Abstract
Upper tract urinary carcinoma (UTUC) is a relatively uncommon but aggressive disease. Recent publications have assessed the prognostic significance of tumor architecture in UTUC, but there is still controversy regarding the significance and importance of tumor architecture on disease recurrence. We retrospectively reviewed the medical records of 101 patients with clinical UTUC who had undergone surgery. Univariate and multivariate analyses were conducted to identify factors associated with disease recurrence and cancer-specific mortality. As our single center study and the limited sample size may influence the clinical significance, we further quantitatively combined the results with those of existing published literature through a meta-analysis compiled from searching several databases. At a median follow-up of 41.3 months, 25 patients experienced disease recurrence. Spearman’s correlation analysis showed that tumor architecture was found to be positively correlated with the tumor location and the histological grade. Kaplan-Meier curves showed that patients with sessile tumor architecture had significantly poor recurrence free survival (RFS) and cancer specific survival (CSS). Furthermore, multivariate analysis suggested that tumor architecture was independent prognostic factors for RFS (Hazard ratio, HR = 2.648) and CSS (HR = 2.072) in UTUC patients. A meta-analysis of investigating tumor architecture and its effects on UTUC prognosis was conducted. After searching PubMed, Medline, Embase, Cochrane Library and Scopus databases, 17 articles met the eligibility criteria for this analysis. The eligible studies included a total of 14,368 patients and combined results showed that sessile tumor architecture was associated with both disease recurrence with a pooled HR estimate of 1.454 and cancer-specific mortality with a pooled HR estimate of 1.416. Tumor architecture is an independent predictor for disease recurrence after radical nephroureterectomy for UTUC. Therefore, closer surveillance is necessary, especially in patients with sessile tumor architecture.
Introduction
Upper tract urothelial carcinoma (UTUC) is relatively rare, accounting for approximately 5% of all urothelial carcinomas.1–3 Despite surgical refinement, oncological outcomes after radical nephroureterectomy (RNU) have not changed substantially over the past three decades. 4 A substantial proportion of patients after RNU with clinically nonmetastatic disease will experience disease recurrence and subsequently die from metastatic UTUC.5,6 Although the benefit of adjuvant therapies remains to be proven,7,8 identification of patients at high risk of failure with RNU alone is important for accurate prognostic assessment, patient counseling, and design of clinical trials using integrated multimodal therapy. Several possible predictive factors for this high incidence of disease recurrence have been reported, including tumor multiplicity, 9 stage,9,10 grade,10,11 concomitant carcinoma in situ (CIS),11,12 ureteric location,12,13 lymphovascular invasion (LVI),10,12,13 and adjuvant chemotherapy.12,14 The prognostic value of potentially important features, such as tumor architecture, remains to be proven.12,14,15
Several studies have reported that macroscopic sessile tumor architecture is independently associated with adverse outcomes after RNU for UTUC in large multi-institutional retrospective studies.7,13,16 Since proof attesting its current role in UTUC is relatively poor and few studies on macroscopic tumor architecture in UTUC of Chinese mainland patients are available, it is of utmost importance to combine these data to reach a reasonable conclusion. Therefore, this study focused on a retrospective evaluation of macroscopic tumor architecture associated with conventional pathological parameters and the prognosis of patients. Because the results of previous studies7,9 are controversial and no reliable conclusions can be drawn to aid surveillance strategies, the influence of tumor architecture upon disease recurrence has not been thoroughly analyzed. Therefore, we performed a meta-analysis of published literature to investigate the role of tumor architecture in predicting disease recurrence after RNU.
Materials and methods
Case control study
Study population
This study was hospital based. In total, 101 patients with UTUC who underwent nephroureterectomy at Affiliated Dalian Friendship Hospital of Dalian Medical University were enrolled in this study between January 2002 and December 2013. Ethical approval was given by the medical ethics committee of Institutional Review Board of Dalian Medical University, and the methods of this study were carried out in accordance with the approved guidelines. Patients with previous or concurrent urothelial carcinoma of the bladder and patients undergoing concomitant cystoprostatectomy were excluded. The administration of neoadjuvant chemotherapy or neoadjuvant radiotherapy was also considered as the exclusion criteria. Surgery was performed by several surgeons according to the standard criteria for RNU. Hospital medical records were reviewed to assess the following parameters: gender, age, tumor architecture, tumor side, tumor location, tumor focality, concomitant CIS, pathologic stage (2002 tumor–node–metastasis (TNM) classification of the American Joint Committee on Cancer), LVI, tumor grade (1998 World Health Organization (WHO)/International Society of Urologic Pathology consensus classification), lymph node status, and adjuvant chemotherapy.
Tumor architecture evaluation
The information on macroscopic architecture was taken from the histopathology reports, which were reviewed by two senior pathologists without knowledge of the clinical outcomes. For the purposes of this study, tumor architecture was defined as being based on the predominant macroscopic feature by pathologic gross examination. If present, macroscopic sessile growth was defined as being predominant. Microscopic tumor architecture, such as trabecular, nodular, infiltrative pattern of invasion, or inverted papillary architecture, was not judged. 14
Surveillance regimen
Patients were generally observed every 3 months for the first year after RNU, every 6 months from the second through to the fifth years, and annually thereafter. Follow-up examinations after surgery consisted of serum chemistry studies, routine urine examinations, radiographic examinations of the urinary system, plain chest radiography, and several selective measures such as cystoscopy. When clinically indicated, bone scans, chest computerized tomography (CT), or magnetic resonance imaging was performed electively.
Statistical analysis
Fisher’s exact test and the χ2 test were applied to evaluate the association between tumor architecture and clinicopathological parameters. The Kaplan–Meier method was used to compare the recurrence-free survival (RFS) among the groups using the log-rank test. Disease recurrence was defined as tumor relapse in the operative field, regional lymph node metastasis, and/or distant metastases. Recurrences in the bladder or contralateral upper urinary tract were not considered in the analysis of the RFS rate. The potential prognostic factors of RFS and cancer-specific survival (CSS) were established by univariate analysis, and only the significant factors were entered into multivariate Cox proportional hazard regression models. All statistical analyses were performed using SPSS version 13.0 (SPSS Inc., Chicago, IL, USA). Differences were considered to be statistically significant when the p value was less than 0.05.
Meta-analysis
Search strategy
We searched the PubMed, Medline, Embase, Cochrane Library, and Scopus databases to identify relevant studies that evaluated tumor architecture and its effects on UTUC prognosis from inception until July 2016. The following text words and medical subject headings that included all spellings were used: “tumor architecture,” “upper urinary tract,” “renal pelvis,” “ureter,” “urothelial cancer,” “urothelial carcinoma,” and “prognosis.” To obtain all relevant articles, further eligible bibliographies including Campbell-Walsh Urology, Surgical Procedures for Core Urology Trainees, Urological Men’s Health: A Guide for Urologists and Primary Care Physicians, and so on were sought. There were no restrictions for language or time of publication.
Selection criteria
Studies were considered if they met the following criteria: (1) the histological type of the tumors was upper urothelial carcinoma, (2) tumor architecture was evaluated in patients with UTUC, and (3) the association between tumor architecture and UTUC prognosis including disease recurrence and cancer-specific mortality was investigated. Accordingly, the following exclusion criteria were applied: (1) studies were reviews, letters to the editor, commentaries, or case reports that lacked the original data; (2) repeated studies included the same author or the same samples; (3) studies did not provide information on UTUC prognosis including disease recurrence and cancer-specific mortality; and (4) the hazard ratio (HR) and its standard error could not be calculated based on the given information.
Data extraction
To minimize the risk of failing to identify a publication, a rigorous process was independently carried out by two investigators to consider potentially relevant studies based on the predefined criteria. A third reviewer checked the resulting extractions and resolved any discrepancies between the independent search results. The data retrieved from the reports included author, year of publication, region, study type, recruitment period, number of patients, demographics (mean age, sex), HR or risk ratio (RR), and 95% confidence interval (CI) of reported endpoints.
Statistical analysis
We estimated the HR with a 95% CI for dichotomous outcomes. The heterogeneity of included studies was tested using the χ2 test, which was a qualitative analysis to judge statistical heterogeneity, and the I2 test, which was a quantitative analysis of measuring the percentage of inconsistency. If a p value for heterogeneity was greater than 0.1 in a χ2 test, indicating no heterogeneity among studies, fixed effects models were applied to estimate the pooled HR of each study. Otherwise, the random effects models were used. The I2 value in the I2 test delineated the proportion of total variation due to heterogeneity rather than errors of sampling, and an I2 greater than 50% presented authentic heterogeneity. The potential for bias was assessed by inspection of a funnel plot and Egger’s test if the number of studies included in each analysis was greater than or equal to 10. Otherwise, publication bias was not tested because of the limited number (below 10). The results were considered to be statistically significant at two-sided p values < 0.05. All statistical analyses were performed using Stata 12.0 (StataCorp, College Station, TX, USA).
Results
Association of tumor architecture with the clinicopathological characteristics of UTUC
Based on the predominant macroscopic feature, tumor architecture was classified as papillary/sessile type, and illustration of two growth types was shown in Figure 1. The descriptive characteristics of the 101 patients are summarized in Table 1. There were 61 males and 40 females, with a median age of 69 years. The median follow-up time was 41.3 months (range: 4.2–106.5 months). Disease recurrence occurred in 25 of 101 patients (24.8%). The primary tumors were located in the renal pelvis in 54.5% (n = 55) of patients and in the ureter in 40.6% (n = 41). Of the patients, 54 (53.5%) had muscle-invasive disease and 76 (75.2%) had high-grade disease. Positive lymph nodes were present in 11 patients (10.9%), and multifocal tumors were present in 10 patients (9.9%). The association between tumor architecture and the clinicopathological factors of UTUC, including age, gender, tumor side, tumor location, tumor focality, concomitant CIS, pathologic stage, LVI, histological grade, lymph node status, and adjuvant chemotherapy are summarized in Table 1. Tumor location was found to be positively correlated with tumor architecture (p = 0.036), and Spearman’s correlation analysis identified a correlation coefficient of 0.245 (p = 0.014). Moreover, the histological grade was also found to be positively associated with tumor architecture (p = 0.019), and the correlation coefficient between tumor architecture and histological grade was 0.232 (p = 0.019).

Illustration of two growth types in upper tract urothelial carcinoma (UTUC). (a) Papillary type always grows toward the tube wall of upper urinary tract with soft texture. (b) Sessile type grows toward the lumen of upper urinary tract regularly with hard texture.
Demographics and clinicopathological characteristics of 101 patients treated with RNU for UTUC.

RNU: radical nephroureterectomy; UTUC: upper tract urothelial carcinoma; CIS: carcinoma in situ; LVI: lymphovascular invasion.
The significance of bold values means that P < 0.05.
The impact of tumor architecture on RFS and CSS in UTUC
To examine if tumor architecture is a significant predictor for RFS after RNU, Kaplan–Meier curves were analyzed between sessile and papillary architecture and RFS. The RFS of patients with sessile architecture was significantly lower than those with papillary architecture (p = 0.001; Figure 2(a)). On univariate analysis, sessile tumor architecture and high pathologic grade were related to a poor RFS for patients with UTUC. Multivariate analysis showed that sessile tumor architecture (HR = 2.648; 95% CI = 1.146–6.122; p = 0.023) was the only independent prognostic factor for RFS (Table 2). Moreover, Kaplan–Meier curves were also used to investigate the relationship between tumor architecture and CSS, indicating that the CSS of sessile architecture was significantly lower than those with papillary architecture (p = 0.009; Figure 2(b)). However, univariate and multivariate analyses showed that high pathologic stage (HR = 2.571; 95% CI = 1.004–6.586; p = 0.049) was the only independent prognostic factor for CSS (Table 3).

Kaplan–Meier curves for (a) recurrence-free survival (RFS) and (b) cancer-specific survival (CSS) according to tumor architecture in patients with UTUC. Patients with sessile tumor architecture exhibited a significantly lower RFS (p = 0.001, log-rank test) and CSS (p = 0.009, log-rank test) rate than patients with papillary tumor architecture.
Univariate and multivariable Cox regression models predicting recurrence-free survival in 101 patients treated with RNU for UTUC.
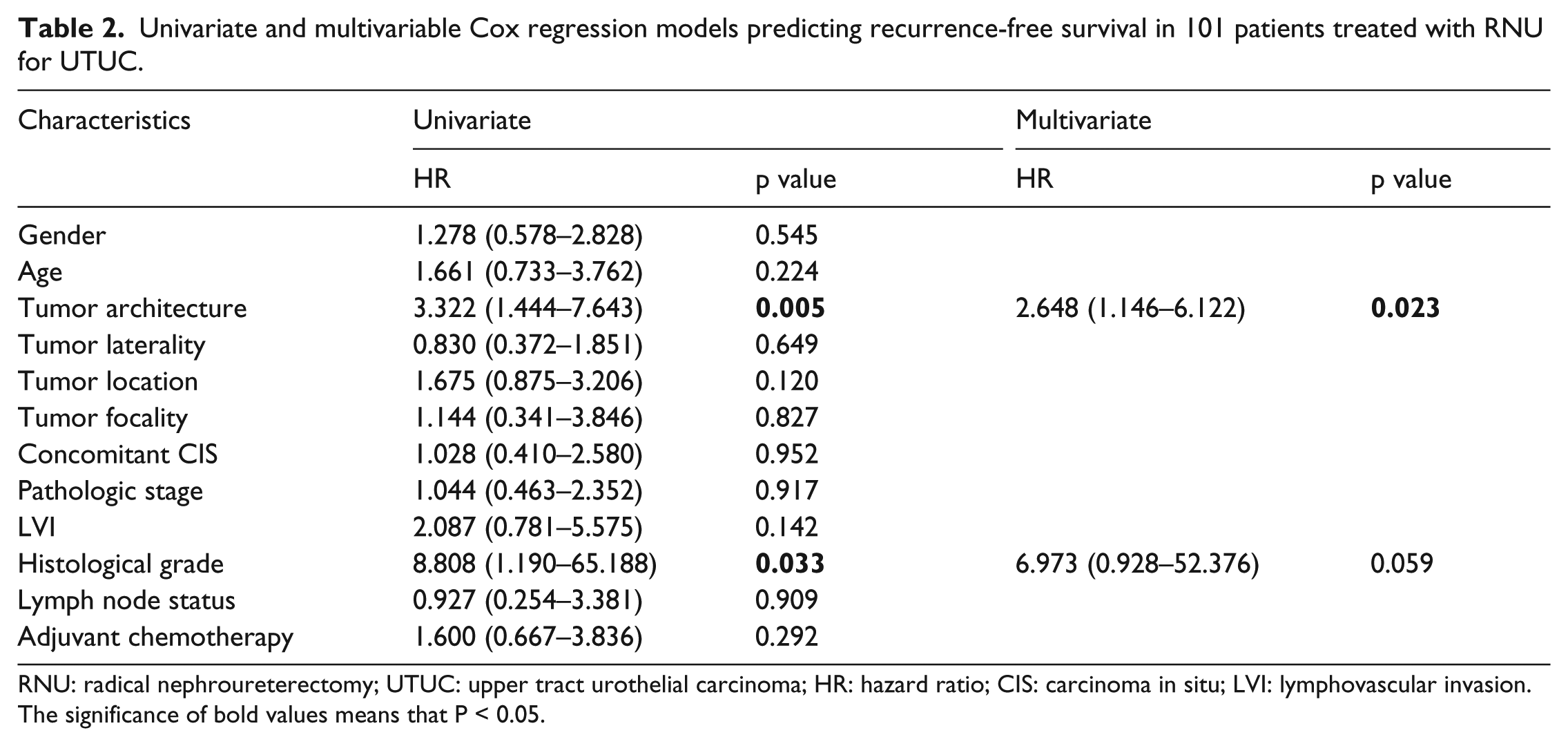
RNU: radical nephroureterectomy; UTUC: upper tract urothelial carcinoma; HR: hazard ratio; CIS: carcinoma in situ; LVI: lymphovascular invasion.
The significance of bold values means that P < 0.05.
Univariate and multivariable Cox regression models predicting cancer-specific survival in 101 patients treated with RNU for UTUC.

RNU: radical nephroureterectomy; UTUC: upper tract urothelial carcinoma; HR: hazard ratio; CIS: carcinoma in situ; LVI: lymphovascular invasion.
The significance of bold values means that P < 0.05.
Identification and eligibility of relevant studies in meta-analysis
The initial search, based upon the title/abstract and full text, yielded 95 relevant publications, of which 68 were excluded for being duplicate studies or for various reasons, such as being reviews, animal studies, or not relevant to our analysis. Figure 3 shows the selection process. Through extensive research, a total of 18 articles, including 17 published articles and our retrospective study, were included in the meta-analysis.7,9–24 It is pointed out that although some of articles6,10,16 selected in their meta-analysis came from multi-institutional collaborative database organized by Shariat, they were characterized by different origin of dataset or recruitment period, so included them as different studies in our analysis. The main characteristics of the 18 included studies are shown in Table 4. They were published from 2009 10 to 2016. 15 The sample size ranged from 101 to 2477 13 patients, and the mean/median follow-up durations varied from 27 11 to 47.5 7 months.
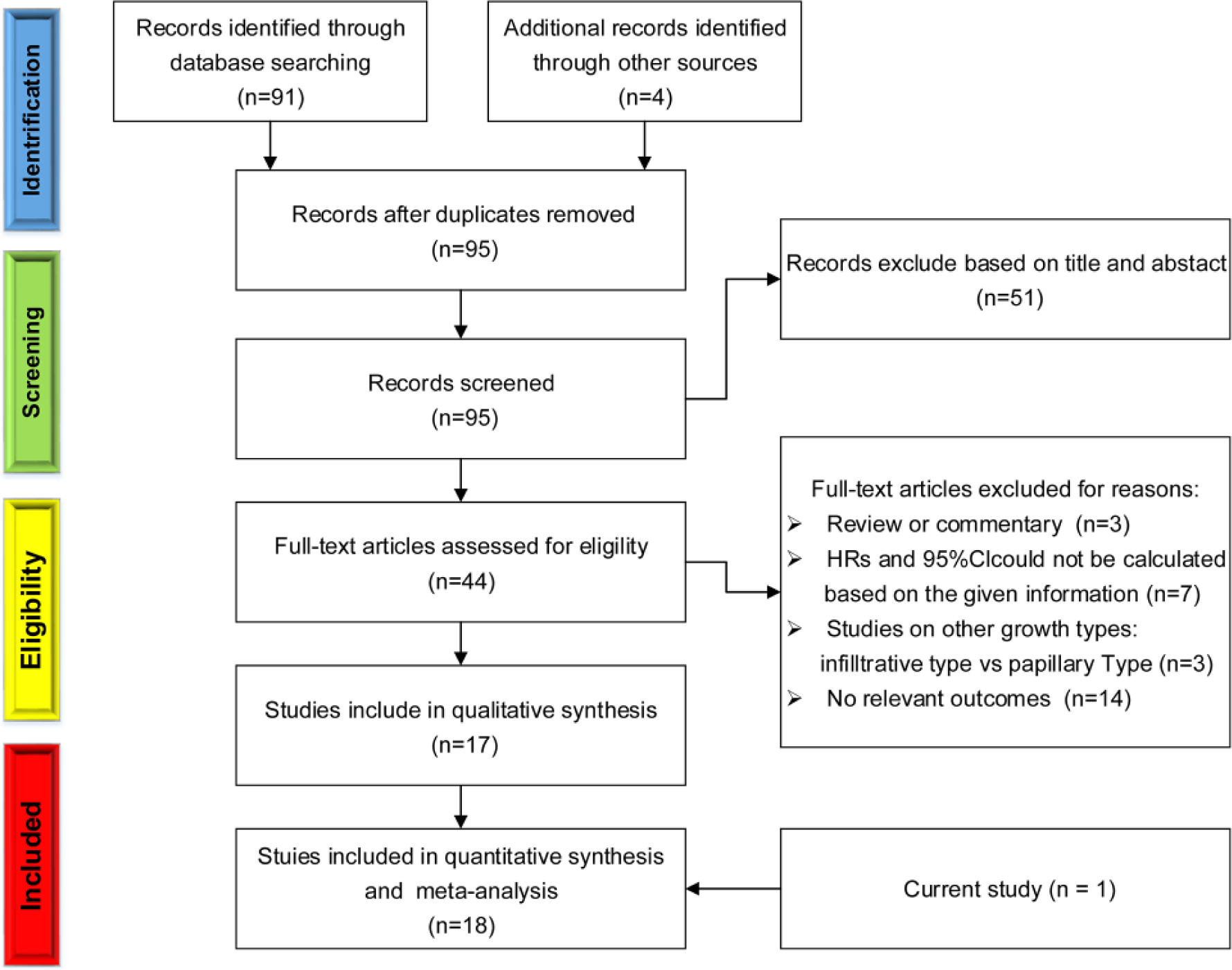
Methodological flow chart for the systematic review.
Main characteristics of 18 studies included in the meta-analysis.

PA: papillary architecture; SA: sessile architecture.
Meta-analysis of tumor architecture and UTUC prognosis of disease recurrence and cancer-specific mortality
Figure 4(a) shows the pooled results from the random effects model combining the HRs for disease recurrence after surgery. Sessile tumor architecture was associated with an increased risk of disease recurrence after surgery in UTUC patients (HR = 1.454; 95% CI = 1.334–1.585; p < 0.001), with no heterogeneity among the studies (I2 = 12.1%; p = 0.312). No publication bias was found between tumor architecture and risk of disease recurrence with the symmetrical Begg’s funnel plot (p = 0.064). Moreover, the p value for Egger’s regression asymmetry test was 0.056 (Figure 4(b)). For cancer-specific mortality after surgery, Figure 5 showed that sessile tumor architecture was associated with an increased risk of cancer-specific mortality in UTUC patients (HR = 1.416; 95% CI = 1.302–1.540; p < 0.001), with no heterogeneity among the studies (I2 = 0.0%; p = 0.506). Mild publication bias was found after Begg’s funnel plot (p = 0.023) and Egger’s regression asymmetry test (p = 0.015).

(a) Forest plot for the association between tumor architecture and UTUC prognosis of disease recurrence. Hazard ratios for each trial are represented by squares; the horizontal line crossing the square represents the 95% confidence interval. The diamonds represent the estimated pooled effect using the Mantel–Haenszel random effects model. (b) Funnel plot for the association between tumor architecture and UTUC prognosis of disease progression. Visual inspection of the Begg’s funnel plot did not identify substantial asymmetry.

(a) Forest plot for the association between tumor architecture and UTUC prognosis of cancer-specific mortality. Hazard ratios for each trial are represented by squares; the horizontal line crossing the square represents the 95% confidence interval. The diamonds represent the estimated pooled effect using the Mantel–Haenszel random effects model. (b) Funnel plot for the association between tumor architecture and UTUC prognosis of cancer-specific mortality. Visual inspection of the Begg’s funnel plot identified mild asymmetry.
Discussion
Previous investigators focused on the risk factors of disease recurrence after total nephroureterectomy—(1) Patient-specific factors: Rieken et al. 25 found that diabetic patients who did not use metformin were at significantly higher risk of disease recurrence. Nuhn et al. 26 suggested that a prior history of muscle-invasive urothelial carcinoma of the bladder independently predicted intravesical disease. (2) Tumor-specific factors: Mathieu et al. 9 revealed that stage, multifocality, and lymph node metastasis were possible risk factors for the recurrence of tumors following surgery for UTUC, whereas Vartolomei et al. 13 reported that tumor necrosis and LVI have a significant impact on the risk of tumor recurrence. (3) Treatment-specific factors: Recently, Jwa et al. 27 reported that adjuvant radiotherapy may significantly decrease the risks of locoregional and disease recurrence. Shariat et al. 12 found that adjuvant chemotherapy had a significant impact on lowering the risk of tumor recurrence. (4) Prognostic biomarkers: immunohistochemical staining for the following biomarkers may have important prognostic roles in UTUC, including Ki-67, 17 insulin-like growth factor messenger RNA binding protein 3 (IMP3), 28 forkhead box A1 (FOXA1), 11 and variant isoforms of CD44 (CD44v). 29
In this study, we found that tumor architecture was associated with adverse clinicopathological features and independently predicted features of biologically and clinically aggressive UTUC, such as pathologic stage and histological grade, indicating that the sessile type was more likely to be found in muscle-invasive and/or higher-grade UTUC. We also found that tumor architecture was a strong predictor of disease recurrence in patients with UTUC treated with RNU (HR = 2.580; 95% CI = 1.113–5.981; p = 0.027). The HR value of 2.58 denoted an increased recurrence rate of 158% in sessile-type UTUC. Our findings confirm those of previous studies in which tumor architecture was associated with unfavorable oncologic outcomes of UTUC. In a multi-institutional study of 2274 patients, Vartolomei et al. 13 concluded that the tumor architecture, stage, and lymph node metastasis had significant impact on the incidence of disease recurrence. Similarly, Bagrodia et al. 19 suggested that tumor architecture was a prognostic factor for disease recurrence after nephroureterectomy.
Whether a parameter can improve the ability of established predictors of cancer outcome requires more than the conventional univariate and multivariate analyses with associated HR and p values. Among the results relating to tumor architecture and its prognostic value in UTUC, different results were found. Fajkovic et al. 21 found no association between sessile tumor architecture and disease recurrence. Also, Favaretto et al. 15 did not identify the tumor architecture as a significant risk factor for disease recurrence in 678 UTUC patients who underwent RNU. Since meta-analysis can combine studies on specific topics, and RNU is less influenced by individual findings from a single study, we performed a systematic review of published articles related to the changes in tumor architecture and their effects on UTUC prognosis. Hence, our work combined 15 independent studies, selected according to the enrollment criteria, which compared disease recurrence in UTUC with sessile tumor architecture in the primary tumor. Our results showed that the HR was 1.460 and that the 95% CI ranged from 1.340 to 1.590, suggesting that sessile tumor architecture increased the risk of disease recurrence after follow-up by 46%, with 95% CI ranging from an increase of 34% to an increase of 59%.
In addition, tumor architecture was found to positively correlate with tumor location. It is interesting to note that a high proportion of the tumors with papillary architecture occurred in the calix or pelvis (62.9%; 44/70), whereas a high proportion of the tumors with sessile architecture occurred in the ureter (61.3%; 19/31). During normal urine flow, ureteral contractile pressure moves boluses of urine that are higher than renal pelvic pressures. In this situation, a closed ureteropelvic junction may be protective for the kidney in dissipating backpressure from the ureter.30,31 Under these physiological conditions, the distribution of different shear stresses induced by urine flow on a vessel wall may be generated locally at the nanoscale level by cell–cell or cell–extracellular matrix (ECM) interactions, thus influencing cell function through actomyosin contractility and actin dynamics.32,33 In some pathological conditions, the weight bearing of hydronephrosis may induce the compressive or tensile stress, leading to biochemical cues that modulate cell and tissue behavior.34–36 Several studies have reported that biomechanical factors may drive cancer cells toward an invasive phenotype in mammary or hepatocellular carcinomas1–3 Therefore, we hypothesized that the formation of different tumor architectures may be related to pressure and flow-rate distributions in different regions of the upper urinary tract, highlighting that more biomechanical research is needed to help identify these effects. Moreover, several studies have reported that tumor architecture was found to correlate with expression of E-cadherin, 15 survivin, 9 Ki-67, 17 and mammalian target of rapamycin (mTOR) pathway biomarkers. 19 Future research should be directed at validating these markers in larger cohorts of patients and perhaps combining clinical prognostics with molecular biomarkers to achieve the most accurate predictive models that enable patient selection for recurrent monitoring of UTUC.
We acknowledge that our study has some limitations. First, this study has a small sample size. This was due to the disease being relatively rare and the study being carried out using data from a single center setting. To overcome these shortcomings and increase the reliability of the conclusions, we performed a systematic review of published articles. We did not limit our literature search to English-language papers only but extended the search to unpublished trials and abstracts that would likely increase the proportion of null results. Potential bias could not be completely avoided. Indeed, publication bias could arise as a result of selective “positive” studies. In addition, the resulting HRs from multivariate survival analysis reported in the selected articles were included. When these results were not available, we extracted the univariate data. 37 Second, with regard to tumor architecture, we focused on the prognostic role of macroscopic architecture (such as sessile or papillary type) on disease recurrence. A macroscopic judgment is easy and is based on a simple aspect compared to microscopic architecture such as trabecular, nodular, or infiltrative patterns of invasion or inverted papillary architecture, which was not assessed in our study. This may minimize inter-investigator variation in our study. Third, we defined disease recurrence as tumor relapse in the operative field, regional lymph node metastasis, and/or distant metastases according to the criteria of related studies.10–12 Recurrences in the bladder or contralateral upper urinary tract were not considered in the analysis of the RFS rate. Future research will be directed at the validation of bladder or contralateral upper urinary tract RFS in larger cohorts of patients.
Conclusion
In conclusion, our retrospective cohort study and meta-analysis concluded that sessile tumor architecture might be a prognostic factor for patients undergoing UTUC, although these findings need to be interpreted with caution. Tumor architecture determination might help identify patients with UTUC at high risk of disease recurrence who might benefit from closer follow-up or more aggressive therapy. The value of tumor architecture for the molecular staging of UTUC also needs to be further confirmed in adequately designed prospective trials involving a larger number of patients before any definitive conclusions can be drawn.
Footnotes
Acknowledgements
B.F., B.H., and Q.Y. contributed equally to this article.
Authors’ Note
Bo Fan is also affilated to Graduate School, Dalian Medical University, P.R. China, Bin Hu is also affiliated to Liaoning Cancer Hospital and Institute, Shenyang, P.R. China, Qingmin Yuan, Xiaofeng Qi and Xin Wang are now affiliated to Dalian Medical University, P.R. China, Shuang Wen and Tianqing Liu are now affiliated to Affiliated Dalian Friendship Hospital of Dalian Medical University, P.R. China, Shanshan Bai is now affiliated to Affiliated Hospital of Dalian Medical University, P.R. China, Deyong Yang and Xishuang Song are now affiliated to First Affiliated Hospital of Dalian Medical University, P.R. China.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was funded by the National Natural Science Foundation of China (Grant numbers: 81271603, 31570953, and 11472074).
