolumab.1,31 Collectively, these studies indicate that an increased MDSC frequency is a valuable predictive biomarker of treatment outcomes.
Utility of the MDSC frequency in the post-treatment follow-up period
Using a mouse model of breast cancer, Crittenden et al. 32 demonstrated that radiotherapy significantly reduced the numbers of PMN-MDSC (CD11b+ Gr-1+ Ly6G+ cells) in the blood. They also found that tumor recurrence from residual disease is correlated with the re-expansion of PMN-MDSC. These results indicate that the number of circulating MDSC might be a useful predictor of recurrence in the post-treatment follow-up period.
Prognostic significance of MDSC in gynecological malignancies
Ovarian cancer
Cui et al. 2 investigated the clinical impact of tumor-infiltrating MDSC using tumor samples obtained from patients with high-grade serous ovarian cancer. They first demonstrated that almost all of the CD33+ cells within the ovarian tumor samples were MDSC. Then, they classified the patients into “CD33+ high” and “CD33+ low” groups according to their CD33+ MDSC density. As a result, they found that an increased number of tumor-infiltrating CD33+ MDSC was significantly associated with shorter overall survival and a reduced disease-free interval. 2 In another study examining the ascites from ovarian cancer patients, Obermajer et al. 33 showed that MDSC are recruited into the ovarian cancer TME through the CXCL12–CXCR4 axis. Moreover, using a murine model of ovarian cancer, Horikawa et al. 34 recently demonstrated that VEGF expression in ovarian cancer stimulated MDSC production, inhibited local immunity, and contributed to tumor progression.
Cervical cancer
Mabuchi et al. found that the number of MDSC (defined as CD11b+ CD33+ HLA-DR− cells) was significantly higher in the peripheral blood of cervical cancer patients than in that of healthy individuals. Importantly, the frequency of MDSC was significantly associated with tumor G-CSF expression. Using a murine model of cervical cancer, they also demonstrated that a high number of tumor-infiltrating MDSC was significantly associated with worse responses to platinum-based chemotherapy 17 and radiotherapy. 16
Endometrial cancer
Using fresh endometrial cancer specimens, Vanderstraeten et al. 35 found that PMN-MDSC (defined as CD11b+ CD14− Lin− HLA-DR− cells) and M-MDSC (defined as CD11b+ CD14+ Lin− HLA-DR− cells) populations expand in endometrial cancer, with PMN-MDSC being the dominant subset.
MDSC as a therapeutic target in cancer patients
Previous preclinical studies have suggested that MDSC-inhibition has been demonstrated to inhibit tumor-growth,2,14,17,34 or enhance the therapeutic efficacy of existing treatments, including chemotherapy 17 and radiotherapy. 16 Moreover, MDSC blockage has also been shown to improve the efficacy of immunotherapies including cancer vaccines 36 as well as immune checkpoint inhibitors nivolumab or ipilimumab.37,38 Thus, MDSC is a promising target in cancer treatment.
Strategies for targeting MDSC
Considering the poor prognosis of cancer patients with increased numbers of MDSC, the development of optimal MDSC-targeting therapies is urgently needed. Given the complicated network of mediators involved in the differentiation, activation, expansion, recruitment, and immunosuppressive activity of MDSC, a variety of different therapeutic strategies have been contrived to target them. These strategies can be broadly classified into eight categories (Figure 3 and Table 1): (a) targeting the membrane proteins of MDSC;39,40 (b) inducing MDSC apoptosis;41–49 (c) deactivating MDSC;20,50–53 (d) promoting the differentiation of MDSC into mature, non-suppressive cells;54,55 (e) blocking the suppressive properties of MDSC;56–58 (f) preventing the recruitment of MDSC from the bone marrow into the TME;59,60 (g) targeting the tumor-derived factors involved in MDSC production;61–66 and (h) inhibiting hematopoiesis.16,17,64,65

Potential MDSC-targeting strategies.
Strategies that therapeutically target MDSC.
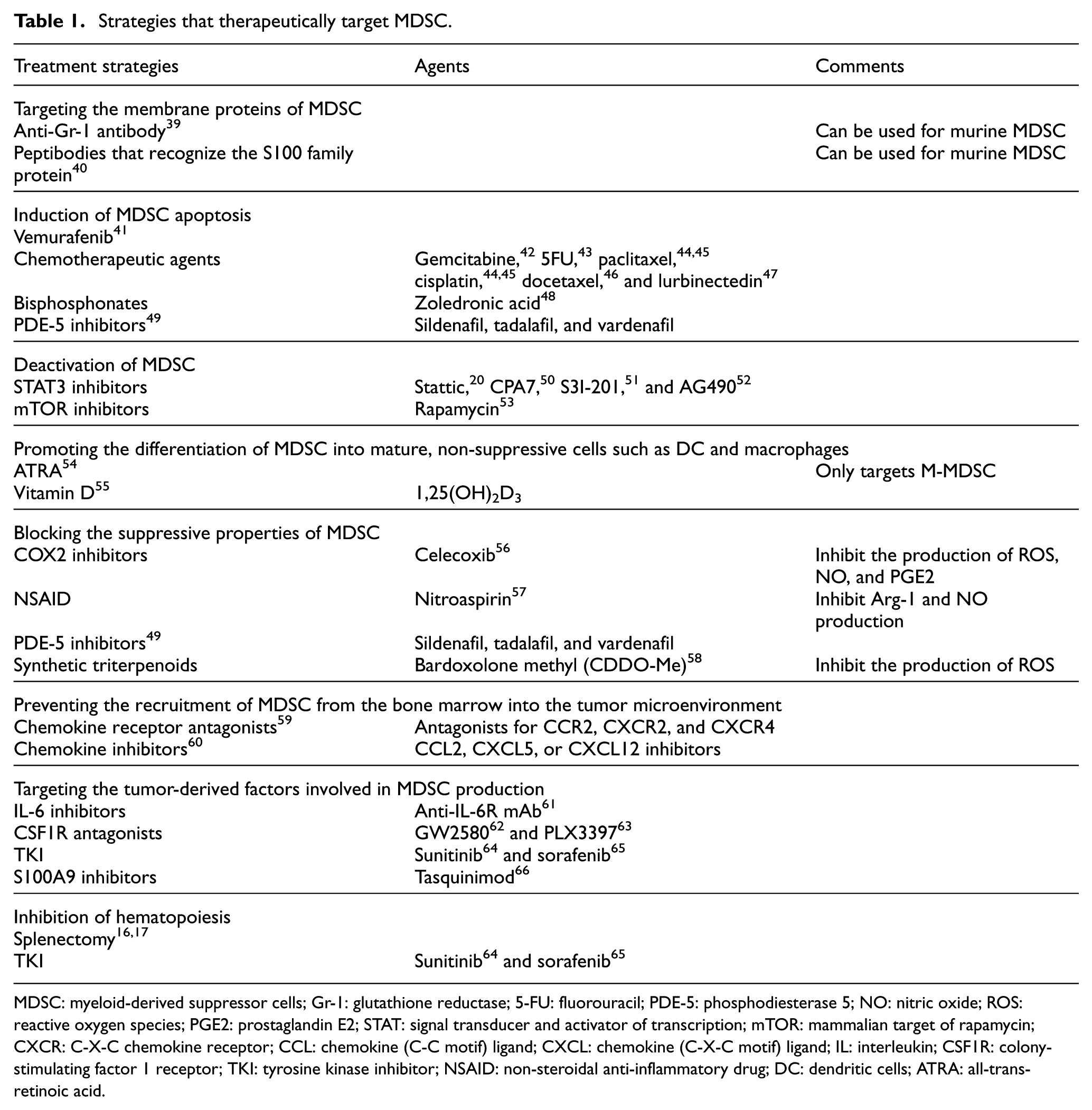
MDSC: myeloid-derived suppressor cells; Gr-1: glutathione reductase; 5-FU: fluorouracil; PDE-5: phosphodiesterase 5; NO: nitric oxide; ROS: reactive oxygen species; PGE2: prostaglandin E2; STAT: signal transducer and activator of transcription; mTOR: mammalian target of rapamycin; CXCR: C-X-C chemokine receptor; CCL: chemokine (C-C motif) ligand; CXCL: chemokine (C-X-C motif) ligand; IL: interleukin; CSF1R: colony-stimulating factor 1 receptor; TKI: tyrosine kinase inhibitor; NSAID: non-steroidal anti-inflammatory drug; DC: dendritic cells; ATRA: all-trans-retinoic acid.
Previous clinical studies have shown that treatments with differentiation agents all-trans-retinoic acid (ATRA) 67 or vitamin D 55 reduced the number of circulating MDSC in lung cancer patients. A phosphodiesterase 5 (PDE-5) inhibitor tadalafil also decreased the number of circulating MDSC as well as the Arg-1 expression in MDSC in head and neck cancer patients. 68 Moreover, conventional chemotherapeutic agent gemcitabine or 5-FU has been shown to reduce circulating MDSC in patients with pancreatic cancer.69,70 These encouraging clinical data as well as the fact that MDSC-targeting agents employed in abovementioned studies are currently available (Food and Drug Administration (FDA)-approved) have facilitated the development of clinical trials.
Currently, phase II studies of the use of chemotherapeutic agents in combination with PDE-5 inhibitors are ongoing in different types of cancer. 71 In a trial recruiting pancreatic cancer patients, the efficacy of CCR2 inhibitor in combination with chemotherapeutic agents are being tested. 72 CXCR2 receptor antagonists are also currently under investigation in prostate cancer patients. 73 Moreover, with an aim to improve the efficacy of immunotherapy, a randomized phase II study is currently comparing the efficacy of ATRA plus ipilimumab versus ipilimumab alone. 74 Hopefully, the next couple of years will bring exciting positive clinical data regarding MDSC-targeting therapies.
Potential of MDSC-targeting therapies for gynecological cancer
Strategies aimed at eliminating MDSC from the TME have shown significant growth-inhibitory activity when used as monotherapies in murine cancer models, including models of cervical and ovarian cancer.2,16,17,34 Moreover, in the setting of combination therapy, MDSC blockade has been demonstrated to enhance the therapeutic efficacy of existing treatments, including chemotherapy 17 and radiotherapy, 16 in a mouse model of cervical cancer. In all studies, the anti-Gr-1 antibody was used to eliminate MDSC from TME. However, two previous studies have shown that splenectomy in mice can reduce the number of tumor-infiltrating MDSC, which induces tumor regression and enhances the anti-tumor effects of chemotherapy and radiotherapy.16,17
Future directions
Development of specific inhibitors of human MDSC
Currently, no specific inhibitors of human MDSC are available. This is mainly due to the highly heterogeneous nature of human MDSC. This is an important issue. In murine studies, the anti-Gr-1 antibody has been widely used to eliminate MDSC from TME. However, this antibody also targets neutrophils; thus, it lacks the necessary specificity. 75 Moreover, due to the absence of a Gr-1 homolog in humans, anti-Gr-1 antibodies cannot be used in the clinical setting. A newly engineered peptibody consisting of S100A9-derived peptides conjugated to antibody Fc fragments successfully eliminated MDSC from various murine tumor models. 40 However, the specificity of this agent and its applicability to human MDSC remain unknown. Until specific inhibitors of human MDSC will be developed, drug repurposing (i.e. the use of gemcitabine, 5-FU, or PDE-5 inhibitor as an MDSC-targeting agent) may be reasonable.
Development of new experimental models
In preclinical studies of MDSC-targeting agents, researchers most commonly use splenic MDSC derived from tumor-bearing animals instead of tumor-infiltrating MDSC because splenic MDSC can be obtained in large numbers. However, as it has been suggested that splenic MDSC might be phenotypically and functionally different from tumor-infiltrating MDSC,4–6 it would be better if novel MDSC-targeting agents were evaluated using tumor-infiltrating rather than splenic MDSC. Thus, the establishment of new experimental models, such as an in vitro culture system for obtaining MDSC that closely resemble tumor-derived MDSC, is needed.
Identification of subgroups of patients who might benefit from MDSC-targeting therapy
Previous studies have suggested that not all cancer patients display increased numbers of MDSC in their TME. In cervical cancer patients, increased numbers of MDSC were only observed in patients who displayed tumor-related leukocytosis (TRL).16,17 Moreover, increased numbers of MDSC were detected in cervical cancer patients whose tumors overexpressed G-CSF.16,17 Consistent with these findings, MDSC-targeting treatments, such as anti-Gr-1 antibody treatment or splenectomy, only had significant anti-tumor effects in a mouse model of G-CSF-expressing, TRL-positive cervical cancer.16,17 As far as we know, these are the only studies that have tried to identify subgroups of cancer patients with increased numbers of MDSC in their TME. Thus, biomarkers of such cancer patients need to be developed, as this would enable physicians to easily identify patients who might benefit from MDSC-targeting therapy.
Conclusion
MDSC populations expand in gynecological cancer patients and play integral roles in disease progression. In order to inhibit their tumor-promoting effects, the efficacy of MDSC-targeting therapies (either as monotherapies or in combination with conventional treatments, including chemotherapy, radiotherapy, or anti-cancer immunotherapeutics) against gynecological cancer is currently being evaluated preclinically. We consider that increasing our understanding of MDSC biology will further aid the development of optimal MDSC-targeting therapies for gynecological cancer. Successful MDSC depletion holds the key to maximizing existing anti-cancer therapies and improving the prognosis of gynecological cancer patients.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by grants-in-aid for General Scientific Research T17K16849 and T17K112760 from the Ministry of Education, Culture, Sports, Science, and Technology of Japan.
