Abstract
M2-like tumor-associated macrophages promote breast tumor growth and survival and may migrate into the peripheral blood. However, the frequency of circulating M2-like monocytes in the peripheral blood of breast cancer patients has not been clarified. The objective of this study was to determine the percentages of circulating M2-like monocytes in patients with breast cancer.
Immunofluorescence staining for CD68 and CD163 was performed to detect M2-like macrophages in pathological tissues. Flow cytometry was used to assess the frequencies of circulating CD14+CD163+/CD14+CD204+/CD14+CD163+CD204+ M2-like monocytes in 99 breast cancer patients, 56 patients with benign breast disease, and 60 healthy controls. Receiver operating characteristic curve analysis was used to compare the diagnostic values of circulating M2-like monocytes, carcinoembryonic antigen, and cancer antigen 15-3. The associations among circulating M2-like monocytes and clinical breast cancer parameters were analyzed.
The number of CD68+CD163+ M2-like macrophages was significantly higher in breast cancer tissues than in benign tissues. In the peripheral blood, CD14+CD163+/CD14+CD204+/CD14+CD163+CD204+ M2-like monocytes were elevated in breast cancer patients compared with normal controls and patients with benign breast disease. The area under the receiver operating curve for circulating CD14+CD163+CD204+ M2-like monocytes was 0.888 (95% confidence interval: 0.839–0.936), a value higher than those for carcinoembryonic antigen and cancer antigen 15-3. High frequencies of circulating CD14+CD204+ and CD14+CD163+CD204+ M2-like monocytes were associated with tumor–node–metastasis stage, lymph node metastasis, histological differentiation, and estrogen receptor expression.
Circulating M2-like monocytes may serve as a diagnostic biomarker in breast cancer and have a potential role in reflecting breast cancer progression.
Introduction
Breast cancer (BC) is one of the leading causes of death among females with malignant tumor diseases. 1 Although a significant proportion of BC patients have been benefitted from the development of effective treatments and early detection, morbidity and mortality remain high. Early detection together with novel treatment strategies is important for increasing the survival of BC patients.2,3 Although serum-based tumor markers, such as carcinoembryonic antigen (CEA) and cancer antigen 15-3 (CA15-3), are commonly used for the detection of BCs, the sensitivity and specificity of these parameters remain questionable. 4 Therefore, it is necessary to identify reliable diagnostic and prognostic biomarkers to guide decision-making during BC treatment.
Circulating monocytes, a heterogeneous population of cells, may migrate into tissues and differentiate into steady tissue-specific macrophages. 5 Tumors attract peripheral blood mononuclear cells (PBMCs) via chemoattractants, and monocytes continuously penetrate the tumor microenvironment, where tumor-derived chemokines and factors released by other infiltrates induce macrophages to become tumor-associated macrophages (TAMs).6,7 Mirroring the Th1 and Th2 dichotomy, polarized macrophages can be generally divided into the following two states: (1) classically activated macrophages (M1-like), which promote inflammation and have high tumor cytotoxicity and immuno-stimulatory functions, and (2) alternatively activated macrophages (M2-like), which suppress inflammation and facilitate angiogenesis, tissue remodeling, and repair.8,9 On the basis of their phenotype, function, and release of inflammatory cytokines, TAMs resemble M2-like macrophages. 10 In BC, M2-like TAMs promote angiogenesis and extracellular matrix breakdown and remodeling, thus enabling breast tumor cells to intravasate into the peripheral blood.11,12 Furthermore, communicating and cooperating with breast tumor cells, M2-like TAMs also migrate into peripheral circulation by intravasating across intratumor capillary barriers.12,13 Previous data have suggested that circulating monocytic cells are involved in tumor cell invasiveness, motility, and metastasis potential.11–15 Thus, circulating M2-like monocytes might be a subset of disseminated TAMs that derived from BC tissue, which can facilitate breast tumor metastasis.12,13
In recent years, some studies have reported that the numbers of circulating M2-like monocytes are elevated in some cancer, such as lung cancer, 16 colorectal cancer, 17 and classical Hodgkin’s lymphoma. 18 These findings highlight the potential of circulating M2-like monocytes as a new tumor biomarker and suggest that circulating M2-like monocytes may be a noninvasive marker for differentiating between benign and malignant tumors and for reflecting tumor progression. To our knowledge, no study of circulating M2-like monocytes has been conducted in BC. Therefore, in this study, we sought to determine whether M2-like monocyte numbers in the peripheral blood of BC patients might be used as a novel biomarker.
Materials and methods
Subjects and samples
Peripheral blood samples were collected from 60 healthy donors, 56 patients with benign breast disease, and 99 patients with BC. The study was approved by the Ethics Committee of Shanghai Jiao Tong University and was performed in accordance with the revised Declaration of Helsinki, 2013. Informed consent was acquired from all individuals. All of the healthy subjects were not taking any medications and did not have cancer, infectious diseases, autoimmune diseases, or other conditions. The enrolled patients were also not taking medication and were histopathologically diagnosed for the first time at the Shanghai Cancer Center. The clinical details of the patients and healthy controls are shown in Table 1.
The baseline demographic and clinical characteristics of subjects.

CEA: carcinoembryonic antigen; CA15-3: cancer antigen 15-3.
p < 0.05 versus the malignant group.
Flow cytometry analysis
Fresh ethylenediaminetetraacetic acid (EDTA)-anticoagulated whole blood samples (100 µL) were labeled with the following anti-human monoclonal antibodies: 7.5 µL FITC-anti-CD14 (Beckman Coulter, France), 4.5 µL APC-anti-CD163 (BioLegend, USA), and 6 µL PE-anti-CD204 (R&D Systems, USA). The samples were incubated for 15 min at room temperature away from light. Subsequently, 2 mL of FACS lysing solution were added to the erythrocytes. Matched isotype controls were used for setting compensation and ascertaining correct gating. The antibody-labeled monocytes were identified using a FACS Navios flow cytometer (Beckman Coulter, USA) and the data were analyzed with FlowJo software (Tree Star, Inc., USA).
Immunofluorescence assay
The formalin-fixed, paraffin-embedded biopsy specimens of BC and benign breast disease were obtained from Shanghai Jiao Tong University Affiliated Sixth People’s Hospital. Sections of 5 µm thickness were cut from the blocks and mounted on silanized slides, which were then deparaffinized and subjected to antigen retrieval for double immunofluorescent (IF) staining as previously described.19,20 The specimens were incubated in blocking solution overnight at 4°C with 1% bovine serum albumin (BSA) in phosphate-buffered saline (PBS). The blocking solution was aspirated, and mouse anti-CD68 (Abcam, UK) primary antibodies were added in blocking solution for 1–2 h at room temperature. The slides were rinsed three times with PBS and then incubated with Alexa Fluor-488 anti-mouse IgG (Abcam) for 1 h at room temperature. After the slides were washed with PBS, rabbit anti-CD163 (Abcam) was added in blocking solution for an additional 4 h, and the slides were subsequently washed. Alexa Fluor-647 anti-rabbit IgG (Abcam) was then added to each slide and incubated for 30 min at room temperature. As a negative control, no primary antibody was added. IF images were acquired using a Confocal Laser Scanning Microscope (Nikon, Japan) to identify the double-positive region of interest.
Plasma concentrations of CEA and CA15-3
All blood samples were centrifuged at 1000g for 5 min at 4°C to collect plasma, which was transferred into tubes and stored at −20°C for further experimentation. CEA (Architect i2000SR; Abbott, USA) and CA15-3 (Cobas e601; Roche, Swit) levels were measured in all the plasma samples using a chemiluminescence method as described in the instructions.
Statistical analysis
Normally distributed data are expressed as the mean ± standard deviation (SD) and were analyzed using an unpaired t-test or one-way analysis of variance (ANOVA) with Tukey’s multiple comparison test. Non-normally distributed values, presented as a median (P25–P75), were analyzed using the Mann–Whitney U test or the Kruskal–Wallis nonparametric test. The correlations among biomarkers were assessed using the Spearman rank correlation test. Receiver operating curve (ROC) analysis was used to evaluate diagnostic efficiency. Logistic regression was performed to acquire the sensitivity and specificity of the optimum combination of circulating M2-like monocytes, CEA, and CA15-3. All of the statistical analyses and figures were performed with SPSS 20.0 (IBM, USA) and GraphPad Prism version 5 (GraphPad Software, Inc., USA) software, and two-sided p values below 0.05 were considered significant.
Results
M2-like macrophages accumulate in BC tissues
To investigate the expression of M2-like macrophages in biopsy specimens of BC and benign breast disease, we used CD68 as a pan-macrophage marker and CD163 as a M2-like macrophage marker. Thus, M2-like macrophages were defined as CD68+CD163+ cells. Our results suggested that the majority of macrophages that infiltrated into BC tissue were M2-like, and there were more of these cells in BC tissues than in benign tissues (Figure 1).

M2-like macrophages in breast cancer and benign breast disease. Immunofluorescence assays were performed as described in “Materials and methods” section to confirm the presence of CD68+CD163+ M2-like macrophages in subjects with breast cancer and benign disease. M2-like macrophages were analyzed on the basis of nuclei (blue), CD68 (green, primarily in cytoplasm), and CD163 (red, cell-surface marker). Confocal planes are shown individually and after merging. Several CD68+CD163+ M2-like macrophages (arrows) were observed within the biopsy specimens from subjects with (a) benign disease and (b) breast cancer. Representative images are shown of tissues from patients with breast cancer (n = 5) and benign disease (n = 5).
Circulating M2-like monocytes are increased in BC patients
The clinical baseline characteristics of the three groups (99 BC patients, 56 patients with benign breast disease, and 60 healthy controls) are summarized in Table 1. As shown in Table 1, there were no significant differences in the distribution of age, white blood cell (WBC) counts, or monocyte counts among BC, benign disease, and control groups.
The number of circulating M2-like monocytes from the peripheral blood of all participants was examined using flow cytometry. A generally accepted definition of M2-like monocytes is CD14+CD163+ cells that express CD204 or CD206. In our study, CD14, CD163, and CD204 were used to identify M2-like tumor-associated monocytes (Figure 2(a)). As expected, the percentages of circulating CD14+CD163+ (40.93 ± 2.38), CD14+CD204+ (10.82 ± 2.83), and CD14+CD163+CD204+ (4.71 ± 1.69) monocytes from BC patients were significantly higher than those in both healthy controls (37.66 ± 2.22, 6.53 ± 2.48, and 2.45 ± 1.05, respectively) and benign patients (38.54 ± 2.44, 7.49 ± 2.63, and 2.90 ± 1.23, respectively; Figure 2(b)–(d)). However, there was no significant difference in the frequency of CD14+CD163+/CD14+CD204+/CD14+CD163+CD204+ monocytes between healthy controls and benign group (Figure 2(b)–(d)). Therefore, our results suggested that the percentages of circulating CD14+CD163+/CD14+CD204+/CD14+CD163+CD204+ M2-like monocytes were significantly increased in BC patients.

Characterization of circulating M2-like monocytes in healthy controls, benign patients, and breast cancer patients. (a) FSCmid/SSCmid fractions primarily include monocytes, which were initially gated on CD14+ cells. Subsequently, M2-like monocytes were gated on CD14+ monocytes and the percentages of CD14+CD163+, CD14+CD204+, and CD14+CD163+CD204+ monocytes were determined. (b) The percentages of circulating CD14+CD163+ M2-like monocytes in patients with breast cancer, benign disease, and healthy controls. (c) The percentages of circulating CD14+CD204+ M2-like monocytes. (d) The percentages of CD14+CD163+CD204+ M2-like monocytes.
Correlations among circulating M2-like monocytes, CEA, and CA15-3
Spearman rank correlation tests were performed to assess correlations among circulating M2-like monocyte, CEA, and CA15-3 in the control, benign, and BC groups (Table 2). The results showed that CA15-3 levels were weakly correlated with CEA levels in the BC group (Spearman r = 0.249, p < 0.05). However, there were no significant correlations between circulating M2-like monocytes and CEA/CA15-3 in any group.
Spearman’s rank correlation coefficients among three biomarkers for distinguishing normal control, benign disease, and breast cancer.

Circulating CD14+CD163+CD204+ M2-like monocytes.
p < 0.05.
Circulating M2-like monocytes have higher diagnostic values for BC than CEA and CA15-3
We used ROC analysis to evaluate the diagnostic value of circulating CD14+CD163+/CD14+CD204+/CD14+CD163+CD204+ M2-like monocytes and then performed a comparison with the diagnostic values for CEA and CA15-3. When comparing BC patients with healthy controls, we found that the areas under the ROC curves (AUCs) for circulating CD14+CD163+/CD14+CD204+/CD14+CD163+CD204+ monocytes were 0.835, 0.868, and 0.888, respectively. These values were higher than those of CEA (0.545) and CA15-3 (0.623; Table 3 and Figure 3(a)). Additionally, ROC curves were used to compare the utility of the percentages of circulating CD14+CD163+/CD14+CD204+/CD14+CD163+CD204+ monocytes in differentiating BC from benign disease. The results indicated that the AUCs were 0.745, 0.804, and 0.816, respectively. The AUCs of circulating CD14+CD204+/CD14+CD163+CD204+ monocytes were also higher than those for CEA (0.605) and CA15-3 (0.649; Table 3 and Figure 3(b)).
Diagnostic values of circulating M2-like monocytes, CEA, and CA15-3 for breast cancer.
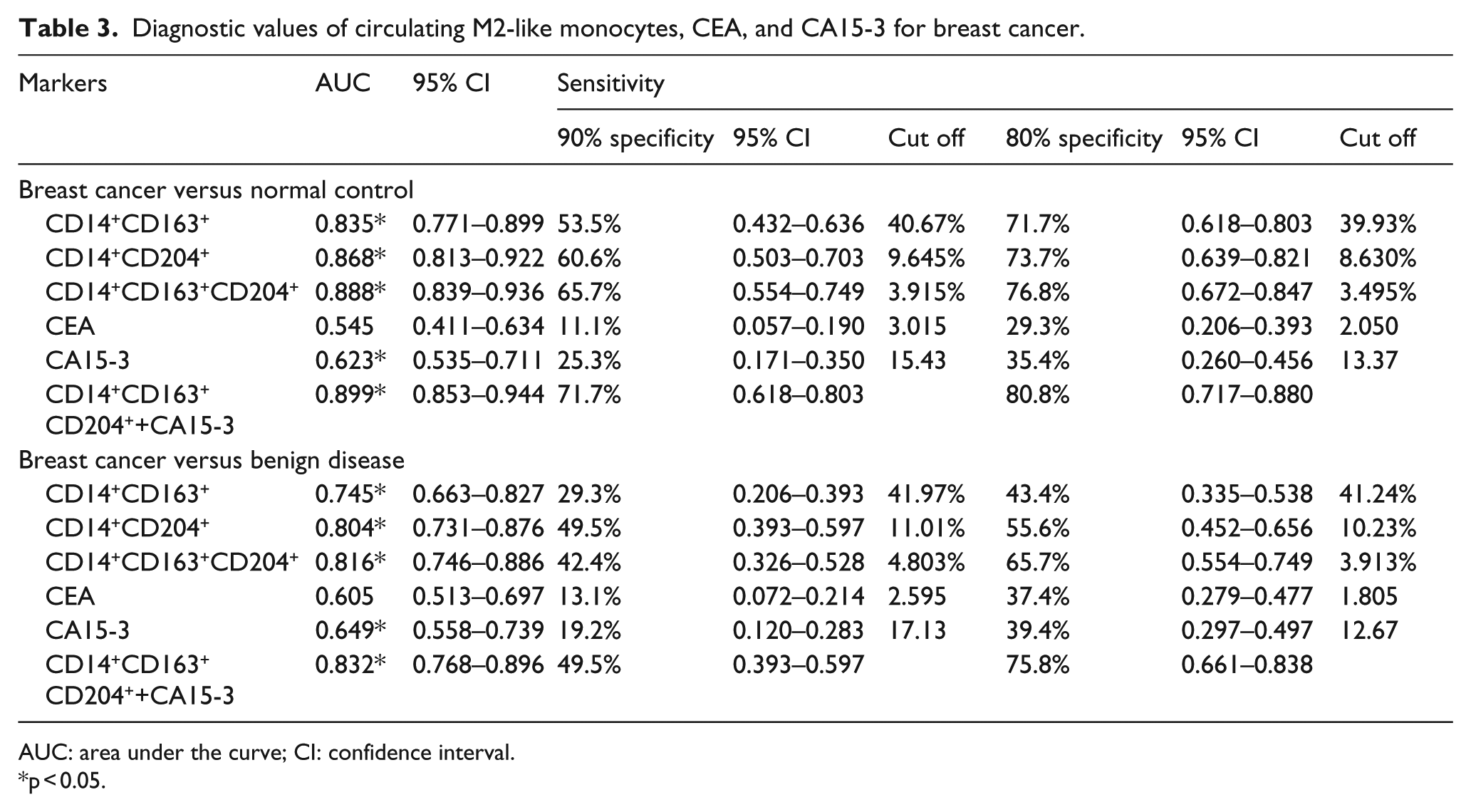
AUC: area under the curve; CI: confidence interval.
p < 0.05.

ROC analysis comparing breast cancer patients, healthy controls, and benign patients. (a) ROC analysis comparing breast cancer patients and normal controls for circulating CD14+CD163+/CD14+CD204+/CD14+CD163+CD204+ monocytes, CEA, and CA15-3. (b) ROC analysis comparing circulating CD14+CD163+/CD14+CD204+/CD14+CD163+CD204+ monocytes, CEA, and CA15-3 in breast cancer patients and patients with benign disease.
We also used logistic regression models to assess the relationship between markers (circulating CD14+CD163+CD204+ M2-like monocytes, CEA, and CA15-3) and BC. Univariate logistic regression analysis showed that higher circulating CD14+CD163+CD204+ M2-like monocyte levels significantly predicted BC versus normal controls (odds ratio (OR) = 4.208; 95% confidence interval (CI): 2.673–6.623; p < 0.01) and benign disease (OR = 2.593; 95% CI: 1.858–3.619; p < 0.01). We also found that CA15-3 but not CEA levels univariately predicted BC versus normal control (OR = 1.115; 95% CI: 1.036–1.200; p < 0.05) or benign disease (OR = 1.107; 95% CI: 1.030–1.189; p < 0.05). In multivariate logistic regression analysis, circulating CD14+CD163+CD204+ M2-like monocytes and CA15-3 but not CEA levels significantly predicted BC versus normal control. Moreover, circulating CD14+CD163+CD204+ M2-like monocytes and CA15-3 also significantly predicted BC versus benign disease, whereas CEA did not. The results from the logistic regression models are given in Table 4.
Results from logistic regression models.

CI: confidence interval; OR: odds ratio.
p < 0.05.
The superior diagnositc value of circulating CD14+CD204+ and CD14+CD163+CD204+ monocytes over CEA and CA15-3 was also demonstrated by their higher sensitivity when specificity was fixed at 80% or 90% (Table 3). For example, when discriminating BC from healthy controls at a specificity of 90%, circulating CD14+CD163+CD204+ monocytes yielded a sensitivity of 65.7% (cut off value = 3.92%), compared with 11.1% for CEA and 29.3% for CA15-3. Likewise, at 80% specificity, circulating CD14+CD163+CD204+ monocytes displayed a higher sensitivity (76.8%, cut off value = 3.495%) than CEA (29.3%) and CA15-3 (35.4%). As shown in Table 3, when used to distinguish BC from benign disease, the sensitivity of circulating CD14+CD204+ monocytes was similar to that of circulating CD14+CD163+CD204+ monocytes when the specificity was 80% or 90%. On the basis of the predictive variables identified in a logistic regression model, combining CA15-3 and circulating CD14+CD163+CD204+ monocytes to discriminate between BC and normal controls yielded a ROC curve with an AUC that was higher (0.899, 95% CI: 0.853–0.944) than either of these markers alone. Moreover, the sensitivity of these two combined biomarkers reached 80.8% at 80% specificity. Similar results emerged when the two markers were used to differentiate BC from benign disease (Table 3). Together, these data strongly suggested that circulating M2-like monocytes may be used as a novel diagnostic marker in BC.
Correlation between circulating M2-like monocyte levels and clinical BC parameters
A preliminary analysis was then performed to identify whether the percentages of circulating M2-like monocytes were associated with patient characteristics (Table 5). The results showed that higher percentages of circulating CD14+CD204+, CD14+CD163+CD204+ M2-like monocytes were significantly associated with tumor–node–metastasis (TNM) stage, lymph node metastasis, and histological differentiation (p < 0.05). Furthermore, the results indicated that the estrogen receptor (ER)-negative group had higher levels of CD14+CD163+CD204+ M2-like monocytes than the ER-positive group (p < 0.05). However, there were no associations between either CEA or CA15-3 levels and clinical parameters. Therefore, circulating M2-like monocytes may present a potential role in reflecting BC progression.
Correlation between the levels of CEA, CA15-3, and circulating M2-like monocytes and clinicopathological characteristics.

ER: estrogen receptor; PR: progesterone receptor; HER2: human epidermal factor 2; SD: standard deviation; TNM: tumor–node–metastasis.
p < 0.05.
Discussion
Macrophages continuously infiltrate into the microenvironment of solid tumors and are induced to form TAMs by cancer cells and other factors. Several studies have reported that the polarized states and levels of TAMs in pretreatment biopsies are altered in BC.21–23 TAMs resemble M2-like macrophages, and M2-like TAMs facilitate tumor progression, angiogenesis, metastasis, matrix remodeling, and treatment resistance.8,21,24 TAMs derived from the tumor tissue disseminate into the peripheral blood circulation in large numbers in conjunction with circulating tumor cell (CTCs) via transendothelial migration.11,13 Recently, circulating M2-like monocytes have been reported as a useful marker for diagnosis of non-small-cell lung cancer and colorectal cancer.16,17 However, until now, no studies have reported the use of circulating M2-like monocytes as a diagnostic marker for BC. Furthermore, data on this cell type are limited, and there have been contradictory results regarding the value of circulating tumor-associated monocytes as potential biomarkers in cancer patients.25–27 This study thus focused on evaluating the diagnostic value of circulating M2-like monocytes in BC. Our data indicated that the numbers of circulating M2-like monocytes were increased in the peripheral blood of BC patients and these monocytes showed promise in differentiating malignancy from benignity.
We first found that the number of M2-like macrophages was increased in histological BC sections. This result was consistent with previous reports suggesting that high expression of CD68 and CD163 can be used to detect M2-like macrophage subpopulations in BC tissue. 28 We next explored whether M2-like monocytes could be detected in the circulating blood of BC patients. We found that the levels of circulating M2-like monocytes were significantly elevated in patients with BC compared with healthy controls and benign patients. This result confirmed previous findings that M2-like TAMs may leak into the circulation from tumor sites and the subsequent increase in circulating M2-like monocyte may serve as a tumor marker. Most of the currently used tumor markers are not suitable as diagnostic tools for the early detection of cancers. CEA and CA15-3 are classical markers of BC; however, they lack sensitivity for early diagnosis.4,29 Additionally, the American Society of Clinical Oncology (ASCO) guidelines do not recommend the use of CEA and CA15-3 for BC screening or directing treatment. 30 In this study, plasma CA15-3 levels were significantly higher in BC patients than in healthy controls and benign patients. In accordance with findings from previous studies, our results showed that both CEA and CA15-3 had relatively low sensitivity in detecting BC. When patients with BC were compared with the healthy population, the frequency of circulating CD14+CD163+CD204+ M2-like monocytes, relative to CD14+CD163+ and CD14+CD204+ M2-like monocytes, had a higher AUC and sensitivity at a specificity of 80.0%. Furthermore, when comparing BC patients with benign patients, we found that the sensitivity of circulating M2-like monocytes was higher than CEA and CA15-3. More importantly, combining circulating M2-like monocytes and CA15-3 increased the diagnostic value in terms of both sensitivity and specificity for BC diagnosis. Therefore, in terms of diagnostic value in BC, circulating M2-like monocytes were superior to both CEA and CA15-3.
In a further analysis, we found that higher percentages of circulating CD14+CD204+, CD14+CD163+CD204+ M2-like monocytes were significantly associated with TNM stage, lymph node metastasis, and histological differentiation. This result was in accordance with previous studies reporting that circulating M2-like monocytes or macrophages contribute to the metastasis and growth of solid tumors.13,16,24 Our results also revealed that the levels of circulating CD14+CD204+/CD14+CD163+CD204+ M2-like monocytes were negatively associated with ER status in patients with BC. The frequency of circulating CD14+CD204+/CD14+CD163+CD204+ M2-like monocytes in ER-negative patients was higher than that in ER-positive patients. It is well known that ER regulates the growth and differentiation of tumor cells and is associated with the survival of BC patients. 31 Furthermore, there were no associations between CEA, CA15-3, and clinical parameters in our study. Therefore, it is reasonable to conclude that there are possible interactions between circulating M2-like monocytes and BC progression.
In conclusion, our data showed that circulating M2-like monocytes may represent a novel blood-based biomarker for BC and may have potential utility as a diagnostic tool. The high sensitivity of circulating M2-like monocytes for detecting BC patients indicates that circulating M2-like monocytes may be helpful to the currently used biomarkers in BC screening. It should be noted, however, that our analyses only focused on CD14+ monocytes and the sample size was relatively small. Additionally, the correlation between intratumoral TAM density and the frequency of peripheral M2-like monocytes in BC patients was not assessed in our study. Therefore, further studies with a larger population and the collection of matching histological specimens are necessary to confirm our results.
Footnotes
Acknowledgements
B.Z. and M.C. are co-first authors of this work. B.Z., M.C., G.Z., and F.G. helped in conception and design of this article; B.Z., Y.H., Y.L., J.X., and J.H. helped in acquisition of data (provided animals, acquired and managed patients, provided facilities, etc.); B.Z., C.Y., and Y.D. carried out analysis and interpretation of data (e.g. statistical analysis, biostatistics, and computational analysis); B.Z., G.Z., C.Y., M.C., and F.G. wrote, reviewed, and/or revised the manuscript; and Y.L. and F.G. supervised the study.
Ethical approval
The study was approved by the Ethics Committee of Shanghai Jiao Tong University in accordance with the Declaration of Helsinki.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (81272479, 81402419, 81572821, 81502490, 81502491, and 81672843), the Natural Science Foundation of Shanghai Municipality (14YF1412200), the Program of Shanghai Leading Talents (2013–038), and the Shanghai Shen-Kang Hospital Development Center (SHDC22014004).
