Abstract
Introduction
Cervical carcinoma (CC) is the third-leading contributor to disability-adjusted life years (DALYs) in females worldwide. 1 Human papillomavirus (HPV) vaccination is used for primary prevention. 2 According to the NCCN Guidelines for Cervical Cancer, patients with early-stage cervical cancer are primarily treated by surgery or radiation therapy (RT), and those with advanced cervical cancer are primarily undergoing chemoradiation (weekly cisplatin given during RT). RT is one of the most common interventions, especially for locally advanced cervical squamous cell carcinoma (LACSCC) in patients. However, resistance is a major therapeutic problem for RT. 3 Enhancing the sensitivity of radiotherapy and reducing the dose of radiotherapy would help the patients to obtain better survival rate and reduce unwanted side effects. Therefore, the identification of new markers to predict radioresistance and the development of an optimal treatment strategy for individual patients who are likely to exhibit resistance are important goals.
In recent years, great researches showed that many factors are associated with RT sensitivity in cervical cancer, including apoptotic proteins, COXs, epidermal growth factor receptor (EGFR), vascular endothelial growth factor (VEGF), and HIF-1α and affect the RT response on apoptosis, tumor growth, angiogenesis, and hypoxia. 4 However, to date, the correlation of several apoptotic proteins such as BCL2, BAX, and p53 with chemoradiotherapy is still controversial. Many candidates, such as HPV status, pAKT, cyclooxygenase, HIF-1α, VEGF, nuclear factor κB (NF-κB), Ku 80, EMMPRIN, and surviving, were detected by immunohistochemical studies and were shown relevant to RT. But, most studies, gene sets vary from study to study. There are still no maturated genetic researches of radiosensitivity and radioresistance to clinical practices. 5
Kruppel-like factor 4 (KLF4) is a zinc finger–type transcription factor that is a member of the evolutionarily conserved specificity protein (Sp)/KLF family of proteins, which are characterized by the presence of zinc finger motifs in their C-terminal domains. 6 KLF4 plays important role in physiological cell processes, including proliferation, apoptosis, and differentiation.6–8 KLF4 functions as a tumor suppressor gene in neuroblastoma, lung cancer, gastric cancer, ovarian cancer, pancreatic cancer,9–13 and colon cancer. 5 Moreover, KLF4 can function as an oncogene in breast cancer. 14
KLF4 also plays an important role in the patient response to RT. In a mouse model of gastrointestinal (GI) syndrome, mice treated with γ-irradiation were protected from radiation-induced gut injury because KLF4 restrained apoptosis during the acute reaction. 15 Moreover, KLF4 modulates the apoptotic response following γ-irradiation by activating the expression of the cell cycle arrest gene p21 and inhibiting the ability of p53 to transactivate expression of the pro-apoptotic gene BAX. 16 Accumulating evidence suggests that KLF4 functions as a tumor suppressor gene in CC; 17 however, the effect of KLF4 expression on the response to radiation is unclear.
The aim of this study was to investigate the potential use of KLF4 expression in LACSCC tissues as clinical biomarkers of tumor RT resistance and prognosis in LACSCC patients. Immunohistochemistry was used to evaluate KLF4 expression in tissue specimens from LACSCC patients.
Materials and methods
Patients and clinical tissue specimens
This study was approved by the Research and Ethics Committee at The Fourth Military Medical University, Xi’an, China. Medical records were obtained from the Institutional Review Board of Xijing Hospital at The Fourth Military Medical University. At first, 121 patients had received primary radical RT, including 117 LACSCC and only 4 cervical adenocarcinoma. To reduce statistical error, we removed the cervical adenocarcinoma cases. This is a retrospective cohort study, so the cases in which information are incomplete or patients refused to follow-up were not included in the study. According to the NCCN Guidelines for Cervical Cancer, patients of IIb–IVa in our hospital need to receive primary radical RT. The patients of stage IIb–IVa were included in this study. From January 2008 to December 2011, 117 patients who received primary radical RT for LACSCC at the Department of Radiation Oncology, Xijing Hospital were enrolled in the study. The inclusion criteria are as follows: (1) patients had LACSCC based on histopathological observations, (2) patients were treated with strategies involving brachytherapy with doses greater than 40 Gy at point A combined with chemotherapy with cisplatin (single-agent cisplatin given weekly), (3) patients had no evidence of distant metastases (International Federation of Gynecology and Obstetrics (FIGO) stages IIb–IVa), and (4) patients had undergone no other cancer treatments before primary RT or surgery after RT. Actually, only one patient was not treated with brachytherapy due to external beam radiation therapy (EBRT) with highly conformal boosts of an additional 16 Gy to RT dosage. At present, radical radiotherapy is mostly combined with concurrent cisplatin-based chemotherapy. The dosage of cisplatin as sensitizer did not reach the therapeutic dose. Radical radiotherapy with concurrent cisplatin-based chemotherapy is an established treatment for cervical cancer patients with stage FIGO IIB and higher. The mean age of the patients was 49.75 ± 8.21 (median: 48; range: 25–69) years. A total of 39 patients had stage II LACSCC, 74 patients had stage III CC, and 4 patients had stage IVa CC.
The follow-up ended in October 2015. The median follow-up time for the survivors was 45 (range: 2.22-78.2) months. The median progression-free survival (PFS) time was 43.5 (range: 2.3-78.2) months.
The patients were divided into a radiation-sensitive group (n = 72) and a radiation-resistant group (n = 45). The patients in the radiation-sensitive group had no local recurrences or distant metastases for at least 3 years (≥36 months) after their primary treatment. The patients in the radiation-resistant group had tumors that did not respond to RT at all or experienced local recurrences or distant metastases less than 3 years (<36 months) after their primary treatment. We further divided the radiation-resistant group into an RT non-response subgroup (n = 6), a local-recurrence subgroup (n = 19) comprising patients whose primary tumor had initially disappeared but subsequently recurred locally less than 3 years after the primary treatment, and a distant-metastasis subgroup (n = 20) comprising patients whose primary tumor had disappeared after treatment but then presented with distant metastasis more than 3 years after the primary treatment.18,19
Immunohistochemistry
Immunohistochemical staining was performed on 4-µm tissue microarray sections of formalin-fixed and paraffin-embedded tissue samples as follows: (1) the slices were deparaffinized in xylene and dehydrated with a graded alcohol series, (2) endogenous peroxidase activity was quenched with 0.3% hydrogen peroxide, (3) antigen retrieval was performed in 0.01 mol/L citric acid buffer, (4) nonspecific binding was blocked with 10% normal goat serum at 37°C for 30 min, (5) overnight incubation at 4°C was performed with an anti-human monoclonal antibody (KLF4, 1:300 dilution; Abcam, Cambridge, MA, USA), (6) a 30-min incubation at 37°C was performed with a horseradish peroxidase–labeled anti-rabbit IgG antibody (1:500 dilution; Zhongshan, Beijing, China); and, finally, (7) the sections were incubated with standard 3′,3-diaminobenzidine (DAB) reagents (Zhongshan) and lightly counterstained with hematoxylin.
Evaluation of staining
The staining was separately evaluated under a microscope by two independent pathologists; if their opinions did not agree, the opinion of a third independent pathologist was solicited. The intensity of KLF4 tissue staining was scored as follows: (1) by the percentage of positively stained cells (≤5% scored as 0, 6%–25% scored as 1, 26%–50% scored as 2, 51%–75% scored as 3, and >75% scored as 4) and (2) by the intensity of the stain (colorless scored as 0, weak (pallide-flavens) scored as 1, moderate (yellow) scored as 2, and strong (brown) scored as 3). The final score of each tissue was calculated by multiplying the scores from (1) and (2). The staining score was stratified as follows: − (0 score, absent), + (1–4 score, weak), ++ (5–8 score, moderate), and +++ (9–12 score, strong). 20 We grouped all of the samples into a low-expression group (overall score of − or +) and a high-expression group (overall score of ++ or +++) according to the overall protein expression levels.
Statistical analyses
The SPSS 20.0 software (IBM, Chicago, IL, USA) was used to analyze the data. A chi-square test, Fisher’s exact test, or t test was used as appropriate to evaluate the correlations between immunohistochemical expression and the clinical variables. Univariate and multivariate survival analyses were performed according to the Cox proportional hazards model. The level of KLF4 expression alone (high vs low), patient age (≥50 vs <50 years), FIGO stage (III + IVa vs II), differentiation grade of the sample (middle + low vs high), tumor diameter (>4 vs ≤4 cm), and tumor shape were included in the regression model. p < 0.05 was considered significant for all statistical tests.
Results
Clinical characteristics of radiation-sensitive and radiation-resistant patients
A total of 117 LACSCC patients were divided into a radiation-resistant group (n = 45) and a radiation-sensitive group (n = 72). The clinical characteristics of the patients are detailed in Table 1. Significant differences were observed in the FIGO stage (p = 0.005), tumor diameter (p = 0.024), and differentiation grade (p = 0.022) between the radiation-resistant and radiation-sensitive groups, but no significant differences in the patient age and tumor shape, total dose at point A, EBRT dose at point A, and BRT dose at point A were found between the two groups.
Patient characteristics.

EBRT: external beam radiation therapy; FIGO: International Federation of Gynecology and Obstetrics.
p value was estimated by the chi-square test.
p value was estimated by the t test.
KLF4 expression in LACSCC tissues and their association with clinicopathological parameters
KLF4 expression was also mostly localized in the nucleus, and the staining was substantially higher in the radiation-resistant group than in the radiation-sensitive group (Figure 1(a)–(c)). Of the enrolled patients, 54.7% (64/117) had low KLF4 expression and 45.3% (53/117) had high KLF4 expression (Table 2). Significant associations were detected between KLF4 expression and the FIGO stage (p = 0.026) and differentiation grade (p = 0.011), but no significant associations were observed between KLF4 expression and the patient age, tumor diameter, and tumor shape.
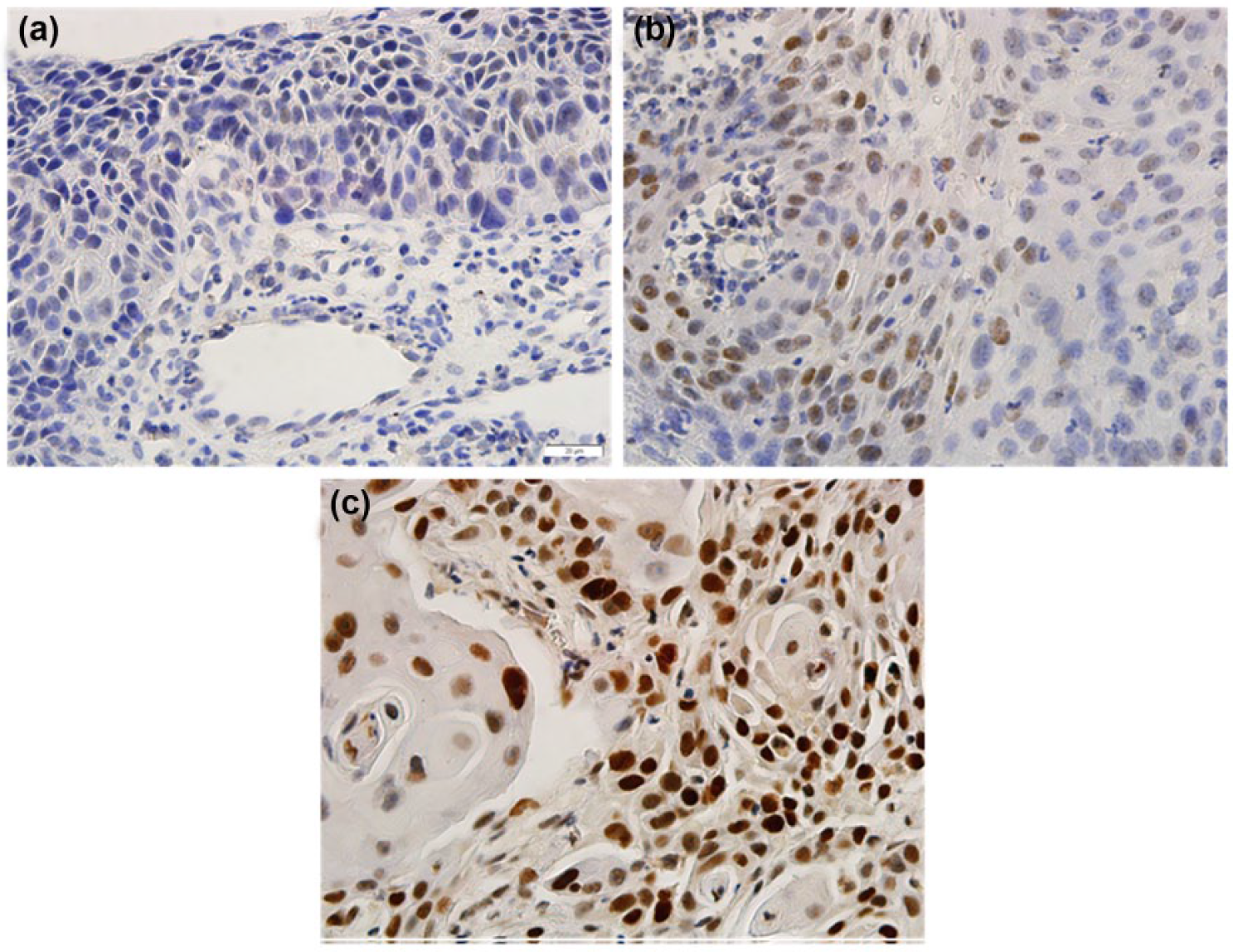
Immunohistochemical micrographs of KLF4 expression in cervical cancer (×400): (a) negative, (b) low, and (c) high. Scale bar: 20 µm.
Correlation between KLF4 expression and clinicopathological parameters.

KLF4: Kruppel-like factor 4; FIGO: International Federation of Gynecology and Obstetrics.
KLF4 expression and the response to radiotherapy
A significant difference in KLF4 expression level was observed between the radiation-resistant and radiation-sensitive groups (p = 0.032; Table 3). The percentages of patients with low and high KLF4 expression were 42.2% (19/45) and 57.8% (26/45), respectively, in the radiation-resistant group and 62.5% (45/72) and 37.5% (27/72), respectively, in the radiation-sensitive group (Table 3). Based on the clinical information obtained for each patient, we further divided the radiation-resistant group into RT non-response subgroup, a local-recurrence subgroup, and a distant-metastasis subgroup. The frequencies of patients with high KLF4 expression were 83.3% (5/6), 68.4% (13/19), and 65% (13/20) in the RT non-response subgroup, local-recurrence subgroup, and distant-metastasis subgroup, respectively. A significant difference in KLF4 expression was observed in RT non-response, local-recurrence, and distant-metastasis subgroups (p = 0.04, p = 0.016, and p = 0.028).
Relationship between KLF4 expression and the response to radiotherapy.
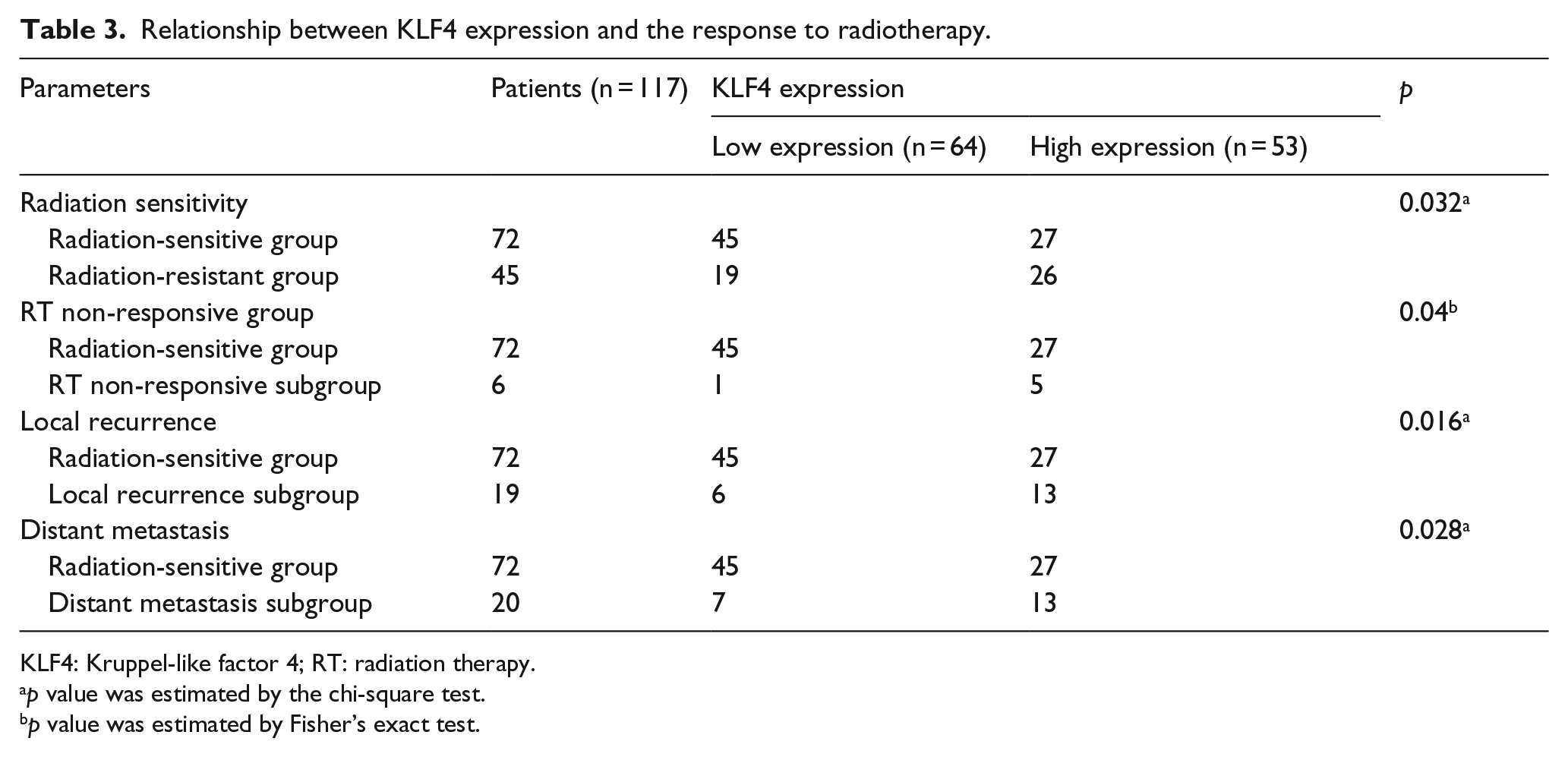
KLF4: Kruppel-like factor 4; RT: radiation therapy.
p value was estimated by the chi-square test.
p value was estimated by Fisher’s exact test.
KLF4 expression and survival
We examined the relationship between KLF4 expression and the prognostic outcomes in LACSCC patients. As shown in Figure 2, the Kaplan–Meier analysis predicted a shorter post-radiotherapy PFS time for patients with high KLF4 expression (n = 53) than patients with low KLF4 expression (n = 64) (p = 0.0019; Figure 2). Similarly, we found that patients with high KLF4 expression had significantly shorter overall survival (OS) than patients with low KLF4 expression (p < 0.0017; Figure 2). This result suggests that high KLF4 expression is significantly associated with an increased risk of death.

Kaplan–Meier survival curves according to the KLF4 protein expression statuses for cervical cancer patients. (a) The progression-free survival (PFS) times were significantly shorter in the high KLF4 expression low KLF4 expression and (b) the overall survival (OS) times were significantly shorter in patients with high KLF4 expression than in patients with low KLF4 expression.
To identify factors that affected PFS and OS, the clinical factors listed in Table 4, the KLF4 expression, age, FIGO stage, differentiation grade, tumor diameter, and tumor shape were included in the Cox regression analysis. The univariate Cox regression analysis revealed that KLF4 expression (hazard ratio (95% confidence interval (CI)): 0.581 (0.405–0.832) p = 0.003), FIGO stage (hazard ratio (95% CI): 0.467 (0.277–0.787) p = 0.004), differentiation grade (hazard ratio (95% CI): 0.648 (0.428–0.983) p = 0.041), and tumor diameter (hazard ratio (95% CI): 2.293 (1.117–4.709) p = 0.024) were significantly associated with PFS and KLF4 expression (hazard ratio (95% CI): 0.579 (0.404–0.83) p = 0.003), FIGO stage (hazard ratio (95% CI): 0.452 (0.268–0.762) p = 0.003), differentiation grade (hazard ratio (95% CI): 0.638 (0.421–0.967) p = 0.034), and tumor diameter (hazard ratio (95% CI): 1.54 (1.075–2.207) p = 0.019) was significantly associated with OS among the LACSCC patients. Multivariate Cox regression analysis revealed that KLF4 expression (hazard ratio (95% CI): 0.520 (0.359–0.752) p = 0.001), FIGO stage (hazard ratio (95% CI): 0.478 (0.278–0.821) p = 0.007), and differentiation grade (hazard ratio (95% CI): 0.622 (0.401–0.965) p = 0.034) were significantly associated with PFS among the LACSCC patients. However, KLF4 expression (hazard ratio (95% CI): 0.503 (0.348–0.727) p = 0.000), FIGO stage (hazard ratio (95% CI): 0.460 (0.269–0.789) p = 0.005), differentiation grade (hazard ratio (95% CI): 0.607 (0.392–0.938) p = 0.025), and tumor diameter (hazard ratio (95% CI): 1.479 (1.006–2.175) p = 0.047) were significantly associated with OS.
Univariate and multivariate Cox regression analyses of the relationships between the clinicopathological outcomes in patients.
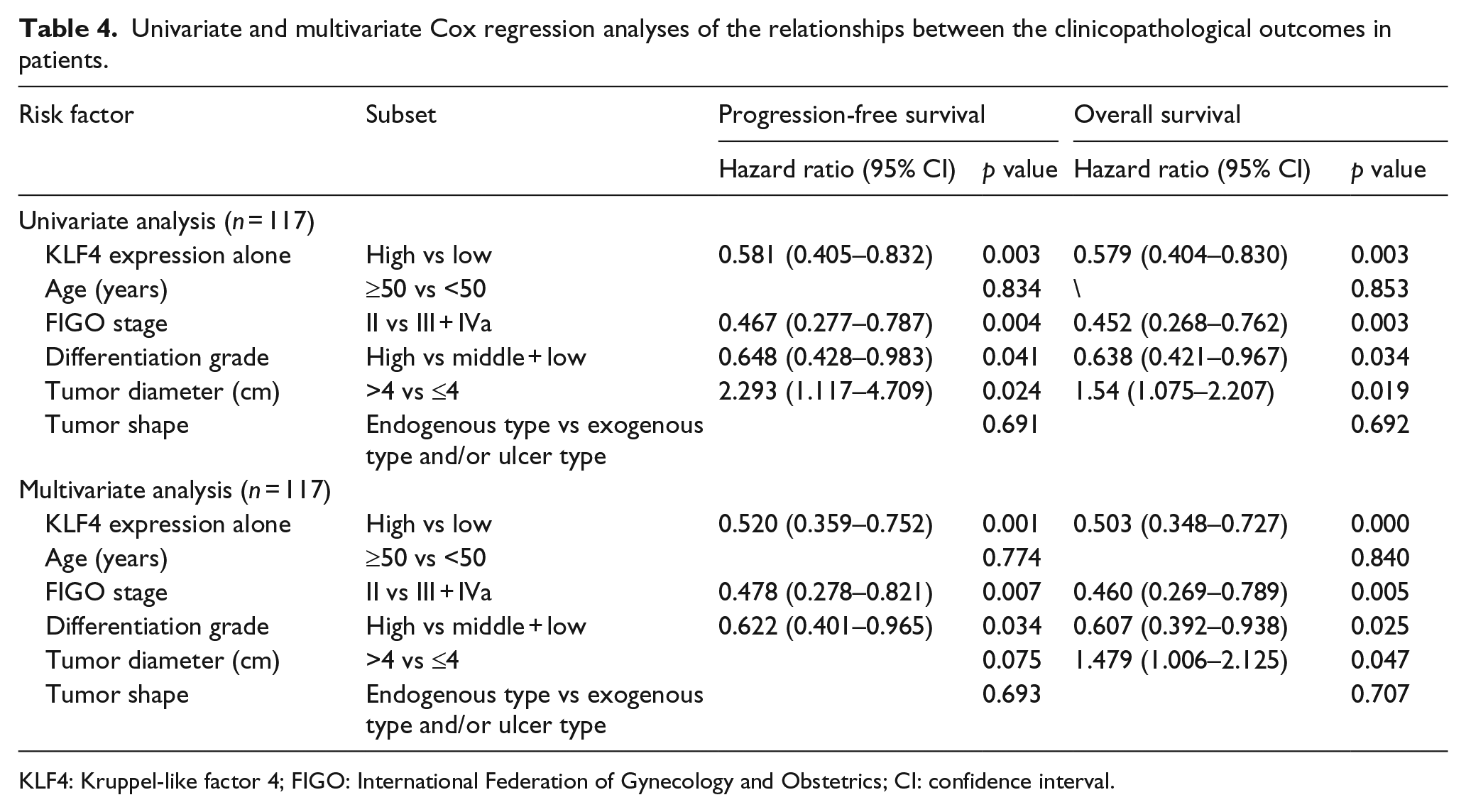
KLF4: Kruppel-like factor 4; FIGO: International Federation of Gynecology and Obstetrics; CI: confidence interval.
Discussion
Radiotherapy plays a key role in the treatment of LACSCC; however, resistance to radiation is a significant problem for treatment failure. 4 According to the literature, 5 radical radiotherapy is a major treatment for FIGO IIB and higher stage cervical cancer patients. The cervical cancer in 40%–80% patients can be controlled after RT. The risk percentage of late postradiation complications with the treatment is about 10%–15%. Thus, detection of molecular markers predicting tumor response to radiotherapy can individualize radiotherapy, help in making treatment decision, and avoid unnecessary postradiation complications. The mechanism underlying radiation resistance is still unclear; therefore, identifying clinical biomarkers that can predict the response to radiotherapy is an important goal. Currently, biomarkers are unavailable in the clinic. In this study, we analyzed KLF4 expression in radiation-resistant and radiation-sensitive LACSCC patient groups and its significance in the radiotherapy outcome. We demonstrated that KLF4 expression correlated with the radiation response in the LACSCC tissues. Importantly, we also assessed the value of KLF4 as prognostic factors to predict radioresistance and to optimize the treatment strategy.
As a zinc finger transcription factor, KLF4 plays important roles in cellular proliferation, differentiation, reprogramming, and the cellular response to oncogenic signals and ionizing radiation. KLF4 can function as a tumor suppressor gene or an oncogene in different cellular contexts. 21 KLF4 functions as a tumor suppressor gene, and KLF4 expression is reduced in different types of cancers. KLF4 expression is decreased in LACSCC tissues and cells compared with normal cervical tissues and functions as a tumor suppressor gene.17,22 Ghaleb et al. 16 demonstrated that KLF4 played an anti-apoptotic role in response to irradiation injury in vivo and might also aid in cell survival secondary to irradiation through either its association with DNA repair, reduced cell death, increased regeneration or all three processes. 15 Kuruvilla et al. 23 showed that KLF4 played a critical role in modulating the BMI1+ intestinal stem cell fate during both homeostasis and the regenerative response to radiation injury. Zhang et al. 24 suggested that RT caused KLF4 overexpression and then induced more double-strand breaks (DSBs) and reduced single-strand breaks (SSBs), which would explain the adverse effects of RT against mouse brain tumors.
Radical radiotherapy with concurrent cisplatin-based chemotherapy is an established treatment for cervical cancer patients with stage FIGO IIb and higher. At present, radical radiotherapy is mostly combined with concurrent cisplatin-based chemotherapy. The dosage of cisplatin as sensitizer did not reach the therapeutic dose. In this study, all patients underwent radical radiotherapy. In our study, first, we found that KLF4 expression was associated with the FIGO stage and differentiation grade. Fadous-Khalifé et al. 25 suggested that KLF4 expression was associated with FIGO stage in non–small-cell lung cancer. But in gastric adenocarcinoma, no association of KLF4 expression with stage and grade was found. 26 Furthermore, consistent with data from previous studies,15,16 our clinical data suggest that KLF4 expression was shown to be associated with radioresistance when the radiation-resistant group was divided into three subgroups (Table 3). The KLF4 expression was associated with the response to RT in each subgroup. This result suggests that KLF4 expression in LACSCC is associated with RT non-response, local recurrence, and distant metastasis and plays a central role in radioresistance. Then, the survival analysis showed that LACSCC patients with high KLF4 expression exhibited significantly poorer PFS and OS than patients with low KLF4 expression after RT. It has been reported that loss of KLF4 expression led to shorter OS and PFS in colon cancer and gastric adenocarcinoma. 26 In our study, the multivariate Cox regression analysis demonstrated that KLF4 expression might be an independent prognostic predictor for CC. KLF4 expression, FIGO stage, differentiation grade, and tumor diameter would be the risk factor for OS. Interestingly, KLF4 expression, FIGO stage, and differentiation grade would be the risk factor for PFS, but tumor diameter would not be the risk factor for PFS.
We also detected the expression of KLF4 in CC cell lines (HeLa, Caski, SiHa, and C33A) using western blot and quantitative real-time polymerase chain reaction (qRT-PCR) analysis and compared the sensitivity of CC cells to radiotherapy by flow cytometry and plate cloning (data not shown). These results suggested that high expression of KLF4 promoted radioresistance. KLF4 induces the expression of p27 and, to a lesser extent, the expression of p21 and function as a tumor suppressor by inhibiting tumor growth. KLF4 synergistically induces the expression of p21, leading to cell cycle arrest and to suppress BAX expression, thus reducing apoptosis. 16 It may explain the discrepancy of the roles of the KLF4. Next, we will further study the specific mechanism of the differences’ discrepancy. The limitations of this study are that the clinical data are not large and the patient information is not a source of multiple centers. Moreover, analysis of expression by immunochemistry is not sufficient to ratify such an effect. In the future, we will further study how KLF4 specifically affects radiotherapy sensitivity from prospective cohort studies and molecular mechanisms research and will help to confirm this finding.
Conclusion
In summary, our results demonstrated that KLF4 might be involved in radioresistance in LACSCC, and that nuclear or nuclear/cytoplasmic KLF4 expression might be related to the pathological type and radioresistance. The high KLF4 expression was significantly associated with poor survival and was an independent predictor of PFS and OS in LACSCC patients; KLF4 expression was associated with radioresistance and was a valuable prognostic factor for LACSCC patients’ response to RT.
Footnotes
Acknowledgements
The authors thank Professor Li-Chun Wei and her team at the Department of Radiation Oncology, Xijing Hospital, for information and technical assistance. They thank members of The State Key Laboratory of Cancer Biology, Biotechnology Center, School of Pharmacy, The Fourth Military Medical University, for discussions. H-X.L. and N.L. performed and analyzed most of the experiments and wrote the manuscript; these authors contributed equally to this work and are joint first authors. B-L.C. and H.S. conceived and designed the project; these authors are co-corresponding authors. L.W. and Y-P.H. performed the histopathological analysis. R.M. and F-X.Z. collected the clinical information. F.X. performed the tumor radiosensitivity studies. W.Z. and Y.Z. analyzed the data.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Young Scientists Fund of the National Natural Science Foundation of China (81301921) and by a scientific research grant from the Ministry of Shaanxi Province, China (2013JM4023).
Informed consent
Informed consent was obtained from all individual participants included in the study.
Research involving human participants and/or animals
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. For this type of study, formal consent is not required.
