Abstract
While human papillomavirus vaccine was recently approved by China Food and Drug Administration, mapping of high-risk human papillomavirus distribution and attribution in cervical precancerous lesions in China becomes critical in development of a high-risk human papillomavirus-based cervical cancer screening and prevention strategy. In total, 1016 patients with cervical precancerous lesions diagnosed in the National Cancer Center/Cancer Hospital, Chinese Academy of Medical Sciences were analyzed retrospectively, including 111 patients with low-grade squamous intraepithelial lesions and 905 patients with high-grade squamous intraepithelial lesions. HPV16, 58, 52, 33, and 31 were the most common high-risk human papillomavirus genotypes in order of decreasing frequency among high-risk human papillomavirus-positive high-grade squamous intraepithelial lesions; this differed from the high-risk human papillomavirus distribution in low-grade squamous intraepithelial lesions (HPV16, 52, 39, 56, and 58). The distribution of high-risk human papillomavirus genotypes in single-type infections for high-grade squamous intraepithelial lesions (HPV16, 58, 33, and 52) was similar to that in multiple-type infections (HPV16, 58, 52, and 33). By contrast, a more diverse distribution spectrum of high-risk human papillomavirus genotypes for low-grade squamous intraepithelial lesions was observed between single-type (HPV16, 52, 39, and 56) and multiple-type infection (HPV52, 68, 58, 59, 39 and 56). A previously published method was adopted to calculate the fractional proportion of individual high-risk human papillomavirus genotypes in multiple infections. For this proportional attribution, HPV16 (48.9%), 58 (10.0%), 33 (5.5%), and 52 (5.5%) were the most frequent among all high-grade squamous intraepithelial lesions, whereas HPV16 (13.2%), 52 (11.6%), 39 (9.5%), and 56 (7.6%) were the most frequent among all low-grade squamous intraepithelial lesions. Differences in high-risk human papillomavirus distribution and proportional attribution in different cervical pathology statuses (high-grade squamous intraepithelial lesions and low-grade squamous intraepithelial lesions) demonstrated the critical role of persistent infection of certain high-risk human papillomavirus such as HPV16, 58, 33, and 52 in carcinogenesis of cervical cancer. Distinctively high prevalence of HPV58, 33 and 52 in Chinese cervical intraepithelial neoplasia population, especially in high-grade squamous intraepithelial lesions, should be taken into consideration in cervical cancer screening strategy and vaccine development.
Introduction
Cervical cancer is the most common cancer of the female genital tract and the leading cause of cancer death among such types of cancer death. 1 Infection with high-risk human papillomavirus (HR-HPV) is an obvious cause of invasive cervical cancer.2,3 In all, 15 HPV genotypes (HPV16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 68, 73, and 82) are classified as high risk and are associated with the development of cervical cancer;2,4 12 (HPV16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, and 59) of these strains are known to cause cervical cancer. 3
A cervical precancerous lesion has the potential to evolve into cervical cancer. More than 80% of patients with cervical intraepithelial neoplasia (CIN) are infected with HPV.5,6 Currently, the attribution of each HPV genotype in cervical precancerous lesion has been reported for several regions in China,5,6 but patients from these studies are mainly confined to southern China. The HR-HPV prevalence and distribution vary across ethnic groups, regions, nations, and histological types.7–11 This study investigated the prevalence, distribution, and attribution of HR-HPV genotypes among women with cervical precancerous lesions in China to develop an HR-HPV-based screening approach and determine the spectrum of HR-HPV genotypes to be covered.12,13
Materials and methods
Study subjects
During September 2008 to October 2016, 1016 women with cervical precancerous lesions from the National Cancer Center/Cancer Hospital, Chinese Academy of Medical Sciences were analyzed retrospectively. All HR-HPV tests were performed before treatment. Cervical precancerous lesions including 111 cases of low-grade squamous intraepithelial lesions (LSILs) and 905 cases of high-grade squamous intraepithelial lesions (HSILs) were confirmed by cervical biopsy or conization. LSILs involved HPV infection (condyloma latum), hollow cell atypia, and CIN1; HSILs consisted of CIN2 and CIN3 according to World Health Organization (WHO) classification of tumors of female reproductive organs in 2014. 14 There were 675 patients from North China, 146 patients from Northeast China, 104 patients from Central China, and 91 patients from other parts of China. In our study, only HR-HPV genotypes were considered. This study was approved by the Local Institutional Ethics Committee.
HPV detecting and typing
Cervical samples were obtained from gynecological examination in our hospital. The 21 HPV GenoArray Diagnostic Kit (HybriBio Ltd., Chaozhou, China) approved by the China Food and Drug Administration was used to detect 21 specific HPV DNA types in cervical specimens. The assay is optimized to detect 14 HR-HPV DNA types (HPV16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66, and 68),4,15 5 low-risk HPV (LR-HPV) DNA types (HPV6, 11, 42, 43, and 44), and 2 unknown-risk HPV (UR-HPV) types (HPV53 and 81 equivalent to CP8304). 6
HPV DNA was extracted from cervical samples using cell lysis; the HPV DNA that was extracted was amplified by polymerase chain reaction (PCR). Amplified DNA amplicons were hybridized with special HPV probes followed by the colorimetric result obtained using the enzyme immunoassay method. All detecting steps were performed in accordance with the manufacturers’ instructions.
Statistical analysis
Irrespective of single-type or multiple-type infection, the prevalence rate of each HPV infection type was calculated as the total number of positive samples divided by total number of samples examined in one disease grade (LSIL or HSIL). The relative distribution of each HR-HPV genotype was calculated using the total number of corresponding HR-HPV genotypes as the numerator and that of all positive cases for one disease grade as the denominator. When more than one HPV genotype was identified in a sample, cases with multiple-HR-HPV infections were counted as each corresponding genotype. Thus, the number of each genotype might be more than that of the positive samples with matching HPV.
We adopted a previously published method to approximate the attribution of multiple HPV genotypes.5,16 Each type of multiple-type infection was believed to have a fractional attribution to the disease development. The attribution factor for each HPV genotype was calculated as the positive samples of corresponding HPV in single-type infections divided by the total number of all positive cases in single-type infections for one histological type. The attribution of an HPV genotype for that disease grade was (the number of single-type infections of the HPV concerned plus the number of multiple-type infections of the HPV concerned multiplied by the attribution factor)/all study cases for that disease grade. 5 We adopted the Wilson score method to estimate the 95% confidence interval. 17 The Chi-squared test was used to compare the HR-HPV prevalence of different cervical pathology statuses. Furthermore, a comparison of the different age groups was performed with a Chi-squared test; p < 0.05 was considered statistically significant. Data were analyzed with SPSS (version 22.0).
Results
The study population was divided into eight age groups (range = 22–77 years). The HR-HPV prevalence in the different age groups is shown in Figure 1. The prevalence of HR-HPV infection among the women with cervical precancerous lesions had one peak at 45–59 years (95.4%). There was a statistical difference between that age group (45–49 years) and all other age groups (30–34, 35–39, 40–44, 50–54, or ≥60 years; Table 1).

The prevalence of HR-HPV infection among women with cervical precancerous lesions in different age groups.
Comparisons between the 45- to 49-year-old age group and all other age groups.

All study samples for each age group are included as a denominator.
Although the HR-HPV-positive rate among patients with cervical precancerous lesions in Northeast China was the highest (91.8%), there was no statistical significance between four different regions (p > 0.05). The single-type HR-HPV infection rate among the women with cervical precancerous lesions in Northeast China was higher than that in East China (p < 0.05; Table 2).
HR-HPV-positive rates of infection types based on population distribution.
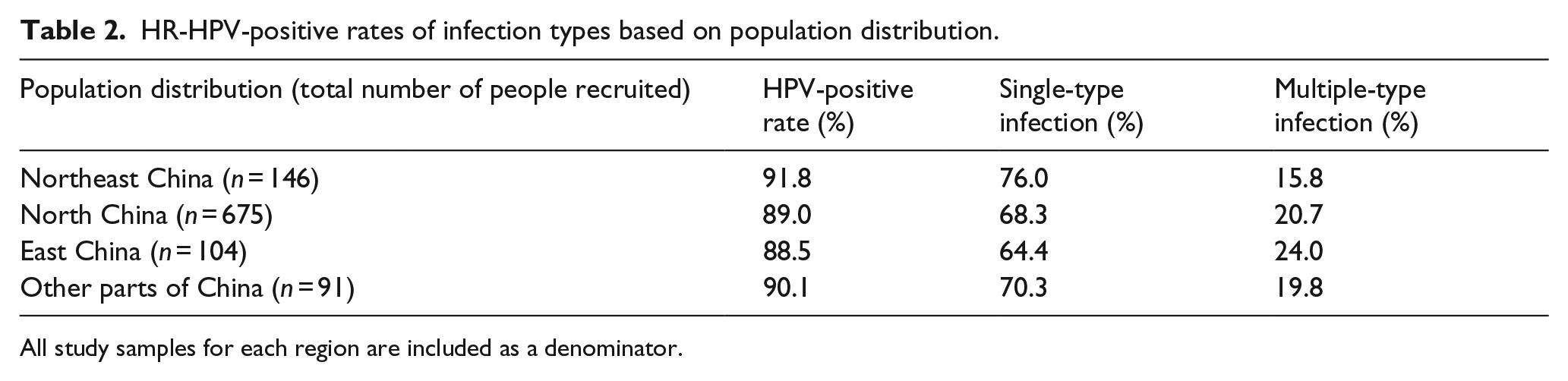
All study samples for each region are included as a denominator.
Of the 1016 women with cervical precancerous lesions, 909 (89.5%) were screened and shown to have an HR-HPV infection. Of the 1016 cases, 111 were LSILs and 905 were HSILs. The HR-HPV-positive rates of single and multiple-type infections (co-, tri, or more) are displayed in Table 3. The prevalence of HR-HPV for LSILs was substantially lower than that for HSILs (p < 0.001). The single-type infection rate increased with the aggravation of cervical squamous intraepithelial lesions (p < 0.001).
HR-HPV-positive rates of infection types based on cervical histological types.

LSIL: low-grade squamous intraepithelial lesion; HSIL: high-grade squamous intraepithelial lesion.
All study samples for one disease grade are included as denominator.
Compared to other HR-HPV genotypes, HPV16 (56.3%) ranked first among HR-HPV-positive women with cervical precancerous lesions, followed by HPV58 (16.0%), HPV52 (12.0%), HPV33 (9.0%), and HPV31 (6.6%; Figure 2).

Relative distribution of HPV genotypes among HR-HPV-positive women with cervical precancerous lesions (n = 909). Participants with multiple HR-HPV genotypes detected are counted for each genotype.
Figure 3 shows the relative distribution of the 14 HR-HPV types for the 909 HR-HPV-positive women with LSILs and HSILs. The most common HR-HPV genotype was HPV16 (60.2%), followed by HPV58 (16.5%), 52 (11.2%), 33 (9.7%), and 31 (6.9%) in HSILs. The percentage of HPV16 accounted for more than 60% of all HR-HPV in HSILs. By contrast, HPV16 (19.5%), 52 (19.5%), 39 (16.1%), 56 (13.8%), and 58 (10.3%) were the five most common types found in LSILs. HPV16 was found in less than 20% of LSILs.

Relative distribution of HR-HPV genotypes among HR-HPV-positive cervical LSILs (n = 87) and HSILs (n = 822). Participants with multiple HR-HPV genotypes detected are counted for each genotype.
HR-HPV infections were divided into single-type infections and multiple-type infections. In single-type infections, HPV16 (16.1%), 52 (13.8%), 39 (11.5%), and 56 (9.2%) were the most common HR-HPV genotypes among HR-HPV-positive LSILs (Figure 4(a)). The distribution of HR-HPV genotypes in single-type infections for LSILs was not as concentrated. HPV16 (45.0%), 58 (10.2%), 33 (5.7%), and 52 (5.6%) ranked the top four places among HR-HPV-positive HSIL (Figure 4(b)). The most common HR-HPV genotype in single-type infections for HSILs was HPV16. In comparison, in multiple-type infections, HPV52 (5.7%), 68 (5.7%), 58 (4.6%), 59 (4.6%), 39 (4.6%), and 56 (4.6%) were the most frequent types found in HR-HPV-positive LSILs (Figure 4(c)). However, HPV16 only ranked seventh in LSILs (3.4%). HPV16 (15.2%), 58 (6.3%), 52 (5.6%), and 33 (4.0%) were the most common genotypes found among HR-HPV-positive HSILs (Figure 4(d)). Figure 4 shows that the top four HR-HPV genotypes for HSILs differed little between single- and multiple-type infections. The spectrum of HPV genotypes for LSILs in multiple-type infections was completely different from the other three scenarios.

Relative distribution of HR-HPV genotypes for different cervical pathology statuses in single- or multiple-type infections. Participants with multiple HR-HPV genotypes detected were counted for each genotype. (a) HPV genotypes in single-type infections for LSILs. (b) HPV genotypes in single-type infections for HSILs. (c) HPV genotypes in multiple-type infections for LSILs. (d) HPV genotypes in multiple-type infections for HSILs.
HPV16 (54.7%), 58 (15.0%), 52 (10.2%), and 33 (8.8%) were the most prevalent types found in HSIL, and their corresponding attribution rate was 48.9%, 10.0%, 5.5%, and 5.5%, respectively. HPV16, 52, 39, and 56 occupied the majority of LSIL with attribution rate of 13.2%, 11.6%, 9.5%, and 7.6%, respectively. The prevalence and attribution of the top four genotypes varied greatly between LSIL and HSIL (Table 4).
Crude prevalence and attribution of each HR-HPV genotype according to histological types.
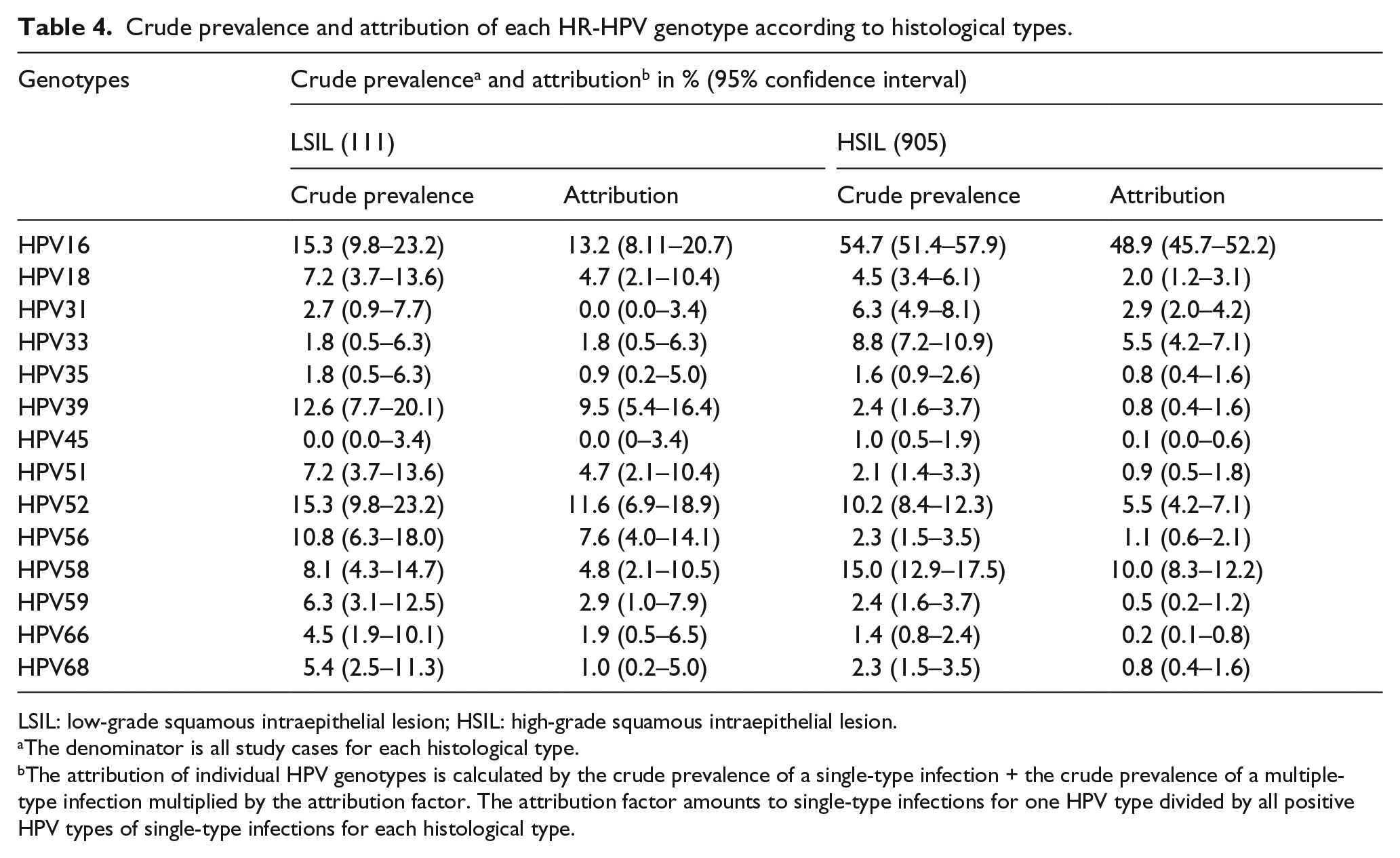
LSIL: low-grade squamous intraepithelial lesion; HSIL: high-grade squamous intraepithelial lesion.
The denominator is all study cases for each histological type.
The attribution of individual HPV genotypes is calculated by the crude prevalence of a single-type infection + the crude prevalence of a multiple-type infection multiplied by the attribution factor. The attribution factor amounts to single-type infections for one HPV type divided by all positive HPV types of single-type infections for each histological type.
In all, 17.9% of LSIL, 50.8% of HSIL, and 46.9% of all histological types combined could be attributed to vaccine-covered high-risk genotypes (HPV16/18). The attribution rate of HPV16 was the highest, regardless of all histological types combined, including LSILs or HSILs. After HPV16, 58, 33, and 52 comprised most histological types in HSILs. In LSILs, HPV52, 39, and 56 accounted for 11.6%, 9.5%, and 7.6%, respectively, after HPV16. The addition of HPV58, 33, and 52 to HR-HPV screening would cover an additional 21.0% of HSILs (Figure 5).

Cumulative attribution rates of 12 (HPV16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58 and 59) HR-HPV genotypes for cervical intraepithelial lesions. Among the 12 HR-HPV genotypes, genotypes with ≤1% were not displayed. Accumulated numbers are represented in boldface. (a) All cervical precancerous lesions. (b) Low-grade squamous intraepithelial lesions (LSILs). (c) High-grade squamous intraepithelial lesions (HSILs).
Discussion
Persistent infection with HR-HPV is the main cause of cervical squamous intraepithelial lesions and cervical cancer.18,19 Therefore, learning about the characteristics of HR-HPV prevalence would contribute to taking effective measures to prevent and control the incidence of cervical intraepithelial lesions and cervical cancer. This study provided more information on the relative distribution and attribution of HR-HPV genotypes for cervical precancerous lesions in China. CIN was classified as LSILs and HSILs according to histological subtypes based on 2014 WHO classification. 14 The prevalence peak of HR-HPV among cervical precancerous lesions occurred in the 45- to 49-year-old age group (95.4%). A study published by the Oxford University Press showed that the peak age of HR-HPV persistent infection resulting in HSILs was generally older than 42 years. 18
The sources of the patients were divided into Northeast China, North China, East China, and other parts of China, where the HR-HPV infection rates in patients with precancerous lesions were 91.8%, 89.0%, 88.5%, and 90.1%, respectively. The difference in the HR-HPV infection rates in four different regions was not significant. Xu et al. 6 reported that the HPV infection rate for CIN2 and 3 in Zhejiang was 81.3% and 86.8%, respectively. A study in East China shows that the HPV infection rate is 81.4% for CIN2 and 88.1% for CIN3. 20 A study in Gansu, China displayed that the HPV infection rate was 87.5% for CIN2/3. 21 These indicate that the difference in the HPV infection rate for CIN2 and 3 in different regions of China was not great. The patients with precancerous lesions in Northeast China (76.0%) had a higher single-type HR-HPV infection rate than that in East China (64.4%; p < 0.05). This might be associated with the different lifestyles and living environment of people in Northeast China and East China. Although the proportion of the multiple-type infection in East China (27.2%) was higher than that in Northeast China (17.2%), no statistical difference was found between two regions.
In this research, the prevalence of HR-HPV for HSILs (90.8%) was significantly higher than that for LSIL (78.4%). The single-type infection rate increased as the level of cervical pathology status increased. The results are the same as Chan et al. 5 and Xu et al. 6 ’s results. Multiple HPV infections are reported to have a higher proportion of LSILs; 5 this result was not supported by our study.
The top five HR-HPV genotypes among HR-HPV-positive HSIL or all precancerous lesions were HPV16, 58, 52, 33, and 31, similar to Shanghai and Zhejiang’s results.6,22 Among HR-HPV-positive LSILs, HPV16, 52, 39, 56, and 58 were the top five genotypes; this differed from the distributions of HR-HPV genotypes for HSILs. Additionally, HPV distributions for LSILs in China are different in various studies.5,6,20 This suggests that women with LSILs usually merge with temporary HPV infections. 23 One study reported that HPV16, 18, 31, and 33 persistent infections were associated with HSILs, particularly HPV16. 24 As this study shows, the HPV16 distribution accounted for more than 60% of all HR-HPV genotypes among HR-HPV-positive HSILs. Among HR-HPV-positive LSILs, HPV16 (19.5%) was not absolutely superior in the proportion of all HR-HPV genotypes although it ranked first.
Based on the study of the relative distributions of HR-HPV genotypes among HR-HPV-positive LSILs or HSILs, we further studied the HR-HPV distributions of single- or multiple-type infections in different pathology statuses. For single and multiple HR-HPV infections in HSILs, the difference in the distribution of the top four HR-HPV genotypes was not significant, indicating that the main HR-HPV genotypes for HSILs had a relatively steady distribution. By contrast, a more diverse spectrum of HR-HPV genotypes for LSIL was observed in single-type (HPV16, 52, 39, and 56) and multiple-type infections (HPV52, 68, 58, 59, 39, and 56). This also indicates that HR-HPV distributions in different infection types for LSILs may not be stable.
It is currently believed that the duration of HPV infection in cervical LSIL is transient. 25 Ciavattini et al. 26 reported that the spontaneous regression rate of LSIL was 88.5%, the women with a persistent lesion accounted for 10.8%, and a progression toward HSIL was reported only in 0.7% of the women. HPV16, 58, 52, and 33 are the most common types among the women with persistent HR-HPV infections in China.27,28 Many LSILs could be caused by transient infection of other HR-HPV types. In addition, some studies reported that multiple HPV infections of LSILs had a higher proportion than that of HSILs.5,20 These may lead to a constant distribution of the top four HR-HPV genotypes in patients with HSILs but not in the LSIL group.
For multiple HR-HPV infections, some main HR-HPV genotypes might play a more dominant role in cervical lesion development than others. 16 Some studies have suggested that multiple HPV infections are not associated with a higher risk of the development of cervical disease.29,30 Research by Depuydt et al. 31 shows that the linear viral load increases in a single HPV genotype in multiple-type infections, indicating the development of cervical cancer. Therefore, we adopted an approach that was previously published to analyze the attribution of each HR-HPV genotype in multiple-type infections. 16 Among all cases with HSILs, HPV16 (48.9%), 58 (10.0%), 33 (5.5%), and 52 (5.5%) were the primary genotypes present. In comparison, HPV16 (13.2%), 52 (11.6%), 39 (9.5%), and 56 (7.6%) were the most frequent genotypes among all cases with LSILs. Compared to the distribution of HR-HPV for HSILs worldwide (HPV16, 31, and 33),32,33 after HPV16, the proportions of HPV58 and 52 were higher in China. Many studies in China have findings consistent with our results.5,6,27,34 HPV58 and 52 had proportions of 15.4% for HSILs and 16.4% for LSILs in China.
HPV16, 58, 33, and 52 belong to HPV16 species group (alpha-9) of the Alpha papillomavirus genus. 35 We aligned the DNA sequences and amino acid sequences of the four types for the minor capsid protein L2 using DNAMAN software and found their consistencies were 80.41% and 77.64%, respectively. It is possible to find a conserved domain on the L2 protein and design a polypeptide that could elicit a novel antibody in humans or animals against these differential genotypes of HR-HPVs. For example, a minimal epitope on the L2 protein could be recognized by an anti-L2 monoclonal antibody, 14H6, which could broadly crossly neutralize at least 11 types of HPV. 36 If a new antibody could be used to detect the four HR-HPV types by enzyme-linked immunosorbent assay (ELISA), approximately 70% of HSIL in our study could be found and got prompt treatment. Previous studies have indicated that HPV-positive oropharyngeal squamous cell carcinomas (OPSCCs) contain significantly higher levels of DNA methylation at some loci compared with the control cells.37,38 If DNA hypermethylation was confirmed in cervical lesions with HPV infections, these lesions should adopt more aggressive treatment.
There are some limitations in our study. First, this study was retrospective; some cases had losses that might cause certain biases. Second, the population distribution in our study was uneven. Third, more study samples and prospective studies are necessary to support our results in the future.
In conclusion, the prevalence of HR-HPV increased with the aggravation of cervical precancerous lesions. The single-type HR-HPV infection rate among the women with cervical precancerous lesions in Northeast China was higher than that in East China. The main HR-HPV genotypes for HSIL had a stable distribution. We should be more concerned about a persistent infection of HPV16, 58, 33, and 52, which have a higher attribution in HSIL. Compared to worldwide rates, HPV58 and 52 had a higher proportion of distribution and attribution in China. 34 The addition of HPV58, 33, and 52 to future screening protocols and vaccines would facilitate the earlier detection of cervical precancerous lesions and prevent them from progressing to cervical cancer. Screening of the main types (HPV16, 58, 33, and 52) of HR-HPV in cervical precancerous lesions would also cut costs and benefit economically underdeveloped areas in China.
Footnotes
Acknowledgements
The authors would like to acknowledge Ms Huang Ying, MD, who had refined English for this paper. W.W. and J.A. have contributed equally to this paper. L.W. provided the critical guidance for the study and, with all co-authors, carried out the design. M.H., W.W., and J.A. took responsibility for recruitment of study participant. Y.S. was responsible for pathological confirmation. M.W. was responsible for the detection of HPV typing. W.W. and J.A. took responsibility for the accuracy of the data analysis. W.W. drafted the manuscript, which was revised by L.W. All the authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by CAMS Innovation Fund for Medical Sciences (CIFMS; item number: 2016-I2M-1-001).
