Abstract
MiR-30 family plays an important role in the tumorigenesis of human cancers. The aim of the study is to investigate the role of miR-30d in human colon cancer cell lines and explore the molecular mechanism in the proliferation of colon cancer cells. The expression of miR-30d was determined by real-time polymerase chain reaction assay in colon cancer cell lines (HCT15, HCT116, HT-29, DLD-1, and SW480) and the results demonstrated that miR-30d level was significantly decreased in human colon cancer cell lines, compared with normal colon epithelial cell line. Transfection with miR-30d mimics inhibited cell proliferation, and transfection with miR-30d inhibitors significantly promoted cell viability of colon cancer cells. Furthermore, TargetScan analysis predicted that miR-30d interacted with messenger RNA on its 3′ untranslated region of ATG5, phosphoinositide 3-kinase, and Beclin1 to negatively regulate cell autophagy in colon cancer cells. Moreover, transfection with miR-30d induced cell arrest at G2/M phase of HT-29 cells. Overexpression of miR-30d mimics inhibited cell viability probably due to the inhibition of cell autophagy and promotion of cell apoptosis. Thus, MiR-30d inhibited cell autophagy by directly targeting messenger RNA of ATG5, phosphoinositide 3-kinase, and Beclin1 and promoted cell apoptosis of human colon cancer cells. It is helpful to clarify the function of miR-30d in tumorigenesis of human cancers.
Introduction
Colorectal cancer is the third most common diseases in the world, which is characterized by abnormal and malignant growth of cells in colon or rectum parts. Colorectal cancer cells normally invade or spread to the other parts of human body.1,2 The incidence of colorectal cancer is higher in developed countries versus developing countries. In developed countries, more than 65% of cases are colorectal cancers. 3 The commonly used treatments for colon cancer are surgery, chemotherapy, radiation therapy, and targeted therapy. It is helpful and beneficial to understand the biology of colorectal cancer in the clinical therapy and prognosis of malignant tumors. 4
MicroRNAs (miRNAs) are a kind of small non-coding RNAs with 20–22 nucleotides length, which regulate various physiological processes.5,6 MiRNAs are reported to be dysregulated in the progression and invasion of various human cancers and play key roles in cell differentiation, proliferation, and cell death by interacting with messenger RNA (mRNA) on the 3′ untranslated region (3′-UTR) sequence of specific targeted proteins. 7 They play an important role as either tumor suppressor or oncogene by participating in epigenetic modification and regulation of the targeted proteins.8,9 MiRNAs have the potential to be used as a biomarker in the clinical therapy of human cancers. 10 MiR-30 family are reported to be involved in the progression and invasion of various human cancers. MiR-30d was reported to be as a novel independent prognostic marker of prostate cancer and as a reference for clinicians to adopt more intensive treatments. 11 Jia et al. have found that expression levels of miR-30d-5p in plasma were higher in patients with acute coronary syndrome (ACS) compared with that in healthy controls (p < 0.001). MiR-30d-5p showed a diagnostic value for early diagnosis of acute myocardial infarction (AMI). 12 The miR-30d upregulation was correlated with stage, metastatic potential, shorter time to recurrence, and reduced overall survival of a highly invasive cancer, human melanoma. 13 Moreover, they also found that miR-30d overexpressed in prostate cancer cells and the overexpression of miR-30d promoted proliferation and invasion in vitro. In human epithelial cancers, miR-30d serves as an oncomir by inhibiting autophagosome formation and LC3B-I conversion to LC3B-II. 14 Conversely, miR-30d was identified as a tumor suppressor and acted as a new potential target for the treatment of renal cell carcinoma through a new Akt/transcription factor forkhead box O 3A (FOXO3A)/miR-30d/MTDH signaling transduction pathway. 15 Zhang et al. 16 also observed that miR-30d level was remarkably decreased in human anaplastic thyroid carcinoma (ATC) and involved in modulating the sensitivity of ATC cells to chemotherapeutic drug, such as cisplatin by negatively regulating the level of beclin1, a key autophagy gene.
In human colorectal cancer cells, miR-30d, miR-181a, and miR-199a-5p were predicted to target GRP78 and the expression was decreased in prostate, colon, and bladder tumors; 17 however, the role of miR-30d was not clearly clarified in human colorectal cancers. In this study, we detected the expression level of miR-30d in human colorectal cancer cells and further investigated the function and molecular mechanism of miR-30d in human colorectal cancer cells, which could provide potential theoretical reference in the clinical therapy for human malignant colorectal tumors.
Materials and methods
Cell lines and agents
FHC (ATCC® CRL-1831™), a normal colon epithelial cell line, was obtained from ATCC. The colorectal cancer cell lines, HCT15 (ATCC® CCL-225™), HCT116 (ATCC® CCL-247™), HT-29 (ATCC® HTB-38™), DLD-1 (ATCC® CCL-221™), and SW480 (ATCC® CCL-228™) were also purchased from American Type Culture Collection (ATCC, Manassas, VA, USA). The cells were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM) medium containing 10% fetal bovine serum (FBS) at 37°C in a humidified 5% CO2 atmosphere. DMEM was purchased from Gibco (Gaithersburg, MD, USA). 3-Methyladenine (3-MA; Cat. No. M9281) and rapamycin (Cat. No. 37094) were obtained from Sigma (St. Louis, MO, USA). 3-MA inhibits autophagy by blocking autophagosome formation via the inhibition of type III phosphoinositide 3-kinases (PI3Ks). In the experiment, 3-MA was used at a concentration of 5 mM to block cell autophagy. Rapamycin was used as an autophagy activator, and the final concentration of rapamycin was 100 n M.
Cell transfection
Hsa-miR-30d miRNA mimic (Cat. No. MCH01699) and miRNA mimic negative control (Cat. No. MCH00000) were purchased from Applied Biological Materials Inc. (Richmond, British Columbia, Canada), which are chemically modified, double-stranded miRNA-like RNA, and designed to copy the functionality of mature endogenous miRNA upon transfection. Hsa-miR-30d miRNA inhibitor (Cat. No. MIH01699) is chemically synthesized, complementary, anti-sense single-stranded oligonucleotides to their target, and endogenous mature miRNA. The inhibitor negative control (Cat. No. MIH000000) is also purchased separately from Applied Biological Materials Inc. The HT-29 and HCT116 cells were transfected with hsa-miR-30d mimics and miRNA mimic negative control and cultured for 24, 48, and 72 h at 37°C in a CO2 incubator. DLD-1 and HCT15 cells were transfected with hsa-miR-30d inhibitors and the inhibitor negative control for 24, 48, and 72 h at 37°C in a CO2 incubator. The transfection agent was Lipofectamine™ RNAiMAX (Invitrogen, Carlsbad, CA, USA) and the colorectal cancer cells were transfected according to protocols.
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay
The human colorectal cancer cells were transfected with hsa-miR-30d mimics and miRNA mimic negative control or hsa-miR-30d inhibitors and the inhibitor negative control using Lipofectamine™ RNAiMAX. The untreated colorectal cancer cells were used as negative controls. The cells were cultured at 37°C in a 5% CO2 incubator for 24, 48, and 72 h. The cell viability was determined by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. Briefly, 10 µL of MTT (5 mg/mL) was added into the cultured medium and cultured for 4 h. A volume of 100 µL of dimethyl sulfoxide (DMSO) was added to dissolve the purple crystal. The absorbance of each well was read at 490 nm with a microplate reader.
Real-time polymerase chain reaction analysis
Total RNA was extracted from colorectal cancer cells with TRIzol reagent (Tiangen, Beijing, China) according to the manufacturer’s instructions. PrimeScript miRNA qPCR Starter Kit (TaKaRa Bio Inc., Kusatsu, Japan) was used according to manufacturer’s protocol. Quantitative real-time polymerase chain reaction (RT-PCR) was performed using SYBR Green fluorescence and the LightCycler Real-Time PCR cycler (Roche, Valencia, CA, USA). The U6 RNA served as an internal control for normalization. The primers used for U6 were listed as follows: 5′-TGCGGGTGCTCGCTTCGCAGC-3′ (forward) and 5′-CCAGTGCAGGGTCCGAGGT-3′ (reverse). The primers for miR-30d were as follows: 5′-CATTTTTGTTTCAAGATGTCAC-3′ (forward) and 5′-GAGAGCACCCTACATTCAAC-3′ (reverse).
Western blotting analysis
The colorectal cancer cells were collected and the samples were lysed using radioimmunoprecipitation assay (RIPA) buffer. The total proteins were separated by polyacrylamide gel electrophoresis (PAGE) as previously described.18–20 The antibodies used were as follows: anti-caspase-3 antibody (Cat. No. ab44976) and anti-active caspase-3 antibody (Cat. No. ab2302) are rabbit polyclonal caspase-3 and are obtained from Abcam (Cambridge, MA, USA). Poly (ADP-ribose) polymerase (PARP) antibody #9542 is a rabbit antibody and detects endogenous levels of full-length PARP1 with molecular weight of 116kDa, which is purchased from Cell Signaling Technology (Danvers, MA, USA). Anti-Cleaved PARP antibody (ab4830) is a rabbit polyclonal cleaved PARP and was purchased from Abcam. ATG5 antibody (Cat. No. NB110-53818) is a polyclonal rabbit ATG5 and was purchased from Novus Biologicals (Littleton, CO, USA). Anti-PI3K p85 antibody (6G10) (ab189403) is mouse monoclonal (6G10) PI3K p85 and was purchased from Abcam. Beclin1 antibody (Cat. No. Q14457) is a rabbit monoclonal antibody and was obtained from Abgent (San Diego, CA, USA). Anti-LC3 (Cat. No. L8918) is obtained from Sigma-Aldrich (St. Louis, MO, USA). The antibody recognizes human and rat LC3A/B-I and LC3A/B-II by immunoblotting and immunoprecipitation, respectively. Anti-β-actin antibody was purchased from TransGen Biotechnology (Beijing, China).
Flow cytometry analysis for cell cycle
Colon cancer cells were plated onto six-well plate. After 6 h of culture, the colon cancer cells were transfected with miR-30d mimics and negative control mimics for 48 h. The cells were digested with trypsin–ethylenediaminetetraacetic acid (EDTA) and washed with phosphate-buffered saline (PBS) twice. Then, the colon cancer cells were fixed with ice-cold 70% ethanol for 1 h at 4°C. The fixative liquid was removed by centrifuging at 1000 r/min for 10 min. Next, the cells were treated with stain buffer (50 µg/mL propidium iodide (PI), 100 µg/mL RNase A, and 0.1% Triton X-100 in PBS) at 37°C for 15 min. DNA contents of cells were subjected to a BD Biosciences FACScan flow cytometer (BD Biosciences, San Jose, CA, USA).
Statistical analysis
The data were analyzed with SPSS 20.0 software (IBM SPSS, Armonk, NY, USA). The data were analyzed by Student’s t test and presented as mean ± standard deviation (SD). *p < 0.05 was considered to be statistically significant.
Results
The expression of miR-30d is significantly decreased in human colon cancer lines
To explore whether miR-30d was aberrantly expressed in colon cancer cells, RT-PCR assay was performed to determine the levels of miR-30d in human colon cancer cell lines, including HCT15 cells, HCT116, HT-29, DLD-1, and SW480 cells. FHC is a normal colon epithelial cell line and used as normal control cell line. As shown in Figure 1, the level of miR-30d was remarkably decreased in colon cancer cell lines compared to the normal colon cells, suggesting that miR-30d might work as a tumor suppressor in the progression of human colon cancers.

The expression of miR-30d is significantly decreased in human colon cancer lines. The colon cancer cells including HCT15, HCT116, HT-29, DLD-1, and SW480 cells were plated onto 48-well plate and cultured for 8 h. The level of miR-30d was determined by real-time PCR assay. The normal colon epithelial cell line FHC was used as normal control cells (*p < 0.05 and **p < 0.01, compared with control cells).
Transfection of miR-30d mimics inhibits cell viability of colon cancer cells
MiR-30d mimics are chemically modified, double-stranded miRNA-like RNA, which are used to test the function of mature endogenous miRNA upon transfection. In order to test the function of miR-30d in colon cancer cell lines, miR-30d mimics and negative control mimics were transfected into HT-29 and HCT116 cells and the cell viability was determined by MTT assay. As shown in Figure 2(a) and (b), the miR-30d mimics and negative control mimics were transfected into HT-29 and HCT116 cells for 48 h, the expression of miR-30d was upregulated in miR-30d mimic–transfected cells compared with negative control mimic–transfected colon cancer cells (**p < 0.01). Moreover, cell viability was determined by MTT in colon cancer cells transfected with miR-30d mimics or negative control mimics. As shown in Figure 2(c) and (d), cell proliferation was remarkably suppressed in HT-29 and HCT116 cells transfected with miR-30d mimics compared with those transfected with negative control mimics (*p < 0.05; **p < 0.01).

Transfection of miR-30d mimics inhibits cell viability of colon cancer cells. The colon cancer cells (a) HT-29 and (b) HCT116 were transfected with miR-30d mimics and negative control mimics for 48 h. The expression of miR-30d was determined by real-time PCR assay. The relative expression of miR-30d is shown in histogram. (c) The cell viability was determined by MTT assay. The inhibitory rate of miR-30d mimic–transfected HT-29 cells and negative control mimic–transfected HT-29 cells are shown in histogram (*p < 0.05 and **p < 0.01, compared with negative control group). (d) HCT116 cells were transfected with miR-30d mimics and negative control mimics for 48 and 72 h. Cell viability was determined by MTT assay. The inhibition rate was shown in histogram (*p < 0.05 and **p < 0.01, compared with negative control mimic group).
Transfection with miR-30d inhibitors promotes cell viability of colon cancer cells
To further confirm the function of miR-30d in colon cancer cell lines, the miR-30d inhibitors and negative control inhibitors were transfected into colon cancer cells, such as DLD-1 cells and HCT15 cells. MTT assay was performed to detect the cell viability of DLD-1 cells and HCT15 cells, which were transfected with miR-30d inhibitors and negative control inhibitors. As shown in Figure 3, miR-30d was obviously downregulated in miR-30d inhibitor–transfected DLD-1 cells and HCT15 cells. Furthermore, inhibition of miR-30d could significantly promote cell proliferation of DLD-1 and HCT15 cells compared with the negative control inhibitor–transfected colon cancer cells (*p < 0.05; **p < 0.01).

Transfection with miR-30d inhibitors promotes cell viability of colon cancer cells. The colon cancer cells (a) DLD-1 and (b) HCT15 were plated onto 48-well plate and transfected with miR-30d inhibitors and negative control inhibitors for 48 h. The level of miR-30d was determined by real-time PCR assay (**p < 0.01, compared with miR-30d inhibitor–transfected cells). The (c) DLD-1 cells and (d) HCT15 cells were transfected with miR-30d inhibitors and negative control inhibitors for 24, 48, and 72 h. Cell viability was determined by MTT assay. The OD490 values were shown in histogram (*p < 0.05 and **p < 0.01, compared with negative control inhibitor–transfected group).
MiR-30d mimics promote cell apoptosis of colon cancer cells
Next, we determined whether the level of miR-30d affected cell apoptosis in colon cancer cells. HT-29 cells were transfected with miR-30d mimics and negative control mimics for 48 h, and western blotting analysis was performed to determine the apoptosis related proteins, such as caspase-3 and PARP. As shown in Figure 4(a), HT-29 cells were transfected with miR-30d mimics for 48 h and the results demonstrated that overexpression of miR-30d obviously increased the levels of cleaved caspase-3, as well as the levels of cleaved PARP. All the data suggested that the overexpression of miR-30d mimics might induce cell apoptosis of colon cancer cells.

MiR-30d mimics promote cell apoptosis and induce cell arrest at G2/M phase of HT-29 cells. (a) MiR-30d mimics promote cell apoptosis of colon cancer cells. HT-29 cells were plated onto 48-well plate and transfected with miR-30d mimics and negative control mimics for 48 and 72 h. The levels of pro-caspase-3 and cleaved caspase-3 were detected by western blotting analysis. PARP and cleaved PARP levels were tested by western blotting analysis. Here, β-actin was used as an internal reference gene. (b) Cell cycles of miR-30d mimics or negative control mimic–transfected HT-29 cells were analyzed by PI staining analysis. The cell cycle distribution was shown in histogram.
Transfection with miR-30d induced cell arrest at G2/M phase of HT-29 cells
HT-29 cells were transfected with miR-30d mimics or negative control mimics for 48 h. The cell cycle distribution was determined by fluorescence-activated cell sorting (FACS) analysis with PI staining method. As shown in Figure 4(b), the results demonstrated that in miR-30d mimic–transfected group, the cell rate was obviously increased in G2/M phase, as well as the decreased in G0/G1 stage, suggesting that cell cycles were arrested at G2/M phase in miR-30d mimic–transfected group. All the results demonstrated that the overexpression of miR-30d remarkably increased the cell apoptosis of colon cancer cells.
MiR-30d regulates cell autophagy pathway by interacting with 3′-UTR of ATG5, PIK3, and beclin1 mRNA in colon cancer cells
To identify the target of miR-30d in colon cancer cells, we used TargetScan to predict the possible targets. As shown in Figure 5(a), we found that miR-30d has binding sites on 3′-UTR of multiple core proteins in autophagy pathway in humans, such as ATG5, PI3K, and Beclin1, all of which play an important role in the induction of cell autophagy. Moreover, HCT15 cells were transfected with miR-30d inhibitors and negative control inhibitors for 48 h, and the levels of ATG5, PI3K, Beclin1, LC3-I, and LC3-II were determined by western blotting analysis. As shown in Figure 5(b), the ATG5, PI3K, and Beclin1 expression were remarkably upregulated in miR-30d inhibitor–treated colon cancer cells. LC3-I was located in cytoplasm and LC3-II, as the autophagy marker, was stably associated with the augophagosome membranes. The results showed that the level of LC3-II was obviously increased in mir-30d inhibitor–transfected HCT15 cells. In the experiment, 3-MA, the autophagy inhibitor, was used as a negative control and rapamycin, autophagy activator, was used as a positive control. All the data suggested that mir-30d regulated the autophagy by interacting with multiple targeted proteins in autophagy pathway in colon cancer cells.
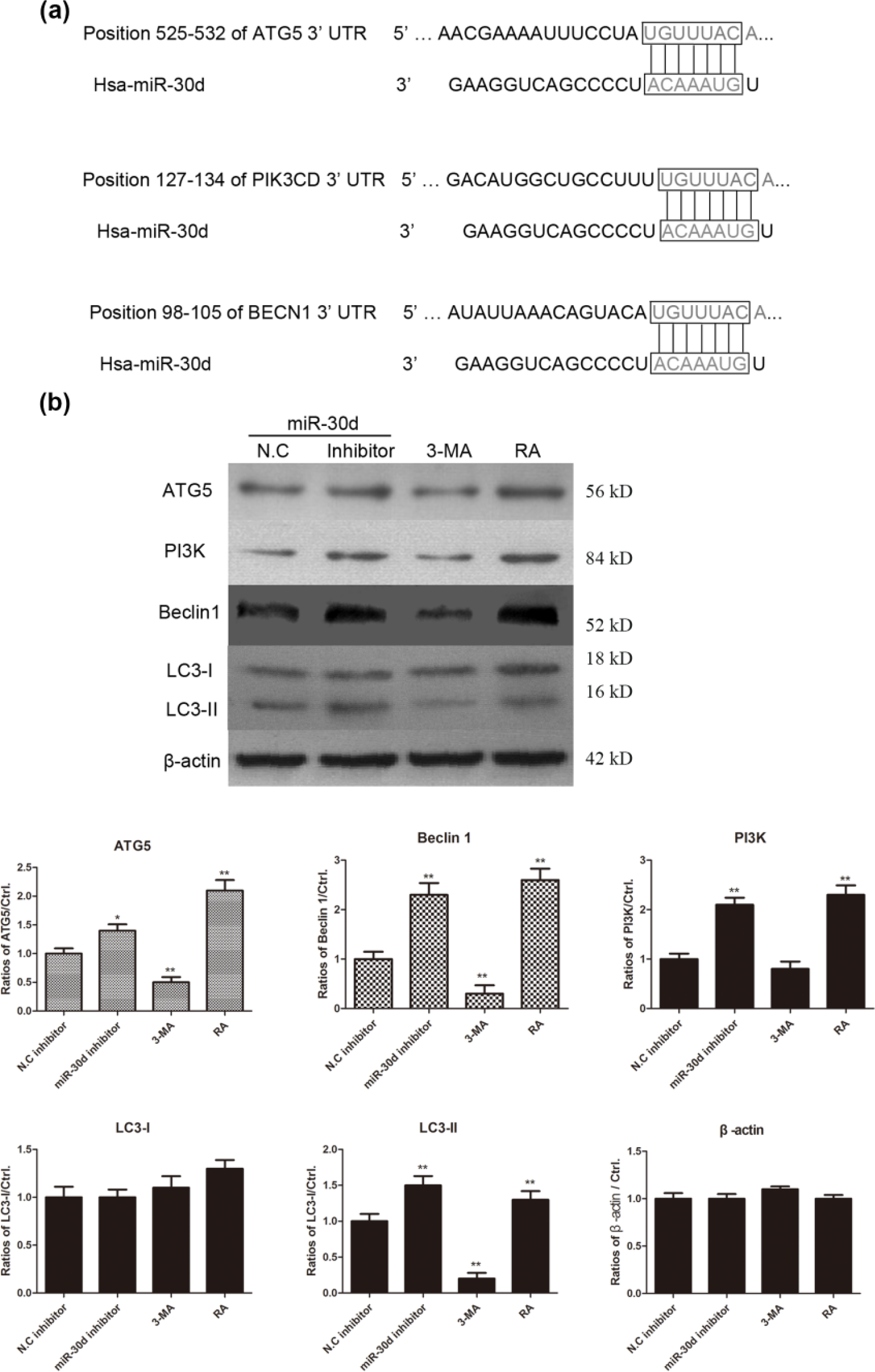
MiR-30d interacts with ATG5, PIK3, and Beclin1 and regulates cell autophagy pathway in colon cancer cells. (a) The possible target of miR-30d was predicted by TargetScan in autophagy signaling pathway. The 3′-UTR of ATG5, PI3K, and Beclin1 in autophagy pathway in humans had binding sites with hsa-miR-30d. (b) The HCT15 cells were treated with miR-30d inhibitors and negative control inhibitors for 48 h. The levels of ATG5, PI3K, Beclin1, LC3-I, and LC3-II were detected by western blotting analysis and the relative expression levels were shown in histogram (*p < 0.05 and **p < 0.01, compared with NC mimic–transfected cells). 3-MA, the autophagy inhibitor was used to treat HCT15 cells as the negative control for autophagy and RA, the activator of cell autophagy was used as the positive control for cell autophagy.
MiR-30d suppresses cell autophagy and promotes cell apoptosis of HT-29 cells
In order to test the association between miR-30d–induced cell autophagy and apoptosis in HT-29 cells, HT-29 cells were transfected with miR-30d mimics and negative control mimics for 48 h. The levels of caspase-3, cleaved caspase-3, LC3-I, and LC 3-II were detected by western blotting analysis. As shown in Figure 6(a), LC3-II level was remarkably decreased in miR-30d mimic–transfected HT-29 cells; however, the level of cleaved caspase-3 was obviously increased, suggesting that transfection with miR-30d mimics could inhibit cell autophagy and increase cell apoptosis of HT-29 cells. This was confirmed in 3-MA treated colon cancer cells.

MiR-30d suppresses cell autophagy and promotes cell apoptosis of HT-29 cells. (a) HT-29 cells were treated with miR-3d mimics and negative control mimics for 48 h plus 5 mM of 3-MA or alone. (a) The levels of LC3B-I, LC3B-II, pro-caspase-3, and cleaved caspase-3 were determined by western blotting analysis. (b) Cell viability of HT-29 cells was determined by MTT assay and OD490 values were shown in histogram (*p < 0.05; **p < 0.01).
Combination treatment with miR-30d mimics and 3-MA remarkably suppresses cell proliferation of HT-29 cells
It has been found miR-30d mimics induced cell apoptosis and reduced cell autophagy of colon cancer cells. A volume of 5 mM of 3-MA was used to inhibit cell autophagy in miR-30d mimic–transfected cells and negative control mimic–transfected HT-29 cells, and the cell viability was tested by MTT assay. As shown in Figure 6(b), cell viability was significantly decreased in miR-30d mimic–treated HT-29 cells alone or in combination with 3-MA. All the data demonstrated that 3-MA promoted cell death in miR-30d mimic–transfected HT-29 cells, suggesting inhibition of cell autophagy would promote cell death of colon cancer cells.
Discussion
MiRNA expression is abnormally expressed in various human cancers. MiRNAs are involved in regulating various cellular processes, such as proliferation, cell apoptosis, cell autophagy, and differentiation. It is reported that about 50% of the miRNA genes are positioned in cancer-associated genomic regions. 21 MiRNAs are widely studied in the progression and invasion of various human cancers, and modulating the expression of miRNAs could reverse the cancer phenotype.22,23 MiRNAs are involved in various cellular processes including proliferation, cell apoptosis, cell autophagy, and differentiation.24,25 Therefore, the clear mechanisms on cancer-related miRNA in the progression of cancers have provided a novel idea for cancer prognosis and clinical therapy. In this study, we detected the role of miR-30d in human colon cancer cell lines, such as HCT15, HCT116, HT-29, DLD-1, and SW480. Importantly, we found that overexpression of miR-30d significantly inhibited cell proliferation of colon cancer cells, which was involved in the promotion of cell apoptosis and inhibition of cell autophagy. As the human colorectal cancer cell lines, such as HCT15, HT-29, DLD-1, SW480, and so on, are obtained from different primary colorectal cancer patients and are not isogenic cell lines, the inherent differences of these cell lines itself might influence miR-30d levels. Thus, we selected more than two colon cancer cell lines to test the role of miR-30d in the following experiments for functional assay.
First, RT-PCR assay was performed to detect the level of miR-30d in several human colon cancer cell lines. We found that miR-30d level was significantly decreased in human colon cancer cell lines compared with FHC, the normal colon epithelial cell line. Moreover, miR-30d mimics and miR-30d inhibitors were transfected into colon cancer cells and the cell viability was determined by MTT assay. The functional studies were performed, and the data demonstrated that transfection with miR-30d mimics suppressed cell proliferation of colon cancer cells and transfection with miR-30d inhibitors promoted cell viability of colon cancer cells. This was consistent with the results from Xuan et al. 26 in prostate cancers, in which they found that the expression of miR-30d was significantly reduced in prostate cancer tissues and the functional study revealed that miR-30d repressed prostate cancer cell proliferation. All the data indicated that miR-30d might be a candidate tumor suppressor miRNA in human colon cancers and prostate cancers. Furthermore, the molecular mechanism was also investigated in colon cancer cells. We wanted to test whether miR-30d was involved in the induction of programmed cell death including cell apoptosis, cell autophagy, and necroptosis. TargetScan tool was used to predict the candidate genes of miR-30d in cell autophagy pathway. Interestingly, the results demonstrated that miR-30d interacted with 3′-UTR of the targeted genes, such as ATG5, PI3K, and Beclin1. The western blotting analysis revealed that overexpression of miR-30d negatively regulated cell autophagy in colon cancer cells. Thus, we speculated that miR-30d-mediated negative regulation of cell autophagy might contribute to the proliferation inhibition of colon cancer cells.
In conclusion, we found the overexpression of miR-30d could inhibit cell proliferation of colon cancer cells by negatively regulating cell autophagy and positively regulating cell apoptosis, suggesting miR-30d might be a candidate tumor suppressor miRNA in colon cancer. It could serve as a potential clinical biomarker and therapeutic target for human colon cancers.
Footnotes
Acknowledgements
J.B. designed the studies and drafted the manuscript. R.Z. performed the experiments. J.X. and J.Z. helped to collect the clinical specimens and to obtain the clinical data.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Fund from the National Natural Science Foundation of China (Grant No. 81672427).
