Abstract
Small leucine-rich proteoglycans are components of extracellular matrix that regulates neoplastic transformation. Among small leucine rich proteoglycans, Decorin, Biglycan and Lumican are most commonly implicated markers, and their expression is well studied in various malignancies. In this novel study, we have collectively evaluated expression of these three molecules in urothelial carcinoma of bladder. Thirty patients of confirmed untreated bladder cancer, 30 healthy controls for blood and 30 controls for adjacent non-tumour tissue were enrolled. Blood was collected from all subjects and tumour/adjacent normal tissue was obtained from the patients. Circulatory levels were estimated by enzyme-linked immunosorbent assay, relative messenger RNA expression by quantitative polymerase chain reaction and protein expression by immunohistochemistry and western-blotting. Circulatory levels of Biglycan (p = 0.0038) and Lumican (p < 0.0001) were significantly elevated, and that of Decorin (p < 0.0001) was significantly reduced in patients as compared with controls. Protein expression by immunohistochemistry and western-blotting showed elevated expression of Lumican and Biglycan and lower expression of Decorin in urothelial carcinoma of bladder. Quantitative polymerase chain reaction for messenger RNA expression from tissue specimens revealed significantly higher expression of Biglycan (p = 0.0008) and Lumican (p = 0.01) and lower expression of Decorin (p < 0.0001) in urothelial carcinoma of bladder. Out of all molecules receiver operating characteristic curve showed that the 0.207 ng/ml cut-off of serum Lumican provided optimum sensitivity (90.0%) and specificity (90.0%). Significant alteration of matrix small leucine-rich proteoglycans in urothelial carcinoma of bladder was observed. Higher expression of Lumican in Bladder cancer patients with the cut-off value of highest optimum sensitivity and specificity shows its importance as a potential non-invasive marker for early detection of UBC following further validation in large patient cohort.
Introduction
Cancer pandemic is growing with an estimated 20 million new cases to occur annually by 2025 where most of the burden would be in the low- and middle-income countries. 1 According to GLOBOCAN 2012, estimated global cancer incidence was 14.1 million new cases and 8.1 million cancer deaths. 2 Overall in both sexes combined, lung, breast and colorectal cancers are having the highest incidences in decreasing order. Urothelial carcinoma of bladder (UBC) is currently ranked as the ninth most common cause of cancer with an estimated incidence of 429,000 cases (both sex combined) in 2012. 2
Understanding the pathophysiology of cancer is very essential for correct diagnosis and treatment. Many elements in the human body restrict cancer growth, proliferation, differentiation and metastasis. They include apoptosis, tumour suppressor gene up-regulation, down-regulation of oncogenes, immunosurveillance, and so on.3–5 Extracellular matrix (ECM) also plays an important role in the microenvironment to regulate cancer formation. ECM is formed by the basement membrane and interstitial matrix. 6 The interstitial matrix is formed of glycoproteins and proteoglycans together with fibrillar collagen. These components provide physical and biochemical characteristics to the matrix. 7 Small leucine-rich proteoglycans (SLRPs) are one such matrix component that is present ubiquitously. 8 Recently, SLRPs have been classified under five major classes (canonical class I–III and non-canonical class IV–V). 9 Biochemically, they consist of a protein core with a different glycosylation pattern characteristic to each component. SLRPs are necessary for matrix assembly, collagen fibril organisation and cell–matrix interactions. 8 A characteristic feature of SLRPs is the presence of leucine-rich repeats (LRRs) in their protein core which permits interaction with other group of proteins carrying similar characteristics. 10 LRRs consist of 20–24 motifs that are rich in leucine. 10 They interact with growth factor receptors leading to down-regulation of cellular growth signals leading to controlled growth of tissue. 11 It has also been seen that low expression of SLRPs leads to various structural defects which correlates with their importance in the matrix as a key structural component. 8
Decorin is the best studied among all the SLRPs. It is closely bound to the collagen fibril and modulates collagen I fibrillogenesis. 12 Decorin interacts with the growth factor receptors and down-regulates their function as seen in some cancers. 13 Decorin-mediated up-regulation of p21 expression acts as an inhibitor of cyclin-dependent kinases that regulates tumour cell overgrowth. 14 Another important effect of Decorin in the matrix is blocking the function of transforming growth factor β (TGF-β). 15 Decorin is also known to cause degradation of Met receptor (receptor for hepatocyte growth factor) and subsequent down-regulation of β-catenin. 16 Low expression of Decorin has been observed in various malignancies which support its function as a key matrix component that regulates cell cycle and hence cancer progression.17–20 Decorin expression has been observed to be lower when the tumour becomes invasive. 17 Experiments with recombinant Decorin have also been explored which demonstrates a similar tumour suppressive effect. Reed et al. 21 had established a reduction in tumour growth both in vitro and in vivo by treating the metastatic breast cancer cell line and an orthotopic mammary carcinoma model with recombinant Decorin.
Biglycan is another member of SLRPs in the matrix that along with its matrix scaffolding property also regulates the immune system through Toll-like receptors (TLRs). 22 High expression of Biglycan in malignancy has been associated with poor prognosis as observed in endometrial cancer, oesophageal cancer and gastric cancer.23–25 Knockdown of Biglycan using small interfering RNA (siRNA) in HCT116 colon cancer cell line inhibited cell proliferation and produced growth arrest. 26 As it also regulates the immune system, Biglycan has been found to be associated with auto-immune diseases like systemic lupus erythematosus (SLE) where it enhances the expression of CXCL13 which is a potent B cell chemo-attractant. 27 Both Decorin and Biglycan belong to class I SLRPs and only differ in the number of glycosaminoglycans around the protein core where Decorin carries single chondroitin sulphate/dermatan sulphate and Biglycan carries two chondroitin sulphate/dermatan sulphate. 28 Lumican is another member classified under class II SLRPs. It consists of four keratan sulphate bound to the LRR. 28 Its structural role in the matrix is well established in tissue like cornea and skin where mice with homozygous defective (mutant) Lumican (Lum) gene developed corneal opacity and skin laxity due to abnormal collagen fibril assembly. 29 One report states that the expression of Lumican is increased in reactive stroma of prostate cancer which reduces its invasiveness as observed by an increased tumour cell invasion in Lumican knock-out. 30 In other cancers, an increase in Lumican expression was observed.31–33 In colorectal cancer, Lumican expression tends to correlate with poor prognosis. 31 Overexpression of Lumican in human colon LS180 cells produced an increased cell migratory potential due to remodelling of actin cytoskeleton. 34 Till date, there is no report that correlates Lumican with UBC. These SLRPs Decorin, Biglycan and Lumican are closely related in ECM assembly, and their expressions are associated with various malignancies.
Though not only required in the matrix for structural framework, these proteoglycans are also effective in controlling various physiological functions like cell cycle regulation which makes them the guardian of the matrix. Although an isolated study of Decorin and Biglycan in bladder cancer has been reported, there is no collective study of Decorin, Biglycan and Lumican together in UBC. It is essential to study their expression in this disease so as to understand their involvement and diagnostic potential in this cancer. Therefore, this study aims to observe the quantitative messenger RNA (mRNA) expression of these SLRPs in tissues and their protein expression in serum and tissue of UBC patients.
Material and methods
Subjects
The subject details documented for the study such as age, sex and types of controls (C1 and C2) are mentioned in Table 1. All the patients were histopathologically proven cases of urothelial carcinoma (transitional cell carcinoma). The tumour specimens were collected from patients undergoing radical cystectomy after informed consent. Blood was also collected from the study subjects and serum was separated. Control samples were obtained from two sources (C1 and C2 groups). The C1 group included patients with urothelial carcinoma of bladder from whom adjacent normal bladder specimen (proven normal by histopathology) was collected during radical cystectomy. C2 comprises apparently healthy volunteers from whom only blood was collected after informed consent. The circulatory level, relative mRNA expression and protein expression were compared statistically between the patient and the control group. Ethical clearance was obtained from the Institute Ethics Committee.
Details of the patients and control groups.

SD: standard deviation.
Blood samples
All the patient samples were collected from patients admitted in the Urology ward of All India Institute of Medical Sciences (AIIMS), New Delhi. Age-matched control samples were obtained from apparently healthy individuals from the local community. Both samples were taken after informed consent.
Tissue samples
In the study, both tumour specimens and adjacent non-tumour specimens (as control) were obtained from the same patient who underwent radical cystectomy from the Urology department of AIIMS, New Delhi. All specimens were collected after informed consent.
Enzyme-linked immunosorbent assay
Enzyme-linked immunosorbent assay (ELISA) was performed on patient (n = 30) and control (n = 30) samples in triplicates. The circulatory levels of Decorin, Biglycan and Lumican were measured using ELISA kits by Sincere™ Biotech (Beijing, China).
Real-time polymerase chain reaction
RNA was extracted from the tissue specimen using TRI reagent® (Sigma-Aldrich, USA). The RNA extracted was quantified using a Nanodrop™ spectrophotometer (Thermo Fisher, USA) at 260 nm. After DNase treatment, 1 µg of RNA was used to synthesise complementary DNA (cDNA) using MuLV Reverse Transcriptase (Fermentas, Thermo Fisher). The cDNA synthesised was used as a template to analyse the gene amplification (using Bio-Rad CFX96 Touch™ Real-Time PCR Detection System, USA) with primers specific to the three SLRPs (Decorin, Biglycan and Lumican) and Maxima SYBR green master mix (Fermentas, Thermo Fisher). The conditions used for polymerase chain reaction (PCR) were as follows: initial denaturation at 95°C for 5 min, followed by 40 cycles of 95°C for 15 s, 60°C for 30 s and 72°C for 30 s. All the primers had an optimal annealing temperature of 60°C. Primers used were as follows: 18S: 5′-GTA ACC CGT TGA ACC CCA TT-3′ (forward) and 5′-CCA TCC AAT CGG TAG TAG CG-3′ (reverse); Decorin: 5′-GGA CCG TTT CAA CAG AGA GG-3′ (forward) and 5′-TCA GAA CAC TGG ACC ACT CG-3′ (reverse); Biglycan: 5′-AAG CTC CTC CAG GTG GTC TAT C-3′ (forward) and 5′-AGA GGC TGA TGC CGT TGT AG-3′ (reverse) and Lumican: 5′-GTC CTG ACA GAG TTC ACA GCA-3′ (forward) and 5′-CCA ATC AAT GCC AGG AAG AG-3′ (reverse).
Immunohistochemistry
Tissue specimens (tumour and non-tumour) were fixed in 10% formalin following which paraffin-fixed tissue-embedded blocks were prepared. Special 3-Aminopropyltriethoxysilane (APES)-coated slides were used for mounting the microtomised tissue specimen. Antigen retrieval was done using citrate buffer (pH 6.0). Washing was performed in Tris-buffered saline (TBS). Primary antibodies for Decorin (mouse monoclonal), Biglycan (mouse monoclonal) and Lumican (rabbit polyclonal) were purchased from Abcam (USA). All the primary antibodies were diluted in TBS at a concentration of 1:150 for both Decorin and Biglycan and 1:300 for Lumican. Anti-mouse and/or anti-rabbit secondary antibodies raised in goat were then added to the tissue section followed by treatment with avidin-conjugated horseradish peroxidase (HRP). After washing, each tissue section was then covered by freshly prepared diaminobenzidine (DAB) chromogen for few minutes until optimum development of brown-coloured peroxidase reactant product forms. Slides were counterstained by haematoxylin. Finally, slides were mounted after dehydrating with alcohol and acetone. The tissue expression was quantified by using immunoreactive score (IRS). 35
Western blotting (immunoblotting)
Tissue samples (tumour and adjacent non-tumour) were lysed in radioimmunoprecipitation assay (RIPA) lysis buffer (150 mM NaCl, 1% Triton X-100, 0.5% deoxycholate, 0.1% sodium dodecyl sulphate (SDS), 50 mM Tris pH 8.0) containing protease inhibitor cocktail and ethylenediaminetetraacetic acid (EDTA). Protein quantification was done by Bradford assay. Thirty micrograms of total protein was then resolved in sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE) and transferred from gel onto a nitrocellulose membrane. Membrane was blocked in 5% bovine serum albumin (BSA)-TBST (Tris-buffered saline, 0.1% Tween 20, pH 7.2) for 2 h. Decorin and Biglycan mouse monoclonal antibodies and Lumican rabbit monoclonal antibody were incubated for overnight at 4°C. HRP-conjugated secondary antibody was then added for 2 h at room temperature. Chemiluminescence detection reagent (Thermo Scientific) was used to develop blots, and band images were acquired with FluorChem M (ProteinSimple, USA) followed by quantification using ImageJ analyser software.
Receiver operating characteristic curve analysis
Receiver operating characteristic (ROC) curves were plotted for serum levels of each molecule to determine optimal cut-points, and their associated sensitivity and specificity for their utility as diagnostic markers.
Statistical analysis
Data were expressed as median (range) for circulatory levels and mRNA fold expression. For IRS by immunohistochemistry (IHC), the data were expressed as mean ± standard deviation (SD). Fold change after densitometry analysis of western-blot was represented as mean ± SD. Comparisons between the patient and control groups were made using the Wilcoxon rank-sum (Mann–Whitney) test for nonparametric data. Student’s t-test was applied for parametric data. Statistical significance was defined at p < 0.05.
Results
Circulatory levels of Decorin, Biglycan and Lumican
The circulatory levels of these proteins were studied using ELISA from the serum samples of the patients (P) and controls (C2). The circulatory levels of Decorin (p < 0.0001) were significantly higher in C2 compared with P (Table 2 and Figure 1). The circulatory levels of Biglycan (p = 0.0038) and Lumican (p < 0.0001) were significantly higher in P as compared with C2.
Circulatory levels of Decorin, Biglycan and Lumican in patients as compared with controls.
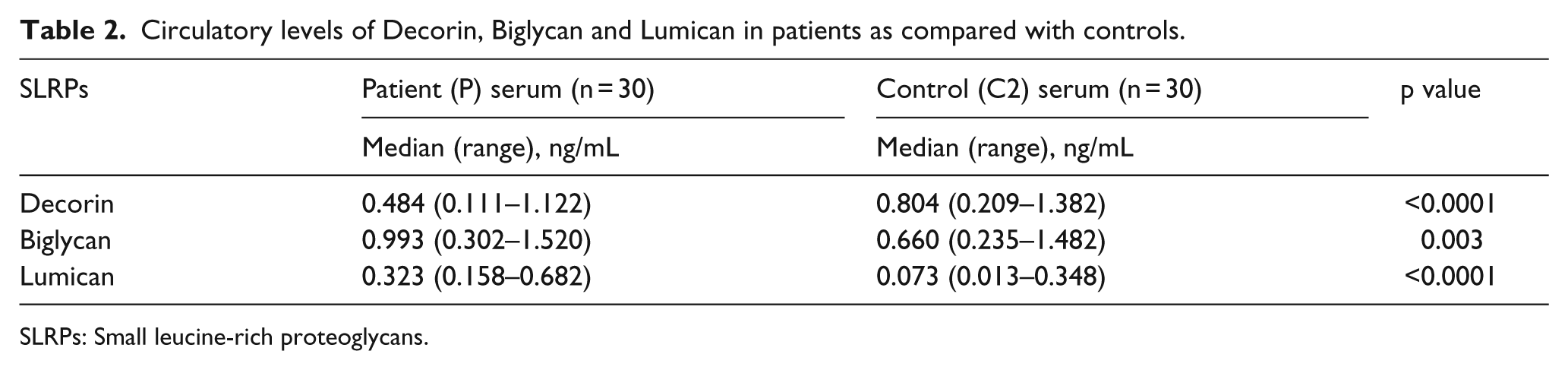
SLRPs: Small leucine-rich proteoglycans.
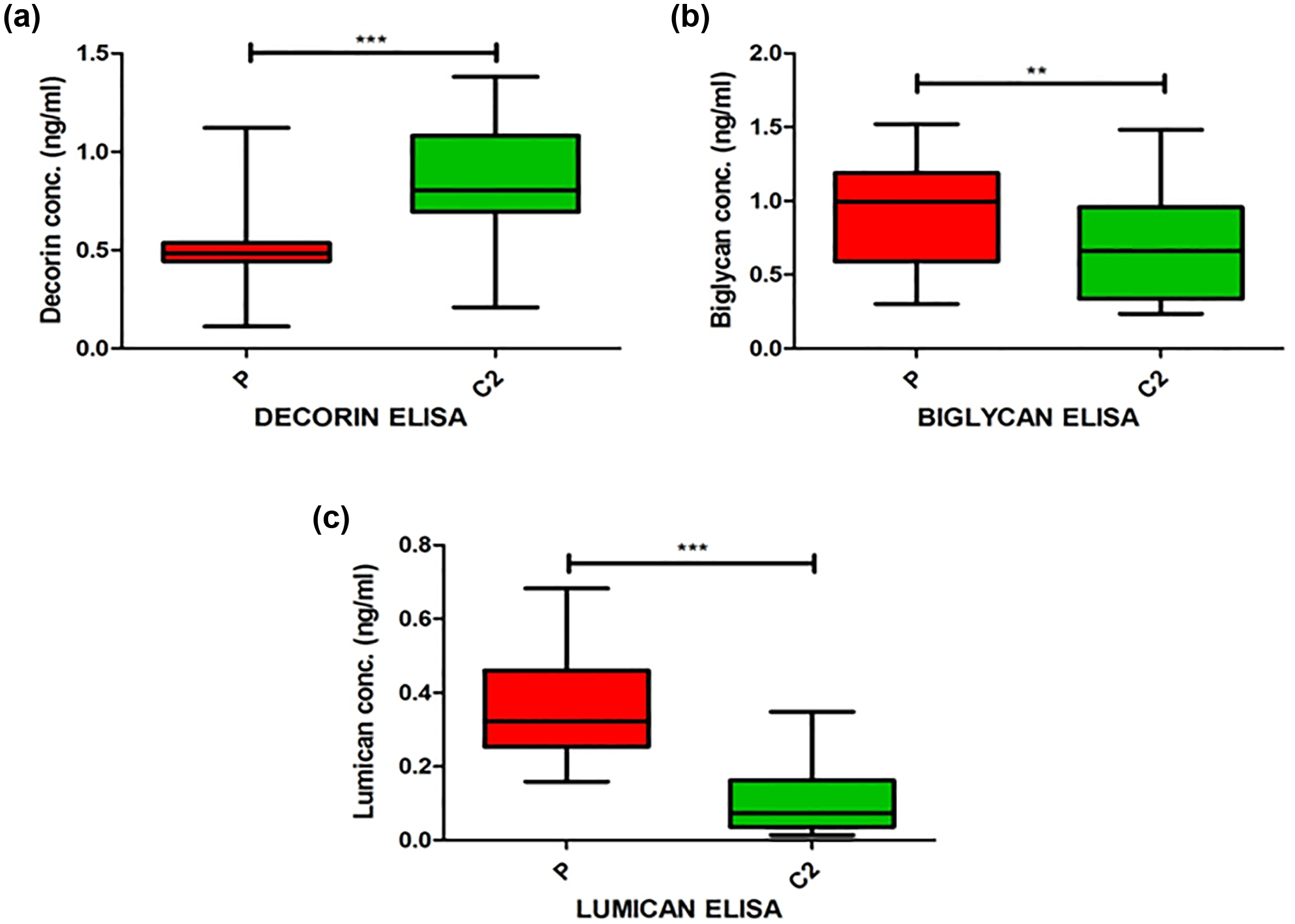
The box-and-whisker plot showing circulatory levels of (a) Decorin, (b) Biglycan and (c) Lumican are shown. The values are taken as median of the total study samples and expressed in ng/mL.
ROC curve analysis of Decorin, Biglycan and Lumican
ROC curves were generated for each molecule with comparison being done between total patients and controls to determine their sensitivity and specificity in serum as shown in Figure 2. The sensitivity and specificity of 0.565 ng/mL cut-off for serum Decorin levels in UBC patients relative to the control group were 83.3% and 83.3%, respectively. Values of sensitivity and specificity of 0.729 ng/mL cut-off for serum Biglycan levels were 70.0% and 66.67%, respectively. Sensitivity and specificity values of 0.207 ng/mL cut-off for serum Lumican level were 90.0% and 90.0%, respectively. The area under the ROC curve for Decorin was 83.67%. Areas under the curve were 71.78% and 96.11% for Biglycan and Lumican, respectively.

The ROC curve for circulatory levels of (a) Decorin, (b) Biglycan and (c) Lumican). The cut-off point and respective sensitivity, specificity and area under the curve (AUC) for each molecule are also mentioned.
Relative mRNA expression of Decorin, Biglycan and Lumican
The relative mRNA expression involved the study group of bladder cancer patients (P) with tumour tissue. For the control (C1) group, adjacent normal bladder mucosa was obtained from surrounding non-tumour tissue of the same bladder cancer patients. The expression of Decorin (p < 0.0001) was significantly lower in patients, and in case of Biglycan (p = 0.0008), the expression was significantly higher in patients as compared with controls. The relative mRNA expression of Lumican (p = 0.015) in patients was found to be significantly higher as compared with controls but not as significant as for Decorin and Biglycan as shown in Table 3 and Figure 3.
Relative mRNA expression of Decorin, Biglycan and Lumican in patients as compared with controls.
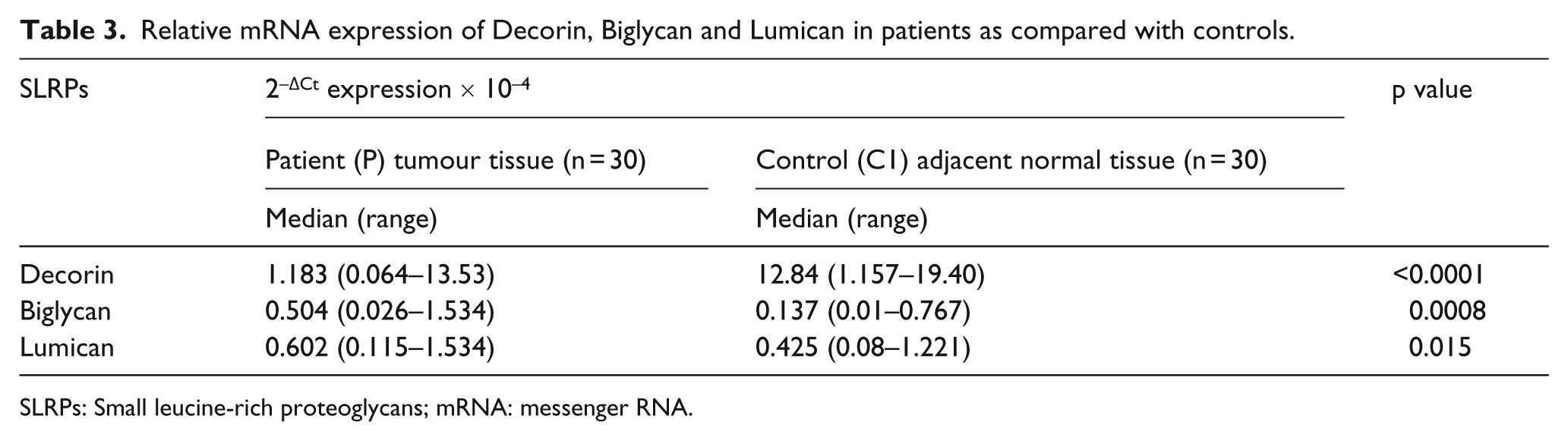
SLRPs: Small leucine-rich proteoglycans; mRNA: messenger RNA.

The box-and-whisker plot showing relative mRNA expression of (a) Decorin, (b) Biglycan and (c) Lumican are shown. The values are taken as median of the total study samples (2–ΔCt expression × 10–4).
Protein expression by IHC
IHC was performed on patient and control samples to study the expression of these proteoglycans at tissue level as shown in Figure 4. IHC was performed on 15 paired tumour and adjacent non-tumour specimens of bladder cancer patients. The tissue expression was quantified by using IRS as represented in Figure 5. Decorin, Biglycan and Lumican are proteoglycans situated in the ECM. In certain cases, cytoplasmic staining was also observed. In this article, we have highlighted the stromal expression of these macromolecules as shown in Figure 4. The expression for Decorin (p < 0.0001) was significantly lower in tumour tissue as compared with adjacent normal tissue. In case of Biglycan (p = 0.005) and Lumican (p = 0.002), the expression was significantly higher in tumour tissue as compared with adjacent non-tumour tissue.

Immunohistochemistry of the (a, c, e) urothelial carcinoma and (b, d, f) adjacent normal urothelium. Decorin (magnification: ×100) expression is shown in (a) and (b). Expression of Biglycan (magnification: ×100) in stromal tissue of urothelial carcinoma and normal urothelium is shown in (c) and (d). Significant Lumican (magnification: ×100) expression found in the stromal region of cancer tissue than in normal urothelium as shown in (e) and (f).

Immunoreactive score (IRS) of (a) Decorin, (b) Biglycan and (c) Lumican calculated by multiplying individual score (i.e. intensity score × percentage score) for analysis of protein expression by IHC.
Western blotting (immunoblotting)
Western blotting was performed on 15 paired tumour (T) and adjacent non-tumour (NT) specimens to study the protein expression of these proteoglycans at tissue level as shown in Figure 6. The tissue expression was quantified by densitometry using ImageJ software as shown in Figure 6(a)–(c). The expression for Decorin (p < 0.0001) was significantly lower in tumour as compared with non-tumour tissue. In case of Biglycan (p = 0.002) and Lumican (p = 0.005), the expression was found to be significantly higher in tumour as compared with non-tumour tissue.
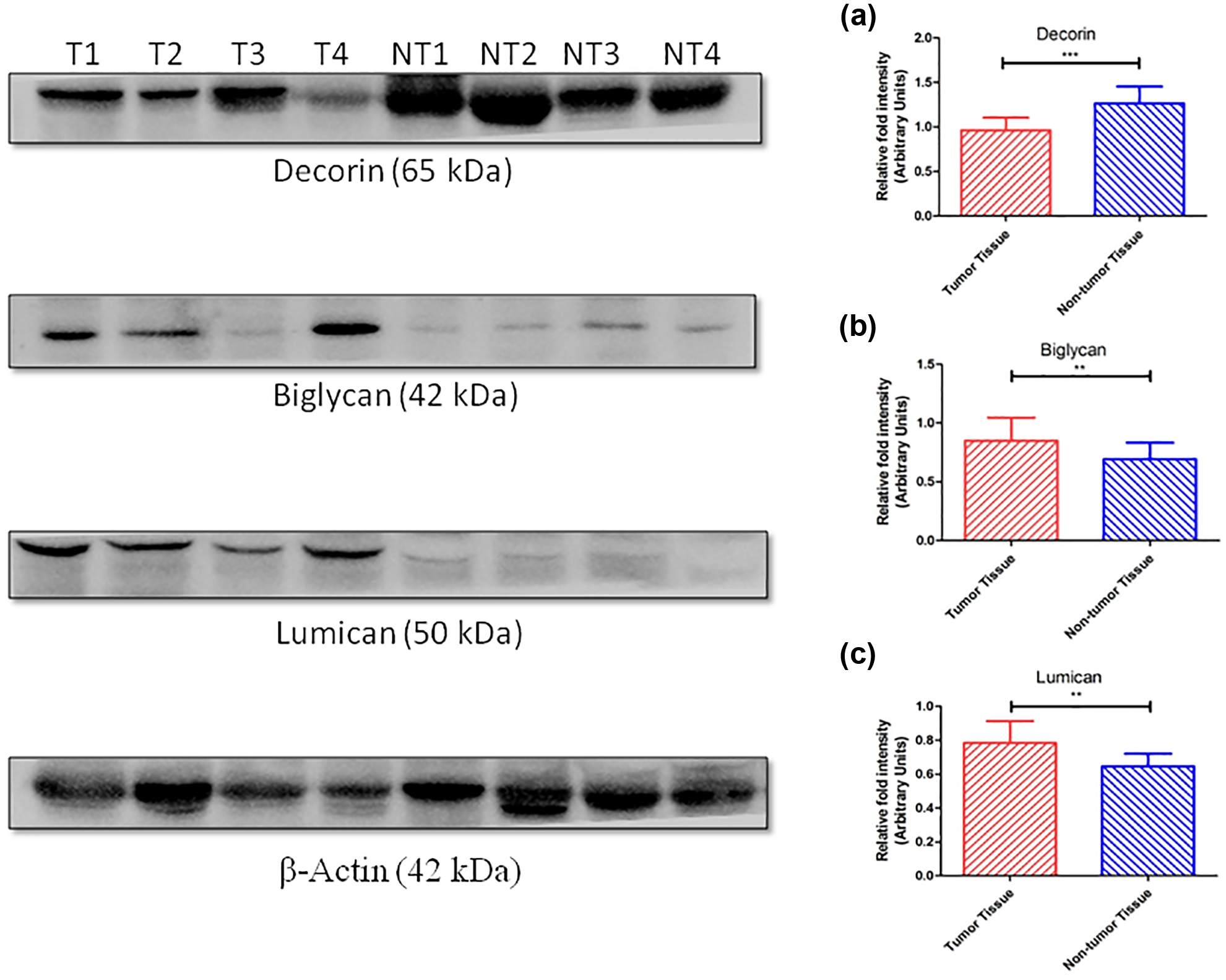
Western blotting of Decorin, Biglycan and Lumican in representative urothelial carcinoma and adjacent normal urothelium of bladder cancer patients. Densitometry analysis of respective molecule (a) Decorin, (b) Biglycan and (c) Lumican has also been done by using ImageJ software.
Discussion
SLRPs are increasingly becoming an important structure studied in the matrix due to its oncoregulatory function. They are present ubiquitously in the ECM. In this maiden study, we collectively aim to evaluate the expression of Decorin, Biglycan and Lumican and their plausible diagnostic potential in UBC. Decorin, a key member of the SLRPs, is known as the guardian of the matrix due to its oncosuppresive effect. 36 Decorin by its inhibitory effect on pan tyrosine kinase (including the ErbB family of kinases) activity decreases cellular proliferation. 37 Thus, it constantly checks for unregulated cell growth which is also observed by its lower expression in cancers. In our study, the circulatory level of Decorin demonstrated a significant reduction in UBC patient samples compared with controls. Very few studies are available on circulatory levels of Decorin in serum. Previously, Wu et al. 38 had shown that mean circulatory Decorin levels were lower in the esophageal squamous cell carcinoma (ESCC) patients compared with controls. We also observed a significantly lower relative mRNA expression of Decorin in tissue of the patients as compared to the controls. Leygue et al. 39 had shown in breast cancer and Suhovskih et al. 40 in prostatic cancer that the mRNA expression of Decorin was significantly lower in tumour tissue compared with normal tissue. On the contrary to the report published by El Behi et al. 41 that states overexpression of Decorin in muscle invasive bladder cancer, we observed a reduced protein expression of Decorin by IHC and western blotting in tumour tissue as compared with adjacent non-tumour tissue. Previously, Sainio et al. 42 had shown a similar low stromal Decorin expression in areas rich in bladder cancer cells as compared with non-malignant bladder tissue, and Oda et al. 43 had observed a reduced immunostaining in stroma of invasive cancer and in situ breast cancer as compared with normal breast tissue. Thus, our findings in bladder cancer for the expression of Decorin are in accordance with the previous studies done in prostate, breast, and oesophageal cancers. These findings highlight the reduced expression of Decorin in tumour microenvironment as compared with normal tissue. In contrast, only limited studies have shown an increased expression of Decorin in cancer as observed by Köninger et al. 44 in which there was a significantly higher expression of Decorin in pancreatic adenocarcinoma at mRNA level.
Biglycan, another class 1 SLRP, is different from Decorin only in having an extra chondroitin sulphate chain. In our study, the circulatory levels of Biglycan in patients were significantly elevated. Liu et al.23 observed a significantly higher circulatory levels of biglycan in endometrial carcinoma which correlated with poor prognosis. Also in our study, Biglycan mRNA fold expression and tissue Biglycan protein expression by IHC and western blotting in the tumour tissue were significantly higher than those in the adjacent normal tissue. Zhu et al.24 observed a twofold rise in the mRNA expression of biglycan in ESCC tissue as compared to their normal counterparts. This finding was also supported by showing a significantly higher immunostaining for Biglycan in ESCC. A single study by Niedworok et al. 45 in urinary bladder cancer had demonstrated an increase in Biglycan expression at mRNA level as well as at protein level by IHC. Thus, our results on Biglycan expression are also in accordance with the previous studies.
Lumican, an important SLRP, has recently been implicated in cancer biology. The circulatory protein levels of Lumican in our study reveal a significant increase along with expression by immunohistochemistry and western blotting in patients, but relative mRNA expression showed a moderately significant increase of Lumican in tumour tissue as compared with non-tumour tissue. Kashyap et al. 46 had shown an increased Lumican expression in stromal and cytoplasmic regions by IHC in ESCC, and Yang et al. 47 had shown an increased mRNA expression and tissue immunostaining in pancreatic adenocarcinoma. There is no report on serum Lumican levels in any cancer. To our knowledge, till now, there is no literature on Lumican circulatory levels, mRNA expression, and protein expression in urinary bladder cancer. On ROC curve analysis of these SLRPs, Lumican in serum showed optimal combinations of sensitivity and specificity. These findings suggest its usefulness as a potential novel diagnostic marker in UBC.
In conclusion, this study reveals that there is significant alteration in the expression of some SLRPs in the matrix of patients with UBC. Out of three molecules (Decorin, Biglycan and Lumican) studied, Lumican shows potential involvement which is reflected by its significantly higher protein expression in bladder cancer patients. ROC curve of Lumican provided the best combination of sensitivity (90.0%) and specificity (90.0%) for the cut-off of 0.207 ng/mL. Our findings provide an insight into the utility of Lumican as a plausible biomarker in bladder cancer which can be further validated by studying in a larger patient cohort for determining the usefulness in diagnosis and/or prognosis of UBC.
Footnotes
Acknowledgements
S.A. and V.A. contributed equally to this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author(s) received no financial support for the research, authorship and/or publication of this article.
