Abstract
Accurate staging of rectal cancer remains essential for optimal patient selection for combined modality treatment, including radiotherapy, chemotherapy and surgery. We aimed at examining the correlation of cell free DNA with the pathologic stage and subsequent risk of recurrence for patients with locally advanced rectal cancer undergoing preoperative chemoradiation. We examined 75 patients with locally advanced rectal cancer receiving preoperative chemoradiation. Blood samples for translational use were drawn prior to rectal surgery. The level of cell free DNA was quantified by digital droplet PCR and expressed as copy number of beta 2 microglobulin. We found a median level of cell free DNA in the AJCC stages I-III of 3100, 8300, and 10,700 copies/mL respectively. For patients with 12 sampled lymph nodes or above, the median level of cell free DNA were 2400 copies/mL and 4400 copies/mL (p = 0.04) for node negative and node positive disease respectively. The median follow-up was 39 months and 11 recurrences were detected (15%). The median level for patients with recurrent disease was 13,000 copies/mL compared to 5200 copies/mL for non-recurrent patients (p = 0.08). We have demonstrated a correlation between the level of total cell free DNA and the pathologic stage and nodal involvement. Furthermore, we have found a trend towards a correlation with the risk of recurrence following resection of localized rectal cancer.
Introduction
Rectal cancer remains a major global health challenge comprising one-third of all colorectal cancers (CRC), thus expected to account for more than 400,000 new cases yearly worldwide. 1 Surgery continues to be the mainstay of treatment for localized rectal cancer with various approaches depending on tumor stage and position in rectum. 2 Preoperative chemoradiotheraphy (CRT), either as long-course concomitant chemoradiotherapy (LCRT) or as short-course radiotherapy (SCRT), has been established as a standard of care for patients presenting with locally advanced rectal cancer (LARC), reducing the risk of pelvic recurrence.3–5
All patients with rectal cancer will undergo a thorough staging procedure including magnetic resonance imaging (MRI) providing accuracy regarding the T-stage and precise visualization of the mesorectal fascia. 6 Assessment of the lymph node status is of major importance both due to the profound prognostic value of nodal involvement and to the fact that the presence of lymph node metastases might alter the treatment strategy. All imaging modalities, however, have limitations especially in terms of nodal staging, 7 so new additional tools are needed. Numerous blood-borne markers have been examined in rectal cancer including carcinoembryonic antigen (CEA) and several inflammatory proteins, failing to contribute to more accurate staging despite holding prognostic value.8,9
Circulating nucleic acids in the bloodstream was first recognized by Mandel and Metasis 10 in 1948 with a further emphasis on cancer patients by Leon et al. 11 in 1977. Today, cell-free DNA (cfDNA) is known to be a mixture of normal DNA fragments and cancer-specific DNA segments with tumor-specific mutations. cfDNA is detectable in the bloodstream in both malignant and non-malignant diseases with a higher concentration in patients with CRC compared to patients with other medical diseases. 12 Quantitation of the total level of cfDNA has shown promising utility in multiple aspects of CRC treatment guidance and is revealed to be a prognostic marker in the metastatic setting, as demonstrated in a recent meta-analysis. 13
Three studies14–16 have found a correlation between the response of preoperative CRT of LARC and cfDNA, suggesting a potential role of cfDNA for therapy monitoring and response assessment. In this study, we aimed at exploring the correlation between the preoperative level of cfDNA and the corresponding pathologic stage and subsequent risk of recurrence for patients with LARC undergoing preoperative CRT.
Material and methods
Patient cohort
Patients with histologically verified locally advanced rectal adenocarcinoma aimed for surgery were collected with correlating biobanked blood samples at the Department of Surgery, Sahlgrenska University Hospital, Gothenburg, Sweden. All patients underwent preoperative treatment with either SCRT (25Gy/5F/5w) or LCRT (52Gy/26F/5w + capecitabine 850 mg/m2 twice a day (BID) with treatment planning and delivery according to institutional guidelines. Patients with primary metastatic disease were excluded. All patients underwent routine preoperative staging including a computed tomography (CT) scan, a rectoscopic exam with biopsy, and a pelvic MRI.
Following neoadjuvant therapy, standardized radical surgical procedures were performed, including rectal resection (n = 26), rectal amputation (n = 42), Hartman procedure (n = 6), and proctocolectomy (n = 1). Blood samples for translational use were taken in the preoperative setting.
LARC was defined as a tumor within 15 cm of the anal verge by rigid proctoscopy and by imaging either a T4 or T3 tumor with close proximity to the circumferential margin (CRM < 5 mm). Decision of preoperative CRT and selection between SCRT and LCRT were taken by multidisciplinary tumor board.
Postoperative specimens were reviewed by pathologist for final staging according to the American Joint Committee on Cancer (AJCC) 17 and the notation ypT denotes the pathologic stage following preoperative CRT.
For evaluation of the lymph node status, we only considered patients with 12 or more sampled lymph nodes, whereas national guidelines considered adequate sampled. 18
The regional ethical review board in Gothenburg approved the study and informed consent was obtained from all patients.
Sample collection and laboratory work
Blood samples were drawn in ethylenediaminetetraacetic acid (EDTA) tubes in the preoperative setting. A volume of 3 mL of the blood was carefully placed on top of 3 mL Histopaque 1077 solution (Sigma-Aldrich, Inc., USA) in a centrifuge tube. The plasma was recovered after centrifugation for 30 min at 400 g. Plasma was obtained and centrifuged within 30 min and stored at −80°C until use. Circulating nucleic acids were purified from 1 mL of plasma using the QIAamp Circulating Nucleic Acid Kit (Qiagen, Germany) according to the manufacturer’s instructions. The quantitative level of cfDNA was measured by droplet digital polymerase chain reaction (PCR), targeting the beta-2 microglobulin (B2M) gene and expressed as copies/mL by an in-house developed assay. B2M is a housekeeping gene not involved in the carcinogenic process and is suitable as a target gene for cfDNA quantification and previously used by our research group. 19 Samples with high degree of contamination from B-lymphocytes were excluded. 20
Statistics
We determined the median survival of the entire cohort by Kaplan–Meier methodology calculating the survival from the date of surgery. With low number of death (n = 2), we did not perform further survival analysis.
Based on the few cases of recurrent disease (n = 11), we do not find the data suitable for multivariate analysis in an attempt to establish a correlation between cfDNA and recurrence.
The concentration of total cfDNA was expressed as median values and 95% confidence interval (CI), and the comparison of cfDNA between groups was analyzed by the Mann–Whitney U test. Calculations were performed by NCSS statistical software 2007 v.07.1.5 (www.nssc.com). The p value was considered significant when <0.05.
Results
A total of 75 patients were selected from the database and translational blood samples available from 74 patients. Patients’ characteristics and corresponding cfDNA levels are presented in Table 1. For the AJCC stages I–III, the corresponding levels of gene copy numbers were 3100 copies/mL (95% CI: 2300–6300), 8300 copies/mL (95% CI: 3600–12,000), and 10,700 copies/mL (95% CI: 4300–19,600), respectively. Data are presented in a box-plot in Figure 1. A comparison between stages revealed significant differences for stage I versus II (p = 0.01) and stages I versus III (p = 0.02), while only a trend toward a higher cfDNA level in stage II versus III (p = 0.30) was observed. The postoperative T stages (ypT) did not show significant differences in cfDNA values.
Patient characteristics.
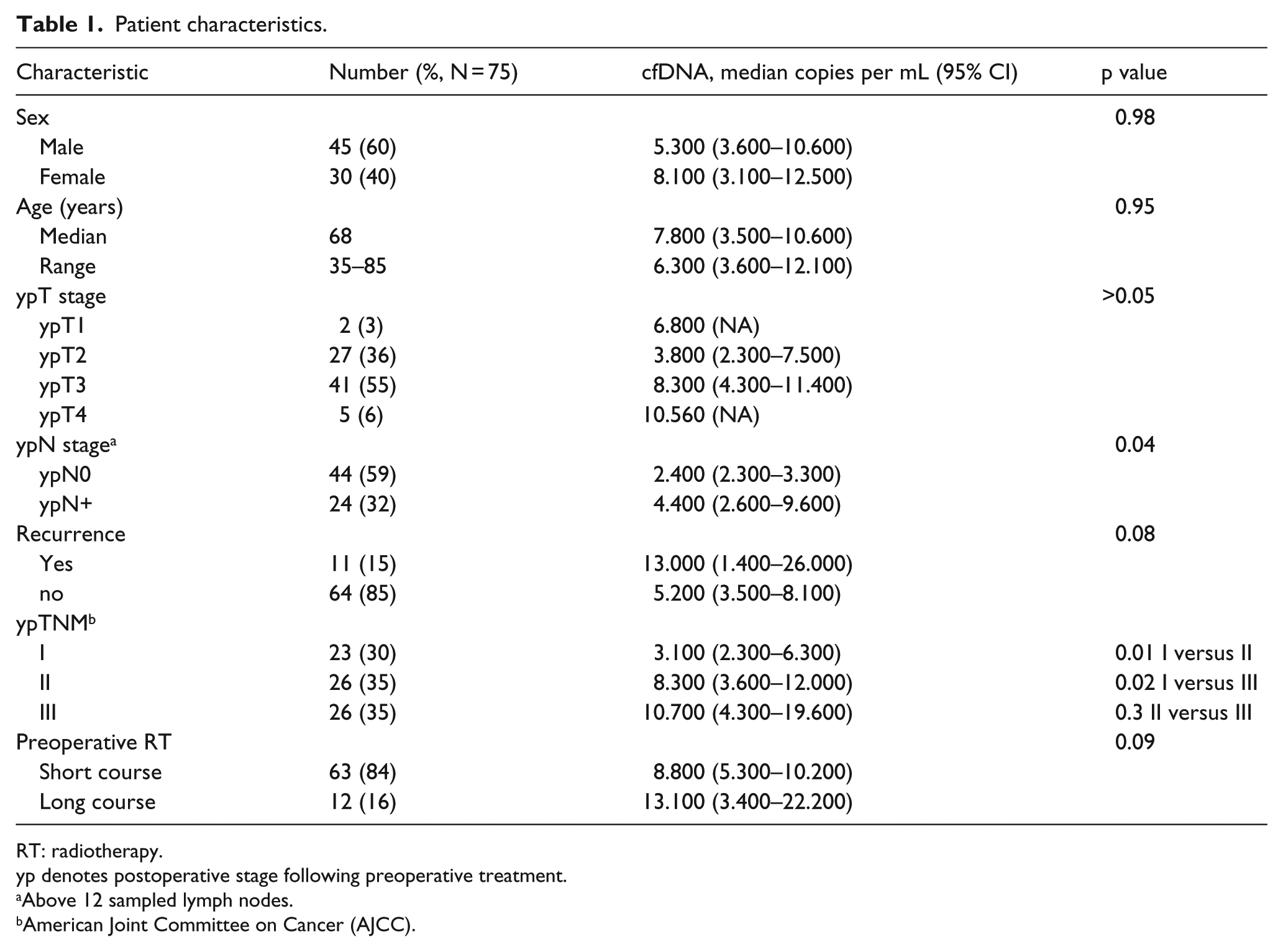
RT: radiotherapy.
yp denotes postoperative stage following preoperative treatment.
Above 12 sampled lymph nodes.
American Joint Committee on Cancer (AJCC).

cfDNA levels in AJCC stages I–III depicted as box plots show the median, upper, and lower quartiles. Data revealed significant differences for the median value of cfDNA between the stages I (n = 23) and II (n = 26; p = 0.01) and stages I and III (n = 26; p = 0.02). Between stages II and III, only a tendency for correlation was seen (p = 0.3).
For patients with 12 or more sampled lymph nodes, postoperative pathology revealed node-positive disease (N+) in 24 patients (32%) and node negative (N0) in 44 (59%). For N0 and N+ stages, the median levels of cfDNA were 2400 copies/mL (95% CI: 2300–3300) and 4400 copies/mL (95% CI: 2600–9600), respectively, (p = 0.04) (Figure 2).

Box plot of nodal involvement and cfDNA levels for patients with 12 or above sampled nodes (n = 68). The median value and upper and lower quartiles are depicted. For node-positive patients (N+; n = 24), a significant higher level of cfDNA was seen compared to the node-negative patients (N–; n = 44; p = 0.04).
With a median follow-up of 39 months, 11 patients (15%) developed recurrent disease. All relapses were distant metastases and no local recurrences. The median level of cfDNA for patients with recurrent disease was 13,000 copies/mL (95% CI: 1400–26000) compared to 5200 copies/mL (95% CI: 3500–8100) for non-recurrent patients (p = 0.08; Figure 3). By the end of follow-up, two patients had died and the median survival of the entire cohort was 98 months (95% CI: 83–100).

cfDNA levels in patients developing recurrent disease (n = 11) and non-recurrent patients (n = 64) depicted as box plots. The median value of cfDNA for patients developing recurrence was 13,000 copies/mL compared to 5200 copies/mL in non-recurrent patients (p = 0.08).
Discussion
LARC is a challenging disease requiring a high degree of multidisciplinary approach for optimal outcome. Preoperative radiotherapy, either LCRT or SCRT, has significantly reduced the risk of local recurrence compared to surgery alone, but has little, if any, impact on the overall survival. 21 The hallmark of optimal patient selection in the preoperative setting remains accurate staging. A pelvic MRI remains the cornerstone of staging rectal cancer supplemented by a clinical exam, endoscopy, and laboratory investigations as recommended by international guidelines.22,23
The major benefit of MRI is precise judgement of the mesorectal fascia, critically for patients scheduled for a total mesorectal excision (TME) as demonstrated in the MERCURY trial examining 408 patients undergoing surgery for a localized rectal cancer. In this study, MRI had a specificity of 92% for the prediction of a clear CRM. 24
Despite the advances in modern imaging, new tools contributing to decision making regarding the therapeutic approach for LARC are needed. Especially for the nodal stage, MRI has limitations, with the size of the mesorectal lymph nodes not being a valid criterion for likelihood of malignancy.25–27
The importance of accurate lymph node staging is well established with lymph node status being the single most important prognostic factor for long-term survival. 28 In the current multidisciplinary treatment for rectal cancer, the preoperative approach could be further personalized by the introduction of reliable biomarkers.
Quantity of cfDNA is gaining wide interest in various scenarios of CRC treatment with a recent systematic review and meta-analysis demonstrating a pronounced prognostic value in the metastatic setting. 13 Three clinical studies14–16 on patients with LARC undergoing preoperative LCRT have examined the correlation of cfDNA and response to treatment, demonstrating a prognostic and predictive potential, but not addressing the issue of stage-dependent variations of cfDNA.
Final staging of rectal cancer is done by pathologic examination postoperative. The increasing interest in non-operative management of selected cases of rectal cancer, treated with definitive chemoradiotherapy and organ preservation,29,30 yields further emphasis on accurate preoperative assessment of disease burden. All imaging modalities have limitations in predicting complete response in restaging patients with rectal cancer following CRT before surgery. 31
In our study, we found a significant correlation between the preoperative level of cfDNA, the nodal involvement, and the pathologic AJCC stage for patients radically operated for LARC following SCRT or LCRT. Furthermore, we found a trend toward a correlation between the level of cfDNA and the risk of recurrence, although this was not significant (p = 0.08). A tendency for correlation between cfDNA and recurrence in our analysis will be biased by a possible influence of other variables with known negative impact, for example, nodal stage.
We did not find a significant difference in cfDNA levels between the postoperative ypT1 and ypT4 stages. The majority of patients were ypT2 or ypT3 (n = 68 (91%)), while six patients (5%) remained ypT4 and only two patients (3%) were regressed to ypT1 following the preoperative CRT. Despite the lack of correlation between the ypT stage and cfDNA, this translates into a significant difference between the AJCC stages I and II/III, respectively.
Several blood-borne markers have been tested in localized rectal cancer, with CEA being the most consistently reported to have prognostic utility, both for response to preoperative treatment and for risk of recurrence.32,33 However, a study from China 9 found no correlation between preoperative CEA level and the Duke’s stages of rectal cancer, in contrast to colonic cancer, for a total of 240 CRC cases.
For patients with LARC, several new molecular biomarkers have been investigated for a potential predictive and prognostic value, as recently reviewed. 34 In surplus to the total level of cfDNA, this include microRNA, circulating tumor cells (CTC), and circulating tumor-specific DNA (ctDNA). None of these have emerged into daily clinical practice and comparative trials are lacking between these new biological markers and traditional evaluation of disease by imaging.
Only three studies14–16 have examined the level of total cfDNA and patients with LARC, focusing on cfDNA and response to the preoperative CRT. The first study in this setting was from Zitt et al. 14 in 2008 evaluating 26 patients undergoing preoperative CRT. They demonstrated a significant decrease in the median level of cfDNA for patients responding to CRT. This finding was confirmed by Agostine et al. 15 and Sun et al., 16 the latter also found decreased ctDNA (KRAS mutations) in 34 patients receiving preoperative CRT.
Compared to these studies, we have demonstrated a stage-dependent variation in cfDNA for patients with LARC and not addressing the kinetics of cfDNA during the multimodal treatment. To the best of our knowledge, this is the first study exploring the correlation between the stage of LARC and cfDNA, supporting the hypothesis of a potential role of cfDNA measurements in this group of patients. We have measured the quantity of the total cfDNA, hence both the DNA harboring tumor-specific mutations and the DNA released from surrounding tissue. The tumor-specific DNA has been the focus of several other CRC research areas,35,36 although the more feasible laboratory investigations in total cfDNA analysis favor this approach. Furthermore, the total cfDNA is expected to be a measurable parameter in the majority of patients, thus with applicability for patients with no detectable mutations in the blood.
The advantage of tumor-specific DNA analysis involves detection of potential genetic alterations with therapeutic consequences, for example, emerging RAS mutations in the bloodstream indicates development of resistance to anti–epidermal growth factor receptor (EGFR) treatment37,38 in the metastatic setting. Circulating tumor DNA (ctDNA) might provide more accurate picture of the current mutational status than archival tissue. 39 The utility of ctDNA was recently tested in four patients with LARC undergoing preoperative LCRT, demonstrating a correlation with treatment outcome and supporting a role for the concept of liquid biopsies for rectal cancer. 40
This study provides new insights into a possible use of cfDNA for more personalized management of rectal cancer and thereby enhancements of patient care. As a limitation, we have no sequential blood samples during the treatment course and therefore unable to monitor the dynamics of cfDNA during the treatment course and follow-up.
In conclusion, we have demonstrated a correlation between the preoperative level of cfDNA and pathologic stage of rectal cancer for patients undergoing preoperative CRT, thus suggesting a new possible applicability of cfDNA.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received grant from the Danish Cancer Society and the Novo Nordisk Foundation.
